Abstract
Objectives
The water distillate derived from
Methods
The chemical constituents of Sid.AS were identified and quantified using Gas Chromatography-Mass Spectrometry (GC-MS) method. The antioxidant potential of Sid.AS samples was assessed
Results
Thymol was identified as the major compound in Sid.AS through GC-MS analysis. Sid.AS demonstrated significant anti-urease, anti-inflammatory, anti-lipase, and antioxidant activities, as evidenced by low IC50 values compared to the positive controls. This suggests its potential in controlling gastric-related disorders, scavenging free radicals, and managing obesity by inhibiting the breakdown and absorption of fats. Additionally, Sid.AS exhibited inhibitory effects against alpha-amylase and alpha-glucosidase enzymes, indicating potential anti-diabetic activity by regulating blood sugar levels. Sid.AS displayed strong antimicrobial activity against tested microorganisms, with higher zones of inhibition and lower MIC and MLC values, indicating its effectiveness in combating microbial infections. Findings from the anti-lipase assay demonstrated activity comparable to that of the positive control, Orlistat.
Conclusion
The findings of Sid.AS suggest its potential as a multi-functional bioactive herbal distillate with various pharmacological activities. Our results highlight that Sid.AS is a promising natural herbal extract with diverse pharmacological properties, including anti-urease, antioxidant, anti-inflammatory, anti-diabetic, anti-obesity, and antimicrobial activities. Further research and development could explore its potential applications in various therapeutic areas.
Introduction
Sri Lanka is truly blessed with a rich heritage of traditional medical systems, including Ayurveda, Siddha, Unani, and Deshiya Chikitsa, collectively known as ‘Traditional Medicine.’ These systems, deeply rooted in our culture, predominantly rely on herbal products for healing. The global interest and recognition of traditional herbal medicine are steadily increasing, particularly within the scientific community.
1
Many chronic metabolic diseases have positively responded to plant-based medicines, highlighting their potential efficacy. Ayurveda and traditional medicine have been integral to Sri Lankan healthcare for over 3000 years, reflecting our enduring commitment to holistic healing.
2
However, despite their longstanding practice, many traditional herbal products’ scientific validation and reproducibility remain unproven. In Sri Lankan traditional medicine, numerous plant species address chronic conditions, notably gastrointestinal issues. One such example is
This well-branched annual plant can reach heights of up to 90 cm and is cultivated across various regions of India and Sri Lanka. The genus
Sri Lanka's rich biodiversity has earned it recognition as one of the world's biodiversity hotspots. Within this diverse ecosystem, aromatic plants play a significant role and are extensively utilized in Ayurveda and traditional medical systems. The rising concern over adverse drug reactions and the development of bacterial resistance associated with synthetic medicines has fueled a growing interest in alternative herbal medicine for managing chronic diseases. This shift in focus has led to increased motivation within indigenous drug industries to develop herbal products with potent bioactivities.
Harnessing the therapeutic potential of aromatic plants offers a natural and sustainable approach to healthcare, aligning with the principles of Ayurveda and traditional medicine. By leveraging the rich botanical resources of Sri Lanka, researchers and practitioners aim to develop effective herbal remedies that can address a wide range of health conditions while minimizing adverse effects and bacterial resistance.
This emphasis on herbal medicine reflects a broader global trend towards holistic and integrative approaches to healthcare, where traditional knowledge is combined with modern scientific research to develop evidence-based therapies. As Sri Lanka continues to explore the medicinal properties of its aromatic plants, it holds the potential to contribute significantly to the advancement of herbal medicine and the well-being of its population.
Fortunately, numerous medicinal plants are known to possess urease inhibition activity, which can help alleviate the symptoms of gastritis. By inhibiting the activity of urease, these plants assist in reducing the production of ammonia by
The prevalence of chronic inflammatory diseases poses a significant threat to human health, with global incidence on the rise. Conditions such as type 2 diabetes, obesity, asthma, arthritis, and cancer are among those affected. While nonsteroidal anti-inflammatory drugs (NSAIDs) are commonly used to manage inflammation by inhibiting cyclooxygenase enzymes, their prolonged use often leads to gastrointestinal and cardiovascular side effects.13,14 In response to this challenge, there is a growing demand for natural alternatives to NSAIDs as anti-inflammatory medicines. Red Blood Cells Stabilization assay offers a promising method for identifying natural NSAIDs. 15 The WHO reports that obesity has reached epidemic proportions globally, with rates tripling since 1975. Type 2 diabetes and obesity are closely linked and are associated with increased risks of cardiovascular diseases and cancer. 16 Efforts to develop new synthetic drugs to combat these non-communicable diseases have had limited success. Therefore, there is a continuous need to explore alternative natural products derived from medicinal plants for the prevention and management of chronic inflammatory diseases. Key enzymes involved in carbohydrate digestion, namely α-amylase and α-glucosidase, and lipase, which facilitates fat absorption in the intestine, are extensively targeted for managing diabetes and obesity. 17 Furthermore, many non-communicable diseases, including neurodegenerative diseases, inflammatory diseases, and cardiovascular diseases, are linked to ailments caused by reactive oxygen species. Thus, the radical scavenging activity of natural substances holds great importance in preventing such diseases in the human body. 18 By harnessing the therapeutic potential of natural substances and targeting key enzymes and reactive oxygen species, we can develop effective strategies for combating chronic inflammatory diseases and promoting overall health and well-being.
In traditional medicine, including Ayurveda, Unani, and Siddha medical systems, the preparation of
The water distillate of
Asamodagam spirit, a popular herbal preparation in traditional medicine, is renowned for its unique pharmacological activities against chronic inflammatory diseases. These activities include antibacterial, anti-inflammatory, antioxidant, and anti-ulcerative properties. Additionally, the Asamodagam spirit is widely recognized in traditional medicine as a remedy for infants and children suffering from stomach aches, bowel disorders, acidity, and flatulence. However, despite its widespread use, limited systematic scientific research has been conducted to confirm the efficacy of these products. Further, it is widely recognized that artificial Asamodagam spirit is manufactured using synthetic thymol crystals and is readily available in the market. There is currently no publication available that systematically distinguishes between Asamodagam spirit prepared using synthetic thymol and the distillate derived from Asamodagam seeds. This gap exists due to the lack of a comprehensive study on the chemical constituents. Therefore, the present study also aims to address this gap by facilitating the differentiation between natural Asamodagam spirit prepared from Asamodagam seeds and synthetic thymol.
Sid. As, prepared from the seeds of
Hence, this study represents the first systematic investigation of the water distillate of
Results and Discussion
Identification of Plant Material

Exterior view of
GC-MS Analysis of the Sid.AS of T. roxburghianum
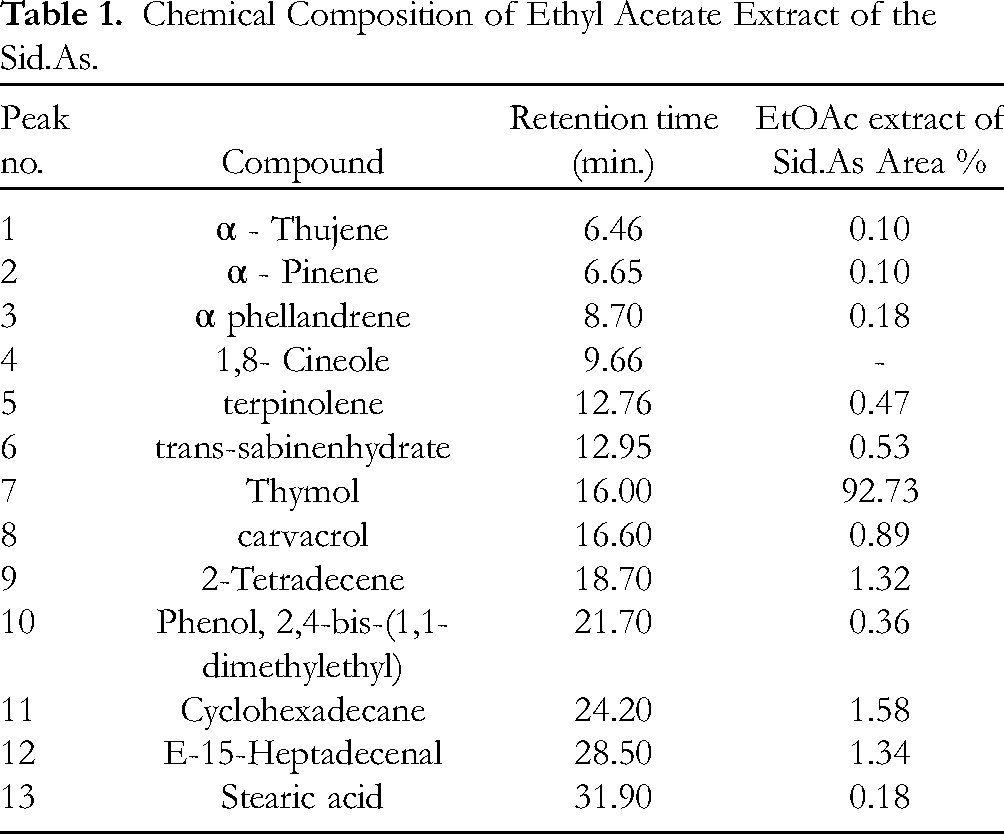
The gas chromatogram of Sid.AS was presented in Figure 2, and the chemical constituents were listed according to their elution on the DB-5 ms column (Table 1). Through GC-MS data and relative retention times, most of the constituents in Sid.AS were identified. Notably, the major constituent was found to be thymol. Thymol accounted for 92.7% of the total composition of Sid.AS. Additionally, 2-tetradecene (1.32%), cyclohexadecane (1.58%), and E-15-heptadecenal (1.34%) were identified as minor constituents. Together, these compounds comprised 100% of the total amount of Sid.AS The water distillate of

GC-MS profile of the ethyl acetate extract of the Siddhaleapa Asamodagam Spirit (Sid.AS) obtained from the seed of
Chemical Composition of Ethyl Acetate Extract of the Sid.As.
Analysis of Heavy Metals and Microbial Detection in Asamodagam Spirit (Sid.AS).

These results provide valuable insights into the chemical composition of Sid.AS, confirming the presence of thymol as the predominant compound. Thymol's significant presence aligns with its well-known therapeutic properties, including antibacterial, anti-inflammatory, and antioxidant effects. Additionally, the minor constituents identified may contribute to the overall pharmacological profile of Sid.AS, further highlighting its potential as a natural remedy for various ailments.
Quality Control Analysis of Sid.AS
The demand for herbal medicine in Sri Lanka is steadily increasing, with approximately 80% of the population relying on herbal products to treat various ailments. Given the widespread use of herbal remedies, the quality standards of these medicinal products must align with the regulations established by the Department of Ayurveda, Sri Lanka. This ensures that these products are safe and effective for human use, contributing to the well-being of individuals. As Asamodagam spirit is frequently employed for gastrointestinal diseases within the Sri Lankan community, comprehensive quality testing was conducted using various accepted analytical techniques (Table 2). The results of these analyses confirmed that Sid.AS meets the stringent quality parameters expected for herbal medicine. Specifically, Sid.AS was free from sugars, fats, proteins, and carbohydrates, as indicated in Table 3. Furthermore, thorough testing demonstrated the absence of hazardous heavy metals or metalloids such as arsenic (As), cadmium (Cd), lead (Pb), and mercury (Hg), which could pose health risks. Additionally, Sid.AS lacked pathogenic microorganisms including
Nutritional Analysis of Distillate of
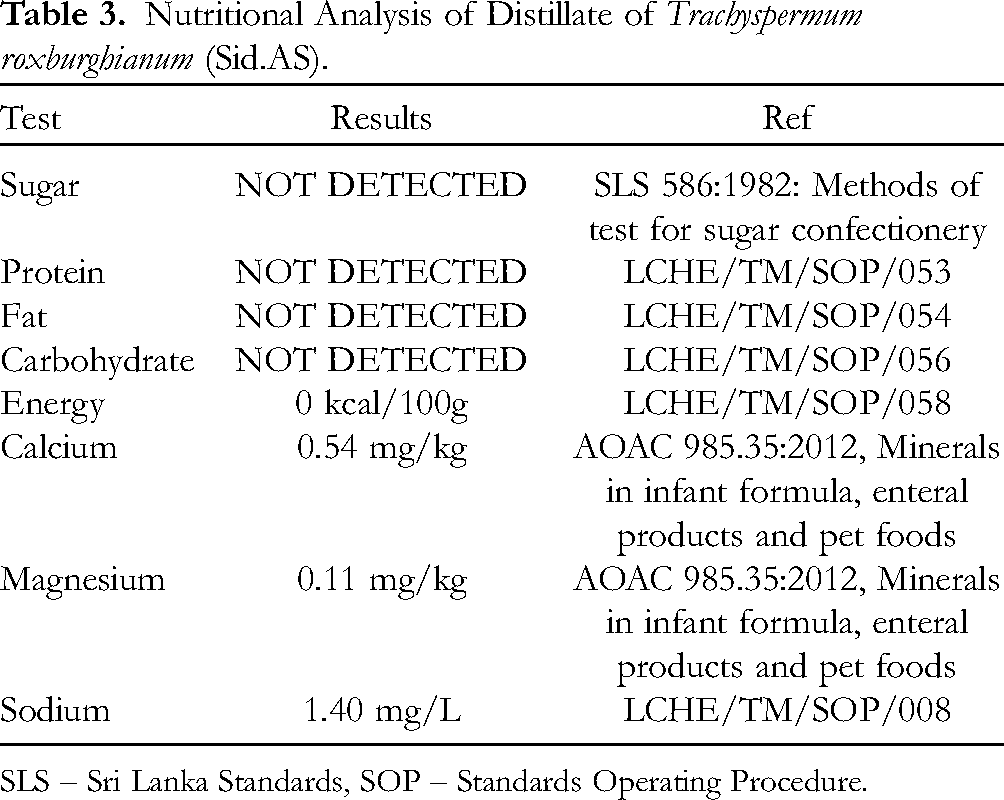
SLS – Sri Lanka Standards, SOP – Standards Operating Procedure.
Quality Control Parameters of Sid. AS. Spirit.

LOQ – Limit of Quantification.
Antioxidant Properties of Sid.AS Obtained from T. roxburghianum Against DPPH and ABTS Assays
The presence of antioxidant compounds with radical scavenging capability in herbal medicine is crucial as they can protect the human body from non-communicable diseases. DPPH and ABTS assays are performed to screen for the scavenging activity of free radicals in natural sources to assess the antioxidant potential of plant extracts.
The DPPH antioxidant activity and ABTS antioxidant activity of the distillate of seeds from

Comparison of the average inhibitory activity of Sid.AS and the positive control (BHT) against DPPH assay.

Comparison of the average inhibitory activity of Sid.As and the positive control (BHT) against the ABTS assay STD (BHT) – Standard compound (Butylated hydroxytoluene).
Urease Enzyme Inhibitory Activity of the Sid.AS
The urease enzyme inhibitory activity of Sid.AS was compared with the positive control, thiourea. Figure 5 illustrates concentration-dependent activities against urease observed between selected concentrations of the test samples, where the inhibitory effect increased with increasing concentration within the range of 0.625-10 mg/mL. The positive control, thiourea, demonstrated the highest inhibitory activity at 10 mg/mL, reaching 96.5% (with an IC50 of 0.03 ± 0.009 mg/mL). In comparison, Sid.AS exhibited inhibitory activity above 78% at the concentration of 5 mg/mL. The study further revealed that the IC50 value of Sid.AS was 0.24 ± 0.01 mg/mL, indicating its potent urease inhibitory activity. This suggests that not only the major compounds but also the minor compounds present in the distillate of
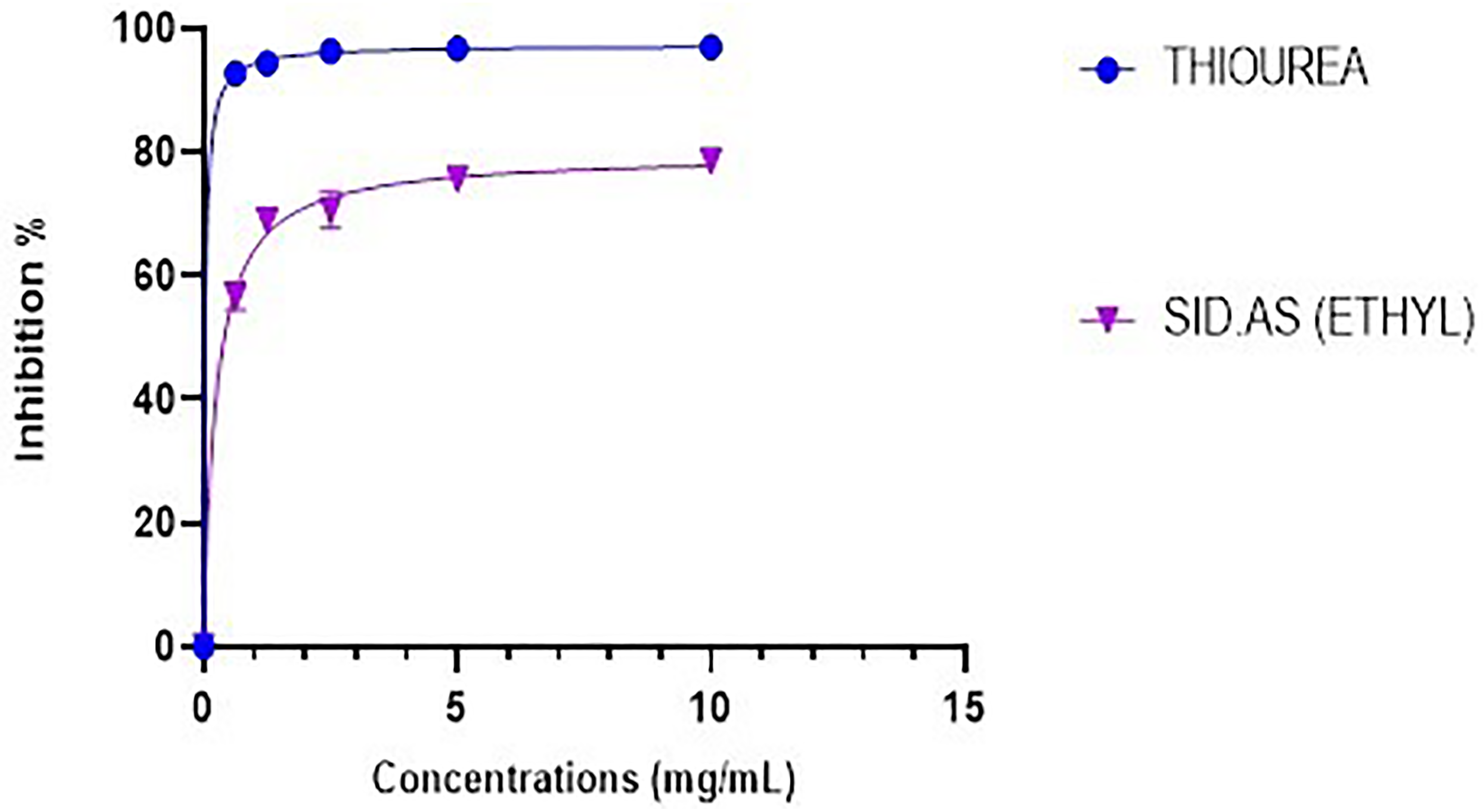
Comparison of the urease enzyme inhibitory activity of Sid.As and the positive control (Thiourea) against the urease inhibition assay.
Anti-Inflammatory Activity of the Sid.AS
The anti-inflammatory activity of Sid.AS and the positive control (Aspirin) were determined using the Human Red Blood Cell Membrane (HRBCM) stabilization assay, and the results are depicted in Figure 6. IC50 values were calculated using GraphPad Prism 7.0 software.

Comparison of the anti-inflammatory activity of Sid.As and the positive control (Aspirin) against the anti-inflammatory assay.
In the assay, the effects of Sid.AS in inhibiting heat-induced hemolysis at different concentrations (ranging from 0.625 to 10.00 mg/mL) were measured. The IC50 values obtained for Sid.AS and the Positive Control (Aspirin) were 0.57 ± 0.09 mg/mL and 0.24 ± 0.03 mg/mL, respectively.
Inflammation is implicated in numerous diseases, often involving the release of lysosomal enzymes that contribute to tissue damage and exacerbate the inflammatory response. Aspirin, a non-steroidal anti-inflammatory drug (NSAID), functions by stabilizing the lysosomal membrane and inhibiting specific lysosomal enzymes. The anti-inflammatory effect of Sid.AS is evidenced by its ability to suppress hypotonicity-induced HRBC membrane lysis, as demonstrated by the HRBCM stabilization assay. This suggests that Sid.AS may exert its anti-inflammatory activity through mechanisms similar to those of Aspirin. These findings underscore the potential of Sid.AS as a natural anti-inflammatory agent, offering a promising avenue for the management of inflammatory conditions and related diseases.
Alpha-Amylase and Alpha-Glucosidase Inhibition Assays
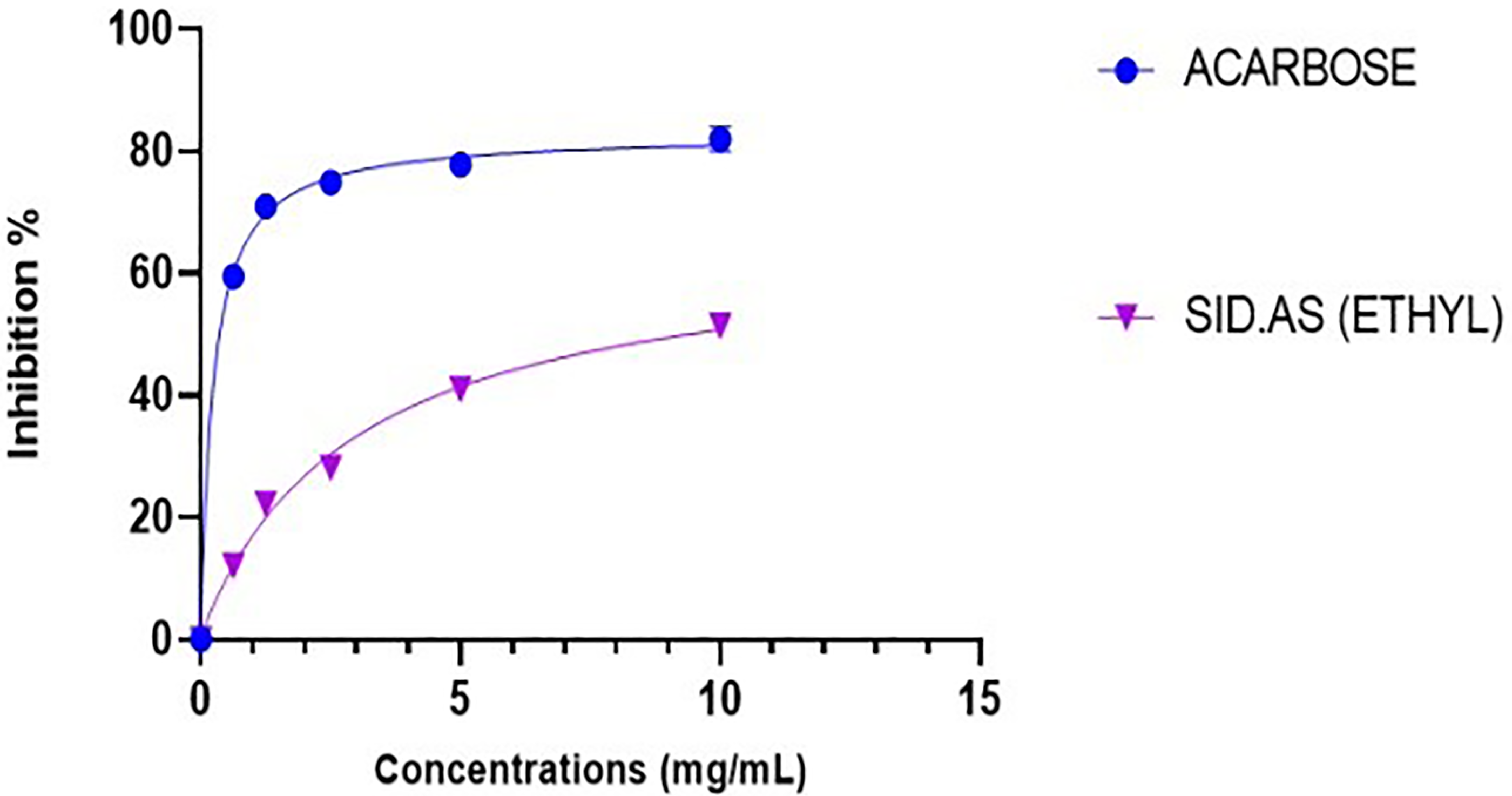
In the present study, Sid.AS was evaluated for inhibiting α-amylase and α-glucosidase enzymes, key targets in diabetes management. At a concentration of 5 mg/mL, Sid.AS exhibited significant enzyme inhibitory activity, demonstrating 77% inhibition of α-glucosidase and 47% inhibition of α-amylase, as shown in Figures 7 and 8, respectively. These results indicate the potential of Sid.AS in regulating postprandial glucose levels by slowing down the breakdown of complex carbohydrates into glucose in the digestive system. Comparatively, the positive control, acarbose, demonstrated robust inhibitory activity, with 80% inhibition of α-glucosidase and 77% inhibition of α-amylase, showcasing its efficacy in diabetes management. Acarbose also exhibited low IC50 values of 0.23 mg/mL for both α-glucosidase and α-amylase inhibitory activities. Sid.AS displayed comparable enzyme inhibitory activity, with IC50 values of 0.51 ± 0.04 mg/mL for α-glucosidase and moderate activity with an IC50 value of 2.95 ± 0.19 mg/mL for α-amylase. These findings suggest that Sid.AS possesses promising potential as a natural remedy for controlling diabetes, particularly in regulating postprandial hyperglycemia. The results of both assays indicate that Sid.AS could be developed as a potential indigenous medicine to complement existing therapeutic strategies for diabetes management.
Comparison of the α-amylase enzyme inhibitory activity of Sid.As and the positive control (Acarbose) against the α- amylase inhibition assay.
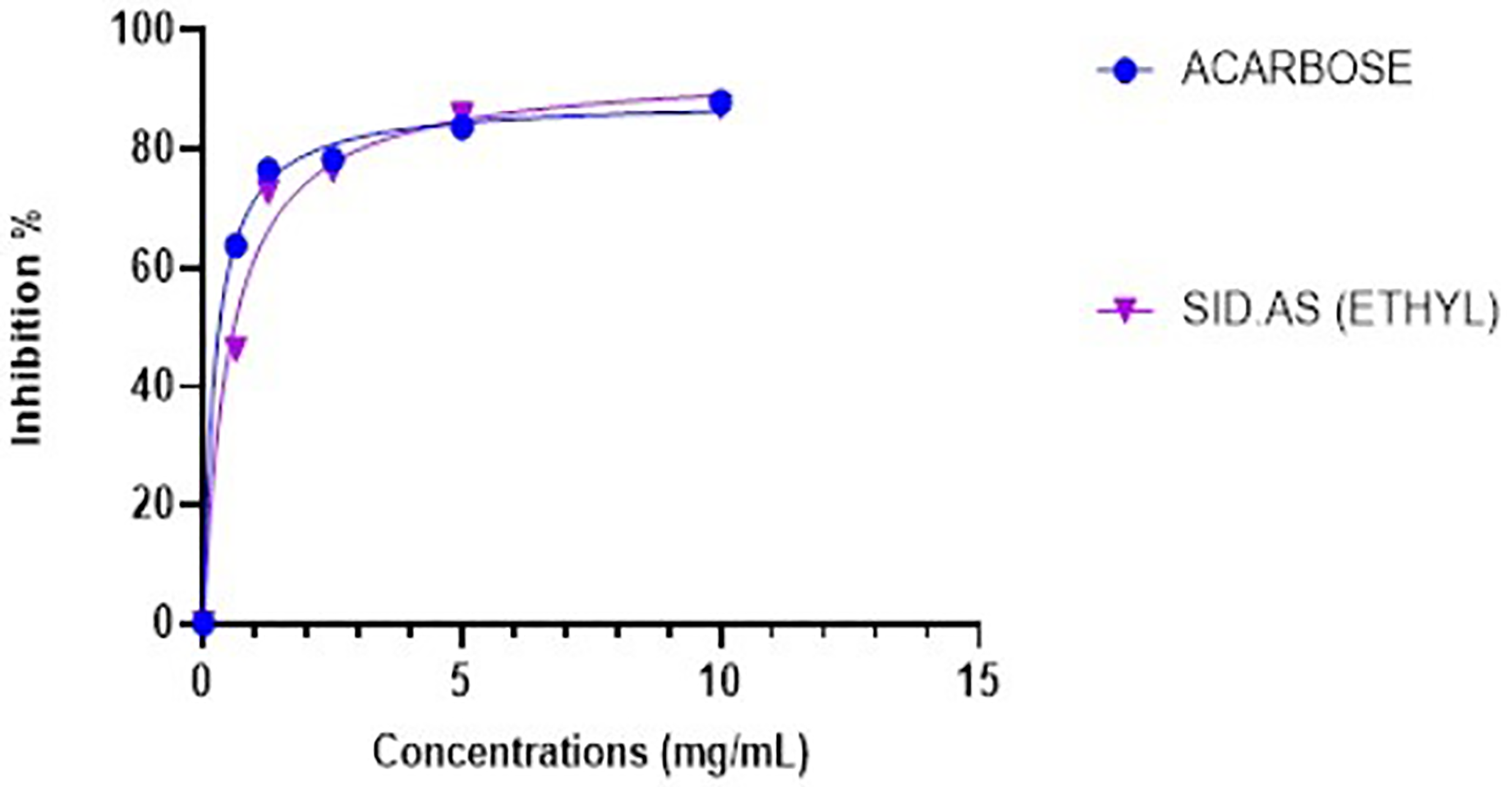
Comparison of the α-glucosidase enzyme inhibitory activity of Sid.As and the positive control (Acarbose) against the α-glucosidase inhibition assay.
Lipase Inhibition Assay
The anti-obesity activity of Sid.AS was investigated using a lipase inhibition assay, employing a series of concentrations ranging from 0.625 to 10.00 mg/mL. The inhibitory activities of Sid.AS and the positive control (Orlistat) are depicted in Figure 9. At a concentration of 10 mg/mL, Sid.AS exhibited significant inhibition of lipase activity, demonstrating 79% inhibition. Similarly, Orlistat, the positive control, demonstrated inhibition of lipase activity, with 89% inhibition at the same concentration. The IC50 values obtained for Orlistat and Sid.AS were 0.38 ± 0.01 mg/mL and 0.40 ± 0.01 mg/mL, respectively. Importantly, these values were not significantly different from each other (

Comparison of the lipase enzyme inhibitory activity of the Sid.As and the positive control (Orlistat) against the lipase inhibition assay.
Antibacterial Sensitivity and Antifungal Sensitivity Analysis of Sid.AS
Different concentrations of Sid.AS were evaluated for their antimicrobial activity against human pathogenic microorganisms, including two strains of Gram-positive bacteria (
Antimicrobial Activity of the Distillate of

NI – NO Inhibitory zone, ZOI – Zone of Inhibition.
Three replicates are used for each analysis.
The significant antimicrobial and antifungal activity demonstrated by Sid.AS aligns with previous findings, which also reported inhibitory effects against
Determination of Minimum Inhibitory and Minimum Lethal Concentrations
Sid.AS showcased robust antibacterial activity against both
MIC and MLC Values of Sid. AS Against Test Pathogenic Microorganisms.

There are several limitations to this study. Firstly, the raw material used was not available in Sri Lanka and was purchased from Pakistan. Additionally, this study only reflects the
Material and Methods
Chemicals and Reagents
Analytical-grade chemicals were used for all the experiments. Urease enzymes from Jack Beans (Sisco Research Laboratories Pvt. Ltd, India), phosphate buffer (Sisco Research Laboratories Pvt. Ltd, India), sodium nitroprusside (Sisco Research Laboratories Pvt. Ltd, India), urea (Sisco Research Laboratories Pvt. Ltd, India), Sodium hypochlorite acid (Loba Chemie Pvt. Ltd (Mumbai, India), Sodium hydroxide pellets (Loba Chemie Pvt. Ltd (Mumbai, India), DPPH (Sigma-Aldrich, USA), Methanol (99.9%) (Sigma-Aldrich, USA), BHT (Himedia Pvt.Ltd, India), ABTS (Sigma-Aldrich, USA), Potassium persulfate (Sigma-Aldrich, USA), Lipase (20,000 units/mg protein) (Sigma-Aldrich, USA), Orlistat (Sigma-Aldrich, USA), p-nitro phenyl acetate (181.15 g/mol, BIOSYNTH) (HiMedia Pvt.Ltd, India), Tris HCl (98%) (HiMedia Pvt.Ltd, India), α-Amylase (300-1500 units/mg protein) (Sigma-Aldrich, USA), 3,5-Dinitrosalicylic acid (Sigma-Aldrich, USA),, Acarbose (Sigma-Aldrich, USA), Sodium monobasic phosphate (Sigma-Aldrich, USA), Hydrochloric acid (35%) (Sigma-Aldrich, USA), Sodium potassium tartrate tetrahydrate (99%) (Loba Chemie Pvt. Ltd, Mumbai, India), Sodium dibasic phosphate dihydrate (99%) (Loba Chemie Pvt. Ltd, Mumbai, India), Starch (Loba Chemie Pvt. Ltd, Mumbai, India), α- glucosidase enzyme (Loba Chemie Pvt. Ltd, Mumbai, India), Para-nitrophenyl glucopyranoside (P-NPG) (Loba Chemie Pvt. Ltd, Mumbai, India), Sodium dibasic phosphate dihydrate (99%) (Loba Chemie Pvt. Ltd, Mumbai, India), Aspirin (USV Pvt. Ltd, Mumbai, India), Red Blood Cells suspension from a volunteer who has not taken any anti-inflammatory drugs, Thymol (Techno Pharmachem Pvt. Ltd, India), Potassium ferricyanide (Himedia Laboratories Pvt. Ltd, India). Trichloroacetic acid (Sigma-Aldrich, USA), ferric chloride (Research-Lab Fine Chem Industries, India), Mueller Hinton Agar (Himedia Laboratories Pvt. Ltd, India), Nutrient Broth (Himedia Laboratories Pvt. Ltd, India), Amoxycillin (Astron Pvt. Ltd, Sri Lanka), Potato Dextrose Agar (Oxoid Ltd, United Kingdom), Streptomycin and Fluconazole (Micro Labs Limited - India), Microplate Reader (Biotek, USA).
Plant Collection and Identification
Preparation of the Distillate of Trachyspermum roxburghianum (Siddhalepa Asamodagam Spirt - Sid.AS)
Sid.AS was prepared in accordance with the method given in the Ayurveda Pharmacopeia. The cleaned, washed, and dried seeds (15 g) were hydro-distilled in a 500 mL round bottom flask for 2 h. The distillate with the essential oil of
Extraction of Chemical Constituents in Sid.AS Samples
Sid.AS (300 mL) was transferred into a separatory funnel and partitioned with ethyl acetate (3 × 100 mL). The ethyl acetate fraction from each partition was then combined and dried using anhydrous sodium sulfate to remove the remaining water. Subsequently, the solvent was completely evaporated using a rotary evaporator set at 40 °C. The remaining viscous solution was weighed and prepared for biological testing.
GC-MS Analysis of the Sid.AS
The Sid.AS extracted into ethyl acetate was examined by gas chromatography-mass spectrometry (GC-MS) using Agilent 7890 B / 5977 B Series GC/MSD with a 30 m×0.25 mm i.d.×0.25 mm DB-5 ms (J & W Scientific, Folsom, CA, USA) column. The analysis parameters were set as: oven temperature program, 50 °C (1 min), 50 °C to 150 °C (5 °C/min), 150 °C (3 min), 150 °C to 200 °C (10 °C/min); Helium gas flow rate, 37 cm/s; injector and detector temperatures, 250 oC. Samples (1 μL) were injected. EIMS, electron energy, 70 eV; ion source and connecting parts temperature, 250 °C. The acquisition was performed in scanning mode (mass range m/z 35-400 u). Compounds were identified using relative retention times and their mass spectra. Each mass spectrum was compared with corresponding reference standard data reported in the mass spectra from NIST 05 in-house libraries.
Quality Control Analysis of Sid.AS
Nutritional analysis, microbial detection, and heavy metal analysis of Sid.AS were investigated using standard AOAC and ISO methods for the quality control aspects, as given in Table 2.
Determination of the Antioxidant Activity of Sid.AS
The antioxidant activity of Sid.AS was evaluated using DPPH (2,2-diphenyl-1-picrylhydrazyl) 27 and ABTS (2,2-Azino-Bis (3-Ethylbenzothiazoline-6-Sulfonic Acid) 28 radical scavenging assays as described below.
Determination of DPPH Radical Scavenging Activity
The assay was carried out in a 96-well plate according to the method described by Upamalika et al, 2015. 27 A concentration series (0.625, 1.25, 2.50, 5.00, and 10.00 mg/mL) was prepared for the samples to be analyzed by dissolving an appropriate amount of Sid.AS, and a positive control (BHT) separately. Absorbance measurements were taken at 517 nm with the microplate reader (Biotek, USA) after 30 min of incubation at room temperature in the dark. The percentage inhibition was calculated and plotted against the concentration to determine the IC50. The IC50 values of all samples were calculated using Graphpad Prism version 8.0.1 (244). Each concentration was replicated three times, and statistical analysis was carried out with One-way ANOVA using Minitab 17.
Determination of ABTS·+ Radical Scavenging Activity
The ABTS radical scavenging ability assay was carried out in a flat bottom 96-well microtiter plate according to the method described by Weerasinghe et al, 2022 with slight modifications. 28 Different doses of the test sample (Sid.AS) and positive control of butylated hydroxy-toluene (BHT) (0.625, 1.25, 2.50, 5.00, and 10.00 mg/mL) were added (10 μL) to 190 μL of freshly prepared ABTS solution in the 96 well plate. All reagents were mixed and incubated for 7 minutes at room temperature under dark conditions. The absorbance of each well was measured at 714 nm with the Microplate Reader (Biotek, USA). The percentage inhibition and the IC50 values were calculated using GraphPad Prism 8.0.1 (244).
Determination of Urease Enzyme Inhibitory Activity
Urease inhibitory activity of Sid.AS was evaluated using a modified Berthelot spectrophotometric method, and the absorbance was measured at 625 nm.
29
The test solutions of Sid.AS, and positive control (Thiourea) were prepared with concentrations of 0.625, 1.25, 2.50, 5.00, and 10.00 mg/mL. The assay solution contained urea (0.125 mol/L, 80 µL) as the substrate, a test sample (20 µL) and phosphate buffer (pH 7.4). Urease enzyme (80 µL) was added to start the enzymatic reaction, and after 30 min, the ammonia concentration in each sample was measured by incubating the mixture with 500 µL of solution A (0.50 g phenol and 2.50 mg of sodium nitroprusside in 100 mL of distilled water) and 500 µL of solution B (0.50 g sodium hydroxide and 840 µL of sodium hypochlorite 5% in 100 mL of distilled water) at 37 °C for 30 min. The absorbance of each well was measured at 625 nm by the plate reader (Biotek, USA). The following equation was generated to calculate the percentage inhibition of the urease enzyme: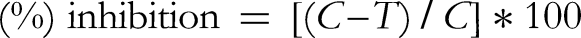
Determination of Anti-Inflammatory Activity
The anti-inflammatory assay used the human red blood cell stabilization method, which uses the heat induced hemolysis principle.30,31 The assay was conducted for Sid.AS, and the positive control separately. The concentrations of test samples used were 0.625, 1.25, 2.50, 5.00, and 10.00 mg/mL. Aspirin at the same concentrations as the test samples was used as the positive control. The samples were dissolved in 0.50 mL of DMSO (dimethyl sulfoxide), and the assay was performed. The absorbance of the supernatants was measured at 560 nm using the Microplate Reader (Biotek, USA) by using aliquots of 200 μL for each well in a 96 well-plate from each mixture. The test was performed in triplicates, and IC50 values were calculated using the statistical software Graphpad Prism version 8.0.1 (244).
Determination of α-Amylase Enzyme Inhibitory Activity
The α-amylase assay was also carried out in a flat-bottom 96-well microtiter plate, according to the method described by Maduranga et al (2018) 17 with slight modifications. The enzyme assay was performed for Sid.AS, and the positive control (Acarbose). The concentrations of test samples used were 0.625, 1.25, 2.50, 5.00, and 10.00 mg/mL and the blank samples were prepared without the test sample or the amylase enzyme separately. The percentage inhibition of α-amylase and the IC50 values were calculated to evaluate the antidiabetic activity of each extract after recording absorbance at 540 nm. The experiment was carried out in triplicate. The IC50 values were calculated using the statistical software Graphpad Prism version 8.0.1 (244).
Determination of α-Glucosidase Enzyme Inhibitory Activity
The α-glucosidase inhibitory activities of Sid.AS, and the positive control (Acarbose) were performed using the method described by Telagari & Hullatti in 2015. 32 The concentrations of test samples used were 0.625, 1.25, 2.50, 5.00, and 10.00 mg/mL. Acarbose at the same concentrations as the test samples was used as the positive control. P-NPG (5 mM, 20 μL) was used as a substrate and incubated further at 37 °C for 20 min. The absorbance was measured at 405 nm to measure the released p-nitrophenol using the Microplate Reader (Biotek, USA). Without a test substance, they were used as control samples, and each experiment was performed in triplicate. The results were expressed as percentage inhibition, and the IC50 values were calculated using the statistical software Graphpad Prism version 8.0.1 (244).
Determination of Lipase Enzyme Inhibitory Activity
Lipase inhibition assay was performed to evaluate the anti-obesity properties of Sid.As. The assay was carried out according to the method described by Maduranga et al, 2018. 17 The concentrations of test samples used were 0.625, 1.25, 2.50, 5.00, and 10.00 mg/mL and Orlistat was used as the positive control. p-Nitrophenyl acetate (4 mM in tris buffer, pH 8.0) was added to each test sample as the substrate and the absorbance was measured at 405 nm after completion of the assay. Each experiment was performed in triplicate. The percentage of inhibition of the lipase enzyme and the IC50 values were calculated using the statistical software Graph pad Prism version 8.0.1 (244).
Determination of Antimicrobial Activity
Preparation of the Inoculum
The microorganisms which were used in this study were separately cultured on sterilized Nutrient agar using the streak plate method and incubated at 36 ± 1 °C for 24 h. Then isolated colonies were inoculated into nutrient broth according to McFarland standards. Bacterial and fungal cultures were prepared as 0.5 McFarland and incubated at 35 ± 2 °C for 12 h. After incubation, cultures were standardized by measuring the absorbance under OD600 0.4 - 0.5 (equal to 12 × 108 CFU/ml) to use as the starter culture.
Antibacterial Sensitivity Test (ABST)
ABST was performed for five selectively prepared concentrations of Sid.As along with positive and negative controls against four microorganisms by using the well diffusion method on Mueller Hinton Agar. Each concentration (200 µL) and controls were poured into wells, and plates were incubated at 36 ± 1 °C for 24 h. Amoxycillin (1.00 mg/mL) and MeOH (70%) were taken as the positive and negative controls, respectively.26,34,35
Antifungal Sensitivity Test (AFST)
AFST was performed against
Determination of Minimum Inhibitory Concentration (MIC) and Minimum Lethal Concentration (MLC)
MIC and MLC of each test sample were evaluated using the following method. A small piece of agar from an inhibitory zone of each concentration was transferred to a freshly prepared Nutrient Agar plate. Then plates were incubated at 36 ± 1 °C, for 24 h, and the growth of each test microorganism was observed after 24 h incubation. The lowest concentration of the test sample, which shows 100% growth inhibition of each microorganism, was taken as the MLC, and the concentration that shows an inhibitory zone, and showed an ability to revive once transferred and incubated on a new agar plate was taken as the MIC. All the concentrations were replicated three times.
Statistical Analysis
Each parameter was tested in triplicate. The values were expressed as the mean ± standard deviation (SD). The Tukey one-way analysis (ANOVA) was used to determine the significant differences among all columns against the control, and a
Conclusion
The Asamodagum Spirit (Sid.AS) was characterized by thymol as its major compound, alongside minor constituents such as long-chain hydrocarbons and fatty acids. Notably, Sid.AS exhibited substantial antioxidant activity as demonstrated by both DPPH and ABTS assays, comparable to the positive control (BHT). Moreover, Sid.AS displayed remarkable inhibitory effects against lipase, α-glucosidase, α-amylase and urease enzymes. In addition to its enzyme inhibition properties, Sid.AS demonstrated potent antimicrobial activity, yielding significant inhibition zones with low MIC and MLC values against tested microorganisms. This suggests its potential as a natural alternative for preventing food poisoning, owing to its antibacterial and antifungal effects. Overall, the findings of this study emphasize the efficacy of Sid.AS in treating gastrointestinal disorders. Furthermore, its promising anti-lipase and anti-diabetic activities suggest its potential for managing obesity and diabetes. Sid.AS emerges as a promising candidate for therapeutic use, offering a multifaceted approach to addressing various health concerns. Additionally, investigating the nutritional and quality parameters according to Ayurvedic standards in Sri Lanka ensures a comprehensive understanding of Sid.AS's potential benefits and safety profile align with traditional practices and regulatory requirements. This multidimensional approach contributes significantly to evaluating Sid.AS's overall efficacy and safety, paving the way for its potential utilization in healthcare or wellness applications.
Footnotes
Acknowledgement
The Authors would like to thank Sujanthe Mauran, Ganushika Mahaliyadda, Hansika Gunathilaka, Dulani Siriwardana and Ureshika Gedara for the technical support to conduct this research and Amila Kannangara, Department of Chemistry, University of Kelaniya for providing GC-MS data. The research was supported by a project for collaboration between the R&D of Industry and the University (HI/UoK 01).
Authorship Contribution Statement
Dilan Jayawantha; performed bioassays, analysis of GC-MS results and preparing the draft manuscript.
Priyani Ashoka Paranagama: Supervision, conceptualization, Methodology, designed, reviewed and editing, visualization.
Lankani Hettigoda: Supervision, Conceptualization, Project administration, review, and editing.
Tharindya Dinethri Mudalige: performed the study, formal analysis, writing – original draft, methodology.
All authors read and approved the manuscript.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
