Abstract
Background
Objectives
The ethanol extracts of these plants were analyzed for qualitative and quantitative phytochemical content, antioxidant potential, and anti-tyrosinase activity.
Methods
The Folin-Ciocalteu technique was used to measure the total phenolic content, and the aluminum chloride method was used to assess flavonoid content.In vitro antioxidant activity was evaluated via 2,2- diphenyl-1-picrylhyrazyl (DPPH), nitric oxide (NO), and hydrogen peroxide (H2O2) assays. A mushroom tyrosinase inhibition assay was employed to assess anti-tyrosinase activity. Molecular docking was performed using the tyrosinase enzyme (PDB ID: 2Y9X) for the five selected phytoconstituents of
Results
The ethanolic extract of
Conclusion
These findings highlight the significant tyrosinase inhibitory potential of
Introduction
Free radicals are unstable reactive chemical species (atoms, molecules, or compounds) with one unpaired electron, seeking stability by pairing with other atoms or molecules. 1 These include hydroxyl radicals (OH−), superoxide anions (O2−), hydrogen peroxide (H2O2), singlet oxygen, hypochlorite radicals, nitric oxide radicals (NO.), and other lipid peroxides as well as reactive oxygen and nitrogen species (ROS or NOS). These free radicals cause biochemical damage to cells and tissues through chain reactions, leading to diseases such as aging, liver disease, inflammation, renal failure, cardiovascular and neurodegenerative diseases. 2
Tyrosinase is crucial for the biosynthesis of melanin, transforming L-tyrosine to levodopa (L-DOPA) and L-DOPA to dopaquinone, the precursor of melanin pigment. 3 Melanin provides color to the skin and hair, but defects in its synthesis can cause dermatological disorders such as hyperpigmentation.4,5 Targeting tyrosinase is an important to reduce melanocyte function and address pigmentation issues, which have cosmetic significance. Tyrosinase is involved in neuromelanin formation in the substantia nigra. 5 High neuromelanin levels are linked to dopamine neurotoxicity and neurodegenerative disorders, such as Parkinson's disease. 6 Consequently, research in cosmetics and medicine has focused on tyrosinase inhibitors, with ethnomedicinal plants being natural sources of effective compounds. Most tyrosinase inhibitors have phenolic structures that interact with the active site of the enzyme. 7
Natural products are important therapeutic agents, comprising approximately a quarter of the treatments for various diseases. Melanogenesis, catalyzed by tyrosinase, leads to melanin pigment formation, which protects the skin from UV radiation and pigmentary disorders. Therefore, by blocking tyrosinase activity, natural products and medicinal plant extracts can be regarded as alternative therapies for hyperpigmentary skin conditions. Molecules derived from natural products are emerging as promising therapeutic candidates for pharmaceutical and cosmeceutical applications. Common Nepalese medicinal plants such as
Materials and Methods
Chemicals and Reagents
Dimethyl sulfoxide (Fischer Scientific, India), distilled water (Thomas Scientific, India), and Absolute ethanol (Changshu Hongshen Fine Chemical Co. Ltd, China), and DPPH (Wako Pure Chemical Co. Ltd, Japan), Aluminum chloride (Fischer Scientific, India), Ascorbic Acid (HiMedia Laboratories Pvt.Ltd, India), Folin Reagent (Sd Fine-Chem Ltd, India), and Gallic Acid (Wako Pure Chemical Co. Ltd, Japan), Hydrogen Peroxide Solution (Fischer Scientific, India), N-(1-Naphthyl) ethylenediamine dihydrochloride (Loba Chemie Pvt. Ltd, India), and Quercetin (Himedia Laboratories Pvt. Ltd, India) were used. For this investigation, only analytical grade materials and reagents were used.
Plant Material
A taxonomist at the National Herbarium and Plant Research Laboratory, Nepal, identified fresh
Preparation of Plant Extracts
Cold maceration was employed for the extraction of different parts of the three selected medicinal plants (300 g each) using absolute ethanol as the solvent. A clean muslin cloth and Whatman grade 1 filter paper were used to filter the final extract. A rotary evaporator (Heidolph, Germany) was used to evaporate the liquid extract at 250–175 mbar pressure, 90 revolutions per minute, a water bath temperature of 40 °C, and a chilling temperature of 5 °C until the solvent was fully evaporated. The concentrated filtrate was stored in a glass vial in a vacuum desiccator until further use.
Qualitative Phytochemical Analysis
The presence of plant secondary metabolites in the extract was determined using standardized testing.8,9
Quantitative Phytochemical Analysis
Total Phenolic Content
The total phenolic content of the ethanolic extract of certain plants was determined using the Folin-Ciocalteu technique.10,11 In summary, 1 mL of Folin-Ciocalteu reagent, 1 mL of 1 mg/mL plant extract, and 5 mL of distilled water were mixed together. 1 mL of Sodium carbonate (10% w/v) was added, and shook for five minutes. After allowing the mixture to stand for 60 min, the absorbance was measured at 725 nm using a UV-Visible spectrophotometer (Model: Cary 60, Agilent, United States).Gallic acid (25-500 mg/L) served as the reference to quantify the overall content of phenolic compounds, and the findings were represented as gallic acid equivalents in mg for each dry weight of the extract in grams.
Total Flavonoid Content
The technique outlined by the authors was used to measure the total flavonoid content.12,13 The flavonoid content of the selected plant extracts was measured using aluminum chloride colorimetric technique. Briefly, 1 mL of 1 mg/mL plant extract was combined with 4 mL of distilled water. After five minutes, 0.3 mL of a 20% aluminum chloride solution was added, and then 0.3 mL of 5% sodium nitrite solution. Two milliliters of one molar sodium hydroxide were added to the mixture after it had stood for six minutes. Using quercetin as a reference, the absorbance was measured at 510 nm using a UV-Visible spectrophotometer. The overall flavonoid contents was expressed as milligrams of equivalent quercetin per gram of dry weight of the extract.
Antioxidant Activity
The antioxidant activity of a medicinal plant extract was assessed by scavenging the free radicals 2,2-diphenyl-1-picrylhydrazyl (DPPH), nitric oxide radicals (NO), and hydrogen peroxide (H2O2).
DPPH Scavenging Activity
The author's methodology was used to assess the capacity of the plant extract to scavenge free radicals of DPPH.14,15 Basically, two milliliters of the plant ethanolic extract (0.1-100 µg/mL) was combined with two milliliters of DPPH solution (60 µM). For half an hour, the combination was left to react at room temperature in the dark.
A decrease in the purple hue of the DPPH solution was observed at 518 nm using a UV-Visible spectrophotometer. As a positive control, ascorbic acid was dissolved in ethanol to equalize the content of the extract.
The negative control was a test solution that did not contain plant extracts.
To determine DPPH radical scavenging capability, the following formula was used:
The antioxidant activity of the extract was calculated using a calibration curve and is shown in relation to the IC50 after the inhibition percentage versus extract concentration was plotted.
Nitric Oxide Scavenging Activity
The capacity of the plant extract to scavenge free radicals was evaluated using the nitric oxide (NO) radical scavenging test, which was previously reported in a research.14,16 Specifically, 1 mL of the plant extract was combined with 1 mL of sodium nitroprusside solution at different doses of plant extract (0.1-100 µg/mL), and the mixture was then incubated for 2.5 h at 29 °C. After the reaction mixture was incubated, 2 mL of Griess reagent was added and a UV-Visible spectrophotometer was used to detect the optical density at 548 nm.
The antioxidant activity of the plant extract was determined by comparing its NO radical inhibition to that of the curcumin benchmark.
Hydrogen Peroxide (H2O2) Scavenging Activity
The authors’ approach was employed to evaluate the ability of the extract to scavenge hydrogen peroxide.
14
In summary, 0.6 mL of 40 mM hydrogen peroxide produced in 0.1 M phosphate buffer (pH 7.4) was mixed with 1 mL of the extract (100 µg/mL). Following a 10-min incubation period, the mixture was diluted with buffer to a final volume of 3 mL. Using a buffer as a blank, a UV-Visible spectrophotometer was used to determine the optical density at 230 nm. The test sample was not included in the control sample, which was the same as that of the reaction combination. The following formula was used to determine the scavenging activity:
Tyrosinase Inhibitory Activity
Preparation of Phosphate Buffer Solution
Following the Indian Pharmacopeia 2010, a 6.8 pH phosphate buffer solution was prepared. This required additional water to obtain a final volume of 500 mL after combining 56 mL of 0.2 M sodium hydroxide and 125 mL of 0.2 M potassium dihydrogen phosphate.
Preparation of Sample and Kojic Acid Solution for Assay
To prepare for the tyrosinase inhibitory assay, 0.2 milliliters of each sample's DMSO solution were used, and the standard tyrosinase inhibitory compound, kojic acid, was dissolved in 100% DMSO to create a 1000 μg/mL solution. The plant extracts were dissolved in 100% DMSO to prepare a 1000 μg/mL sample solution.
Preparation of L-DOPA Solution and Mushroom Tyrosinase Solution
Phosphate buffer solution (pH 6.8) was used to dissolve 30.0315 mg of L-DOPA (MW: 200.21) to create a 1.5 mM solution. The final volume was adjusted to 100 mL.To create the mushroom tyrosinase solution, 6.85 mg of the mushroom tyrosinase enzyme (activity: 730 U/mg) was dissolved in phosphate buffer solution (pH 6.8), and the volume was changed to 5 mL to produce a 1000 U/mL solution.
Assay for Tyrosinase Inhibitory Activity
The technique used to measure the inhibitory activity of mushroom tyrosinase was slightly modified from that used in a previous study. In a test tube, two milliliters of 1.5 mM L-DOPA solution, two milliliters of plant sample solution (1000 μg/mL), and one milliliter of phosphate buffer solution (pH 6.8) were combined for this experiment. The mixture was then incubated for ten minutes at room temperature.
17
After adding 0.2 mL of mushroom tyrosinase solution (1000 U/mL), a UV-Visible spectrophotometer was used to detect the absorbance at 475 nm. The proportion of tyrosinase inhibition was determined using the following formula. A = the absorbance difference of the control sample measured at 30 and 60 s. B = The test samples’ absorbance difference as seen at 30 and 60 s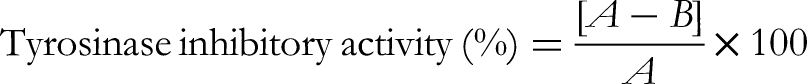
A test solution without the sample served as the control. A positive control solution containing 1000 μg/mL kojic acid was used.
Molecular Docking

Two-dimentional structure of selective phytocontituents from
Using AutoDock Vina software, molecular docking was performed inside a predetermined region of tyrosinase, which was represented by a grid box of 60 × 60 × 50 sizes with coordinates of x = -8.888, y = -25.444, and z = -40.793, spaced 0.375 Å to include residues from the catalytic site. Using BIOVIA Discovery Studio Visualizer 2020, the predominant binding complex (greater negative value in kcal/mol) was chosen, and its interacting amino acid residues and bond nature were evaluated (Figure 2).

3D structure and grid box enclosing the docking site (catalytic site) of target enzyme tyrosinase.
Statistical Analysis
The average (mean) and standard error of the mean (SEM) of the results are shown in a tabular form. Each
Results
The present study assessed the ethanolic extracts of three medicinal plants from Nepal in terms of phytochemical analysis, calculation of total phenolic and flavonoid content, in vitro antioxidant activity, and in vitro anti-tyrosinase activity. Phytochemicals have garnered significant interest because of their wide range of applications. Medicinal plants are recognized as reservoirs of drugs in traditional medicine systems, allopathic medicine systems, folk medicine, food supplements, intermediates for pharmaceuticals, nutraceuticals, and chemical compounds for various synthetic modern medicines. 18
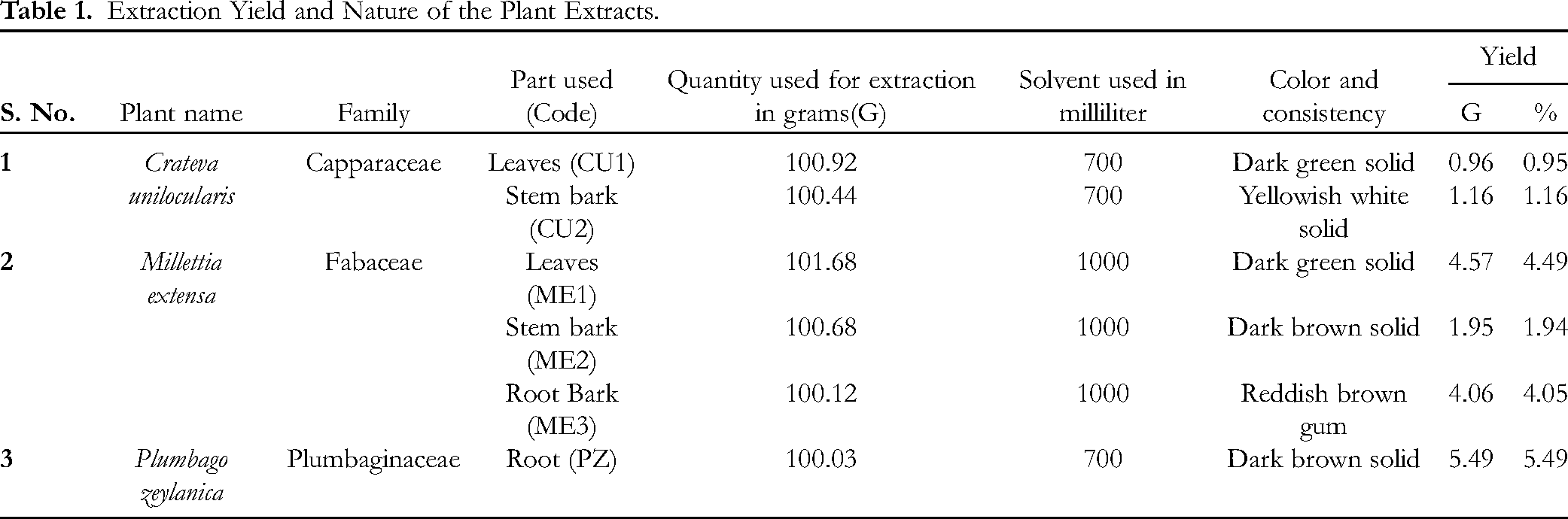
Extraction Yield and Nature of the Extracts
As shown in Table 1, different plant parts were used for extraction in this study.
Extraction Yield and Nature of the Plant Extracts.
Phytochemical Analysis
Phytochemical Screening of Ethanolic Extract of Plant Samples
The extracts of the three distinct plants showed the presence of alkaloids, terpenoids, flavonoids, carbohydrates, and amino acids, but glycosides were absent. This was determined using qualitative phytochemical analysis. Saponin, Tannin, and phenolic compounds were absent in both the stem bark and leaf extracts of
Phytochemical Screening of the Ethanolic Extracts of Plant Samples.

Note: (+) presence; (−)absence.
Total Phenolic and Flavonoid Content
The Folin-Ciocalteu (FC) reagent method was usedto evaluate the total phenolic content (TPC). The calibration curve of the gallic acid standard was drawn as illustrated in Figure 3 and 4 at seven different concentrations ranging from 25 to 500 mg/L.The equivalent TPC was calculated based on the standard regression line for gallic acid (y = 0.006x + 0.122; R² = 0.992) (Table 3).

Gallic acid standard calibration curve for calculation of total phenol content.

Quercetin standard calibration curve for calculation of total flavonoid content.
Total Phenolic and Flavonoid Content of Plant Samples.

All data are expressed as mean ± standard error of mean (SEM) (n = 3).
The flavonoid content in the plant extracts was expressed as milligram quercetin equivalents (QE). A quercetin standard calibration curve was constructed at seven concentrations ranging from 25 to 500 mg/L, as shown in Figure 4. The total flavonoid content was calculated based on the standard regression line for quercetin (y = 0.000 × 0.005; R ² = 0.993) (Table 3).
In-Vitro Antioxidant Activity
DPPH Free Radical Scavenging Activity
For the DPPH radical, all ethanolic extracts showed proton-donor activity. The
Percentage DPPH Radical-Scavenging Activity of Plant Extracts at Different Concentrations.

All data are expressed as mean ± standard error of mean (SEM) (n = 3).
NO Free Radical Scavenging Activity
Plant extracts demonstrate antioxidant activity by scavenging the nitrite radical produced by sodium nitroprusside at physiological pH in an aqueous medium by competing with oxygen. NO radical-scavenging activities of the plant samples are shown in Table 5. With an increase in concentration, all ethanolic extracts of the plant samples showed an increase in antioxidant activity. The one with the greatest NO radical scavenging activity among them was found in
Percentage NO Radical-Scavenging Activity of Plant Extracts at Different Concentrations.
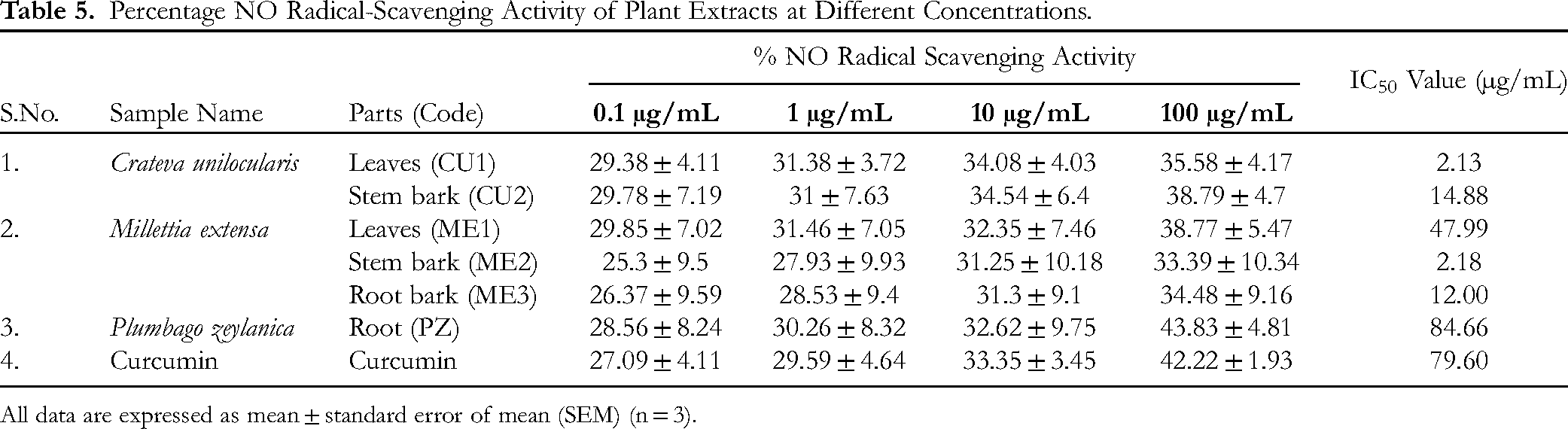
All data are expressed as mean ± standard error of mean (SEM) (n = 3).
Hydrogen Peroxide Radical Scavenging
In hydrogen peroxide radical scavenging activity,
Hydrogen Peroxide Radical Scavenging Activity.

All data are expressed as mean ± standard error of mean (SEM) (n = 3).
Mushroom Tyrosinase Inhibitory Activity
The enzyme tyrosinase limits melanin synthesis. Melanin protects the skin from UV rays; however, too much melanin can cause age spots and melasma. Therefore, tyrosinase inhibitors are crucial for reducing skin abnormalities caused by excess melanin.19,20 L-tyrosinase is transformed into dopaquinone by tyrosinase, which is then transformed into dopachrome. The amount of dopachrome produced in the presence of the test chemicals was measured using the tyrosinase inhibitory assay. 21
After testing for tyrosinase inhibitory action, ethanolic extracts from various plants showed the highest levels of tyrosinase inhibitory activity, as shown in Table 7.
Mushroom Tyrosinase Inhibitory Activity.

All data are expressed as mean ± standard error of mean (SEM) (n = 3).
Molecular Docking
In comparison to ordinary Kojic acid (−5.6 kcal/mol), all of the chosen phytoconstituents had a substantial binding affinity ranging from −8.3 to −9.9 kcal/mol, as shown in Table 8. auriculatin showed the highest binding affinity with tyrosinase enzyme via interacting to its amino acid residue including GLN 307, ASP 354, ASP 357, TRP 358, LYS 376 and LYS 379 through conventional hydrogen bond, carbon hydrogen, pi-Anion and pi-Alky bond as illustrated in Table 8, Figure 5.

Two-dimentioanal (2D) molecular docking interaction mode of phytoconstituents from
Binding Energy and Amino Acid Interactions on Phytoconstituent Docking Against Tyrosinase.

Discussion
Phytochemicals have recently gained significant attention owing to their broad applications in traditional and modern medicine, food supplements, and pharmaceuticals, among others.
18
Consistent with earlier research, alkaloids, terpenoids, flavonoids, carbohydrates, and amino acids were found in extracts from the four medicinal plants using qualitative screening.22,23 Plants are rich in phenolic chemicals produced from phenylalanine and tyrosine.
24
The phenolic and flavonoid contents of the ethanolic extract of
One common type of phenolic chemical is flavonoids, which have a variety of biological actions, such as anti-inflammatory, antibacterial, antioxidant, and anticancer effects.26,27 Our study corroborates that higher phenolic and flavonoid contents correlate with enhanced free radical scavenging activity, as observed in DPPH and nitric oxide assays.28,29 However, the correlation was not significant for hydrogen peroxide scavenging, likely due to structural differences affecting the antioxidant potential.30,31
The evaluation of antioxidant potential across various assays underscores the complexity of antioxidant mechanisms and assay methodologies.
25
The ethanolic extracts of
A single antioxidant assay is insufficient to assess the antioxidant capacity of plant extracts. Furthermore, the assay principles and experimental procedures used in different free radical scavenging tests differ. Examples include the DPPH free radical scavenging assay, NO radical scavenging assay, and hydrogen peroxide assay method. The capacity of antioxidants, time factors, diverse methodologies, and experimental settings are all related to the ability of a material to scavenge free radicals. 33 In addition, natural antioxidants are polar in nature, such as phenolics and flavonoids, or polar compounds, such as vitamin E. Antioxidants possess free radical scavengers via hydrogen or electron donating mechanisms. 34
The DPPH test is a commonly employed technique for evaluating the antioxidant capacity of plant extracts because of its short analysis time. 35 DPPH is a stable, nitrogen-centered free radical that produces a violet color in ethanol. The addition of the fractions in a concentration-dependent manner reduced this to a yellow-colored product (diphenylpicryl hydrazine). The quantity of accessible hydroxyl groups is associated with a decrease in DPPH molecules.35,36 All extracts had a positive correlation with the total phenolic and flavonoid contents and exhibited a significantly greater inhibition percentage (stronger hydrogen-donating ability).
Antioxidants demonstrate radical scavenging in the DPPH assay, which may be related to their ability to donate hydrogen.25,37The capacity for radical scavenging is essential for preventing several diseases, including diabetes, cancer, neurological disease, and liver diseases. Comparing the ethanolic extracts of
Polyphenol content and ascorbic acid scavenge DPPH radicals through their hydrogen-giving capacity.25,38 The findings of this study indicate that the ethanolic extracts of
Nitric oxide (NO) or reactive nitrogen species are responsible for altering the structural and functional nature of many cellular components. The nitrite radical generated by sodium nitroprusside (2.5 h) in phosphate-buffered saline at 25 °C was scavenged by the antioxidant component from the test samples. Compared to curcumin, the standard antioxidant, the ethanolic extract of
Hydrogen peroxide is naturally present in the air, water, plants, microorganisms, human body, and food
42
at low concentrations. Hydrogen peroxide itself is not reactive, but it can sometimes be toxic to cells. In cells, it may trigger the formation of hydroxyl radicals (●OH) when decomposed into oxygen and water.
43
Hydroxyl radicals can interact with DNA and cellular proteins to initiate lipid peroxidation, which is associated with cancer formation and different disease conditions.44,45 The hydrogen-donating ability of the plant extracts was measured using hydrogen peroxide radicals as the hydrogen acceptor. The hydrogen peroxide test revealed that the ethanolic extract of
In recent years, there has been a lot of interest in natural anti-tyrosinase medicines because they can provide tremendous skin whitening effects. 46 Plant extracts contain active phytoconstituents with antioxidant and anti-tyrosinase activities, such as polyphenols, alkaloids, flavonoids, terpenoids, saponins, and tannins. These results are consistent with previous research demonstrating that these phytochemical have anti-tyrosinase action.47–49
Hyperpigmentation is a common skin problem prevalent in middle-aged and elderly individuals. Cosmetically, it has great implications, as it detracts individuals from both appearance and quality of life. Individuals with hyperpigmentation feel ugly and unattractive, which makes them socially incompetent and lowers their confidence. 50 Therefore, different methods have been applied to prevent hyper-pigmentation, involving the wide use of various cosmetic products. The cosmetic products currently used consist of different ingredients that enhance skin lightening, among which depigmenting agents are the most commonly used. Depigmenting (hypo-pigmenting) agents help lighten the skin through various mechanisms. An ideal depigmenting agent should have a potent, rapid, and selective depigmenting effect on hyperactivated melanocytes, act at one or more steps of the pigmentation process, and should not have any side effects. 46 Mushroom tyrosinase is a popular depigmentation technique used to examine tyrosinase inhibition.
Molecular docking is an essential tool for predicting the binding site, affinity, and manner of interactions between drugs and proteins. It helps uncover the binding process and makes it easier to create and find new therapeutic compounds.51–53 The docking methodology was first validated using docking and re-docking approaches. When the redocked ligand was superimposed with an earlier ligand, an RMSD value of 0.88 Å (< 2) was observed, indicating that the docking process was validated. Insights into the binding location, type of bonding, bond length, and corresponding binding energy (kcal/mol) were obtained from docking visualization in three dimensions. Considering the noteworthy anti-tyrosinase activity of
This study highlights the diverse bioactive properties of medicinal plant extracts, suggesting their potential as natural antioxidants and tyrosinase inhibitors. Future studies should explore synergistic effects and clinical applications to effectively harness their therapeutic benefits. Since this study primarily examined ethanol extracts, it might not fully capture the variety of phytochemicals seen in other solvent extracts. Furthermore, although molecular docking validates the
Conclusion
In conclusion, the extract from
Footnotes
Abbreviations
Acknowledgements
The authors are grateful to Pokhara University for offering the research facilities and a full postgraduate scholarship.
Author Contributions
Data Availability Statement
All study data and materials will be made available to the corresponding authors upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not required for this study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Statement of Human and Animal Rights
Our study did not include any studies that involved human or animal subjects.
Statement of Informed Consent
There were no human subjects in this study, and informed consent was not required.
