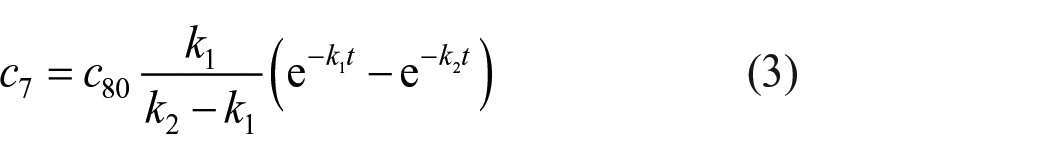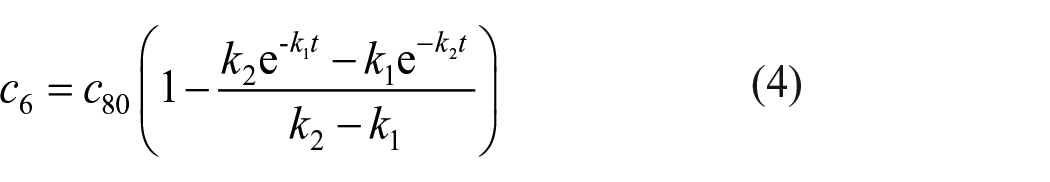Abstract
Various isomers can be observed in the Friedel–Crafts alkylation of benzene. However, the mechanism and influence of these isomerization reactions were still unclear. In this work, several methyl phenylundecanoate were analyzed by gas chromatography–mass spectrometry to explore the isomerization of the Friedel–Crafts alkylation. The results showed that there were three isomerization reactions in the Friedel–Crafts alkylation reaction: five isomers of methyl 6–10-phenylundecanoate due to carbocation rearrangement, five isomers of methyl 6–9-methyldecanoate and methyl undecanoate due to methyl migration, and the loss of neutral alcohol molecular fragments due to hydrogen transfer. At higher reaction temperature, carbocation migration was more active and the formation of methyl undecanoate isomers was observed. There was a competitive relationship between the production of methyl undecanoate isomers and methyl phenylundecanoate isomers. In addition, the hydrogen transfer could be initiated in the presence of unsaturated bonds and alcohol molecules lost as a fragment. The hydrogen from the phenyl groups, which were at a different position on the carbon chain of fatty acid esters, could be transferred to the ester group. It was found that hydrogen could be transferred intramolecularly over a long range. This work revealed the isomerization process in the Friedel–Crafts alkylation and further deepened the understanding of alkylation.
Keywords
Introduction
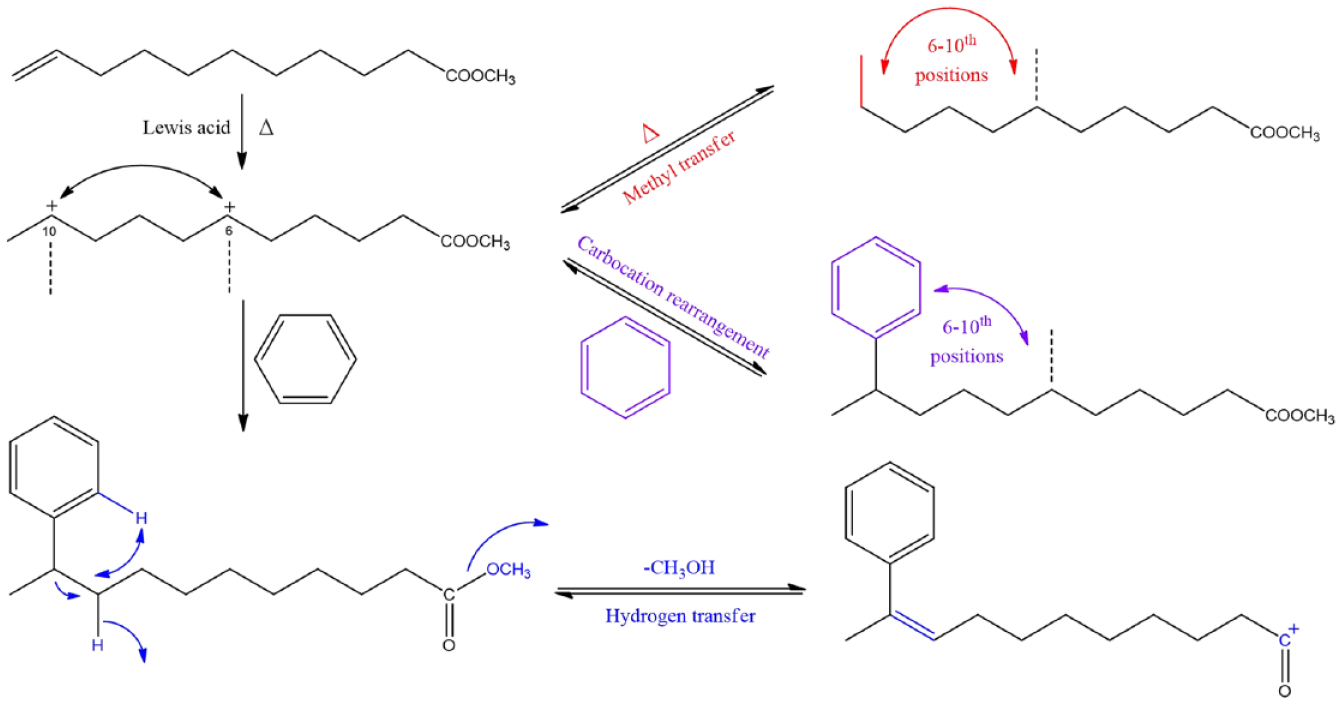
Hydrogen transfer, as a common rearrangement phenomenon in compounds, commonly exists in chemical reactions such as isomerization of unsaturated compounds,1,2 biological metabolism,3,4 and protein decomposition.5,6 In the mass spectra of fatty acid methyl esters, in addition to the M-31 ions are generated by simple cleavage of the ester group, hydrogen transfer can also induce the formation of other fragment ions. A well-known example of hydrogen transfer is the McLafferty rearrangement, which is widely used as an important law of ion rearrangement in mass spectrometry analysis. 7 One of the main ideas is that γ-hydrogen transfers to electron-withdrawing groups (such as carbonyl) after β-bond breaking, forming a large number of rearranged ions and losing neutral molecules. Furthermore, abundant M-32 fragment ion peaks are also observed in the mass spectra of unsaturated fatty acid esters (UFAEs),8,9 and the same fragment ions are also present for fatty acid esters after phenyl substitution. 10 Such fragments result from the loss of a certain hydrogen atom of the fatty acid chain together with an alkoxy group, 11 but are not fully explored by the original position of the transferred hydrogen. Several studies12–14 had shown that hydrogen can be derived from the α-position of phenyl or the α-position of benzyl, but it was also observed that the hydrogen atom at other positions would be lost along with the alkoxy group, and even the hydrogen atom of the benzene ring could be transferred. In the process of transfer, the transferred hydrogen might undergo a hydrogen-bridged cyclic intermediate or transition state, 15 the hydrogen at the benzene ring was first transferred to the α hydrogen of the phenyl group by [1,4-H] transfer and then dropped off with the ester group. 16 In contrast, the transfer of hydrogen farther from the carbonyl group had rarely been mentioned. 17
Numerous studies have analyzed a range of compounds prepared from benzene and unsaturated fatty acids.18–21 A series of compounds were generated by the Friedel–Crafts alkylation which was confirmed to be phenyl octadecanoic acid isomers of phenyl at different positions under the influence of carbocation rearrangement, and most of the phenyl existed at the positions between C-6 and C-17.22,23 The products were repeatedly crystallized at low temperature with solvents such as acetone and n-hexane, and pure 17-, 16-, 15- and 13-phenyl methyl stearate could be isolated, accounting for 3.6%, 2.3%, 2.6%, and 1.5%, with a purity of over 98%.24,25 Research has shown that the position of the phenyl group had some influence on the performance of the products, and the performance of methyl esters of phenyl fatty acids located in the middle position is better. 26 In this work, a new understanding of Friedel–Crafts alkylation was reported. Carbocation rearrangement, methyl transfer, and hydrogen transfer existed in the alkylation process. The changes caused by the three isomerization reactions were confirmed, respectively, and the causes and influencing factors of the isomerization reaction were explored.
Results and discussions
The carbocation rearrangement isomers of methyl phenylundecanoate
The total ion chromatograms (TIC) of methyl 10-undecenoate reacted with benzene at 40 and 70 °C for 60 min was determined (Figure 1). The results showed that five isomers were formed, and the mass spectrometry showed that the five peaks were methyl 6-phenylundecanoate to methyl 10-phenylundecanoate (Figure 2). But there were obvious differences in their relative contents at different temperatures. When the reaction temperature was lower (40 °C), the relative abundance of each isomer followed 6th < 7th < 8th < 9th < 10th. However, when the reaction temperature was higher (70 °C), the content of methyl 6-phenylundecanoate increased significantly, and the content of seventh to ninth positions isomers was comparable. The isomer of second to fifth positions were almost undetectable, which indicated the interference factor appeared after the migration of carbocation to the sixth position.
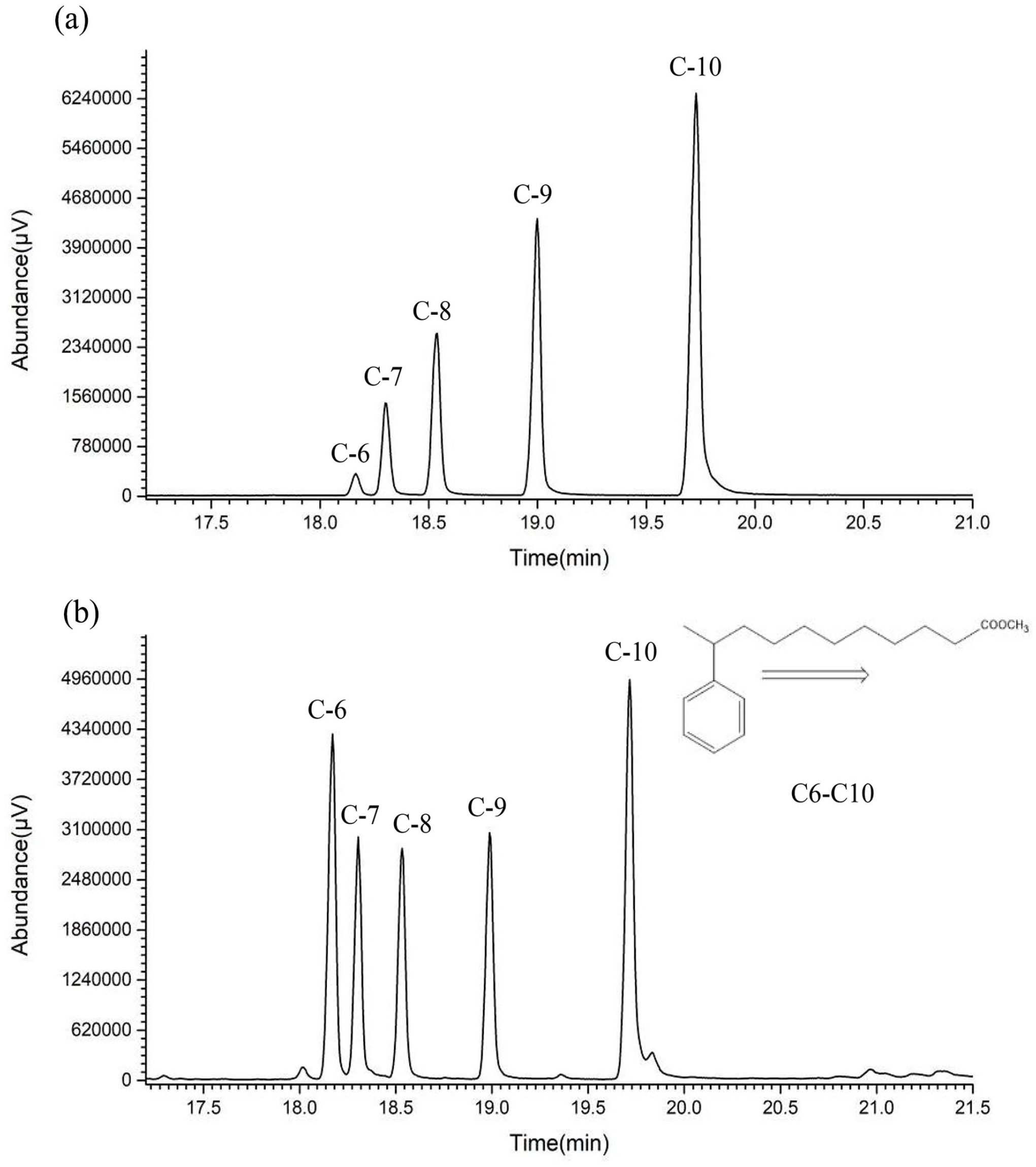
The TIC of methyl phenyl undecanoate after the reaction at 40 °C for 60 min (a) and the reaction at 70 °C for 60 min (b) (the original version were shown in Supplemental Figures S5 and S6).

The mass spectra of methyl phenylundecanoate isomers (the original version was shown in Supplemental Figure S23).
The structural analysis revealed that since the fifth carbocation could form a six-membered ring with oxonium ion, the benzene could not continue the addition reaction. 27 The benzene with octadecenol, oleic acid, and methyl oleate was performed to the Friedel–Crafts alkylation 24 and the product of octadecenol could detect the phenyl at the fifth position, while the oleic acid and methyl oleate products only to sixth position (Scheme 1). This means that in the absence of carbonyl oxygen, carbocations can still migrate to farther positions, and the presence of carbonyl oxygen prevents this process.

The reason why it is difficult to form methyl 5-phenylundecanoate—carbocation ions can form a six-membered ring structure with oxonium ions.
The methyl migration isomers of methyl undecanoate
In the Friedel–Crafts alkylation reaction between methyl 10-undecenoate and benzene, in addition to the formation of methyl phenylundecanoate, methyl undecenoate also underwent hydrogenation and isomerization. Five peaks with shorter retention times were detected after 30 min of reaction at 70 °C (Figure 3). The structures of the five peaks were determined by mass spectrometry (Figure 4). The results showed that the five peaks were methyl undecanoate, methyl 6-methyldecanoate to methyl 9-methyldecanoate. Compared with methyl 8-methyldecanoate, methyl 9-methyldecanoate has better symmetry and lower boiling point, so the retention time is earlier than methyl 8-methyldecanoate. However, when the reaction was performed at 40 °C, the methyl undecanoate isomer was not observed.

The TIC of five isomers at 70 °C for 30 min (the original version was shown in Supplemental Figure S6).

The mass spectra of methyl undecanoate isomers (the original version was shown in Supplemental Figure S26).
In this reaction, the system did not have a large amount of hydrogen source but produced a lot of saturated methyl ester. In order to explore the source of hydrogen, benzene-D6 was used to react with methyl 10-undecenoate. Figure 5 showed that when benzene was used in the Friedel–Crafts reaction, the molecular ion peak of methyl 9-methyldecanoate is m/z = 200, while that of deuterated benzene is m/z = 201. This means that benzene might participate in the methyl migration of methyl 10-undecenoate and provided a deuterium atom. When the bond “a” had broken, the molecular weights of the fragment ion peaks produced by the two substances were the same (m/z = 143 and smaller fragment peaks). With the bond “b” broken, methyl 9-methyldecanoate produced the fragments ion peak of m/z = 157 and methyl 9-methyldecanoate-D produced the fragments ion peak of m/z = 158. Similar fragmentation peaks were found in other isomers (Supplemental Figure S27). It could be speculated that the deuterium is in the eighth position of the carbon chain, which results from the hydrogen transfer.

The mass spectra of methyl 9-methyldecanoate (a) and methyl 9-methyldecanoate-D (b) from the reactions of methyl 10-undecanoate with benzene-D6 and benzene, respectively.
The kinetic rate of carbocation rearrangement
According to the above results, at the initial stage of the reaction, phenyl was preferentially added to the tenth position of methyl 10-undecenoate, while the content of the isomer at the sixth position was minimal. Due to the influence of carbonyl oxygen on carbocation, the addition of phenyl to the fifth position was very difficult. Here, we assumed that the carbocation at the sixth position did not exchange with the fifth position, so the migration rate of the sixth position carbocation could be determined by the formation rate of methyl 6-phenylundecanoate at 70 °C. The kinetic process of carbocation migration from the tenth position to the sixth position is a consecutive reaction
Since the migration rate of carbocations at the sixth position mainly depends on the number of carbocations at the seventh and eighth position, the above formula can be simplified
According to the kinetic theory, the migration rate and concentration of the carbocation at position eighth can be easily obtained
Since the carbocation of the sixth position is mainly formed by the migration of the seventh and eighth carbocations. Therefore, the rate equations for the sixth and seventh position can be substituted into c6+c7+c8 = c80, and the trend of the sixth position concentration over time can be calculated
In order to calculate the transfer rate of a carbocation in the Friedel–Crafts alkylation, the abundance of methyl 6-phenylundecanoate was plotted over time and fitted according to the Expassoc function (Figure 6), the concentration–time equation of the sixth position isomer was obtained
Taking the derivative of the above function to obtain the formation rate function of the sixth position product
The rate function indicated that the migration rate of carbocation gradually decreased with the progress of the reaction at 70 °C. When the reaction progressed to a certain extent, the abundance of each isomer remained relatively balanced. The experimental results of the concentration–time variation of the sixth position isomer were consistent with the theoretical calculation results.

The relative content and generation rate of methyl 6-phenylundecanoate isomer (internal standard methyl laurate).
The relationship between carbocation rearrangement and methyl migration
The relative abundances of each methyl phenylundecanoate isomer and methyl undecanoate isomer were determined at different reaction times (Figure 7). The methyl 6-phenylundecanoate had a low content at the initial stage of the reaction, and the phenyl was preferentiously added to the tenth position. With the progress of the reaction, the carbocation gradually migrated to the sixth position, and the relative content of methyl 6-phenylundecanoate increased. However, the relative contents of the methyl undecanoate isomers remained in relative equilibrium at the beginning of the reaction and did not change significantly as the reaction progressed.

Changes in the isomer content of methyl phenylundecanoate ((a) extracting ion m/z = 276) and methyl undecanoate ((b) extracting ion m/z = 200) at different reaction times (normalization method).
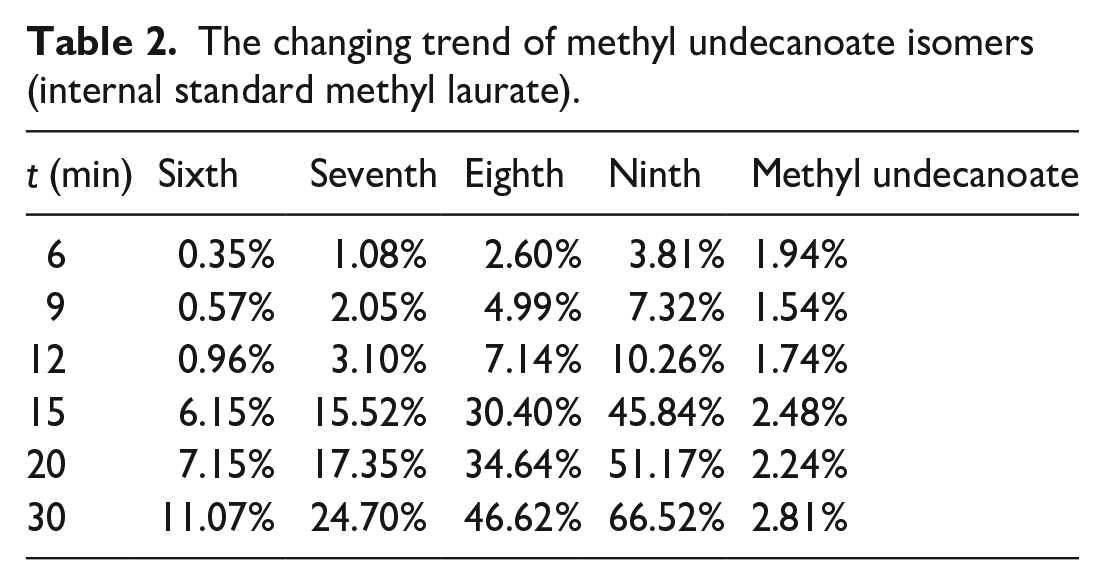
The abundances of methyl phenyl undecanoate isomers (Table 1) and methyl undecanoate isomers (Table 2) with the reaction were calculated. The results showed that the abundance of methyl undecanoate isomers gradually increased with the reaction. The abundance changes of methyl undecanoate isomers were compared with methyl phenylundecanoate, and the results were shown in Figure 8 (taking methyl 9-phenylundecanoate and methyl 9-methyldecanoate as examples). As the reaction progressed, the content of methyl undecanoate isomer increased and the content of methyl phenylundecanoate gradually decreased. Between 12 and 15 min, the abundance of methyl 9-methyldecanoate increased significantly, accompanied by a decrease in the abundance of methyl 9-phenylundecanoate decreased. This indicated that there was a competitive relationship between methyl phenylundecanoate and methyl undecanoate isomers, their abundances also changed with the reaction times and temperatures.
The changing trend of methyl phenyl undecanoate isomers (internal standard methyl laurate).

The changing trend of methyl undecanoate isomers (internal standard methyl laurate).

The relative abundances of methyl 9-phenylundecanoate and methyl 9-methyldecanoate at 70 °C (internal standard methyl laurate).
The hydrogen transfer in unsaturated fatty acids
The lost fragments of saturated fatty acid esters (SFAEs) and UFAEs were analyzed by the mass spectrometer. All methyl laurate (Figure 9(a)), methyl myristate, and methyl palmitate (Supplemental Figures S14 and S15) have a high content of M-31 (M-CH3O, m/z = 183, 211, 239) fragment peaks produced by cleavage of the ester methoxy group. However, high content of M-32 (M-CH3OH, m/z = 166, 264) fragment peaks was generated from methyl 10-undecenoate (Figure 9b) and methyl oleate (Supplemental Figure S16). It could be speculated that the hydrogen transfer occurred during the cleavage of the UFAE’s ester bond, the C–O bond accepted the hydrogen atom and then dropped the neutral molecular fragment. Based on the above exploration, benzene was added to methyl 10-undecenoate by the Friedel–Crafts alkylation, the gas chromatogram of the alkylation product was shown in Figure 10. Carbocation rearrangement triggered the position isomerism of the phenyl group in the carbon chain and formed five position isomers, methyl 6–10-phenylundecanoate. The mass spectra of all five peaks were collected and the structure was determined (Figure 2); there was also a high content of M-32 (M-CH3OH, m/z = 244) fragment peak in each methyl phenylundecanoate isomer (Figure 9(c) and Supplemental Figure S23). The mass spectra of methyl 3-phenylundecanoate and methyl 10-phenyldecanoate could also confirm the above results (Supplemental Figures S17 and S18). In all, whether the C–C double bonds in the main chain or attached chains promoted the hydrogen transfer process.
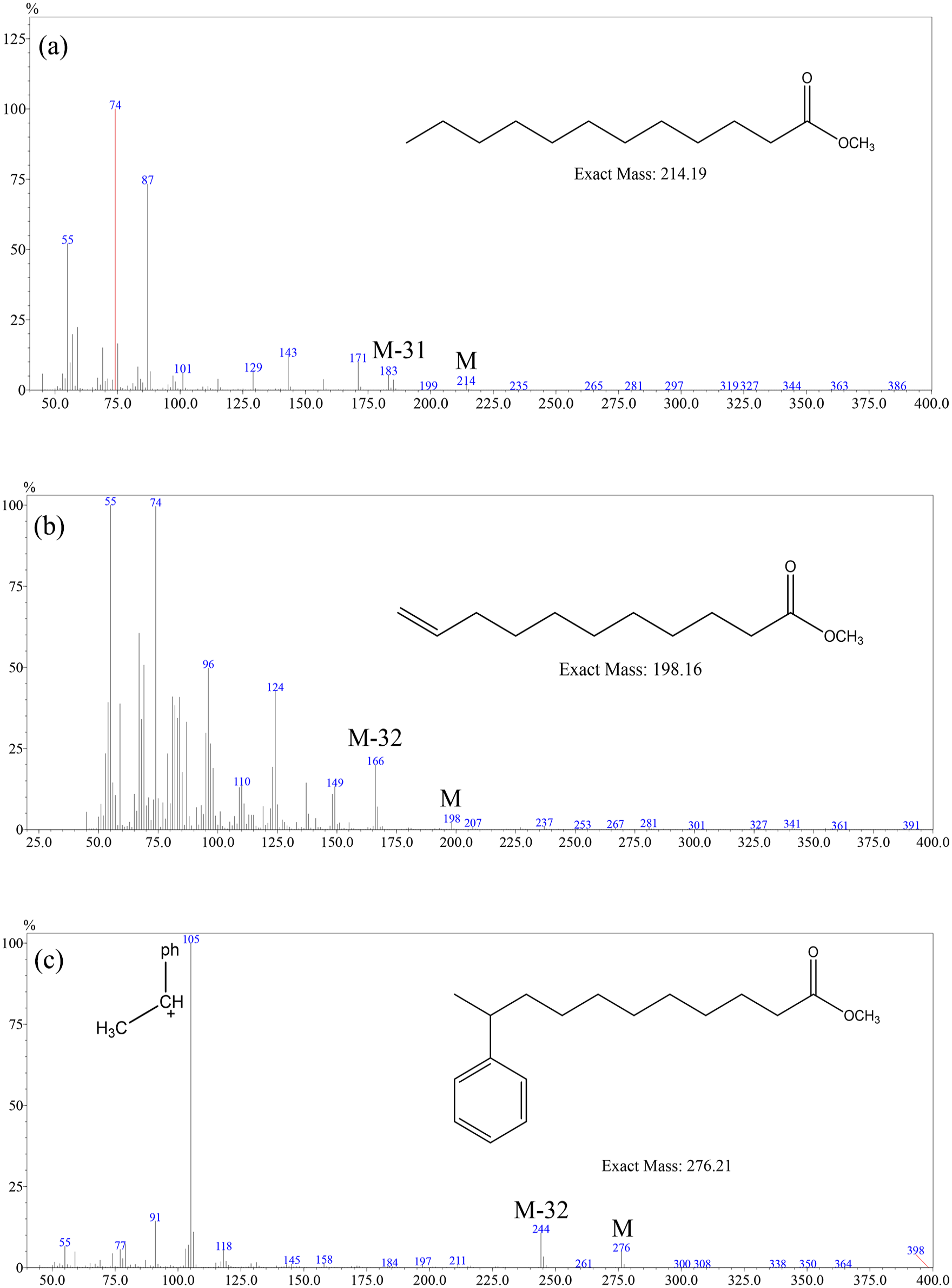
The mass spectra of methyl laurate (a), methyl 10-undecenoate (b), and methyl 10-phenylundecanoate (c).

The TIC of methyl phenylundecanoate.
Sources of transferred hydrogen atoms
Identifying the donor of the hydrogen atom could provide a deeper understanding of the mechanism of hydrogen transfer. As shown in Table 3, the relative abundances of M-31 (M-CH3O, m/z = 183), M-32 (M-CH3OH, m/z = 166, 244), and M-33 (M-CH3OD, m/z = 249) fragment peaks in methyl laurate, methyl 10-undecenoate, and methyl 10-phenylundecanoate were calculated. The fragment peak content of M-31 in methyl laurate was higher than M-32, while the M-32 fragment peak of methyl 10-undecenoate accounts for 82.66%. After the Friedel–Crafts alkylation, the proportion of the M-32 fragment peak in methyl 10-phenylundecanoate was reduced to 75.25%. It is reasonable to speculate that the cleavage of ester bonds in SFAEs was mainly by simple fragmentation, while the hydrogen transfer rearrangement dominated UFAEs.
The ratio of lost methoxy to neutral molecular fragment peaks in several fatty acid esters.

In addition, benzene-D6 was used for alkylation, and the integral area of M-33, M-32, and M-31 fragment ions in extraction ion chromatography were compared to confirm the effect of benzene on hydrogen transfer. In methyl 10-phenyl-D5-undecanoate, the integrated area of the M-33 fragment accounted for 47.13% and that of the M-32 fragment was 35.19%. It was indicated that the hydrogen atoms in the entire carbon chain and the benzene were involved in the hydrogen transfer process, and the hydrogen atoms in benzene were more readily transferred. Meyerson and Leitch 8 analyzed the pyrolysis mass spectra of methyl 6-phenylhexanoate, and the mechanism of M-32 fragment formed by methyl 6-phenylhexanoate was explained. The isotopic tracer method indicated that the hydrogen atom at C-5 has participated in the formation of bicyclic hydrogen-bridged transition states then has been lost with the breaking of the ester bond. Amick et al. 16 analyzed the mass spectra of ethyl 3-phenylpropionate and six deuterium-labeled isotopomers by mass-analyzed ion kinetic energy (MIKE) spectrometry. The deuterium atom in the phenyl ring could undergo H/D exchange with the hydrogen atoms originating from the ortho-positions of a benzene ring and the ortho-positions of the benzylic methylene group. But in this work, since the position of benzene was far from the carbonyl group, it was challenging to complete the hydrogen transfer through a single transfer. Therefore, this process possibly followed a stepwise mechanism: the hydrogen in the phenyl group was exchanged with the carbon chain and then gradually transferred to the C–O bond. Scheme 2 describes the possible steps of hydrogen transfer, accomplished by intramolecular transfers.

A possible steps for the transfer of deuterium from benzene and CH3OD lost from methyl 9-phenylundecanoate.
As mentioned above, benzene-D6 was added to methyl 10-undecenoate by the Friedel–Crafts alkylation. In the process, phenyl was added to one side of the double bond, and a hydrogen atom was also needed on the other side, which was supplied by benzene. In general, the Friedel–Crafts alkylation of end alkenes follows Markovnikov’s rule, hydrogen will be preferentially added to the position with more hydrogen. The mass spectrum of methyl 10-phenyl-D5-undecanoate showed that the fragment of C8H3D6+ (m/z = 111) was the main fragment peak, and the content of C7H2D5+ (m/z = 96) fragment peak was low; it indicated that the phenyl was mainly added at the tenth position, and the hydrogen atom was added at the terminal position. In addition, the fragment peak M-16 (M-CH2D, m/z = 266) was observed from the mass spectrum, and the fragment peak M-15 (M-CH3, m/z = 267) was not detected, but the peak of M-15 could be detected in the mass spectrum of methyl phenylundecanoate (Supplemental Figure S23). There were also observed the fragment peaks with loss of deuterium-containing groups (M-30, M-C2H4D+, m/z = 252; M-44, M-C3H6D+, m/z = 238; M-58, M-C4H8D+, m/z = 224; M-72, M-C5H10D+, m/z = 210) in the mass spectra of methyl 6-phenyl-D5-undecanoate to methyl 9-phenyl-D5-undecanoate (Supplemental Figure S24). Furthermore, the Sun et al. 28 had analyzed the mass spectra of several methyl 2,2-dideuterio unsaturated esters, a significant proportion of the M-32 fragment peak was observed, whereas the M-33 fragment peak accounted for 12%–18%. It indicated that the deuterium atom in the unsaturated fatty acid chain could participate in the hydrogen transfer process, but it was not the main factor affecting the loss of neutral molecules.
The mass spectrum of methyl 7-phenyl-D5-undecanoate (Figure 11) showed that there was not only the fragment peak of C7H2D5+ (m/z = 96) but also the fragment peaks after the exchange of deuterium and hydrogen atoms C7H3D4+• (m/z = 95), which accounted for 25.95%. However, the peak content of the C7H6+• (m/z = 94) fragment was extremely low in methyl phenyl undecanoate (Supplemental Figure S23); this means that losing a hydrogen atom and forming the fragment peak C7H6+• was very difficult. For methyl phenyl-D5-undecanoate, it is more likely that the phenyl deuterium atom exchanged with the carbon chain hydrogen atom rather than dropped directly. This is also a piece of strong evidence to support the participation of phenyl hydrogen in hydrogen transfer.

The mass spectrum of methyl 7-phenyl-D5-undecanoate.
In addition, the fragment ions were formed in the high vacuum state of gas chromatography–mass spectrometry (GC-MS), the molecules were far apart from each other, and there should be only intramolecular transfer and no intermolecular transfer. It means that other molecules did not affect the lost neutral molecular fragment.
Activity of the transferred hydrogen
The mass spectra of methyl phenyl-D5-undecanoate showed that hydrogen in benzene also participated in hydrogen transfer. To explore the relationship between hydrogen transfer and the additional position of benzene, the integrated area of M-31, M-32, and M-33 fragments in methyl phenylundecanoate and methyl phenyl-D5-undecanoate (phenyl at seventh to tenth positions) was calculated (Table 4). For methyl phenylundecanoate, the position of benzene had little effect on hydrogen transfer; the ratio of the M-32 and M-31 fragment peaks was approximately 3:1. However, when benzene-D6 was substituted, the closer the addition position of benzene to the carbonyl group, the lower the relative proportion of M-33 fragments, and the total amount of transferred hydrogen atoms also decreased slightly. The proportion of carbon chain hydrogen transfer increased, while the growth of deuterium transfer slowed down in methyl 10-phenyl-D5-undecanoate. The dissociation energy of different C–H bonds was satisfied by
The relative content of each fragment when the phenyl group in 7–10 positions.

Conclusion
In this study, carbocation rearrangement, methyl migration, and hydrogen transfer in the Friedel–Crafts alkylation of benzene with methyl 10-undecenoate were investigated by GC-MS. The results showed that carbocation rearrangement resulted in the formation of five methyl phenyl undecanoate isomers, and the abundance of each isomer varied greatly at different reaction temperatures and times. Since the migration of carbocation to the fifth position will form a six-membered ring with oxonium ion, the migration rate of carbocation at 70 °C could be estimated by calculating the formation rate of methyl 6-phenylundecanoate. Five methyl undecanoate isomers with methyl groups at different positions were also observed at 70 °C, the relative content did not change significantly as the reaction proceeded. By exploring the source of hydrogen addition, it was found that benzene also participated in the formation of methyl undecanoate and provided a hydrogen atom. The formation of carbocation rearrangement isomers and methyl migration isomers was competitive. The increase in methyl undecanoate isomer was accompanied by the decrease in methyl phenylundecanoate isomer. In addition, all the hydrogen atoms from the carbon chain and phenyl attached at different locations of the fatty acid carbon chain could participate in hydrogen transfer, which indicated the long-range transfer of hydrogen in unsaturated and phenyl fatty acid esters. By comparing several C–H bond dissociation energies, the closer the addition position of deuterated benzene is to the carbonyl group, the easier the hydrogen atom of the carbon chain is to transfer to the ester bond. This work furthered the understanding of the Friedel–Crafts alkylation and provided assistance in controlling the species and content of the phenyl fatty acid ester isomers.
Materials and methods
Materials
The following reagents were used, 10-undecenoic acid (Meryer Chemical Technology Co., Ltd., China), methyl laurate, methyl myristate, methyl palmitate, methanol-D4 (Nine-Dinn Chemistry (Shanghai) Co., Ltd., China), aluminum trichloride (Shanghai Lingfeng Chemical Reagent Co., Ltd., China), benzene (Aladdin Chemical reagent Co,. Ltd., China), benzene-D6 (Sigma-Aldrich Corporation), and methanol (Shanghai Macklin Biochemical Co., Ltd., Shanghai, China).
The synthesis of phenyl fatty acid esters
The synthetic procedures for several products are shown in Scheme 3. Four target compounds were prepared from 10-undecenoic acid by the Friedel–Crafts alkylation and esterification. The reaction was conducted in a closed system, and the molar ratio was 1:5:1 (10-undecenoic acid (0.01 mol):benzene (benzene-D6):AlCl3), ran at 70 °C for 6 h. After the reaction, the remains of benzene were evaporated, an appropriate amount of 10% methanol (methanol-D4)-sulfuric acid solution was added, and the reaction was conducted at 60 °C for 1 h. After the reaction time was completed, the methanol was evaporated, and the organic phase was collected after extraction with ethyl acetate and evaporated to dryness to obtain the product.

The synthetic route of four kinds of methyl phenylundecanoate, which were subjected to mass spectrometry.
Calculation of the abundance of fragment ions
The TIC, mass spectra, extracted ion chromatograms (EICs), and integral areas of several products were determined by GC-MS (SHIMADZU GC-MS-TQ8040 NX, Agilent 6890, and mass spectrometer 5978). The gas chromatograph was equipped with a capillary column HP-5MS (30 m × 0.25 mm × 0.25 μm). The ion source, injector, and detector temperatures were 200, 280, and 300 °C, respectively. The temperature program was 80 °C as the initial temperature for 3 min, and then raised to 300 °C at 5 °C min−1 for 5 min. The TICs were reanalyzed by extracted ion chromatography, selecting the corresponding target molecular fragment weights (M-31, M-32, and M-33) and integrating, counting the peak height and integral peak area of each peak, and the relative abundance was calculated. Scheme 4 shows the result of losing fragments before and after hydrogen transfer, with the loss of methoxy fragments in the absence of hydrogen transfer and the loss of neutral molecular fragments in the presence of hydrogen transfer.
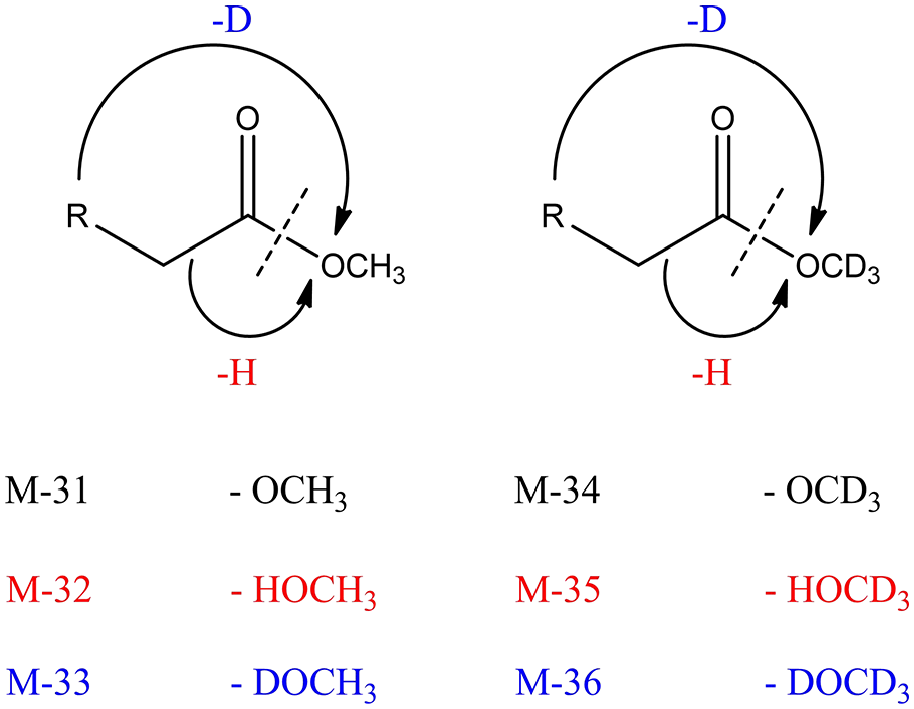
Different fragments of fractured methyl ester before and after hydrogen transfer.
Supplemental Material
sj-docx-1-chl-10.1177_17475198231195806 – Supplemental material for Carbocation rearrangement, methyl migration, and long-range hydrogen transfer in phenyl fatty acid esters
Supplemental material, sj-docx-1-chl-10.1177_17475198231195806 for Carbocation rearrangement, methyl migration, and long-range hydrogen transfer in phenyl fatty acid esters by Jian-Qiao Lang, Hong-Ze Gang, Bo-Zhong Mu and Shi-Zhong Yang in Journal of Chemical Research
Footnotes
Author contributions
J.-Q.L. contributed to data curation, formal analysis, and writing—original draft. H.-Z.G. and B.-Z.M. contributed to supervision. S.-Z.Y. contributed to formal analysis, writing—review and editing, and supervision. All authors have read and agreed to the submitted version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by funding from the National Key Research and Development Program of China (no. 2022YFC2105200), the National Natural Science Foundation of China (no. 51574125), and the Fundamental Research Funds for the Central Universities of China (no. JKJ01231714).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.