Abstract
Objective
Cardiovascular diseases (CVDs) remain a primary reason for death, and current thrombolytic agents face significant limitations due to side effects and high costs. Plant-derived alternatives, particularly from the Curcuma genus, show promise as safer and more accessible thrombolytic options. However, the thrombolytic potential of Curcuma wallichii specifically remains largely unexplored. The aim of this study is to evaluate the thrombolytic and anti-inflammatory potential of the chloroform fraction of Curcuma wallichii rhizomes (CFCWR) through in vitro models and in silico molecular docking with ADMET predictions.
Methods
The anti-inflammatory activity of the CFCWR was studied through the HRBC stabilization method and thrombolytic activity was tested using the clot-lysis assay. Phytochemicals were detected using GC-MS and the compounds were subjected to in silico docking against tissue plasminogen activator (tPA), along with ADMET evaluation by computational methods.
Results
The CFCWR exhibited a wide range of membrane-stabilizing capabilities, showing 65.23 ± 0.02%stabilization at a concentration of 100 µg/mL. In contrast, it demonstrated clot lysis activity of 62.23 ± 1.56% at the same concentration in comparison with the standard streptokinase (89.14 ± 0.03%) in the in vitro thrombolytic model. GC-MS analysis identified more than 100 phytochemicals, among which 2-(1-(Beta-d-glucopyranosyloxy)-1-methylethyl)-2,3-dihydro-7-oxo-7H-furo(3,2-g) exhibited the highest binding affinity (score -9.0 kcal/mol) toward the tPA protein. ADMET studies indicated that these compounds are non-carcinogenic, well-absorbed, and possess good oral bioavailability.
Conclusion
This study reports for the first time the thrombolytic activity of Curcuma wallichii and identifies bioactive phytochemicals with potential mechanistic interaction at the tPA active site. These findings justify further exploration of Curcuma wallichii as a potential therapeutic agent for cardiovascular diseases. Future studies should include in vivo evaluation of clot-dissolving efficacy and the isolation and characterization of bioactive compounds.
1. Introduction
Cardiovascular diseases (CVDs) persist as a major global health challenge contributing to an estimated 19.8 million deaths in 2022 and accounting for about one-third of total deaths worldwide. 1 The primary cause of death and long-term disability in many of these conditions is thrombosis, the formation of blood clots that block blood vessels. 2 While clot formation is a natural component of the body’s hemostatic process, excessive or dysregulated clotting can lead to severe vascular events like myocardial infarction, ischemic stroke, and venous thromboembolism. 3 Therefore, the timely dissolution of these clots to restore blood flow is essential for reducing associated mortality and morbidity.
Inflammatory cytokines are now recognized as key mediators bridging inflammation and coagulation, often acting as primary triggers for coagulation activation. 4 During systemic inflammation, increased levels of pro-inflammatory cytokines such as IL-6 and IL-1 shift the hemostatic balance toward coagulation by suppressing natural anticoagulant pathways and enhancing endothelial activation and tissue factor expression. 5 These changes may potentiate thrombus formation and accelerate pathologies such as atherosclerosis, extensive tissue damage, and thrombosis. Persistent inflammation, regardless of its origin, can contribute to the development of serious complications, including portal hypertension, impaired hepatic perfusion, ischemic cardiac events or stroke, and tissue anoxia. 6 Thrombosis is clinically associated with various disorders such as pancreatitis, cirrhosis, diverticulitis, and cholangiocarcinoma, and may also contribute to elevated mortality risk. 7 Consequently, thrombosis represents a significant global health burden.
Existing anti-thrombotic therapies, such as antiplatelet agents and anticoagulants, are effective in preventing new clot formation but are not designed to break down existing clots. 8 For this purpose, fibrinolytic agents like streptokinase and recombinant tissue plasminogen activators (rtPAs) are the current standard of care. 9 Despite their life-saving capabilities, these agents come with significant drawbacks, notably a high risk of life-threatening bleeding complications, allergic reactions, and a short plasma half-life.10-12 Moreover, their high cost and limited availability restrict their use in many resource-constrained settings, highlighting the critical need for safer, more affordable, and universally accessible thrombolytic drugs.
This urgent demand has driven a renewed interest in natural products, which are abundant sources of bioactive compounds with potential therapeutic properties against various human diseases. Natural products have become an alternative and supplementary treatment technique due to their diverse pharmacological and biological applications.13-15 Traditional plant-based medicines serve as the primary health care source for nearly 80% of the world’s population, especially in developing countries. 16 Medicinal plants are highly valued for their wide chemical diversity, bioavailability, and safety compared to synthetic drugs. 17 In recent decades, numerous studies have reported the thrombolytic and anticoagulant properties of various phytochemicals, thereby fueling the search for plant-derived fibrinolytic agents. 18 Several plants, including Allium sativum (garlic), Allium cepa (onion), Curcuma longa (turmeric), Ocimum sanctum, and Azadirachta indica have demonstrated promising clot-dissolving activities in preclinical studies.19,20 Notably, garlic extracts and curcuminoids from turmeric have been associated with enhanced fibrinolysis, reduced platelet aggregation, and improved cardiovascular outcomes. 21 These findings justify the systematic evaluation of unexplored medicinal plants for their thrombolytic potential. 22
The genus Curcuma (family Zingiberaceae) includes more than 133 species distributed all over the world, many of which are important in traditional medicine systems like Ayurveda, Unani, and Chinese medicine. 23 The genus has long been used as a medicinal agent in Islamic traditional medicine and other folk healing practices. 24 Traditionally, species of the Curcuma genus have been used in the treatment of various ailments, such as leprosy, cancer, hemorrhoids, asthma, inflammatory conditions, and wound healing. 25 However, phytochemical investigations of Curcuma species have identified a wide range of bioactive compounds such as curcuminoids, sesquiterpenes, and essential oils. 26 These compounds are known for their antioxidant, anti-inflammatory, liver-protective, anticancer, antimicrobial, and heart-protective properties. 27 Notably, Curcuma longa and Curcuma zedoaria have demonstrated fibrinolytic activity and the ability to reduce platelet aggregation, supporting their traditional use in treating circulatory problems.28,29
Ethnopharmacologically, Curcuma wallichii (C. wallichii) has traditionally been used in the treatment of leucoderma, asthma, tumors, piles, rheumatism, and digestive disorders. Preliminary phytochemical analyses of C. wallichii rhizomes indicate the existence of carbohydrates, flavonoids, steroids, alkaloids, phenols, tannins, amino acids, terpenoids, and glycosides. The chloroform fraction of C. wallichii exhibited the highest antioxidant potential. In the DPPH free-radical scavenging assay, this fraction demonstrated a strong antioxidant effect, indicating potent radical-quenching activity. 30
However, despite a diverse array of bioactive compounds and an extensive history of traditional use, the cardiovascular and thrombolytic potential of C. wallichii remains largely unexplored for thrombolytic potential. Although related Curcuma species exhibit fibrinolytic activity. This study bridges this gap by integrating phytochemical profiling with in vitro and computational analyses, providing mechanistic insights into active compounds. In addition to experimental assays, computational approaches such as molecular docking and in silico ADMET analysis can predict interactions with key proteins and evaluate pharmacokinetic and safety properties. 31 The molecular docking simulation technique has been evaluated for the exploration of the possible binding modes of receptors with selected ligands which reveals binding energetics and molecular interactions. 32
This study employed in vitro anti-inflammatory, clot lysis assays, GC-MS phytochemical profiling, molecular docking and in silico ADMET analysis to evaluate the thrombolytic potential of the chloroform fraction of C. wallichii rhizomes. The results are expected to provide insight into its potential as a safe and affordable thrombolytic agent and to aid the development of plant-based therapeutics for cardiovascular disorders.
2. Materials and Methods
2.1. Chemicals and Reagents
Analytical-grade methanol, chloroform, and other chemicals were sourced from Merck (India). Streptokinase (SK) was obtained from Beacon Pharmaceutical Ltd., Bangladesh, while acetylsalicylic acid was provided by ACME Laboratories Ltd., Bangladesh.
2.2. Collection, Preparation and Extraction of Plant Material
The C. wallichii rhizomes were collected from the National Herbarium, Bangladesh, in November 2022 and were authenticated by an expert taxonomist, where a voucher sample (accession no.: 104,530) was deposited. After washing and drying the plant materials were subjected to grinding. The extraction was performed according to the method described earlier by Hossain et al. 23 The coarse powder (1.7 kg) was kept in a light-protected bottle and soaked with 100% methanol (1 L × 5 times) at RT, with intermittent agitation. The mixture was initially filtered through cotton, followed by filtration using Whatman filter paper (11 μm). The crude methanolic extract (CME) was obtained (56 g) using a rotary evaporator under reduced pressure at 40°C. The CME (46 gm) was suspended in methanol (90%) 200 mL and then sequentially partitioned with n-hexane, chloroform and finally water using the modified Kupchan method. 33 This yielded the n-hexane fraction, chloroform fraction (CHCWR, 26.3 gm), and aqueous fraction.
2.3. In vitro Thrombolytic Assay
The study was performed following the method described by Prasad et al.
34
Initially, 0.5 mL of fresh blood samples was carefully transferred into pre-weighed microcentrifuge tubes obtained from the non-contraceptive and nonanticoagulated donors. Following incubation of the blood at 37°C for 45 minutes, serum was carefully removed without disturbing the clot, and the tubes were reweighed. A volume of 100 μL of the test sample, at concentrations of 6.25, 12.5, 25, 50, and 100 µg/mL, was added to each tube. In the assay, positive control tubes contained 100 µL of streptokinase (30,000 IU), whereas negative control tubes were filled with 100 µL of distilled water (DW). The tubes were incubated at 37°C for 90 minutes, after which the supernatant was removed and the tubes were reweighed. Finally, the percentage of clot lysis was estimated from the weight differences before and after lysis, and the experiment was performed in triplicate.
2.4. Hypotonicity-Induced HRBC Membrane Stabilization Assay
This study was performed using the method described earlier by Shinde et al with slight modifications.
35
Initially, a 5 mL blood sample was obtained from a healthy donor, having refrained from NSAID intake for two weeks prior to donation. Then it was anticoagulated with dipotassium EDTA (final concentration: 2.2 mg/mL). The collected blood was centrifuged at 3,000 rpm for 10 min and washed repeatedly (three times) with an isotonic NaCl solution (154 mM) in 10 mM sodium phosphate buffer at pH 7.4. The cell pellet was finally resuspended in an equal volume of isotonic buffer. Subsequently, 0.5 mL of the resulting cell suspension was added to a mixture containing 5 mL of hypotonic solution (50 mM NaCl) and 100 µL of either the test sample or standard solution at concentrations of 6.25, 12.5, 25, 50, and 100 µg/mL in 10 mM sodium phosphate-buffered saline (pH 7.4). The control tube consisted of 0.5 mL of cell suspension, 5 mL of hypotonic solution, and 100 µL of distilled water (DW) in the same buffer. All reaction mixtures were incubated at room temperature for 10 minutes, followed by centrifugation at 3,000 × g for 10 minutes. After incubation and centrifugation, the absorbance of the supernatant was measured at 540 nm using a spectrophotometer (Centurion Scientific Ltd, UK). Acetyl salicylic acid served as the reference standard. The percentage of HRBC membrane stabilization was estimated utilizing the following formula:
2.5. GC-MS Analysis
The CFCWR was analyzed using gas chromatography-mass spectroscopy, 36 using a combined 8890 gas chromatography system (Agilent 19091S-433UI:2489267H, USA) coupled with an Agilent 7010B GC/TQ MS system, EI Ionization technique. An HP-5MS UI column (5% phenyl methyl siloxane, 30 m × 250 μm × 0.25 μm) was utilized for chromatographic separation. As the carrier gas, Helium was used. The flow rate was 1.0 mL/min, the injection volume was 1 μl, and a split ratio of 1:50. The injector temperature was precisely regulated at 250°C, with a maintained pressure of 11.653 psi. The thermal gradient program began at 120°C for 2 minutes. The system temperature was raised at 10°C/min until reaching 320°C, where it was held for 5 minutes. This resulted in a total elution time of 27 minutes. The mass spectrometric analysis was conducted under electron impact ionization at 70 eV, with a scanning mass range of 40–700 m/z. The acquired mass spectra were cross-referenced against a computer-assisted mass spectral database. The molecular fragmentation patterns of the separated constituents were analyzed and identified by comparing them with spectral chromatograms available in the 2020 edition of the National Institute of Standards and Technology (NIST) GC-MS library software. Compounds identified by GC-MS were selected for molecular docking based on their relative abundance in the chloroform extract and reported or predicted thrombolytic or cardiovascular activity, ensuring that the docking study focused on the most relevant bioactive phytochemicals.
2.6. Protein and Ligand Preparation
The RCSB Protein Data Bank (https://www.rcsb.org) provided the tPA 3D crystal structure (PDB ID: 1A5H). The selected 3D structures of ligands were obtained in SDF format from the PubChem database. These ligand structures were optimized through energy minimization and protonation using Avogadro software and then converted to PDB format for docking analysis. 37 For protein preparation, BIOVIA Discovery Studio Visualizer was used to clean the tPA structure by removing unnecessary chains, heteroatoms, and water molecules. Using the Open Babel software, the refined protein was then converted into PDBQT format.
2.7. Molecular Docking
AutoDock Vina from PyRx was employed to conduct all docking calculations on the active sites of the tPA protein, using the identified molecules from C. wallichii. 38 A grid box was selected to encompass the maximum surface area of the prepared protein. The grid center points were set to X = 28.6846, Y = 28.6031, Z = 9.2936, and the dimensions (Å) were X = 58.2800, Y = 76.9576, Z = 64.7700. The dimensions of the grid box were strategically defined to enclose the active site of the protein, specifically targeting its substrate-binding domain. Visualization was performed using BIOVIA Discovery Studio. 37
2.8. Pharmacokinetic Properties Analysis
Swiss ADME (https://www.swissadme.ch/index.php) and pkCSM-pharmacokinetics (https://biosig.unimelb.edu.au/pkcsm/prediction) were the online tools used for the prediction of molecular properties and pharmacokinetics (ADMET) of the molecules identified from C. wallichii. 39 During the generation phase, the Simplified Molecular Input Line Entry System (SMILES) was utilized for the selected molecules. 40
2.9. Statistical Analysis
Differences between streptokinase (SK) and plant extracts were analyzed using a paired Student’s t-test. Data are presented as mean ± standard deviation. All experiments were performed in triplicate. Comparisons between positive and negative controls were performed using one-way ANOVA, followed by Tukey’s post-hoc test for multiple comparisons. Normality and homogeneity of variance were assumed for parametric tests. Exact p-values are reported in the Results section, and significance was considered at p < 0.05. Statistical analyses were conducted with SPSS software (version 18.0, IBM, New York, USA).
3. Results
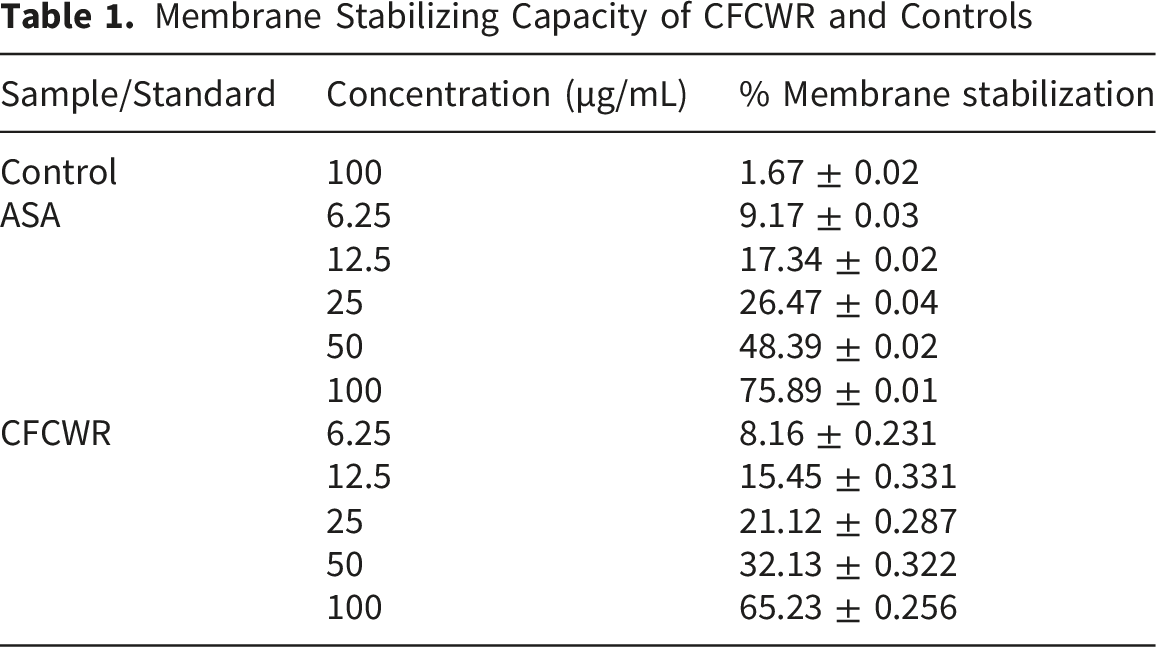
3.1. Membrane-Stabilization Test
Membrane Stabilizing Capacity of CFCWR and Controls
3.2. In vitro Thrombolytic Assay
Clot Lysis Capacity of CFCWR and Controls

3.3. Gas Chromatography-Mass Spectrometry (GC-MS) Analysis
The chromatographic spectra depicted in Figure 1 display well-defined peaks that represent the individual compounds detected, each corresponding to specific retention times and mass spectral patterns. GC-MS chromatogram of CFCWR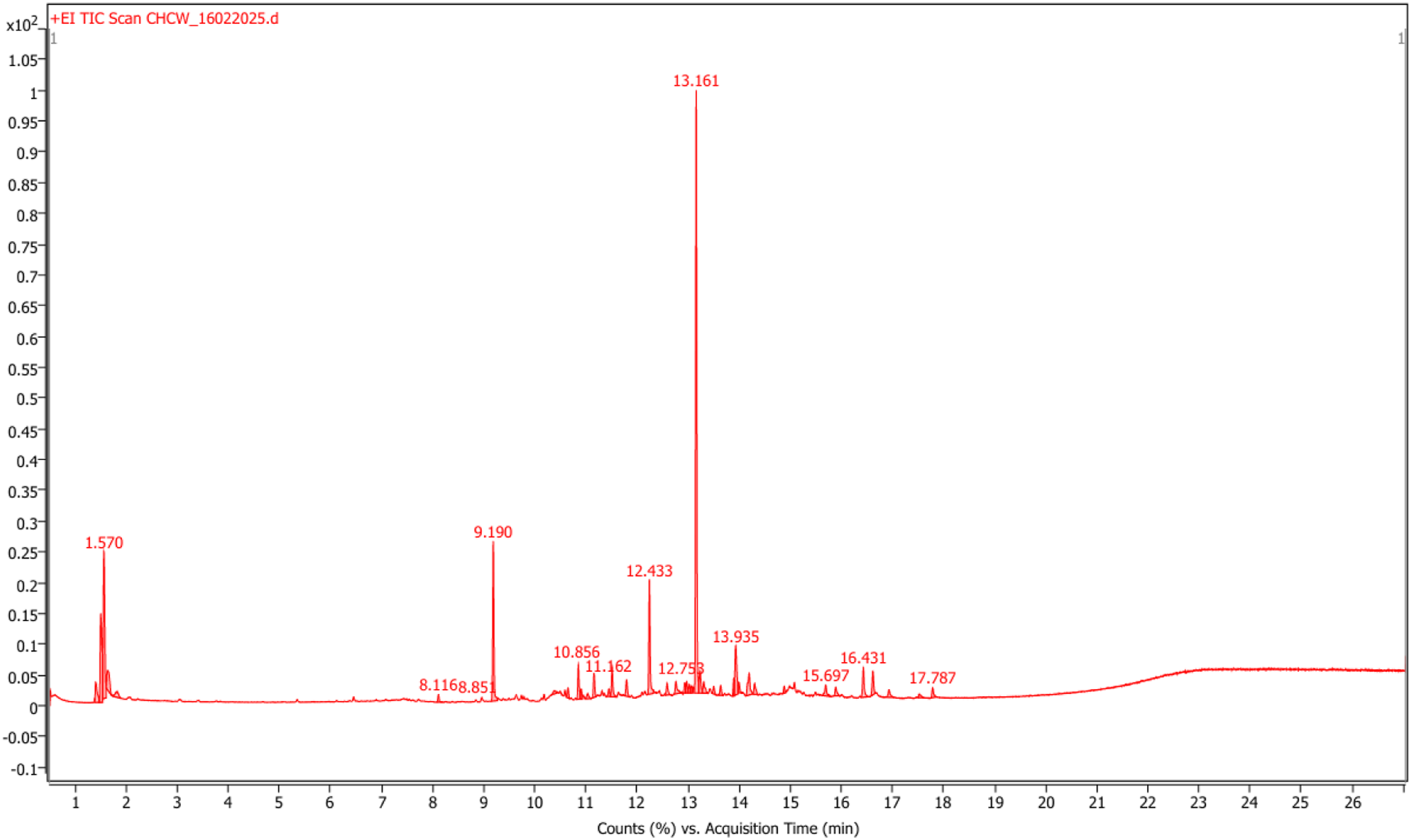
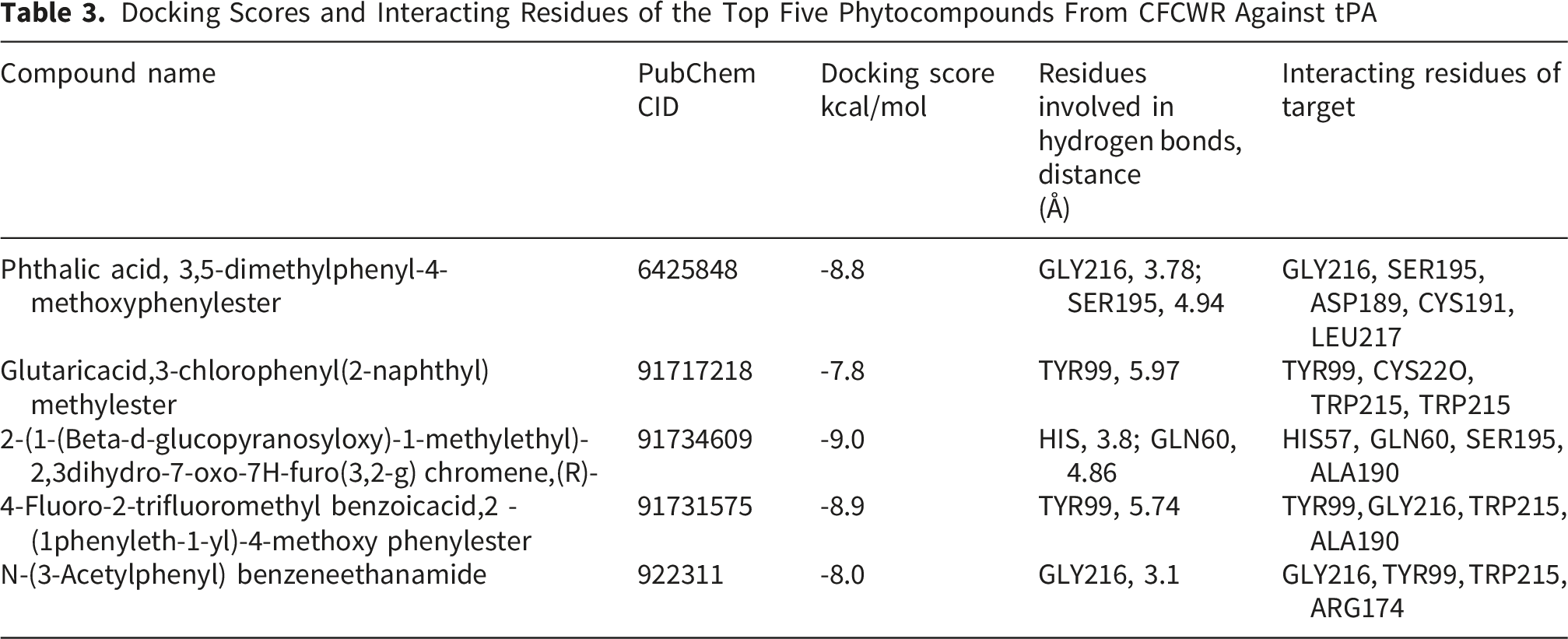
3.4. Molecular Docking and in Silico Analysis
Molecular docking analysis of selected bioactive compounds (Figure 2) identified from the GC–MS profile of CFCWR was performed against the human tissue plasminogen activator (tPA) (PDB ID: 1A5H). Among these, five molecules exhibited the most favorable binding affinities, with docking scores ranging from −7.8 to −9.0 kcal/mol (Table 3). The compound 2-(1-(β-D-glucopyranosyloxy)-1-methylethyl)-2,3-dihydro-7-oxo-7H-furo(3,2-g)chromene (CID: 91734609) demonstrated the strongest interaction (−9.0 kcal/mol), forming hydrogen bonds with HIS57 (3.8 Å) and GLN60 (4.86 Å) within the catalytic domain of tPA. Other key residues involved in the ligand–protein interactions included SER195, GLY216, TYR99, TRP215, and ALA190, which are essential for the catalytic function and substrate (fibrin/plasminogen) recognition of tPA. The next top-ranked ligands, 4-fluoro-2-trifluoromethylbenzoic acid, 2-(1-phenyleth-1-yl)-4-methoxyphenyl ester (CID: 91731575; −8.9 kcal/mol) and phthalic acid, 3,5-dimethylphenyl 4-methoxyphenyl ester (CID: 6425848; −8.8 kcal/mol), also formed stable hydrogen bonds with active-site residues, suggesting a strong potential to modulate the fibrinolytic activity of tPA (Figure 3). Structures of (A) 2-(1-(Beta-d-glucopyranosyloxy)-1-methylethyl)-2,3dihydro-7-oxo-7H-furo(3,2-g) chromene,(R)-, (B) Glutaricacid,3-chlorophenyl(2-naphthyl) methylester, (C) Phthalic acid,3,5-dimethylphenyl-methoxyphenylester, (D) 4-Fluoro-2-trifluoromethylbenzoicacid,2-(1phenyleth-1-yl)-4-methoxyphenylester, (E) N-(3-Acetylphenyl) benzeneethanamide Docking Scores and Interacting Residues of the Top Five Phytocompounds From CFCWR Against tPA 2D and 3D representations of ligand interactions with the tPA active site (PDB: 1A5H). Panels (A–E) show 2D interaction diagrams of five selected ligands, where magenta circles indicate polar amino acids, green circles denote nonpolar amino acids, and orange circles represent positively charged residues. Panels (A–H) depict the corresponding 3D binding poses, with ligands shown in ball-and-stick representation. The protein surface is colored according to electrostatic potential: blue for positive, red for negative, and white for neutral regions. Mechanism of clot degradation by CFCWR

Molecular Properties of the Top Five Docked Phytocompounds From CFCWR

Drug-Likeness Assessment of the Selected Five Phytocompounds From CFCWR

In silico Toxicity Prediction of the Selected Five Phytocompounds From CFCWR

Pharmacokinetic Parameters of the Top Five Phytocompounds From CFCWR

4. Discussion
Thrombosis represents a critical final common pathway leading to morbidity and mortality in several major cardiovascular disorders, including myocardial infarction and ischemic stroke. Despite considerable advances in elucidating the molecular mechanisms underlying thrombus formation and the complex pathophysiology of thrombosis, current pharmacological therapies for its prevention and management have largely remained unchanged for decades, with only incremental improvements achieved through the development of newer drug variants. 41 Increasing evidence indicates that natural products possess significant antithrombotic potential, as demonstrated in both preclinical and clinical investigations. These bioactive compounds may offer promising preventive or adjunctive strategies to complement existing pharmacotherapies for thrombotic diseases. The membrane stabilization assay of erythrocytes is a widely accepted model for evaluating the anti-inflammatory potential of plant extracts.42,43 Exposure of erythrocytes to harmful agents, such as hypotonic solutions,44,45 induces membrane rupture leading to hemoglobin release and oxidative degradation. 42 These phenomena result in the extravasation of serum proteins and fluids into surrounding tissues, thereby triggering an inflammatory response. 46 The compounds capable of stabilizing erythrocyte membranes hold promise as anti-inflammatory agents. In the present study, CFCWR exhibited considerable protection against hypotonic-induced hemolysis, demonstrating superior membrane stabilization compared to the standard ASA and the control.
Effective clot lysis is maintained through a dynamic balance between plasminogen activators promoting fibrin degradation and inhibitors such as PAI-1 that prevent overactive fibrinolysis.
47
It is the mechanism through which coagulation-induced fibrin clots are dissolved, ensuring proper blood flow.
48
Fibrinolysis is mainly driven by plasmin and modulated by activators such as tPA and urokinase (uPA). In this context, the CFCWR showed significant clot-dissolving potential, suggesting the presence of bioactive phytoconstituents capable of facilitating fibrinolysis or plasmin activation (Figure 4). Mechanism of clot degradation by CFCWR
While GC-MS identified numerous compounds, no isolation, quantification or bioactivity-guided fractionation was performed, molecular docking was therefore used as a preliminary mechanistic tool to identify potential bioactive compounds for future studies. The computational evaluation of CFCWR phytocompounds revealed promising thrombolytic potential through efficient binding to the tPA active site. 2-(1-(Beta-D-glucopyranosyloxy)-1-methylethyl)-2,3-dihydro-7-oxo-7H-furo(3,2-g)chromene, displayed the highest binding affinity (−9.0 kcal/mol), engaging catalytically relevant residues such as HIS57, SER195, and GLN60. These residues are well-known for their involvement in fibrinolytic mechanisms, suggesting that this compound could mimic or enhance natural thrombolytic processes by destabilizing fibrin networks.49,50 However, these docking results represent predicted ligand–protein interactions and should be interpreted only as supportive computational evidence complementing the observed in vitro thrombolytic activity.
Comparative binding energies indicated that other compounds, particularly 4-fluoro-2-trifluoromethylbenzoic acid,2-(1-phenyleth-1-yl)-4-methoxyphenyl ester, and phthalic acid,3,5-dimethylphenyl-4-methoxyphenyl ester, also exhibit significant potential. Polar interactions, such as hydrogen bonding or dipole contacts with residues like Tyr99, Gly216, and Trp215, may further stabilize ligand binding in the fibrin binding pocket. In thrombin and related serine proteases, tryptophan residues (like Trp215) have been shown to contribute significantly to ligand binding and substrate recognition. 51 Moreover, surface loop residues adjacent to active sites are known to modulate binding specificity through polar interactions in serine proteases. 49 These interactions are consistent with previous computational studies, where hydrophobic substitution improved ligand affinity toward tPA.
In terms of molecular characteristics, the selected top five candidates adhered closely to drug-likeness standards, with acceptable Log P and molecular weight values, minimal rule violations, and absence of PAINS alerts. 52 Such profiles reinforce the notion that these compounds could possess both bioactive potency and pharmacokinetic suitability. The bioavailability scores (0.55) and rotatable bond counts (<10) are typical of molecules with good absorption and oral delivery prospects.53,54
The predicted toxicity and ADMET results supported the safety of these phytochemicals. The low AMES mutagenicity and carcinogenicity probabilities, along with moderate clearance and non-inhibitory CYP3A4 profiles, indicate a low risk of adverse drug reactions. Moreover, the combination of strong human intestinal absorption and optimal plasma protein binding suggests efficient systemic exposure while minimizing toxicity accumulation.51,55
While the present findings are promising, this study is limited to in vitro assays and computational analyses and lacks in vivo validation. Furthermore, the individual phytoconstituents identified were not isolated and experimentally verified for their specific thrombolytic activities. Therefore, further in vivo investigations and detailed mechanistic studies are necessary to confirm and substantiate the therapeutic potential of Curcuma wallichii.
The integrated molecular docking, pharmacokinetic, and toxicity results indicate that several phytocompounds from CFCWR can be promising lead compounds for further in vivo thrombolytic studies. The observed pharmacological potential of Curcuma wallichii supports its ethnomedicinal relevance and highlights the utility of in silico screening in identifying natural thrombolytic agents.
5. Conclusion
The present study demonstrates that the chloroform extract of Curcuma wallichii exhibits moderate thrombolytic activity in vitro. Molecular docking identified 2-(1-(β-D-glucopyranosyloxy)-1-methylethyl)-2,3-dihydro-7-oxo-7H-furo(3,2-g)chromene as the top bioactive compounds, showing favorable binding to key residues of tPA. The combined in vitro and in silico findings indicate that C. wallichii contains phytoconstituents with potential thrombolytic activity. Further studies, including compound isolation and mechanistic evaluation, are needed to better understand its biological effects and therapeutic relevance.
Footnotes
Acknowledgments
The authors thank the Department of Pharmacy, University of Rajshahi, Bangladesh, for providing facilities to carry out the research. Also, the authors would like to thank Dr. A. H. M. Mahbubur Rahman, a specialist taxonomist from Rajshahi University, for his assistance in authenticating the plant species.
Ethical Considerations
The experimental procedure was approved both for animal and human models by the Institutional Animal, Medical Ethics, Biosafety and Biosecurity Committee of Rajshahi University, Bangladesh (Approval No. 218/320(41)/IAMEBBC/IBSc). All experimental procedures involving both animal and human subjects were carried out in accordance with relevant institutional, national, and international guidelines and regulations.
Consent to Participate
Informed consent was obtained from the healthy volunteers prior to human blood sample collection.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.


