Abstract
We have attempted a green alternative to reuse the spent fluid catalytic cracking (FCC) catalyst that is used in petroleum refining industry for the upgradation and purification of various petroleum streams and residues. The spent FCC zeolite–based catalyst modified by enhancing the acidic properties by incorporating Zn and In metals in the matrix. The various prepared catalysts were systematically characterized by X-ray powder diffraction and Brunauer–Emmett–Teller (BET; adsorption isotherm) surface area. The acidity of the materials was studied by temperature-programmed desorption of ammonia (NH3-TPD). The well-characterized catalysts were applied for liquid phase benzylation of o-xylene using benzyl chloride.
Keywords
Introduction
Green chemistry, also called sustainable chemistry, is a philosophy of chemical research and engineering that encourages the design of products and processes that minimize the use and generation of hazardous substances. 1 In the last decade, it has been recognized as a new approach to scientifically based environmental protection, and catalysis has manifested its role as a fundamental tool in pollution prevention. 2,3
Large variety and volume of fluid catalytic cracking (FCC) catalysts are used in the petroleum refining industry for the upgradation and purification of various petroleum streams and residues. The catalysts deactivate with time and when the activity of the catalyst declines below the acceptable level, it is usually regenerated and reused. But regeneration is not always possible and after a few cycles of regeneration and reuse, the catalyst activity may decrease to very low levels and further regeneration may not be economically feasible. Hence, the spent catalysts are eventually discarded as solid wastes. The quantity of spent catalysts discharged from different processing units depends largely on the amount of fresh catalysts used, their life as well as the effect of reaction conditions like temperature, pressure amount, and type of the deposits formed during use. In most refineries, a major portion of the spent catalyst waste comes from hydrotreating, FCC, and hydroprocessing units. This is because the catalysts used in these processes deactivate rapidly by coke and metal (V and Ni) deposits and have a shorter life. 4,5 Furthermore, technology for regeneration and reactivation of the catalysts deactivated by metal fouling is not available to the refiners. Much information or literature was not available for utilization of the spent catalyst in fine chemicals synthesis.
Friedel–Crafts alkylations are a very important class of reactions commonly used in organic chemistry. 6,7 Among these reactions, the liquid phase benzylation of benzene and other aromatic compounds by benzyl chloride (BC) or benzyl alcohol is important for the production of diphenylmethane and substituted diphenylmethane which are industrially important compounds used as pharmaceutical intermediates or fine chemicals. 7,8 These reactions are generally known to proceed over homogeneous Lewis acid catalyst using AlCl3, FeCl3, BF3, ZnCl2, and H2SO4. 9 However, these acid catalysts are toxic and corrosive. Moreover, the isolation of the product is very difficult and causes severe pollution. The new environmental legislations urge for the replacement of all liquid acids by solid acid catalysts that are environmentally friendly and allow minimizing pollution and waste. 10 Indeed, several solid acid catalysts with a good efficiency, which can be easily separated from the reaction mixture and have high catalytic activity, have been proposed for the benzylation of o-xylene and other aromatic compounds.
Due to their shape selectivity, thermostability, the easy separation from the products, and the possibility of regeneration of the deactivated catalysts, zeolite catalysts are used extensively in the petroleum refining and petrochemical industries for various cracking, hydrocracking, isomerization, alkylation, dehydrogenation, and rearrangement reactions of hydrocarbons and their derivatives. The use of zeolite catalysts in the synthesis of fine chemicals is an increasing area of application of growing importance in recent years. 11,12
The study has undertaken to modify the zeolite-based spent FFC catalyst for Friedel–Crafts alkylation reaction. The various catalysts were synthesized by incorporating Zn+2 and In3+ ions to enhance Lewis acid sites into the pores of the spent catalyst by wet incipient technique. In this work, an attempt has been made to study the benzylation of benzene, toluene, and mainly o-xylene with benzylchloride (BC) using the modified spent catalyst as heterogeneous catalyst.
Experimental
Chemicals and Catalyst
Commercially available chemical reagents (AR Grade, Sigma Aldrich & Labort Fine Chem Pvt. Ltd.) are of high purity and were used directly without further purification. The spent catalyst received from Reliance Industries Limited was used in this study. It mainly contains Zeolite-Y, ZSM-5, additives, and clay binders. The spent catalyst calcined in the air at 550 °C for 6 h to remove the adsorbed material and coke was named as decoked spent catalyst (DSC).The DSC is used for metal impregnation and further study.
Preparation of Zn and In-supported Catalyst
The Zn and In containing various amounts of ZnO (10 and 20 wt %) and In2O3 (5, 10, and 20 wt %) were prepared by a standard wet impregnation. In this method, the required quantity of zinc chloride (source of zinc) and indium nitrate monohydrate (source of indium) were dissolved in methanol. The solution was added in small aliquots of 1 mL each time to the 10 gm of decoked catalyst with constant stirring using a glass rod or kneading it properly. The paste on further kneading for 10 min resulted in a free flowing powder. The preformed catalyst was dried at 120 °C for removal of water and other occluded volatiles and subsequently calcined at 550 °C for 4 h.
The analysis of the spent catalyst was performed by using microwave digestion followed by estimation using Inductively Coupled Plasma Optical Emission Spectroscopy (ICP-OES), and the data are presented in Table 1.
Chemical Composition of Spent Catalyst (Mass %).

Catalyst Characterization
Physical and chemical properties of the spent and modified catalyst were determined using a number of different methods and techniques. Surface area and pore volume of the catalysts were determined using QUANTACHROME (Model Autosorb-I) physical adsorption series based on the BET method. Philips PW1840 type X-ray diffractometer was used to determine the silver crystal planes and phases. Diffused reflectance ultraviolet–visible (UV–vis) spectra (UV-DRS) measurements were carried out on a Purkinje TU-1901 UV/Vis spectrophotometer (Beijing, China) equipped with a diffused reflectance accessory with an IS19-1 integrating sphere, and BaSO4 powder was used as reference.
Temperature-programmed desorption of ammonia (NH3-TPD) was carried out to evaluate acidic properties of the DSC and the metal-loaded DSC using Micromeritics (Chemisorb 2750) system.
Experimental Procedure and Analysis
The liquid phase benzylation of o-xylene with BC was carried out in a 50 mL two-necked flask attached to a condenser and a septum. The temperature of the reaction vessel was maintained using an oil bath. In a typical run, o-xylene and BC were added in the required molar ratio to the activated catalyst (Zn and In/DSC). The catalysts were activated at 200 °C in air for 2 h with a flow rate of 50 mL/min and cooled to room temperature prior to their use in the reaction. The reaction mixture was magnetically stirred and heated to the required temperature under atmospheric pressure. The temperature of the reaction vessel was maintained using an oil bath at 80 °C. In a typical reaction mixture consist of o-xylene to benzyl chloride mole ratio 1:20 with a catalyst loading of 0.5 g at 80°C temperature was used. The reaction mixture was allowed to reach the desired temperature, the initial/zero time samples was withdrawn, and the catalyst was added thereafter. The product samples were analyzed periodically by a gas chromatograph (Thermo Finnigan Trace GC) equipped with a flame ionization detector and a capillary column (Restek-5). The products were also identified by injecting authentic sample and GC/MS (Thermo scientific Trace GC ultra). Finally, the percentage conversion (wt %) of BC is defined as the total percentage of BC transformed. The reaction scheme is as shown in Figure 1.

Reaction scheme.
Results and Discussion
Catalyst Characterization
The prepared catalysts were systematically characterized by various techniques.
Surface Area and Acidity Measurement
The BET surface area, pore volume, and acidity strength by ammonia TPD of the prepared catalyst are presented in Table 2. The total surface area and pore volume of DSC is 156.6 m2/g and 0.22 cm3/g, respectively, which reduced to 49.9 m2/g and 0.19 cm3/g upon 20% Zn loading. The similar type of phenomena was observed in case of indium-supported catalyst. It indicates that insertion of metal into the aluminosilicate framework leads to a decrease in the BET surface area and pore volume, which can be explained by a change of the pore shape due to the change of the cell parameters.
Surface Acidity, Surface Area, and Pore Volume.

NH3-TPD is used to estimate the acid property of the catalysts qualitative and quantitatively. It is known that ammonia is an excellent probe molecule for testing the acidic properties of solid catalysts. Its strong basicity and smaller molecular size allow detection of acidic sites located in the narrow pores of the solids. Hence, the total amount of NH3 desorbed relates to the number of acidic sites; and from the desorption temperature, the strength of acidic sites is suggested. The results of NH3-TPD of the catalysts are tabulated in Table 2. The amount of acid or acidity is expressed as moles of NH3 desorbed per unit weight of the solid. It is also clearly seen from Table 2 that the acidity of decoked catalyst is 0.20 mmol/g and it increases with 10% of zinc loading. However, further increase in zinc loading did not increase the acidity. This could be due to shift in strong acid sites leading to NH3 desorption at higher temperature.
X-ray Diffraction Study
Figure 2 shows the X-ray diffraction (XRD) of DSC, 10% and 20% Zn-supported catalyst. The crystalline peaks of DSC (MP008 sample) have been diminished by zinc loading. Chen et al. 13 reported that in case of ZSM-5 there is no change in its crystalline phase for more than 4% zinc loading on zeolites. They have demonstrated that ZnO clusters have been formed in the pores of zeolite and no macrocrystalline ZnO exists. Similar results were obtained by Choudhary et al. 14 with no phase change with metal (Fe, Zn, In, and Ga) loading. Mondal et al. 15 reported that the (002) peak appears with maximum intensity at 34.55, and the other peaks at 31.85°, 36.35°, 47.6°, and 56.85° can be associated with (100), (101), (102), and (110) peaks of ZnO, as is expected for hexagonal ZnO structure. But in this study, the peaks of ZnO did not appear, and phase change that had taken place with zinc loading is quite unpredictable due to complex structure of spent catalyst.
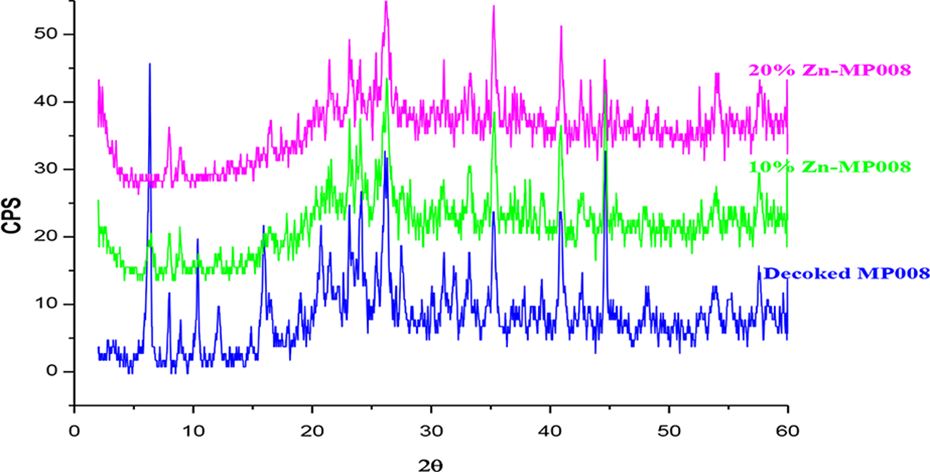
XRD of DSC, 10% and 20% Zn/DSC.
In case of pure zeolite, Choudhary et al. 14 have reported that there is no structural or crystalline phase change with indium loading. When indium 16,17 supported DSC, the crystalline phases of the spent catalyst have diminished and new peaks are formed at d-spacing of 2.92, 1.79, and 2.53 Å, which are attributed to the major peaks of In2O3 at 222, 400 (cubic structure), and 440 planes, respectively. These results indicate that indium is present on the surface only and has not entered inside the pores of the spent catalyst as shown in Figure 3.

XRD patterns of DSC, 5, 10, and 20% In/DSC.

Diffused reflectance ultraviolet–visible spectra (UV-DRS) of DSC and 20% Zn/DSC.
UV–vis Diffusive Reflectance Spectroscopy (DRS)
UV–vis diffusive reflectance study showed that the absorption spectra in the UV–vis region for DSC and Zn- and In-supported DSC. From figure 4, it is observed that DSC and Zn-supported DSC exhibit strong absorption ligand-to-metal charge transfer (LMCT) broadband at 295 nm. It indicates that Zn is available on the surface and not strongly bonded with support so there is no shift in absorption band due to d–d transition. The similar type of pattern is also observed in the case of indium-supported DSC. A faint absorption band around 440 nm was observed due to interaction of indium with support on weaker side. Whereas according to Chen et al., 13 band gap of macrocrystalline ZnO appears at absorption band at about 370 nm with more than 4 wt % ZnO loading. However, no absorption band was observed at higher ZnO loading on DSC. This suggests that Zn exchange has not occurred inside the pores.
Effect of Zinc Loading on DSC
The DSC, 10% and 20% Zn-modified DSC are compared for their benzene benzylation activity at 80 °C. The BC conversion versus reaction time was plotted (Figure 5). DSC as such did not catalyze the reaction but with 10% and 20% zinc loading on DSC, conversion was 30% and 75%, respectively, which was attained in 6 h of reaction time.

Effect of zinc metal loading.
Activity of In over Zn supported on DSC
The activities of 20% In and 20% Zn supported on DSC are compared under identical reaction conditions for benzylation reactions and are shown in Figure 6 (temperature = 80 °C, catalyst = 0.5 g, mole ratio = 20:1), and benzylation of o-xylene with BC is carried out. It was observed that the rate of reaction is higher in case of In compared to Zn metal. In general, the benzylation activity of the zeolite catalysts is in the following order: In2O3/DSC > ZnO/DSC. Also the order of the redox potential for the metal oxides in zeolite catalysts is

Effect of Zn and In metal on benzylation.
Effect of Different Aromatics
The effect of three different aromatics was investigated under optimum reaction condition (Catalyst = 0.5 g, Temperature = 80 °C, stirring speed = 600 rpm, and mole ratio 20:1) using 20% Zn-loaded DSC as the catalyst. The results are shown in Figure 7. The rate of BC conversion was higher in case of o-xylene followed by toluene and benzene. According to the classical mechanism of the Friedel–Crafts type acid catalyzed benzylation reaction, the benzylation of an aromatic compound will be easier if one or more donating groups are present in the aromatic ring. These reactions show similar trend. In the present case, the activity of the zinc-loaded DSC for the benzylation of the aromatic compounds with donating groups is as expected as that of the classical mechanism.

Effect of different aromatics.

Reaction mechanism.
Reaction Mechanism
The possible reaction mechanism for the benzylation of o-xylene catalyzed on indium-loaded DSC is shown in Figure 8. As it can be observed, indium species have a key role in the catalytic activity. Considering the trend followed for the adsorption of o-xylene and BC (Figure 8), it seems that the high catalytic activity of the indium-loaded DSC is related to the strong interaction of the BC with indium oxide active sites.
According to Choudhary and Jana, 14 the adsorption takes place on tetrahedral indium atoms by the formation of a benzyl cation (C6H5CH2 +) and a chloride ion in a process involving the homolytic rupture of the C–Cl bond of this molecule catalyzed by In3+, which suffer a redox cycle (In3+/In2+). This effect can explain that in this work, increasing the amounts of the metal leads to most active catalysts.
Concerning the interaction between o-xylene and metal species, although Choudhary and Jana propose that indium oxide also participates in o-xylene activation, 14 this possibility is not clear according to our experimental results, since both the strength of the adsorption and the specific interaction (which provides an insight into the chemical interaction between o-xylene and catalyst surface) are lower for the metal-loaded spent catalyst. This result led to think that there is a high surface concentration of o-xylene in all the cases due to the formation of reactive species from BC, the determinant step for this reaction. In this reaction, the metal support interaction is weak, so it undergoes homogenous catalysis mechanism as shown in Figure 8.
Conclusion
A series of Zn- and In-loaded DSC catalysts were prepared and systematically characterized. The acidity of the materials increases with the increase in ZnCl2 loading; however, the surface area and pore volume decreased due to effective loading of ZnCl2 on the channels of zeolite. Friedel–Crafts alkylation of benzene with BC was studied systematically using 20% zinc-supported DSC as solid acid catalyst. The catalyst reusability and enhancement of interaction of Zn and In with support using various techniques are in progress. The green alternative of utilization of spent catalyst by further modification for Friedel–Crafts alkylation reaction has been attempted.
Footnotes
Acknowldgment
Authors wish to acknowledge technical support rendered by Mr. D.P. Patel, Mrs. Sangita Rathod, Mrs. Rashmi Dave, and Mr. J.T. Vora.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
