Abstract
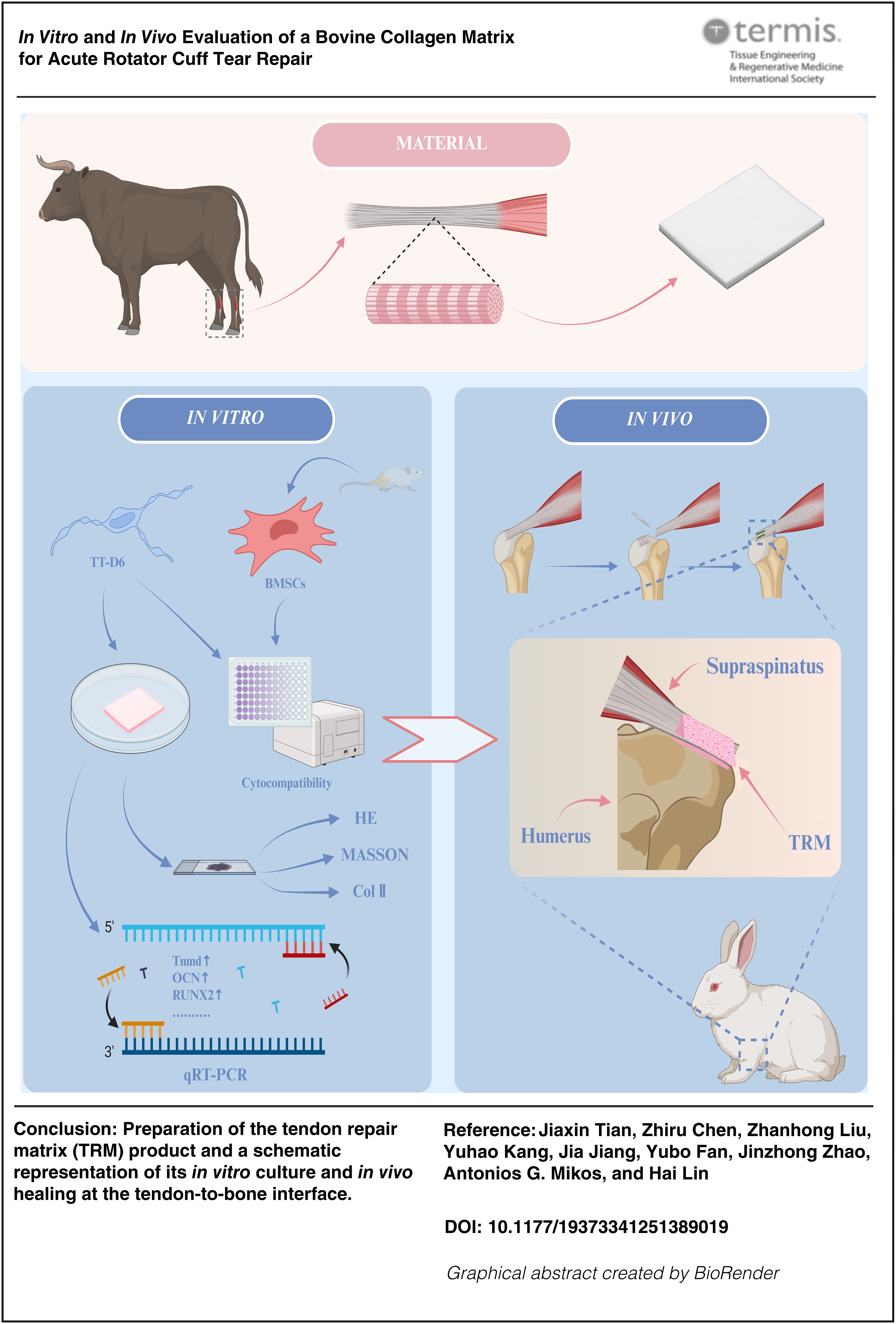
Full-thickness rotator cuff tears (RCTs) represent a musculoskeletal damage that severely affects shoulder function and quality of life. Current surgical interventions are hindered by limited regenerative capacity of rotator cuff repair implants and high retear rates postoperatively. In this study, we investigated a tendon repair matrix (TRM) product prepared from bovine tendon collagen. The TRM was designed as a regenerative scaffold to improve the healing of damaged rotator cuff. In vitro results showed excellent cytocompatibility of TRM, with significantly enhanced adhesion, proliferation, and spreading of bone marrow stromal cells and tenocyte-like mouse tendon precursor cells,mouse tendon-derived cell line, clone D6 (TT-D6) cells (mouse tendon-derived cell line, clone D6). In a rabbit model of acute full-thickness supraspinatus tendon tear, TRM promoted type I collagen deposition, improved interface tissue formation, and enhanced tendon-to-bone integration. Furthermore, biomechanical test results revealed load-bearing capacity of the TRM group compared with both the empty and native tissue control groups. These findings support the clinical potential of TRM as a regenerative scaffold for the functional reconstruction of RCTs.
Impact Statement
This study addresses a critical clinical need in sports medicine by evaluating a novel bovine collagen-based tendon repair matrix (TRM) for the repair of acute full-thickness rotator cuff tears (RCTs). The TRM exhibited excellent biocompatibility and significantly enhanced tendon-to-bone healing, as demonstrated by improved fibrocartilaginous tissue formation and biomechanical strength in a rabbit model. These promising results underscore TRM’s potential to reduce postoperative retear rates by promoting effective regeneration of the tendon-bone interface. Consequently, this research represents an important advancement toward improving clinical outcomes for RCT patients, offering substantial potential for translation into clinical practice.
Keywords
Get full access to this article
View all access options for this article.
