Abstract
Objective
This study aimed to characterize the phytochemical profile, mineral composition, and biological activities of aqueous and ethanolic extracts prepared from the aerial and underground parts of Cerinthe major L.
Methods
Phytochemicals and mineral elements were analyzed using UPLC-ESI-MS/MS and ICP-OES, respectively. Antioxidant activity was evaluated by DPPH●, ABTS●+, β-carotene–linoleic acid, and reducing power assays, while anti-cholinesterase, anti-urease, anti-inflammatory, antimicrobial, antifungal, cytotoxicity, and in vitro sun protection factor (SPF) were also assessed.
Results
The aqueous extract showed the strongest butyrylcholinesterase inhibition (IC50 = 58.80 ± 1.43 µg/mL), whereas the ethanolic extract exhibited higher acetylcholinesterase inhibition (IC50 = 665.87 ± 13.01 µg/mL). Antibacterial activity against Escherichia coli ATCC 25922 was weak, requiring relatively high extract concentrations to inhibit growth. In the yeast cytotoxicity assay, neither extract showed detectable toxic effects on Saccharomyces cerevisiae within the tested concentration range, suggesting favorable biocompatibility in this model. ICP-OES analysis revealed that Cerinthe major extracts are rich in macro- and microelements, with several essential elements present at levels close to FAO dietary recommendations, highlighting their nutritional relevance. Cerinthe major provided moderate UV protection, with SPF values ranging from 32.82 ± 0.17 to 35.78 ± 0.12, and displayed strong antioxidant potential in all assays.
Conclusion
The ethanolic extract of Cerinthe major, characterized by a richer LC-MS/MS phytochemical profile than the aqueous extract, together with the high levels of macro- and microelements revealed by ICP-OES, suggests that this species is a promising source of minerals and bioactive compounds with potential nutraceutical and pharmaceutical applications.
Keywords
Introduction
Medicinal plants have been utilized since prehistoric times as vital resources for the prevention and treatment of a wide range of diseases and health conditions. Although the popularity of herbal medicines waned in Western societies with the advent of synthetic pharmaceuticals over the past two centuries, the global resurgence of interest in plant-based therapies has marked a significant shift towards natural and holistic healthcare approaches. 1 The World Health Organization (WHO) estimates that more than 80% of the global population depends largely or exclusively on medicinal plants for their primary healthcare needs. 2 This reliance underscores the critical importance of ethnopharmacological research to validate traditional uses and explore new therapeutic agents derived from plants. The Mediterranean region, including Algeria, represents one of the world's richest biodiversity hotspots, home to thousands of plant species with substantial endemism and ethnobotanical relevance.3,4 This region harbors around 25,000 vascular plant species, nearly 50% of which are endemic, making it one of the top biodiversity hotspots globally. Algeria alone hosts over 3000 plant species, including hundreds with recognized medicinal and traditional uses, particularly within the Lamiaceae, Asteraceae, and Fabaceae families.
These plants are used extensively in traditional medicine, reflecting a rich ethnobotanical heritage rooted in Berber and Arab healing traditions. However, threats from urbanization, climate change, and overexploitation have made conservation efforts increasingly urgent.5–7 Cerinthe major L., belonging to the Boraginaceae family, is a perennial herbaceous plant native to the Mediterranean basin and Algeria in particular. 8 Traditionally, it has been used by local communities for its medicinal and nutritional properties, although it remains relatively understudied in terms of its chemical constituents and biological activities. 9 The genus Cerinthe (Boraginaceae) includes species producing various useful compounds such as flavonoids and phenolic acids, including rosmarinic acid, 10 as well as pyrrolizidine alkaloids and terpenoids that are widely documented in Boraginaceae species. 11 These phytochemicals have demonstrated antioxidant, antimicrobial, anti-inflammatory, and enzyme-inhibitory activities.10,11 These 1,2-unsaturated pyrrolizidine alkaloids (PAs) are known to alkylate DNA, leading to hepatotoxicity, genotoxicity, and carcinogenicity. A 2014 open-access review analysed PAs across Boraginaceae genera and emphasized their structure, distribution, toxicity mechanisms, and risks to food safety—underscoring the necessity of combining safety profiling with bioactivity evaluation.12,13
Phytochemical profiling of medicinal plants has emerged as a central approach in modern natural products research, as it provides a comprehensive picture of the small-molecule constituents responsible for therapeutic effects. Plant extracts typically contain hundreds of structurally diverse secondary metabolites, including phenolic acids, flavonoids, coumarins, lignans, alkaloids, saponins, and terpenoids, many of which occur at low concentrations and in complex mixtures that cannot be resolved by classical colorimetric tests alone. Advanced hyphenated techniques, particularly liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS), are therefore indispensable for resolving this chemical complexity. In LC-MS/MS, chromatographic separation on reversed-phase or HILIC columns reduces matrix interferences and allows compounds with similar masses to be distinguished based on retention behavior, while high-sensitivity mass spectrometers provide accurate mass measurements and characteristic MS/MS fragmentation patterns that enable tentative or unambiguous identification of individual metabolites, even in the absence of authentic standards. Targeted LC-MS methods can be used to precisely quantify known bioactive molecules, whereas untargeted metabolite profiling and metabolomics workflows capture a broad range of detected features, enabling comparative studies between species, plant organs, extraction solvents, or treatment conditions and facilitating the discovery of chemomarkers associated with specific biological activities. In parallel, inductively coupled plasma optical emission spectrometry (ICP-OES) facilitates accurate and simultaneous analysis of both major and trace mineral elements in plant samples.14,15 Minerals such as potassium, calcium, magnesium, and trace elements contribute significantly to the nutritional and therapeutic value of medicinal plants, often enhancing their biological efficacy. 16 Moreover, the quantitative extraction of these mineral and bioactive constituents is strongly influenced by the choice of solvent, which can differentially solubilize polar and less polar compounds from the plant matrix.
In addition to evaluating the beneficial biological activities of Cerinthe major extracts, it is also important to obtain preliminary information about their safety at the cellular level. Saccharomyces cerevisiae is widely used as a simple eukaryotic model organism for toxicity and cytotoxicity assessment because it shares many conserved cellular processes with higher eukaryotes, is easy to culture, and allows rapid, low-cost screening of toxic effects without ethical concerns. In this context, we also performed an in vitro cytotoxicity (growth sensitivity) assay on S. cerevisiae to gain initial insight into the biocompatibility of the aqueous and ethanolic extracts of Cerinthe major.
Several studies emphasize the integral role of natural antioxidants from plants in mitigating oxidative stress, which is implicated in the pathogenesis of chronic diseases, including cancer, cardiovascular disorders, and neurodegenerative conditions. Additionally, antimicrobial resistance poses a major global health threat, prompting research into novel plant-derived antimicrobials as potential alternatives. 17 In this context, evaluating the antimicrobial properties of Cerinthe major extracts is both timely and necessary. This study aims to thoroughly analyse the chemical makeup and mineral content of Cerinthe major collected from Algeria and to test its biological activities in the lab, such as its ability to fight oxidation, bacteria, and inhibit enzymes. By combining chemical and biological information, this research aims to confirm the traditional uses of Cerinthe major, encourage its responsible use, and investigate its potential as a source of beneficial compounds for medicine and health products.
To the best of our knowledge, this is the first comprehensive study to characterize the phenolic profile of Cerinthe major L. by LC–MS/MS, together with an integrated evaluation of its antioxidant, enzyme-inhibitory, anti-inflammatory, antimicrobial and photoprotective activities, while also addressing safety-relevant pyrrolizidine alkaloids. This combined phytochemical–bioactivity–safety approach provides novel insights into the medicinal potential and risk–benefit profile of this traditionally used yet understudied Boraginaceae species.
Materials and Methods
Extraction of Secondary Metabolites
Plant Material
In April 2022, when the plants were blooming, Cerinthe major L. plants were taken from their native habitat in Bougaa, Setif, Algeria (36°19′59.9″ N, 5°08′29.5″ E). The roots, stems, leaves, and flowers of the whole plant were picked from the field. We used the usual botanical keys given by Quezel and Santa 18 to figure out what kind of plant it was, and the Center for Scientific and Technical Research on Arid Regions’ Dr Halis Yousef (CRSTRA) confirmed our findings. A voucher specimen with the code (202204Bo/CerMaj) was put in the local herbaria. The plants were gently washed with distilled water to get rid of any dirt that was still on them. They were then dried in the shade at room temperature and then ground up using an electric mixer. Before it was used again, the powdered stuff was kept in dry, dark places in paper bags that were sealed shut.
Preparation of Extracts
Ethanol extraction was performed by soaking 100 g of dry plant powder in one liter of ethanol (100%) for 72 h at room temperature. After 72 h of contact, the process was repeated three times, the ethanol mixture was filtered, and it was concentrated at 40 °C under reduced pressure using a rotary evaporator to obtain a crude ethanol extract (EE), 19 Ethanol was selected as extraction solvent due to its intermediate polarity, safety, and ability to solubilize a broad spectrum of phytochemicals (phenolics, flavonoids, and moderately non-polar constituents), while remaining acceptable for food and pharmaceutical applications 20 .Aqueous extraction: Aqueous extraction was performed by macerating 100 g of dry plant powder (Sétif region, Algeria) in 1 L of distilled water for 24 h at room temperature (approximately 22-25 °C). At the beginning of the extraction, the mixture was heated for about 5 min in a covered vessel in order to limit microbial growth and potential fermentation. After cooling to room temperature, the maceration was continued in closed, clean glass containers, with occasional stirring. After 24 h, the mixture was filtered and the procedure was repeated three times under the same conditions. The combined aqueous filtrates were then frozen and lyophilized to obtain a crude aqueous extract (AE). 21
Determination of Extraction Yield
The extraction yield was expressed as a percentage of the weight of the obtained extract relative to the weight of the dried plant material,
22
as described in the equation (1).
Inductively Coupled Plasma ICP-OES
Sample Digestion for ICP-OES Mineral Analysis23–26:
Digestion: A 500 mg aliquot of powdered Cerinthe major was transferred to a 200 mL flask. Acid Addition: 3 mL of 65% HNO₃ and 3 mL of 37% HCl (Sigma Aldrich, Germany) were added to the flask. Agitation & Heating: The mixture was agitated for 90 min and then heated to 105 °C until complete dissolution to liquid was achieved. Dilution & Preparation: The digest was diluted to 50 mL with distilled water, transferred to a new tube, and allowed to settle. Analysis: The resulting supernatant was analyzed by ICP-OES in triplicate for each sample to determine mineral concentrations. The mineral composition was determined by ICP-OES after sample mineralization, and all elements were quantified as aqueous ionic species rather than as neutral elemental forms.
Elemental analysis was performed using an ICP-OES spectrometer (PlasmaQuant 9000, Analytik Jena, Jena, Germany).
Liquid Chromatography-Mass Spectrometry Analysis Conditions LC-MS-MS
Ultra-Fast Mass Spectrometry (UFMS) technology and a Nexera XR LC-20AD binary pump. Chromatographic separation was performed on a Restek Ultra C18 column (150 × 4.6 mm, 3 µm particle size), using a mobile phase composed of solvent A (water containing 0.1% formic acid) and solvent B (methanol) at a flow rate of 0.2 mL/min, with an injection volume of 5 µL. The gradient elution program followed this sequence: 98% A from 0.0 to 0.2 min, decreased to 25% A from 0.2 to 2.5 min, then to 0% A from 2.5 to 4.0 min, maintained at 0% A until 7.0 min, returned to 98% A at 7.1 min, and held until 12.0 min and operated in multiple reaction monitoring (MRM) mode. Mass spectrometric detection was performed using an electrospray ionization (ESI) source under the following conditions: collision-induced dissociation (CID) gas pressure, 230 kPa; conversion dynode voltage, −6.00 kV; desolvation line (DL) temperature, 250 °C; nebulizing gas flow, 3.00 L/min; heat block temperature, 400 °C; and drying gas flow rate, 10 L/min. 27 In this study, LC–MS/MS was used for qualitative metabolite profiling only; individual compounds were not quantified.
Phytochemical profiling was carried out on an LC-MS/MS system (LCMS-8040 triple quadrupole, Shimadzu, Kyoto, Japan) equipped with an electrospray ionization (ESI) source.
Assessment of Total Phenolics, Flavonoids Compounds
Total Phenolic Content (TPC)
The Folin-Ciocalteu method was used to measure TPC spectrophotometrically. 28 In short, 100 μL of Folin-Ciocalteu reagent (diluted ten-fold v/v) and 75 μL (75 g/L) of sodium carbonate were combined with 20 μL of the extract. The absorbance measurement at 765 nm was recorded using a 96-well microplate reader (Perkin Elmer EnSpire, Singapore) during a two-hour incubation period in the dark. Micrograms of gallic acid equivalents per milligram of extract (µg GAE/mg extract) were used to express the results. 29
UV–Vis absorbance was measured using a microplate spectrophotometer (EnSpire, PerkinElmer, Singapore)
Total Flavonoid Content (TFC)
The colorimetric method of Topcu et al (2007) was used to measure TFC. 30 10 μL of aluminum nitrate (10% w/v), 10 μL of potassium acetate (1 M), and 130 μL of methanol were combined with 50 μL of extract. The absorbance was determined spectrophotometrically at 415 nm following a 40-min incubation period. The results were reported as micrograms of quercetin equivalents per milligram of extract (µg GAE/mg extract), with quercetin serving as the reference ingredient. 31
Antioxidant Activity
The antioxidant power of plant products is likely to be viewed in multiple ways due to the complexity of phytochemicals. We used DPPH●, ABTS●+, β-carotene and reducing power tests to find out how well Cerinthe major L. fought free radicals. The tests were done, and the results are shown in Table 1. This methodology makes the data more complete, easier to understand, and more relevant to other chemicals. The A0.5 and IC50 values of the extracts’ antioxidant activity were determined using linear regression analysis. The activity effectiveness goes down as the IC50 and A0.5 numbers go up.
Total phenolics and flavonoid contents of different extracts of Cerinthe major.

Data are presented as the mean ± SEM (n = 3). Significant differences (p < 0.05) determined by Tukey's test are indicated by different lowercase letters within a column. Units: * μg GAE/mg extract, ** μg QE/mg extract
DPPH● Scavenging Activity
The DPPH● radical scavenging activity of Cerinthe major extracts was evaluated according to the method of Blois.
32
A fresh 0.1 mM DPPH● solution in methanol was prepared and mixed with 40 µL of the plant extracts at different concentrations. The mixtures were incubated for 30 min in the dark at room temperature, and the absorbance was then measured at 517 nm using a microplate reader. The synthetic antioxidants BHA and BHT were used as reference compounds. The DPPH● radical scavenging activity (%) was calculated using equation (2):
Abs517 blank: absorbance of control reaction.
Abs517 sample: absorbance of test sample.
The percentage inhibition curve displayed against different concentrations was used to calculate the IC50 value. The extract concentration in μg/mL that results in 50% enzyme inhibition is known as the IC50.
ABTS●+ Scavenging Activity
We evaluated the extracts of Cerinthe major . The extracts of Cerinthe major were tested to see how well they could remove free radicals using a method called ABTS●+, which was created by Re and others. 33 A 7 mM water solution of ABTS●+ was combined with 2.45 mM potassium persulfate (K₂S₂O₄) and allowed to sit for 16 h to create the cation ABTS●+. After that, it was kept out of the light and at room temperature. The absorbance value of the diluted ABTS●+ solution was 0.700 ± 0.020. We combined a well microplate with 160 μL of the ABTS●+ solution. After adding 40 μL of each sample—prepared in methanol at different concentrations—to a 96-well plate, the mixture was left to incubate for 10 min. A wavelength of 734 nm was used to test absorbance. The antioxidant standards that were employed were BHA and BHT. The given formula was used to determine the percentage of inhibition (2).
β-Carotene-Linoleic Acid Bleaching Activity
According to Marco, a system consisting of β-carotene and linoleic acid was used to test the efficacy of Cerinthe major extracts in inhibiting β-carotene.
34
Dissolve 0.5 mg of beta-carotene in 1 mL of chloroform to start the process. This process blends beta-carotene and linoleic acid. This combination was added to a tube that already contained 200 μL of Tween 40 and 20 μL of linoleic acid. A vacuum extractor was used to remove the chloroform. The liquid was thoroughly stirred after 50 μL of hydrogen peroxide was added. After adjusting the mixture's absorbance at 470 nm to between 0.8 and 0.9, 160 μL of the emulsion was mixed with 40 μL of plant extracts or synthetic antioxidants (BHA and BHT) in different concentrations. The 96-well microplate's wells are then filled with the mixture. The absorbance at 470 nm was measured every 30 min while the microplate was being incubated at 50 °C. The duration varied between 0 and 120 min. The equation was used to show the percentage blockage (3), which follows:
I (%): percentage of inhibition. AH0: Absorbance of β-carotene in extract at t0. AC0: Absorbance of β-carotene in negative control at t0 . AHt: Absorbance of β-carotene in extract at 120 min. ACt: Absorbance of β-carotene in negative control at 120 min.
Reducing Power Activity
The reducing power of the samples was evaluated by their ability to reduce ferric (Fe3⁺) to ferrous (Fe2⁺) ions in K₃[Fe(CN)₆], according to the method of Oyaizu 35 with minor modifications. The reaction mixture contained 10 μL of Cerinthe major extract or standard (ascorbic acid or BHA) at different concentrations, 40 μL of 0.2 M phosphate buffer (pH 6.6), and 50 μL of 1% potassium ferricyanide. The mixture was incubated at 50 °C for 20 min, followed by the addition of 50 μL of 10% trichloroacetic acid, 40 μL of distilled water, and 10 μL of 0.1% ferric chloride. After thorough mixing, the solution was left to stand for 15 min to allow color development, and the absorbance was then measured at 700 nm using a microplate reader. The equation was used to show the percentage blockage (4), 36 which follows:

Enzymatic Activity
Anticholinesterase Activity
We employed spectrophotometers to assess how well pharmacological inhibitors affected the activity of acetylcholinesterase and butyrylcholinesterase. Our inquiry was based on previous work, with some modest modifications.37,38 The components listed below were mixed together: We combined 20 μL of enzyme buffer solution (BChE or AChE), 10 μL of sample extract solution with varying amounts of ethanol, and 130 microliters of sodium phosphate buffer (pH 8.0, 100 mM). After that, we let the mixture cool to 25 °C for fifteen minutes. Next, 0.5 mM DTNB (5,5′-dithio-bis(2-nitrobenzoic) acid) was added to 20 μL. The procedure was started by adding either acetylthiocholine iodide (20 μL, 0.71 mM) or butyrylthiocholine chloride (20 μL, 0.2 mM). By monitoring the light at 412 nm, we were able to determine how much of the yellow chemical 5-thio-2-nitrobenzoate was produced during the interaction between DTNB and thiocholine using a 96-well microplate reader. Enzymes that break down acetylthiocholine iodide and butyrylthiocholine chloride simplified this process. The results were shown as the percentage of enzyme inhibition at a 200 μg/mL extract concentration. We used the following formula to compare the sample response rates to the blank sample response rates in order to calculate inhibition (5):
Enzyme activity was measured both in the absence (E) and presence (S) of the sample. Galantamine, a known acetylcholinesterase inhibitor, was used as a positive control to benchmark the inhibitory activity of the tested samples. I% : Inhibition%.
Urease Inhibition Potential
The indophenol method was used to measure the amount of ammonia produced in order to determine urease's blocking capacity. 39 We combined 10 µL of extract solution, 50 µL of urea solution, and 25 µL of 5 U/mL jack bean urease for the reaction. It was then allowed to rest at 30 °C for 15 min. We then added 70 µL of the main solution, which was generated by combining 0.7125 g of NaOH with 1.175 mL of NaOCl in 25 mL of water, and 45 µL of the phenol solution, which was made by combining 2 g of phenol in 25 mL of water with 25 mg of Na₂[Fe(CN)₂NO]·2H₂O in another 25 mL of water. The absorbance at 630 nm was measured following a 50-min incubation period. The value discovered is the half maximal inhibitory concentration, or IC50.
Sun Protection Factor (SPF)
The approach described by Mansur et al has been used to evaluate Cerinthe major's SPF.
40
A methanol stock solution was mixed with 2000 parts per million of Ethanolic Extract and Aqueous Extract. The absorbance was then calculated at seven distinct wavelengths, separated by 5 nm, between 290 and 320 nm. The SPF was computed using the following formula, and all measurements were performed in triplicate (6):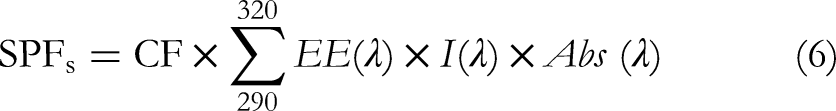
SPFs: Sun protection factor spectrophotometric; CF: correction factor (=10); EE(λ): erythemal effect spectrum; I(λ)
The values of EE × I are constants that were determined by Sayre et al (1979) and are listed in (Table 2).
Normalized product function (EE × I) used to calculate the SPF. 41

In addition to the formulated plant-based products, two commercial sunscreens were included for comparison: Nivea SPF 60 (Beiersdorf, Germany) and Vichy SPF 50 (L’Oréal, France), purchased from local pharmacies.
Anti-Inflammatory Activity Assay (BSA Denaturation Method)
The effectiveness of the plant extracts in preventing heat from degrading bovine serum albumin (BSA), a typical method of evaluating how well proteins stabilize in the laboratory, was used to assess their anti-inflammatory properties. A Tris-HCl buffer (0.05 M, pH 6.6), which was created by combining 1.2144 g of Tris with 200 mL of filtered water and then correcting the pH with HCl, was used for the test. Plant extracts were produced from a stock solution of 10,000 ppm and tested at different concentrations. The comparator anti-inflammatory medication was diclofenac sodium (injection form), and varying concentrations were prepared from a 500 ppm stock solution in distilled water. The following methods were used to prepare blank solutions: Take out the blanks: To adjust the background absorbance in colored extracts, 1 mL of plant extract and 1 mL of Tris-HCl buffer were combined. BSA blank: To demonstrate what occurs to the protein in the absence of an inhibitor, 1 mL of a 0.2% BSA solution was mixed with 1 mL of the solvent used for the extracts. The 0.2% BSA solution was created by combining 0.2 grams of BSA with 100 milliliters of pH 6.6 Tris-HCl buffer. 0.1 mL of plant extract or diclofenac standard was combined with 0.1 mL of BSA solution for each test concentration. To cause denaturation, the mixtures were heated in a water bath at 72 °C for five minutes after being incubated at 37 °C for fifteen minutes. Once the samples had cooled to room temperature, turbidity was measured at 660 nm using a UV-visible spectrophotometer. A 96-well microplate was then filled with 200 µL of each sample. 42
Agar Diffusion Method (Well Method) for Evaluating Antimicrobial Activity
In addition to a typical strain of Candida albicans ATCC 10231, we employed two non-staining bacteria (Pseudomonas aeruginosa ATCC 27853 and Escherichia coli ATCC 25922) and two staining bacteria (Staphylococcus aureus ATCC 25923 and Bacillus subtilis ATCC 25973). The agar diffusion or well method is frequently used to assess a substance's capacity to eradicate bacteria. Scientists filled Petri dishes with Sabouraud dextrose agar with 2% glucose for yeast and Mueller-Hinton agar for bacteria with a specific liquid. The liquid had 106 cells/mL from a new culture of bacteria or yeast. Swabbing the agar surface evenly is how inoculation is done. Use the tip of a Pasteur pipette to make a well in the middle of each plate after the agar surface has dried. This ensures that the size is always the same. Scientists put 50 µL of water-based (Aq) or alcohol-based (Eth) plant extracts into each well. The concentrations are 40, 20, 10, and 5 mg/mL. Plates with yeast cultures are kept at 37 °C for 48 h, while plates with bacterial cultures are kept at 37 °C for 24 h. The fact that an inhibitory zone was formed around the well shows how well each extract kills bacteria. To establish how well the antimicrobial works, the diameter of the inhibition zones is measured in millimeters. If the inhibitory zone is larger than 6 mm, the plant extract is deemed antibacterial. This approach allows you to easily assess the effectiveness of plant extracts against bacteria and yeast when combined with alcohol and water. This is an effective approach to assess how efficiently they destroy microorganisms. 43
Cytotoxicity Assay Using Yeast Cells
The effect of the aqueous (Aq) and ethanolic (Eth) plant extracts on the growth of Saccharomyces cerevisiae was evaluated using a growth sensitivity (cytotoxicity) assay. Each extract was initially prepared at 1.0 mg/mL and then serially diluted two-fold to obtain final concentrations of 1.0, 0.5, 0.25, and 0.125 mg/mL. Yeast cells, adjusted to 2 × 107 cells/mL in 100 mM potassium phosphate buffer (pH = 7), were treated with the different extract concentrations in a 96-well plate for 30 min at 37 °C. Untreated cells were used as controls.
For the semi-quantitative spot assay, 2.5 µL of each treated culture from the 96-well plate was spotted onto solid YPD agar plates containing 2% glucose, with or without the corresponding plant extract concentrations. The plates were incubated at 37 °C for 48 h. After incubation, yeast growth was recorded using an Epson® scanner, and the density of colonies in treated samples was visually compared with the untreated control to assess potential cytotoxic effects of the aqueous and ethanolic extracts.44,45
Statistical Analysis
The results are expressed as mean ± standard deviation (SD) (n = 3). In accordance with EAT Khodja et al,, 46 one-way analysis of variance (ANOVA) was performed to assess differences among groups, followed by Dunnett's multiple comparison test to compare each treatment group with the control. Differences were considered statistically significant at p < 0.05, whereas p < 0.001 was considered highly significant.
Reagents and Chemicals
Chemicals were obtained from Merck (Darmstadt, Germany), analytical standards from Sigma-Aldrich (St. Louis, MO, USA), and the solvents used in this study (methanol and ethanol) were purchased from VWR Chemicals and diclofenac sodium manufactured by Laboratoires SAIDAL, Algeria. All chemicals were of analytical grade.
Results
The Yield of Aqueose Extract and Ethanolic Extract
Comparing with other studies brings out the significant variation in phytochemical profiles of Cerinthe major . For example, a comparative investigation on Echinacea purpurea roots showed that varying the ethanol-to-root ratio (eg, 1:5 vs 1:11) significantly affected the yield of total dissolved solids and, more importantly, the concentration of lipophilic alkylamides, indicating that ethanol efficiently extracts hydrophobic bioactives and highlighting the importance of matching extraction solvents to plant profiles. Consequently, water extracts enriched in hydrophilic constituents are ideal for oral delivery, while ethanol extracts with higher lipophilic content are more suitable for topical applications due to enhanced skin penetration. 47 It also means that a variety of methods should be used to extract phytochemicals from plants if they are to be fully investigated as medicinal agents. In the future, more solvents with dissimilar polarities should be considered in order to identify all the bioactive constituents present in Cerinthe major. It is important to mention that further studies on extracts’ biological activities could provide insight into potential therapeutic uses and give rise to other applications of Cerinthe major for various diseases.48,49 The extraction yields of the aqueous and ethanolic extracts of Cerinthe major are presented in (Table 3).
The yield of AE and EE.

Mineral Analysis
Elemental concentrations of the studied plant Cerinthe major were determined using the ICP-OES technique and are presented in Table 4. The principal mineral concentrations in Cerinthe major are expressed as mean ± SD from triplicate analyses. Sixteen elements were investigated, including Ca, Fe, Mg, Mn, Na, Co, Cd, Cr, Ni, Pb, Ag, and Cu. To compare variability independent of measurement units, the coefficient of variation (CV) was calculated. This revealed that magnesium content was the most stable (CV = 2.15%), whereas lead concentrations were the most variable (CV = 15.79%), which may reflect heterogeneous environmental exposure, possibly related to local soil or atmospheric contamination. Silver, nickel, cobalt, and cadmium were not detected above the low-level detection limit of the method (ND in Table 4), suggesting very low contents of these elements in the analysed samples, The mineral composition was determined by ICP-OES after sample mineralization, and all elements were quantified as aqueous ionic species rather than as neutral elemental forms. These differences in extraction solvents are expected to markedly influence the antioxidant and enzyme inhibitory properties of Cerinthe major extracts, since polar phenolics are better recovered in aqueous media, whereas more lipophilic constituents are enriched in ethanolic extracts and may contribute to additional biological effects.
Mass fractions of chemical elements in the plant samples. Values are presented as mean ± standard deviation (SD) in mg/kg on a dry mass basis (n = 3).

* SD standard deviation (dry weight basis for all data).
** V is the coefficient of variability. Nd: low-level identification
ND : not detected; +/−: positive/negative; Rt: retention time; ESI: electrospray ionization; Voltage CE: capillary electrospray.
Analysis of Liquid Chromatography-Mass Spectrometry LC-MS-MS
Table 5 summarizes the results of the UPLC-ESI-MS-MS analysis of aqueous extract (AE) and ethanolic extract (EE) by comparing the retention times of the standards injected in positive and negative modes (Figure 1 and Figure 2). The UPLC-ESI-MS study of the aqueous and ethanolic extract of Cerinthe major revealed the presence of a variety of bioactive chemicals, as shown in Figure 3, mainly classified into phenolic acids, flavonoids, and other important secondary metabolites. (−)-Catechin, chrysin-6-C-glucoside, myricetin-3-O-arabinoside, myricetin-3-O-rhamnoside, myricetin, orexilin A, quercetin-3-O-glucoside, telluride, hispidulin, ferulic acid, luteolin, oleanolic acid, oleuropein, quercetin, resveratrol, riboflavin, rutin, kojic acid, naringenin, thymol, vanillin, caffeic acid, p-coumaric acid, and salicylic acid were identified. This detailed chemical profile highlights 23 of the 24 compounds in the aqueous extract and 18 of the 24 compounds in the ethanolic extract.

UPLC–ESI–MS/MS chromatogram of the aqueous extract (AE) of Cerinthe major showing the main identified phenolic and non-phenolic constituents.
UPLC–ESI–MS/MS chromatogram of the ethanolic extract (EE) of Cerinthe major showing the main identified phenolic and non-phenolic constituents.
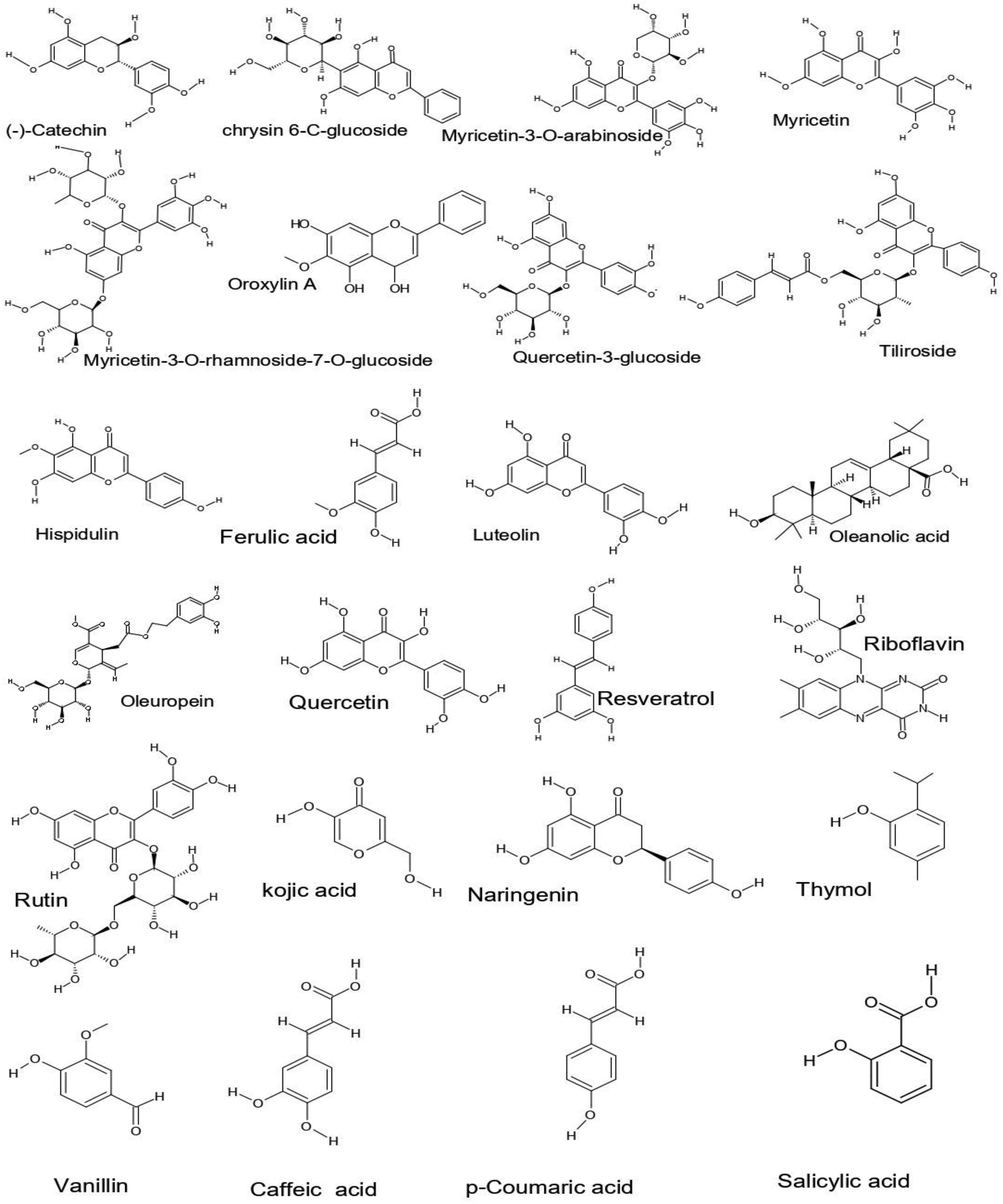
Chemical structures of the main phytochemicals identified in Cerinthe major L.
UPLC-ESI-MS-MS-Determined Bioactive Components Profile of AE and EE.

The identification of numerous phenolic acids and flavonoids, such as catechin, quercetin, myricetin derivatives, rutin, luteolin, and caffeic and ferulic acids, suggests a strong potential for antioxidant activity, as these classes of compounds are well-known radical scavengers and metal chelators. Moreover, the presence of flavonoids like naringenin, hispidulin, and chrysin derivatives, together with triterpenoids such as oleanolic acid, may also contribute to enzyme inhibitory and anti-inflammatory effects reported for related plant species. Thus, the rich and diverse phenolic profile of Cerinthe major extracts provides a plausible mechanistic basis for their expected antioxidant and enzyme-modulating properties.
Assessment of Total Bioactive Compounds
The total phenolic contents of the ethanolic and aqueous extracts of Cerinthe major were determined to be 50.86275 ± 12.68576 mg QE/g and 175.3725 ± 5.505057 mg GAE/g, respectively. The amounts of flavonoids in the ethanolic and aqueous extracts of Cerinthe major were determined to be 57.77778 ± 3.682848 mg QE/g extract and 145.9722 ± 0.883883 mg GAE/g extract, respectively. The ability of these substances to function as strong antioxidants is one of their most significant characteristics, 50 anti-inflammatory, 51 anti-atherogenic, 52 and anti-carcinogenic agents. 53 The markedly higher total phenolic and flavonoid contents in the aqueous extract compared with the ethanolic extract suggest that polar phenolics are preferentially extracted into water, which may partly explain any superior radical scavenging or reducing activities observed for this extract. These polyphenolic constituents are known to act as chain-breaking antioxidants by donating electrons or hydrogen atoms to reactive species, as well as modulating key signaling pathways involved in inflammation, atherogenesis, and carcinogenesis.
Antioxidant Activity
Given the complex nature of phytochemicals, the antioxidant activity of Cerinthe major extracts was evaluated using multiple assays: DPPH●, ABTS●+, β-carotene bleaching, and reducing power. The results are summarized in Table 6. Antioxidant activity was quantified using the IC₅₀ and A₀.₅ indices, which represent the concentration required to achieve 50% scavenging and half-maximal reducing power, respectively. These values were calculated by linear regression analysis. It is important to note that both IC₅₀ and A₀.₅ exhibit an inverse relationship with antioxidant efficacy; lower values indicate more potent activity.
Antioxidant Activity of Different Fractions of Cerinthe major.

* Standard compounds. NT: not tested.
DPPH● Scavenging Activity
The DPPH● assay measures the free radical-scavenging capacity of antioxidants via hydrogen atom transfer. 54 All Cerinthe major extracts demonstrated notable DPPH● radical scavenging activity. The IC₅₀ values, indicating potency, established the following order of effectiveness: BHA > BHT > AE > EE (Table 6). The aqueous extract (AE) showed strong activity with an IC₅₀ of 41.81 ± 2.76 µg/mL, which, while considerably higher than that of BHA (6.14 ± 0.41 µg/mL) and BHT (12.99 ± 0.40 µg/mL), confirms its appreciable antioxidant potential.
This pronounced DPPH● scavenging activity of the aqueous extract is consistent with its higher total phenolic and flavonoid contents, supporting the view that these polyphenols are the main contributors to its hydrogen atom donating capacity. Although synthetic antioxidants such as BHA and BHT exhibit lower IC₅₀ values, the substantial activity of the plant extract suggests that Cerinthe major may provide effective, naturally derive d antioxidant protection, particularly in systems where complex mixtures of phenolics can exert synergistic effects.
ABTS●+ Scavenging Activity
The ABTS●+ assay, which operates on a similar electron-transfer principle as DPPH●, was used to further evaluate antiradical activity. Based on IC₅₀ values, the scavenging capacity of the extracts and standards decreased in the order: BHT > BHA > AE > EE (Table 6). The ethanolic extract (EE) exhibited an IC₅₀ of 18.80 ± 1.37 µg/mL, demonstrating its excellent capacity to scavenge ABTS●+ radicals.
The stronger ABTS●⁺ scavenging ability of the ethanolic extract compared with its DPPH● performance suggests that some of its constituents may interact more efficiently with this relatively more hydrophilic radical cation, reflecting differences in solubility and reaction kinetics between the two assays. Together with the aqueous extract data, these findings indicate that both polar and moderately lipophilic phenolics in Cerinthe major contribute to antiradical activity, with their relative importance depending on the specific radical system evaluated.
β-Carotene-Linoleic-Acid System Assay
The β-carotene–linoleic acid experiment revealed that the aqueous extract was mostly inert (IC•₀ ≥ 800 µg/mL), whereas the ethanolic extract showed considerable antioxidant activity (IC⋅₀ = 235.23 µg/mL). Standard antioxidants BHT and BHA, on the other hand, showed high action with IC2₀ values of 32.08 µg/mL and 16.94 µg/mL, respectively. These results imply that the plant's antioxidant capacity is greatly enhanced by ethanol-soluble substances. 55
The superior performance of the ethanolic extract in the β-carotene–linoleic acid system suggests that more lipophilic antioxidants, likely including certain flavonoids, phenolic acids in esterified forms, and terpenoids, are responsible for delaying lipid peroxidation in this emulsion model. In contrast, the weak effect of the aqueous extract indicates that its predominantly hydrophilic phenolics are less efficient at partitioning into the lipid phase, highlighting the importance of ethanol-soluble constituents of Cerinthe major for protecting lipid-rich environments against oxidative damage.
Reducing Power Activity
The reducing power of the extracts was determined by their capacity to reduce Fe3⁺ to Fe2⁺ 56 . A lower A₀.₅ value indicates greater reducing activity. Among the samples tested, ascorbic acid demonstrated the strongest activity (lowest A₀.₅), followed by BHA. The aqueous extract (AE) was the least effective, with an A₀.₅ value of 49.22 ± 3.43 µg/mL. The rank order of efficacy was: Ascorbic acid > BHA > AE.
The relatively weak reducing power of the aqueous extract, compared with ascorbic acid and BHA, indicates that although Cerinthe major contains redox-active phenolic compounds, their electron-donating capacity in the Fe3⁺/Fe2⁺ system is modest, which may reflect lower concentrations of highly reducing phenolics or structural features less favorable for direct electron transfer.
Enzymes Inhibition Activity
Cholinesterase Inhibitory Activity
The inhibitory activity of the extracts against acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) was evaluated, as these enzymes are relevant targets for enhancing cognitive function. 57 The results are presented in Table 7. The ethanolic extract (EE) exhibited significant inhibitory activity against AChE, whereas the aqueous extract (AE) showed no effect. Both extracts inhibited BChE, with EE demonstrating a markedly stronger effect than AE.
Anti-Cholinesterase and Urease Activities of Different Extracts of Cerinthe major.
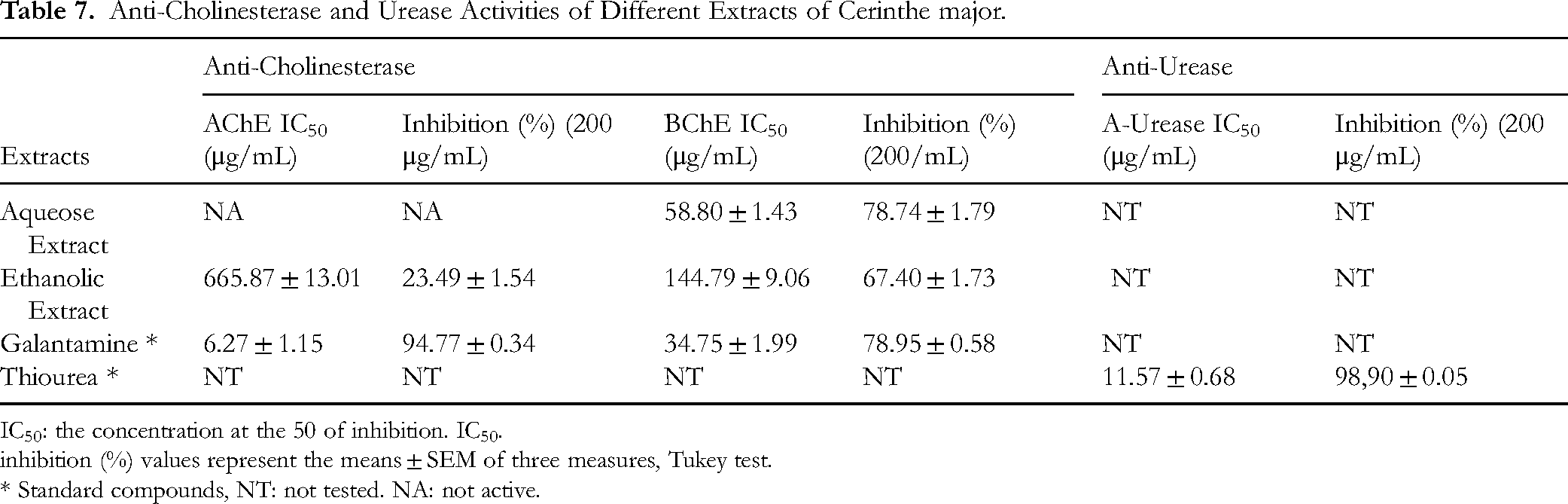
IC50: the concentration at the 50 of inhibition. IC50.
inhibition (%) values represent the means ± SEM of three measures, Tukey test.
* Standard compounds, NT: not tested. NA: not active.
The selective AChE inhibition observed for the ethanolic extract, together with its stronger effect on BChE, is consistent with the presence of more lipophilic flavonoids and related phenolics, which are known to interact with the catalytic and peripheral anionic sites of cholinesterases through hydrogen bonding and π–π stacking, thereby reducing substrate hydrolysis. In contrast, the lack of AChE inhibition by the aqueous extract suggests that its predominantly hydrophilic phenolic profile is less suited for effective binding within the active gorge of these enzymes, further emphasizing the importance of ethanol-soluble constituents of Cerinthe major as potential neuroprotective agents.
Urease Inhibition Potential
The ability of the ethanol extract of Cerinthe major to block enzymes was tested by looking at how it affects urease. The results are presented in Table 7. Enzyme inhibitors reduce the biological activity of enzymes, which may play a vital role in correcting any metabolic dysfunction.
Therefore, many enzyme inhibitors are available as medications in pharmacies today. 58 According to the results of the current study, extracts of Cerinthe major did not exhibit any enzyme inhibitory activity.
The absence of urease inhibition by Cerinthe major extracts indicates either a low abundance of urease-targeting constituents or an unfavorable structural fit of the present phenolics and flavonoids for interaction with the nickel-containing active site of the enzyme, suggesting that urease is not a primary pharmacological target for this species despite its other promising bioactivities.
Photoprotective Activity
The sunscreen activity of Cerinthe major was assessed using the sun protection factor (SPF) calculation. For SPF values between 2 and 12, 12 to 30, 30 to 50, and higher than 50, sunscreen activity was classified as low, moderate, or high, accordingly. The SPF values of the extracts utilized in Cerinthe major L. research are displayed in Table 8. The range of the EE and AE values was 35.78 ± 0.12 and 32.82 ± 0.17, respectively. These findings showed that both extracts had a noticeable sunscreen effect. Long-term UV exposure raises the risk of skin conditions such photoallergic responses and melanoma. The primary cause of skin conditions is UV-B radiation, which is released at wavelengths between 280 and 320 nm. By examining their absorption in the UV-B spectrum, recent research has concentrated on investigating the possible application of natural substances with antioxidant properties as sunscreens. The phenolic content of plant extracts and their sun protection factor (SPF) have been found to be clearly and strongly correlated.59,60 So, both EE and AE showed strong sun protection levels that are similar to the SPF values found in regular commercial and cosmetic sunscreens (Table 9).
Results of antimicrobial tests:.

NI = No Inhibition.
Photoprotective activity of different extracts/fractions of Cerinthe major.

* Reference compounds. ** variability coefficients (%).
The SPF values of the photoprotective activity showed very low coefficients of variation (V = 0.13-1.00), indicating a very high degree of accuracy in their determination (Table 9).
The high SPF values obtained for both extracts are in line with their rich phenolic profiles, since many phenolic acids and flavonoids absorb strongly in the UV-B region and can also neutralize UV-induced reactive oxygen species, providing dual photoprotective effects. These results therefore suggest that Cerinthe major extracts may serve as promising natural UV filters or adjuvants in sunscreen formulations, potentially reducing reliance on purely synthetic filters while maintaining effective protection against photo-induced skin damage.
SPF values were calculated using the EE × I constants reported by Sayre et al (Table1), which allowed us to compare our plant-based formulations with commercial sunscreens.
Anti-Inflammatory Activity Assay
Anti-inflammatory characteristics—Preventing denaturation of albumin: One well-established cause of inflammation is protein denaturation. The capacity of various solvent plant extracts to denaturate proteins was examined as part of the inquiry into the mechanism of the anti-inflammatory effect. It was successful in preventing albumin denaturation brought on by heat (Table 10). Aqueous extract had the highest IC50 of 90.48 ± 2.68 µg/mL, whereas ethanolic IC50 was 195.25 ± 2.99 µg/mL. The albumin denaturation was prevented by all solvent extracts, although the aqueous extract outperformed the ethanolic extracts. The typical anti-inflammatory medication diclofenac demonstrated the lowest inhibition (IC50 = 658.66 ± 4.58 µg/mL). The effectiveness of each sample in lowering inflammation was determined by calculating the proportion of protein denaturation that was averted. Cerinthe major L. contains bioactive chemicals. The chemical structures of the identified compounds are shown in Figure 4.

Diclofenac (standard) and AE and EE's in vitro anti-inflammatory effects.
In vitro anti-inflammatory effects of AE, EE and the reference drug. IC₅₀ values (µg/mL).

The values are expressed as mean ± SD of three replicates. Means sharing the same lowercase letter are not significantly different according to Tukey's honestly significant difference (HSD) post hoc test at p < 0.05.
The superior ability of the aqueous extract to prevent heat-induced albumin denaturation, even surpassing diclofenac, suggests that its polar phenolic constituents can effectively stabilize protein structure by forming hydrogen bonds and other non-covalent interactions that counteract unfolding. Such inhibition of protein denaturation is consistent with the proposed anti-inflammatory potential of Cerinthe major, as several of the identified phenolic acids and flavonoids are known to attenuate inflammatory cascades triggered by protein damage and oxidative stress
Antimicrobial Activity
The antimicrobial potential of the aqueous (AE) and ethanolic (EE) extracts was evaluated against selected Gram-positive, Gram-negative, and fungal strains using the agar well diffusion method. The results are presented in Table 8 as inhibition zone diameters (in mm).Among the tested bacterial strains, Escherichia coli ATCC 25922 exhibited the highest sensitivity to the ethanolic extract. A noticeable increase in inhibition was seen, with the sizes of the inhibition zones measuring 23 mm, 13 mm, 11 mm, and 8 mm at concentrations of 40, 20, 10, and 5 mg/mL, respectively. In contrast, the aqueous extract showed no inhibitory effect against this strain at any tested concentration. Staphylococcus aureus ATCC 25932 was only slightly affected by the ethanolic extract, showing an inhibition zone of 11 mm only at the highest concentration (40 mg/mL). No inhibition was recorded with the aqueous extract or at lower concentrations of the ethanolic extract. The other strains, like Pseudomonas aeruginosa ATCC 27853, Bacillus subtilis ATCC 25973, and Candida albicans ATCC 10231, did not show any sensitivity to either extract, as there were no inhibition zones at any of the concentrations tested.The negative control (0.45% NaCl) produced no inhibition, confirming the validity of the test conditions. In this assay, 0.45% NaCl was used as an inert negative control to reproduce the osmotic conditions of the extract solutions without providing any antimicrobial effect, ensuring that the observed inhibition zones could be attributed solely to the plant extracts. These results indicate that the ethanolic extract is effective against E. coli, but the aqueous extract does not seem to have much effect on germs in the tests done.
The ability of the water (AE) and alcohol (EE) extracts to fight germs was tested on certain types of bacteria and fungi using the agar well diffusion method. The results are presented in Table 8 as inhibition zone diameters (in mm). Among the bacterial strains tested, Escherichia coli ATCC 25922 showed the highest sensitivity to the ethanolic extract; however, this antibacterial effect was weak and observed only at relatively high concentrations (5-40 mg/mL). A noticeable increase in inhibition was seen, with the sizes of the inhibition zones measuring 23 mm, 13 mm, 11 mm, and 8 mm at concentrations of 40, 20, 10, and 5 mg/mL, respectively. In contrast, the aqueous extract showed no inhibitory effect against this strain at any tested concentration. Staphylococcus aureus ATCC 25932 was only a little impacted by the ethanolic extract, showing an inhibition zone of 11 mm only at the highest concentration of 40 mg/mL. No inhibition was recorded with the aqueous extract or at lower concentrations of the ethanolic extract. The other strains, like Pseudomonas aeruginosa ATCC 27853, Bacillus subtilis ATCC 25973, and Candida albicans ATCC 10231, did not show any sensitivity to either extract, as there were no inhibition zones at any of the concentrations tested. The negative control (0.45% NaCl) produced no inhibition, confirming the validity of the test conditions. These results indicate that the ethanolic extract is effective against E. coli, but the aqueous extract does not seem to have much effect on germs in the tests done.
Antimicrobial Activity
The cytotoxicity assay performed on Saccharomyces cerevisiae to preliminarily assess the biocompatibility of the aqueous and ethanolic extracts revealed no detectable cytotoxic effects under the tested conditions. The spot assays on YPD agar plates showed comparable yeast growth across all concentrations tested, indicating that the extracts did not visibly reduce cell viability or proliferation relative to the untreated control.
For the aqueous extract, concentrations from 1.0 to 0.125 mg/mL (corresponding to 10-1.25 mg/mL in the stock preparations) produced no observable reduction in colony size or density. Similarly, the ethanolic extract, tested over the same range of concentrations, did not affect yeast growth, with treated cultures displaying growth patterns similar to those of the control cells.
The absence of cytotoxic effects in Saccharomyces cerevisiae across the tested concentration range suggests that both aqueous and ethanolic extracts of Cerinthe major are largely biocompatible under these in vitro conditions, at least toward non-mammalian eukaryotic cells. This lack of growth inhibition supports the notion that the bioactive phenolic and flavonoid constituents can exert antioxidant, anti-inflammatory, and enzyme-modulating activities without overtly compromising basic cellular viability, which is encouraging for their potential safe use in future pharmacological or cosmetic applications (Figure 5 and Table 11).

Cytotoxic assay for aqueous and ethanolic plant extracts on Saccharomyces cerevisiae using cytotoxicity spot-test assay.
Cytotoxicity of Aqueous and Ethanolic Cerinthe major Extracts on Saccharomyces cerevisiae Evaluated by the Spot-Test Assay.

C: cytotoxic; NC: no cytotoxic.
Discussion
This study provides the first ICP-OES-based characterization of the mineral profile of Cerinthe major . The plant proved to be a rich source of macroelements, particularly calcium (26,375.65 ± 1580.70 mg/kg), magnesium (4096.87 ± 88.14 mg/kg), sodium, and iron (263.05 ± 6.28 mg/kg), all of which play key roles in bone structure, neuromuscular function, and enzymatic reactions in human nutrition.61–63 The remaining elements followed the order Cr > Mn > Cu > Pb, with appreciable levels of chromium (98.93 ± 3.49 mg/kg) and manganese (18.25 ± 1.43 mg/kg), suggesting that Cerinthe major could contribute to the dietary intake of these essential trace elements.
From a safety perspective, the copper content of Cerinthe major exceeded the FAO/WHO guideline value of 4.5 mg/kg for fruits, indicating that excessive consumption should be avoided, although typical intake amounts are likely too low to pose a major risk . In contrast, the lead concentration (0.19 ± 0.03 mg/kg) was well below the WHO limit of 10 mg/kg for medicinal plants, indicating that Cerinthe major complies with international recommendations regarding Pb contamination. Overall, the mineral profile highlights Cerinthe major as a nutritionally interesting species, provided that its elevated copper levels are carefully considered in future applications.
LC–MS/MS profiling of the aqueous and ethanolic extracts of Cerinthe major revealed marked qualitative differences in their phytochemical composition, highlighting the influence of solvent polarity on extraction efficiency . The ethanolic extract contained a broader range of polyphenols, particularly flavonoids and phenolic acids, with catechin accounting for 36.7% of the chromatographic area, whereas rutin predominated in the aqueous extract (51.7%), in agreement with previous reports on solvent-dependent extraction patterns . Hydrophilic phenolic acids such as caffeic acid and p-coumaric acid were enriched in the aqueous fraction, while less polar compounds including quercetin-3-O-glucoside, luteolin, tiliroside, thymol and hispidulin were comparatively more abundant or exclusive to the ethanolic extract.
Some metabolites exhibited pronounced solvent-specific distributions. Myricetin-3-O-arabinoside was detected only in the aqueous extract, whereas myricetin and ferulic acid were markedly enriched in the ethanolic fraction (40.37% vs 4.19% for ferulic acid), consistent with their higher solubility in moderately polar organic solvents.61,64,65These findings confirm that ethanol is more efficient than water for extracting flavonoid- and phenolic-rich fractions from Cerinthe major, which likely contributes to the distinct bioactivities observed for the two extracts.
Interestingly, kojic acid was tentatively identified only in the aqueous extract. Since kojic acid is typically produced by fungi and is also soluble in ethanol, this finding should be interpreted with caution. We cannot exclude the possibility that it may arise as a processing artefact or from trace microbial activity during extraction, and further targeted analyses would be required to confirm its genuine occurrence in Cerinthe major.
Nterestingly, the LC–MS/MS data suggested the presence of a secoiridoid with a fragmentation pattern reminiscent of oleuropein, a compound characteristically associated with the family Oleaceae (eg, Olea europaea L.). However, iridoids (and secoiridoids) have not been reported from Boraginaceae so far (Riedl, 1997), 66 and it cannot be excluded that this peak corresponds to an undescribed or otherwise uncommon iridoid-derived structure. This unexpected finding therefore needs to be interpreted with great caution and should be confirmed by isolation and detailed structural elucidation (including comprehensive NMR studies).
From a biogenetic perspective, secoiridoids usually arise from Route I (loganic acid-derived) pathways that are highly expressed in Gentianales, whereas Route II, involving 8-epi-loganic acid and more typical of Lamiales, only rarely leads to secoiridoid derivatives. Such chemotaxonomic considerations further support a cautious interpretation of our tentative assignment.67,68
Regarding pyrrolizidine alkaloids and meroterpenoids, these classes have been reported in several Boraginaceae species, but our LC-MS/MS method was optimized mainly for phenolic compounds and did not specifically target the full spectrum of PAs and meroterpenoids. Their apparent absence in the present profiles should therefore not be taken as evidence of their true absence in Cerinthe major, and dedicated targeted analyses would be needed to address this point.
Together, these findings confirm that solvent polarity strongly influences the selectivity of phytochemical extraction. The ethanolic extract, which was richer in flavonoids and several phenolic acids, is therefore expected to exhibit stronger anti-inflammatory and free radical–scavenging effects, in line with the well-documented bioactivities of these compound classes. In contrast, the aqueous extract, characterized by higher levels of specific hydrophilic phenolic acids, may underlie complementary bioactivities, particularly in hydrophilic environments. Consistent with previous work showing that ethanol efficiently recovers phenolic-rich fractions from plant materials, our LC–MS/MS data support the use of ethanolic Cerinthe major extracts as promising candidates for the development of nutraceutical or phytopharmaceutical formulations.
The higher extraction yield obtained with water compared to absolute ethanol can be attributed to the ability of the aqueous solvent to solubilize a wide range of highly polar constituents (eg, sugars, organic acids and other hydrophilic metabolites), which substantially increases the crude mass of the extract but does not necessarily reflect a higher content of bioactive polyphenols. By contrast, ethanol selectively recovers less polar and medium-polar compounds such as many flavonoids and certain terpenoids, resulting in a lower overall yield but an extract that is more enriched in specific bioactive constituents, in agreement with our LC–MS/MS and bioactivity data.
Phenolic compounds are a large group of plant secondary metabolites that exert important pharmacological effects in humans, mainly through their antioxidant and other bioactive properties. 69 The total polyphenol content of Cerinthe major was determined in extracts obtained with solvents of different polarities, and this quantitative analysis revealed varying levels of flavonoids and other polyphenols among the extracts. In this study, polar organic solvents proved to be more effective for extracting flavonoids and phenolic compounds from the plant material. The amounts of these chemicals can be influenced by several factors, including the choice and polarity of the extraction solvent, geographical origin, extraction and analytical methods, and environmental and genetic conditions.
Antioxidant capacity assays rely on different underlying mechanisms, mainly hydrogen atom transfer (HAT) and electron transfer (ET), so the response of a given extract can vary between tests. In our study, the aqueous and ethanolic extracts of Cerinthe major showed clearly distinct profiles across the DPPH●, ABTS●⁺, β-carotene–linoleic acid, CUPRAC and reducing-power, underscoring the role of solvent polarity in selecting specific antioxidant constituents .
The aqueous extract exhibited stronger radical-scavenging activity in the DPPH● (IC₅₀ = 41.81 µg/mL) and ABTS●⁺ (IC₅₀ = 18.80 µg/mL) assays than the ethanolic extract (IC₅₀ = 203.29 and 46.15 µg/mL, respectively), which is consistent with its higher content of hydrophilic phenolic acids such as caffeic acid acid and ferulic acids identified by LC–MS/MS; these acids are well known for their efficient hydrogen-donating and radical-quenching properties. In contrast, the ethanolic extract was more effective in protecting lipids in the β-carotene–linoleic acid model (IC₅₀ = 235.23 µg/mL vs ≥800 µg/mL for the aqueous extract) and showed a lower IC₅₀ in the CUPRAC assay (152.23 vs 264 µg/mL), in line with its enrichment in less polar flavonoids such as catechin, rutin and quercetin derivatives that are particularly efficient ET-type antioxidants.
The aqueous extract also showed higher activity in the phenanthroline and reducing-power assays (IC₅₀ = 26.45 and 49.22 µg/mL, respectively), confirming its strong electron-donating and metal-reducing capacity, likely driven by its phenolic acid–rich profile. In contrast, both extracts displayed relatively weak responses in the TAC assay at ≥400 µg/mL compared with ascorbic acid, indicating that, despite their measurable activity, they should not be considered high-potency antioxidants when benchmarked against standard reference antioxidants.
Overall, the selective behavior of the two extracts across the different antioxidant models is consistent with their distinct phytochemical fingerprints. The aqueous extract, richer in polar phenolics, performs better in radical-scavenging and certain ET-based assays, whereas the ethanolic extract, enriched in less polar flavonoids, is more effective in lipid-phase protection and other ET-dominated systems. These complementary profiles suggest that combining aqueous and ethanolic Cerinthe major fractions, or tailoring their use to specific biological environments (hydrophilic vs lipophilic), may be a more rational strategy than relying on a single extract, in line with the view that no single assay—or single extract—can fully capture antioxidant potential..
Cerinthe major extracts exhibited a clear selectivity towards butyrylcholinesterase (BChE) over acetylcholinesterase (AChE). The aqueous extract showed negligible inhibition of AChE, whereas the ethanolic extract was weakly active (IC₉₀ = 665.87 ± 13.01 µg/mL; 23.49% inhibition at 200 µg/mL), far below the potency of galantamine (IC₉₀ = 6.27 ± 1.15 µg/mL; 94.77% inhibition). In contrast, both extracts significantly inhibited BChE, with the aqueous extract displaying particularly strong activity (IC₉₀ = 58.80 ± 1.43 µg/mL; 78.74% inhibition), approaching that of galantamine (IC₅₀ = 34.75 ± 1.99 µg/mL; 78.95% inhibition), while the ethanolic extract showed moderate inhibition (IC₉₀ = 144.79 ± 9.06 µg/mL; 67.40% inhibition).
This BChE-selective profile is consistent with the LC-MS/MS data, which revealed that the aqueous extract is enriched in flavonoids and phenolic acids, classes of compounds frequently reported as BChE inhibitors. Selective BChE inhibition is considered particularly relevant in later stages of Alzheimer's disease, when AChE activity declines and BChE activity increases . Thus, the strong BChE inhibitory effect of the aqueous Cerinthe major extract identifies it as a promising lead for further neuroprotective investigation, although isolation of the active constituents and in vivo confirmation will be required before any therapeutic conclusions can be drawn.6-Some phenolic compounds that can stop urease from working are quercetin, myricetin, and luteolin 70 . As expected, the extract did not work against urease either. So, the lack of effectiveness is because these compounds are either not present at all or are present in very small amounts.
The in vitro SPF values obtained for the aqueous and ethanolic extracts of Cerinthe major place both in the “high protection” category according to the European Commission/SPF labeling guidelines, which classify products with SPF ≥ 30 as providing high or very high protection.71–73 This strong photoprotective effect is consistent with the high levels of phenolic compounds and flavonoids detected in the extracts, as these classes are known to absorb UV radiation and to quench UV-induced reactive oxygen species.
A positive relationship was observed between total phenolic content and SPF, suggesting that the phenolic fraction contributes substantially to the UV-blocking capacity of Cerinthe major, in agreement with previous reports on herbal extracts. Given that the plant grows in Saharan environments characterized by intense solar exposure, the accumulation of UV-absorbing secondary metabolites may represent an adaptive response that can be exploited for cosmetic or pharmaceutical applications. Overall, our data indicate that Cerinthe major extracts have promising potential as natural ingredients in sunscreen formulations, although further formulation, stability and in vivo efficacy studies will be required before practical use.
The anti-inflammatory potential of Cerinthe major was evaluated using the protein denaturation model, a widely used in vitro assay in which compounds that prevent heat-induced protein denaturation are considered to possess anti-inflammatory properties relevant to rheumatic conditions.74–76 In this assay, the extracts of Cerinthe major significantly inhibited protein denaturation in a concentration-dependent manner, indicating that they can stabilize protein structure under stress conditions.
This effect is likely linked to the high content of flavonoids and other phenolic compounds in the extracts, as these classes have been repeatedly reported to exert anti-inflammatory actions through membrane stabilization and interference with pro-inflammatory signaling pathways.77,78 In our study, the aqueous extract, which contained higher levels of flavonoids, showed stronger inhibition of protein denaturation than the ethanolic extract, suggesting that its phenolic profile may be particularly effective in this model. These findings support the view that Cerinthe major could provide promising lead compounds for the development of anti-inflammatory agents, although isolation of the active molecules and in vivo validation will be required.
Antimicrobial tests showed that only the ethanolic extract of Cerinthe major exhibited significant, but weak, antibacterial activity. The concentrations required to inhibit microbial growth in our assays (5-40 mg/mL) are much higher than the MIC ranges that are generally considered indicative of strong or clinically relevant antimicrobial activity for plant extracts. Several authors classify plant extracts as highly active or active only when their MIC values are below 1-2 mg/mL (and often much lower).. 79 ranging from 8 to 23 mm in a dose-dependent manner, whereas the aqueous extract was inactive under the tested conditions. This pattern is consistent with numerous reports that ethanol more efficiently extracts phenolic compounds and other antimicrobial secondary metabolites, including flavonoids, alkaloids and terpenoids, than water.80–82 The higher activity against E. coli may be attributed to lipophilic or moderately polar constituents in the ethanolic extract that can interact with and disrupt the outer membrane or key metabolic enzymes of Gram-negative bacteria.
In contrast, Pseudomonas aeruginosa, Bacillus subtilis, Staphylococcus aureus and Candida albicans were not inhibited, suggesting either an intrinsic resistance of these strains or insufficient levels of active compounds in the extracts at the tested concentrations. The well-known multidrug resistance of P. aeruginosa, related to low outer-membrane permeability, efflux pumps and biofilm formation, may explain its lack of susceptibility. The absence of activity of the aqueous extract is likely related to its more polar composition, favoring extraction of sugars and glycosides, which generally lack direct antimicrobial effects.83–85 Overall, these findings reinforce that solvent polarity is a key determinant of both phytochemical composition and antimicrobial activity, and they identify the ethanolic Cerinthe major extract as a more promising candidate for further antibacterial investigations, particularly against E. coli and potentially other Gram-negative pathogens.
Although Pseudomonas aeruginosa, Bacillus subtilis, Staphylococcus aureus and Candida albicans were not inhibited at the tested concentrations, this negative result is still informative because it helps to define the antimicrobial spectrum of Cerinthe major L. extracts and suggests that these highly resilient strains may require higher doses, different extract fractions or alternative extraction procedures. The study remains significant as it (i) provides a detailed phenolic/chemical profile of the extracts, (ii) demonstrates antioxidant, enzymatic inhibitory, anti-inflammatory and photoprotective activities, and (iii) identifies selective antibacterial effects against more susceptible strains, thereby guiding future fractionation and optimization efforts rather than suggesting a broad, non-specific antimicrobial action.
Phenolic compounds such as flavonoids (eg, quercetin, myricetin, catechin, rutin) and phenolic acids (eg, caffeic acid, ferulic and p-coumaric acid) are known to exert multiple bioactivities through well-characterized mechanisms. Their antioxidant effects mainly arise from hydrogen atom and single-electron transfer from phenolic hydroxyl groups to reactive oxygen species, as well as from metal-chelating properties that limit Fenton-type radical generation. In addition, several of the flavonoids identified in Cerinthe major (quercetin, myricetin, luteolin, naringenin, hispidulin) have been reported to inhibit key enzymes such as cholinesterases and pro-inflammatory targets by binding to catalytic or allosteric sites and modulating redox-sensitive signaling pathways (eg, NF-κB, MAPKs). With regard to antimicrobial activity, phenolic compounds can disrupt microbial membranes, interfere with energy metabolism and nucleic-acid synthesis, or act synergistically with other metabolites, which may explain the selective activity observed against E. coli and the lack of effect on more intrinsically resistant strains such as P. aeruginosa.
The apparently contrasting behavior of the aqueous and ethanolic extracts across the different bioassays can be rationalized by their distinct phytochemical fingerprints rather than by a simple “more or less active” dichotomy. The aqueous extract, enriched in highly polar phenolic acids such as caffeic acid and ferulic acids and in certain flavonoid glycosides, performed better in radical-scavenging and electron-transfer-based assays (DPPH●, ABTS●⁺, phenanthroline and reducing-power), in line with the well-known hydrogen-donating and metal-reducing properties of these hydrophilic phenolics. In contrast, the ethanolic extract, which contained higher levels of less polar flavonoids (eg, catechin, quercetin derivatives, tiliroside, hispidulin) and other medium-polar constituents, showed superior performance in lipid-phase protection (β-carotene–linoleic acid model), CUPRAC and antibacterial assays, where more lipophilic or moderately polar molecules are expected to interact more efficiently with biological membranes and hydrophobic microenvironments.
These findings indicate that, within the concentration range tested, both the aqueous and ethanolic extracts of Cerinthe major did not exhibit detectable cytotoxic effects on Saccharomyces cerevisiae, suggesting a favorable biocompatibility profile in this yeast model. Yeast cells are widely used as a simple eukaryotic bioassay system for assessing general cellular toxicity, given their ease of handling and sensitivity to a variety of toxic agents. Nevertheless, the absence of cytotoxicity in yeast does not substitute for toxicity testing on mammalian (especially normal human) cells, which remains essential to define the safety and therapeutic window of Cerinthe major extracts. Therefore, in vitro cytotoxicity assays on relevant normal human cell lines are still required in future studies to complement these preliminary results and to better characterize the safety profile of the extracts.
Although the plant material was collected in 2022 and properly dried and stored until analysis, the present study provides, for the first time, an LC–MS/MS-based phenolic profile of Cerinthe major L. combined with a broad panel of bioactivity assays and a consideration of safety-relevant pyrrolizidine alkaloids, which had not been jointly investigated before for this species.
A major limitation of this study is the absence of cytotoxicity testing on normal human cell lines, which is necessary to define the safety profile and therapeutic window of Cerinthe major extracts. Future work will therefore include in vitro cytotoxicity assays on appropriate human cells to complement the present findings
This study also has some limitations that should be acknowledged. First, the LC–MS/MS analysis was primarily qualitative and focused on phenolic compounds; absolute quantification of individual metabolites and a comprehensive assessment of other classes such as pyrrolizidine alkaloids and meroterpenoids were beyond the scope of the present work. Second, all bioactivity data (antioxidant, enzyme inhibition, photoprotective and antimicrobial assays) were obtained in vitro using crude extracts, without fractionation, toxicity assessment or in vivo confirmation. As a result, the observed activities cannot yet be directly extrapolated to clinical efficacy or safety. Future studies should therefore include quantitative LC–MS/MS profiling, bioassay-guided fractionation, toxicity testing and in vivo models to better define the therapeutic potential and safety margins of Cerinthe major preparations.
Conclusions
In conclusion, aqueous and ethanolic extracts of Cerinthe major L. from Algeria showed a complex phytochemical profile rich in phenolic compounds, together with a characteristic mineral composition. The extracts exhibited noteworthy antioxidant, anti-cholinesterase and anti-inflammatory activities, which may support some of the traditional uses of this species and highlight its potential as a source of bioactive constituents. In addition, the in vitro cytotoxicity assay on Saccharomyces cerevisiae indicated no detectable toxic effects within the tested concentration range, suggesting a favorable biocompatibility in this simple eukaryotic model. However, an important limitation of the present work is the absence of cytotoxicity assessment on normal human cell lines, which is essential to define the safety profile and therapeutic window of these extracts. Therefore, further studies, including in vitro cytotoxicity assays on relevant normal human cells, in vivo evaluations, and isolation of the most active compounds, are warranted to clarify the pharmacological relevance and safety of Cerinthe major.
Footnotes
Abbreviation
Acknowledgments
The authors gratefully acknowledge the technical and logistical support provided by the team of the Centre de Recherche en Biotechnologie (C.R.B.T), Constantine 25000, Algeria; the staff of the VTRS Laboratory, University of El Oued, B.P. 789, 39000 El Oued, Algeria; and the team of CRAPC Ouargla, Algeria. Their assistance with sample preparation, instrumental analyses, and laboratory facilities was essential for the completion of this work.
Ethical Approval
This study is not applicable to both human and/or animal studies that require ethical approval.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data that supports the findings of this study are available within the article.

