Abstract
Introduction
Reactive oxygen species (ROS) represent a group of one-electron oxygen reduction products
Dietary polyphenols are involved in the regulation of lipid and protein homeostasis and longevity, and exhibit antiinflammatory and antioxidant functions. Some polyphenols, such as curcumin, were even demonstrated to have significant neuroprotective potentials due to their antiamyloid function.
4
Curcumin is a natural polyphenol that is mainly isolated from turmeric. In recent decades, curcumin has drawn increasing attention and its biological activities were extensively studied
The rat pheochromocytoma cell line PC12 was derived from a transplantable male rat adrenal pheochromocytoma. PC12 cells differentiated and showed neurite outgrowth in response to nerve growth factor or other stimuli, while removal of stimuli led to degeneration processes and cells resumed to proliferate. 12 PC12 cells have been developed to specific differentiated states with different morphologies and characteristics, namely nondifferentiated, low-differentiated (LD) and high-differentiated (HD) PC12 cells. As cell differentiation is enhanced, the PC12 cell synapses increase and its neuronal properties are improved, exhibiting tighter connection and synaptic intertwining, as well as exacerbation in drug susceptibility. 13 Nondifferentiated PC12 cells do not show neurogenic characteristics, however, the differentiated PC12 cells showed improved neurogenic characteristics along with cell differentiation. 14 The cell differentiation level may in part reflect the different development stages of neurodegenerative diseases. 15 H2O2 can diffuse across cellular membranes to generate hydroxyl radicals. H2O2-induced oxidative damages may cause apoptotic changes and/or eventually lethality of a variety of cells, including neurons. 16 In recent years, PC12 cells have been applied to establish various neurodegenerative disease cell models upon exposure to H2O2 or other stimuli to explore the antioxidants’protective effects. 17 - 21 For instance, Siddiqui et al reported that curcumin pretreatment had a better protective effect than co- and post-curcumin treatment again H2O2 damage in PC12 cells. 18 Recently it was also demonstrated that curcumin alleviated arsenic-, lidocaine-, and rotenone-induced cytotoxicity in PC12 cells.19- 21
Neurodegenerative diseases are characterized by impaired neurogenesis, which includes the deterioration of proliferation, differentiation and migration of nerve cells. 22 The degree, rate, and quality of neuronal differentiation are crucial to the progression of neurodegenerative diseases. However, the association between diverse cell differentiations and cellular responses to external stimuli in PC12 cells was ignored in the majority of studies. This may have hampered the application of antioxidants in alleviating neurodegenerative diseases. The responses of distinct differentiated PC12 cells to external oxidative stimuli varied. And it remained to be elucidated how variant differentiated PC12 cells respond to the same dietary antioxidant. The current study aimed to investigate the oxidative stress responses in PC12 cells with varied differentiation conditions, which may mimic specific aspects of neurodegenerative diseases. Thus, the study may provide insights into the mechanism underlying the progression of neurodegenerative diseases and the potential therapeutic application of polyphenols. Therefore, taking advantage of the LD and HD PC12 cell models, the current study investigated how curcumin protects different PC12 cells against H2O2-induced oxidative damage and the possible functional mechanism. Aging will cause dysfunction of synaptic mitochondria, which show increased ROS generation and oxidative stress, so old mice were expected to have elevated levels of oxidative stress.23,24 Therefore, the antioxidant properties of curcumin were further examined in old-aged mice. This may provide insights into the development and application of curcumin as a food-borne antioxidant for ameliorating neurodegenerative diseases.
Results
Curcumin Pretreatment Prevented H2O2-Induced Oxidative Damages in PC12 Cells
Curcumin has been used for the treatment and/or prevention of neurodegenerative diseases with low cytotoxicity. Although the cell differentiation degree was not mentioned in most of the studies, PC12 cells have been used as a model to investigate the protective effect of curcumin on nerve injury and the potential mechanism.25,26 In the current study, H2O2 was used to induce oxidative damage in distinct differentiated PC12 cells. As shown in Figure 1A, the viability of LD and HD PC12 cells remained constant in the presence of 10 μM H2O2. However, the cell viability began to decrease along with elevated H2O2 amount. After treatment with 20 μM H2O2 for 4 h, the viability of both cell types was reduced by about 10% (
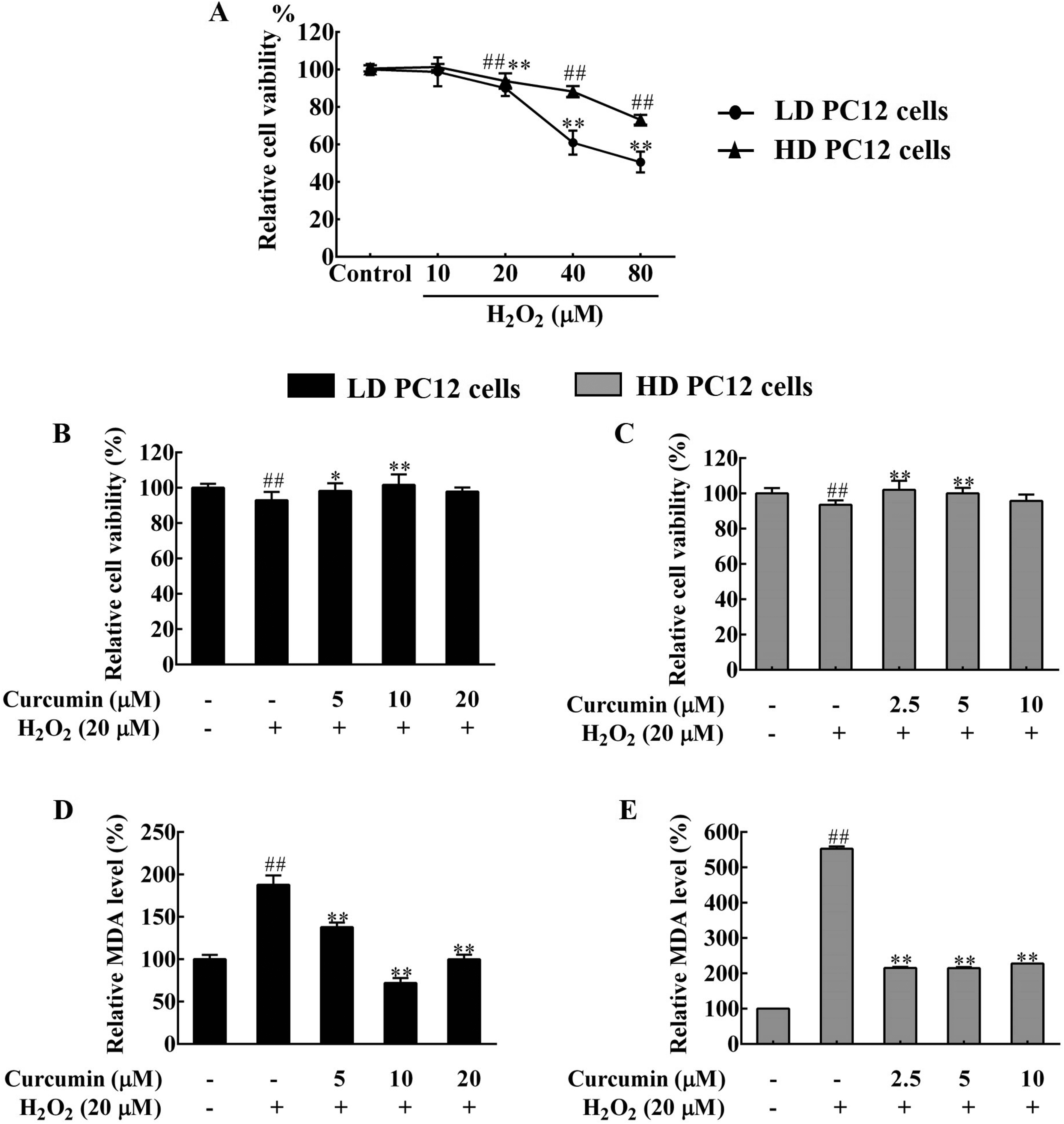
Curcumin alleviated H2O2-induced oxidative damages in differentiated PC12 cells. The cells were treated with different amounts of curcumin as specified for 24 h, followed by exposure to 20 μM H2O2 for 4 h. (A) H2O2 treatment reduced the viability of LD and HD PC12 cells. The cells were treated with 10 to 80 μM of H2O2 for 4 h before measurement of cell viability by the MTT assay. The viability of control cells (control) was set as 100%. (B) and (C) Curcumin pretreatment restored cell viability in H2O2-treated LD (B) and HD (C) PC12 cells. (D) and (E) Curcumin pretreatment partially reduced lipid peroxidation in H2O2-treated LD (D) and HD (E) PC12 cells. The cell viability and the MDA level of the negative control cells were set as 100%. ##
To address how curcumin protects H2O2-treated PC12 cells, PC12 cells were treated with curcumin for 24 h and then exposed to H2O2 for 4 h, followed by measurement of cell viability and lipid peroxidation level. LD PC12 cells were treated with 5, 10, or 20 μM curcumin, and HD PC12 cells were treated with 2.5, 5, or 10 μM curcumin, due to the fact that these dosages did not affect cell viability (Supplemental Figure S1). The results showed that 20 μM H2O2 significantly reduced the viability of both LD and HD PC12 cells. However, curcumin pretreatment prevented H2O2-induced reduction in cell viability and instead elevated PC12 cell viability. Pretreatment with 10 μM curcumin in LD cells or 2.5 μM curcumin in HD cells restored cell viability by around 10% (
Curcumin Enhanced the Antioxidant Capacity of H2O2-Treated PC12 Cells
Cellular oxidative stress may result from excessive accumulation of ROS in response to different stimuli. The mitochondria are the main organelles responsible for ROS production and elimination. As shown in Figure 2A and B, the intracellular ROS levels in LD and HD PC12 cells were significantly increased upon H2O2 treatment. Consistently, the mitochondrial ROS levels were increased by around 20% (
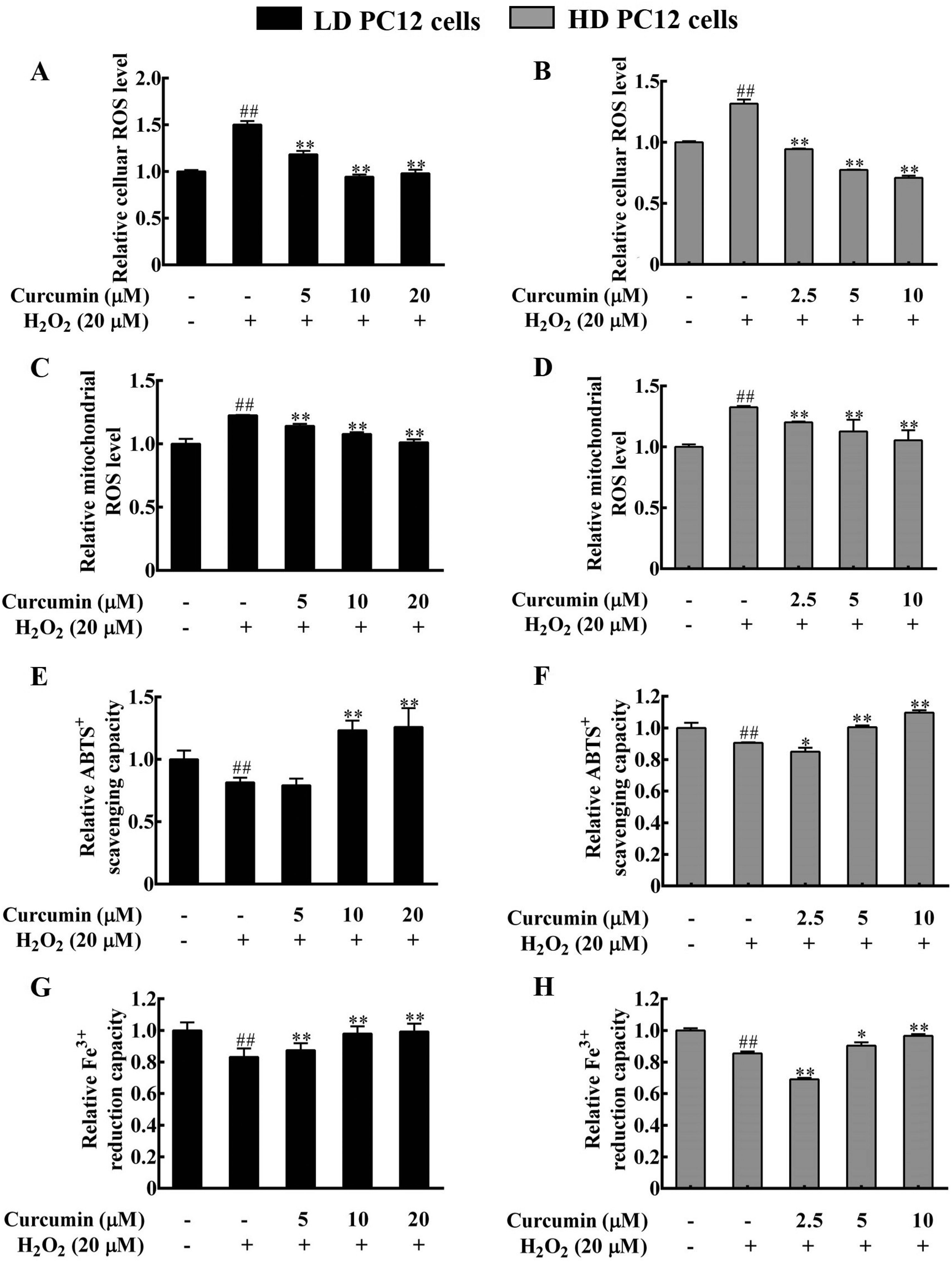
Curcumin pretreatment prevented H2O2-induced decrease of cellular antioxidant capacity in PC12 cells. PC12 cells were treated with different amount of curcumin as specified for 24 h, followed by exposure to 20 μM H2O2 for 4 h. (A-D) Curcumin treatment prevented H2O2-induced increment of cellular and mitochondrial ROS levels. (A) and (B) the intracellular ROS levels, (C) and (D) the mitochondrial ROS levels. The ROS levels of the negative control cells were set as 1.0. (E-H) Curcumin restored the total cellular antioxidant capacity of H2O2-treated PC12 cells, as indicated by the enhanced ABTS+ free radical scavenging capacity (E, F) and the Fe3+ reducing capacity (G, H) in LD (E, G), and HD (F, H) PC12 cells. The total antioxidant capacity of the negative control cells was set as 1.0. ##
There are a variety of antioxidants inside the cells, which reflects the cell's ability to scavenge free radicals. The total cellular antioxidant capacity of PC12 cells was further examined by the ABTS and the Fe3+ reduction ability (FRAP) methods. In line with increased ROS levels, the total antioxidant capacity of LD and HD PC12 cells was significantly decreased upon 20 μM H2O2 treatment, indicating reduced cellular antioxidant capacity (Figure 2E-H). However, 10 or 20 μM curcumin pretreatment significantly increased the total cellular antioxidant capacity of H2O2-treated LD PC12 cells (Figure 2E and G). Similarly, the cellular antioxidant capacity was significantly improved upon 5 or 10 μM curcumin pretreatment in H2O2-treated HD PC12 cells (Figure 2F and H), based on the ABTS and the FRAP assay results. Therefore, curcumin treatment enhanced the cellular antioxidant capacity of PC12 cells, which prevented H2O2-induced oxidative stress. In addition, HD cells seemed to be more sensitive than LD cells to curcumin treatment and required a lower dosage of curcumin to reduce oxidative stress.
Curcumin Prevented H2O2-Induced Deterioration of Antioxidant Enzymes in PC12 Cells
The cells contain both enzymatic and nonenzymatic antioxidant systems to scavenge excessive ROS. The activity of major cellular antioxidant enzymes, including SOD, CAT, and GPx, can be used as an effective indicator of the body's free radical scavenging ability. SOD mainly catalyzes superoxides into O2 and H2O2, CAT and GPx further decompose H2O2 into H2O molecules. 27 Compared with the negative control cells, LD PC12 cells showed significantly reduced activities of CAT, SOD, and GPx upon exposure to 20 μM H2O2 for 4 h (Figure 3A-C). Similarly, the enzyme activities of CAT, SOD, and GPx in H2O2-treated HD PC12 cells were significantly decreased by around 35%, 20%, and 50%, respectively (Figure 3D-F). Thus, H2O2 treatment deteriorated the antioxidant enzyme system of both PC12 cells. In addition, the response of different antioxidant enzymes to H2O2 treatment varied, as SOD was the least sensitive and GPx was the most sensitive. Furthermore, curcumin attenuated the effect of H2O2 on antioxidant enzymes, as H2O2-reduced activities of antioxidant enzymes were significantly elevated upon curcumin pretreatment (Figure 3A-F), which was consistent with the response of cellular antioxidant capacity to combined curcumin and H2O2 treatment.

Curcumin significantly restored the activity and expression of major antioxidant enzymes in H2O2-treated PC12 cells. PC12 cells were treated with different amounts of curcumin for 24 h before exposure to 20 μM H2O2 for 4 h. (A-F) H2O2-reduced antioxidant enzyme activities were restored by 24 h pretreatment with curcumin. (A, D) CAT, (B, E) SOD, (C, F) GPx. (A-C) LD PC12 cells, (D-F) HD PC12 cells. (G) Representative western blot images of mitochondrial antioxidant enzymes. The activity and expression level of antioxidant enzymes in the negative control cells were set as 1.0. ##
The varied enzyme activity could be attributed to altered gene expression. CAT, SOD, and GPx were all distributed in the mitochondria and their mitochondrial isoforms were encoded by
Curcumin Restored Mitochondrial Oxidative Phosphorylation in H2O2-Treated PC12 Cells
ROS are the byproducts of cellular oxidative phosphorylation, which not only provides the cells with the most ATP energy to support basal life requirements, but is also responsible for maintaining metabolic homeostasis. 28 The present study further evaluated oxidative phosphorylation in PC12 cells with distinct differentiation types. As shown in Figure 4, 4 h of H2O2 treatment significantly reduced the ATP level (Figure 4A and B), and increased the ADP/ATP ratio in both LD and HD PC12 cells (Figure 4C and D). However, a certain amount of curcumin pretreatment significantly restored the ATP level and the ADP/ATP ratio. It is worth noticing that a low dose of curcumin (2.5 μM and 5 μM) significantly reversed H2O2's deteriorated effect on oxidative phosphorylation in HD PC12 cells, while a higher dose of curcumin (10 μM) did not show further recovering activity. In addition, a higher amount of curcumin seemed to restore oxidative phosphorylation in LD PC12 cells. The exact regulatory effect of curcumin on mitochondria function during oxidative stress remained to be further studied. In summary, the above data indicated that an appropriate amount of curcumin restored oxidative phosphorylation in H2O2-damaged PC12 cells.

Curcumin prevented the reduction of mitochondrial oxidative phosphorylation in H2O2-treated PC12 cells. PC12 cells were treated with different amounts of curcumin for 24 h before exposure to 20 μM H2O2 for 4 h. (A) and (B) The ATP level, (C) and (D) The ADP/ATP ratio. The ATP level and the ADP/ATP ratio of negative control cells were set as 1.0. ##
Curcumin Improved the Antioxidant Capacity of KM Mice
The antioxidant effect of curcumin on old-aged mice were further examined. The change in mouse body weight reflects the influence of curcumin on mouse health condition. 29 During the 30-day experimental period, the weight of mice was recorded weekly, and the results were as shown in Figure 5A. After 30 days of continuous gavage, there was no significant change in the body weight of mice in each group, indicating that the applied curcumin dosage and experimental period had no obvious effect on mouse body weight and were least toxic to mice. Curcumin's effect on mouse antioxidant capacity was further analyzed. As shown in Figure 5B and C, the total antioxidant capacity of mouse liver tissues was significantly increased upon curcumin administration, compared to that of the carboxymethylcellulose (CMC)-treated group. However, there was no significant induction by 200 mg/kg curcumin in mouse ferric reduction ability. And there was no obvious dose-dependent effect of curcumin on mouse antioxidant capacity. The exact reasons are still under investigation. The elevated liver antioxidant capacity may be attributed to enhanced enzyme activities of CAT, SOD and GPx in the liver (Figure 5D-F), which were significantly increased upon curcumin supplement. In addition, no gender-specific results for curcumin treatment were observed in this study, as no specific difference was observed between male and female mice tested (data not shown). Therefore, curcumin has antioxidant properties and may alleviate age-related oxidative damage by enhancing the antioxidant enzymes of mice.

Curcumin improved the antioxidant capacity of KM mice. The mice underwent continuous gavages of curcumin at 0.1 mL/10 g/d for 30 days. The body weight of mice was recorded weekly, followed by subsequent assays. (A) Variations in mouse body weight. (B) and (C) The total antioxidant capacity of liver tissue, which was measured by the ABTS (B) and FRAP methods (C). (D-F) The relative enzyme activities of CAT (D), SOD (E), and GPx (F). The total antioxidant capacity and the antioxidant enzyme activities of mouse liver in untreated mice (Con) were set as 1.0. *
Discussion
Although the precise mechanisms of oxidative stress-induced neuronal damage were not completely characterized, searching for effective antioxidants to relieve neuronal damage has been a hot issue. Dietary polyphenols, especially curcumin, have significant potential for the treatment of neurodegenerative diseases. 4 It was shown that curcumin inhibited the formation and promoted the disaggregation of amyloid plaques, as well as restoring their redox balance. 30 In addition, curcumin inhibited Aβ-induced apoptosis and ROS-mediated DNA damage to promote the regeneration and recovery of neurons. 31 However, the underlying mechanisms remained largely unknown. The current study proved the protective effect of curcumin in PC12 cells, which have both high and low differentiated types, against H2O2-induced oxidative damage. Meanwhile, the antioxidant enzymes were enhanced by curcumin to improve the total cellular antioxidant capacity. Curcumin pretreatment also improved mitochondrial oxidative phosphorylation, indicating enhanced mitochondrial functions to scavenge excessive ROS. Moreover, PC12 cells of different differentiated types showed varied sensitivities to curcumin. And HD PC12 cells seemed to be more sensitive to curcumin than LD PC12 cells, as lower-dosed curcumin exhibited a protective effect against H2O2-induced oxidative damages in HD PC12 cells via enhancing antioxidant enzymes and mitochondrial phosphorylation. This may be related to the structure, characteristics and interaction of PC12 cells, however, the exact reasons remain to be clarified. This study also demonstrated that curcumin improved the antioxidant capacity of KM mice, which further strongly proved that curcumin was an effective natural antioxidant that have great potential to ameliorate age-related neurodegenerative diseases.
ROS are involved in the development and progression of neurodegenerative diseases. Under physiological conditions, the balance between ROS production and elimination is essential for normal cellular function and metabolism. The current study showed that H2O2 deteriorated the cellular redox balance by increasing the intracellular ROS levels and lipid peroxidation, which was prevented by curcumin pretreatment. This was similar to the results of Fu et al who demonstrated that curcumin prevented H2O2-induced cytotoxicity and apoptosis of PC12 cells by reducing the ROS levels. 32 Later Farkhondeh et al also demonstrated that curcumin attenuated glucose/serum deprivation-induced oxidative stress in PC12 cells by reducing the cellular ROS levels. 25
The antioxidant defense system serves as the frontline of cells to scavenge free radicals. The cellular antioxidant enzymes mainly include SOD, CAT, and GPx, which catalyze different radical reactions and are distributed in various cellular compartments. This study showed that H2O2 treatment led to reduced activities of CAT, SOD, and Gpx to different extent, which was in consistent with previous reports.33,34 The reduced antioxidant enzyme activity may be attributed to an increment of intracellular free radicals by H2O2, which may attack proteins and/or other biological macromolecules and eventually led to protein peroxidation and damage or decreased enzyme expression. 35 It was of note that H2O2 showed less reduction effect on SOD than other antioxidant enzymes in PC12 cells. Although the exact mechanism remained to be clarified, this might be explained by that SOD catalyzed H2O2 production, which facilitated SOD homeostasis and prevented its deterioration by H2O2. Thus, the protective effect of curcumin on SOD was diminished in H2O2-treated cells. The results showed that curcumin pretreatment reversed the deterioration of H2O2 on antioxidant enzyme activities in both LD and HD PC12 cells. Consistently, the expression of mitochondrial antioxidant enzymes was restored by curcumin, without significant difference in LD and HD PC12 cells. These findings were similar to a previous study, which showed that curcumin reversed H2O2-induced antioxidant enzyme reduction in IPEC-J2 cells. 36 In summary, curcumin facilitated the maintenance of the antioxidant enzyme system, increased the tolerance against H2O2 and protected the cells against oxidative damages.
Mitochondria are the “energy factories” of cells and supply the cells with the most energy through oxidative phosphorylation to fuel essential cellular functions. 37 The aberrant mitochondrial oxidative phosphorylation system is closely associated with mitochondrial dysfunction, which represents a common feature of many neurodegenerative diseases. To determine curcumin's protective effect on mitochondrial function in oxidative-damaged PC12 cells, mitochondrial oxidative phosphorylation was further measured in H2O2-treated cells with or without curcumin. H2O2 treatment reduced ATP production and increased the ADP/ATP ratio, indicated decreased oxidative phosphorylation in PC12 cells. However, a certain amount of curcumin (5-20 μM for LD cells and 2.5-5 μM for HD cells) alleviated the deterioration effect of H2O2, suggesting enhanced mitochondrial function to match the cellular energy demands during oxidative stress. In addition, it was shown that 10 μM curcumin treatment did not restore, but deteriorated oxidative phosphorylation in HD PC12 cells. The exact response and mechanism are still under investigation.
As the differentiation level of PC12 cells increases, more neuron-like cells gradually appear, which facilitates the study of neurite growth and drug sensitivity. Compared with LD PC12 cells, HD PC12 cells in physiology are more similar to dopaminergic neurons of the midbrain. 38 The drug sensitivity of PC12 cells varies and is associated with their differentiation level. Tian et al demonstrated undifferentiated LD and HD PC12 cells had different responses and HD cells were more sensitive to corticosterone. 39 In this study, LD and HD PC12 cells also showed different responses to curcumin. Curcumin seemed to significantly enhanced antioxidant enzymes and mitochondrial oxidative phosphorylation at lower dose (2.5-5 μM) in HD PC12 cells, while higher amount of curcumin (10 μM) may be required to reach a similar effect in LD PC12 cells. It is possible that high differentiated PC12 cells are more sensitive to external stimuli, thus showing quick response to even low-dosed curcumin. However, low differentiated cells may be more capable to buffer or reduce the damaging effect from external stimuli, thus being less sensitive to curcumin protection. The exact reason requires further investigation.
Ageing is the main risk factor for most neurodegenerative diseases, and it is closely related to inflammation and oxidative stress. It is a complex process that differentially impacts multiple cognitive, sensory, neuronal and molecular processes.
24
Radulescu et al reported that mice showed neuronal circuit dysfunction and antioxidant dysfunction as early as 4 to 10 months old.
40
Olesen et al showed that mice experienced significant oxidative stress and premature dysfunction in synaptic mitochondria at 6 months old, as indicated by increased ROS generation and reduced ATP production.
23
As the body's metabolic center, liver cells are rich in mitochondria which makes the liver vulnerable to oxidative stress. To further evaluate curcumin's antioxidant activity
Conclusion
In summary, curcumin had a protective effect in both H2O2-damaged LD and HD PC12 cells by reducing lipid peroxidation and oxidative stress, and eventually improved cell viability.
Materials and Methods
Cell Culture and Treatment
The LD (TCR 8) and HD (TCR 9) PC12 cell lines were obtained from the cell bank of the Chinese Academy of Sciences (Shanghai). The cells were grown in Hyclone's RPMI 1640 medium (Logan) supplemented with 10% Gemini's fetal bovine serum (FBS) (Calabasas) and 1% Gibco's penicillin/streptomycin antibiotic mixture (Grand Island) in a humidified cell incubator at 37 °C with 5% CO2. Curcumin from Sigma-Aldrich (Saint Louis) was dissolved in dimethyl sulfoxide (DMSO) (Amresco, Solon) and added to cells at 1% (v/v) for 24 h before subsequent assays, DMSO served as the solvent control and was added to the medium at 1% (v/v). To induce oxidative damages, H2O2 (Xilong, Shantou) was added to the cells for 4 h. Except for specifications, PC12 cells were treated with curcumin (1%, v/v) for 24 h, and then incubated with H2O2 for 4 h before subsequent assays.
Cell Viability Assay
Cell viability was measured by the MTT assay. Briefly, cells were plated in a 96-well plate at a density of 1 × 104 cells per well overnight and then treated as specified. The MTT powder (98%) from Sigma − Aldrich was dissolved in phosphate buffered solution and then added to the cells to final 0.5 mg/mL at the indicated time point, followed by further incubation at 37 °C for 4 h. The medium was then discarded and 150 μL DMSO per well was added for an additional 10 min with continuous shaking. The optical absorbance was determined at 490 nm on a Biotek microplate reader (Winooski). The relative cell viability was presented as the percentage of viable cells over the original control cells.
Lipid Peroxidation Measurement
Cellular lipid peroxidation was assessed by quantifying the amount of cellular malondialdehyde (MDA), a lipid peroxidation product, using the MDA assay kit from Beyotime. Briefly, 100 μL cell lysate was incubated with 200 μL TBA working solution at 100 °C for 15 min. The reaction mixture was centrifuged at 1000 g for 10 min, followed by measuring the supernatant's absorbance at 532 nm on a Biotek microplate reader according to the manufacturer's instructions. The MDA level of control cells was set as 100%.
Cellular ROS Level Measurement
The 2′, 7′−dichlorofluorescein diacetate (DCFH − DA) was used for quantification of the intracellular ROS levels according to a previously described method. 41 Briefly, the cells were harvested and labeled with 1 μM DCFH-DA in Opti-MEM for 30 min at 37 °C. The cells were then washed three times with and finally resuspended in Opti-MEM. The green fluorescence intensity was determined as the ROS level with a Millipore's Guava EasyCyte 6-2L flow cytometer (Billerica) using GuavaSoft 3.1.1 software.
Mitochondrial ROS Level Measurement
The MitoSOX Red reagent from ThermoFisher was used to detect mitochondrial superoxide formation as previously described. 42 Briefly, the cells were harvested and labeled with 5 μM MitoSOX reagent in PBS for 30 min at 37 °C. Followed by three washes with PBS, the red fluorescence intensity was measured as the mitochondrial ROS level with a Millipore's Guava EasyCyte 6-2L flow cytometer using GuavaSoft 3.1.1 software.
Total Cellular Antioxidant Capacity Detection
The assays for total cellular antioxidant capacity, including the ABTS free radical scavenging ability (ABTS) and the Fe3+ reduction ability (FRAP), were carried out following the manufacturer's instructions using the assay kits from Beyotime.
Measurement of Antioxidant Enzyme Activities
The activities of major antioxidant enzymes were measured using the superoxide dismutase (SOD) and the glutathione peroxidase (GPx) assay kits from Jiancheng Biotechnology, and the catalase (CAT) assay kit (Beyotime), respectively, following the manufacturers’ instructions.
Measurement of Protein Expression Level
The cellular protein level was measured by western blot. The cell lysate was collected by lyzing the cells with Beyotime's Cell lysis buffer for Western and IP on ice for 20 min, followed by centrifugation for 12 000 g for 10 min (4 °C) to recover the supernatant. A certain amount of lysate was subjected to 12% SDS − polyacrylamide gel electrophoresis and then transferred to Millipore's polyvinylidene difluoride membrane (Danvers). The membrane was blocked with 5% nonfat milk for 1 h at room temperature and hybridized with the primary antibody overnight at 4 °C, followed by incubation with the horseradish peroxidase − conjugated secondary antibody (Boster) at room temperature for 45 min. The membrane was finally developed using ZetaLife's WesternLumaxLight Superior Kit. The primary antibodies used in this study included anti-CAT (Cell Signaling Technology, #12980); anti-SOD2 (Abcam, ab13533); anti-GPx1 (GeneTex, GTX116040) and anti-GAPDH (Affinity, AF7021).
Cellular ATP and ADP/ATP Measurement
Cellular ATP and ADP/ATP ratio were measured based on Sigma's bioluminescent assays. Briefly, cells were first incubated with the ATP reagent in a 96-well assay plate for 1 min at room temperature and luminescence was taken as the ATP level (RLUA) on a Biotek microplate reader as previously described. 42 Upon 10 min of addition incubation, luminescence was taken as the residual ATP level (RLUB) prior to ADP assay, and the ADP level (RLUC) was quantified upon addition incubation with the ADP reagent for 1 min. The ADP/ATP ratio was calculated as (RLUC-RLUB)/RLUA.
Animal Experiments
KM mice (SPF level) at 6-month-old were obtained from HFK Bioscience Co. Ltd. All animals were maintained under a controlled environment (22 ± 0.5 °C, 50%-70% humidity, and 12 h light–dark cycle). The mice were fed with a standard pellet diet and clean drinking water. Upon one week of acclimatization, the mice were randomly divided into 6 groups: control group (Con), CMC sodium-treated group (CMC), 50 mg/kg body weight curcumin-treated group, 100 mg/kg body weight curcumin-treated group, 200 mg/kg body weight curcumin-treated group and 200 mg/kg body weight vitamin C (VC)-treated group (VC). 43 - 45 Curcumin was dissolved in CMC and administrated by gavage and VC served as a positive control. There may be sex differences across different oxidative stress-related neurodegenerative diseases.46- 48 To validate the potential gender-specific antioxidant effects of curcumin in old-aged mice, each group included 3 male and 3 female mice. All procedures were carried out in accordance with the guidelines for the care and use of laboratory animals. All experiments were approved by the Animal Ethics Committees of Jimei University.
The experimental groups were administered with curcumin (Solarbio) suspension at different dosages of 50 mg/kg, 100 mg/kg, and 200 mg/kg by gavage, respectively. The CMC group and VC group were administered with 0.5% CMC (Sinopharm) and 2 mg/mL VC solution (Xiya) via gavage, respectively. All groups of mice were given continuous gavages at 0.1 mL/10 g/d for 30 days and the weight of mice was recorded weekly. Mice were sacrificed and the liver tissues were obtained to measure the total antioxidant capacity and antioxidant enzyme (CAT, SOD, GPx) activities as described above.
Statistical Analysis
The data were presented as the mean ± SD from three independent experiments and analyzed using the SPSS 23.0 statistical software. The differences between samples were evaluated by one-way analysis of variance (ANOVA).
Supplemental Material
sj-doc-1-npx-10.1177_1934578X231206228 - Supplemental material for Curcumin Protects Pheochromocytoma Cells Against Hydrogen Peroxide-Induced Oxidative Stress
Supplemental material, sj-doc-1-npx-10.1177_1934578X231206228 for Curcumin Protects Pheochromocytoma Cells Against Hydrogen Peroxide-Induced Oxidative Stress by Xiaoting Sun, Lu Chen, Lingli Lin, Kuo-Ting Ho, Shi-Ying Huang, Jian Li, Jingwen Liu, Zhengxiao Zhang and Guiling Li in Natural Product Communications
Footnotes
Authors Contributions
Xiaoting Sun: methodology, investigation, data curation, and writing—original draft preparation; Lu Chen: methodology, investigation, and data curation; Lingli Lin: methodology and writing—original draft preparation; Kuo-Ting Ho, Shi-Ying Huang, and Jian Li: resources and data curation; Jingwen Liu: resources, data curation, and funding acquisition; Zhengxiao Zhang: supervision, writing—original draft preparation, projection administration, and funding acquisition; Guiling Li: conceptualization, supervision, writing—original draft preparation, projection administration, and funding acquisition.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The experimental protocols were approved by the local ethics committee following the regulations for the treatment of live animals at Jimei University (Xiamen, China) with the approval number of JMU202203005.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant numbers: 31771972, 32202068, 42076086), Natural Science Foundation of Fujian Province (grant numbers: 2017J01447, 2020J01676, 2022J01332), the High-level Talent Innovation Project of Quanzhou (grant number 2019CT007) and the Science and Technology Project of Quanzhou (grant number 2017Z003), .
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
