Abstract
As the aging phenomenon continues to increase, the incidence of neurodegenerative diseases continues to increase annually. As one of the significant contributive factors of neurodegenerative diseases, oxidative stress damage has received extensive attention in recent years. Oxidative stress plays an important role in neuronal damage through various apoptotic mechanisms related to neurodegenerative diseases. The use of natural antioxidants to combat oxidative stress may be a useful approach in delaying disease progression. In this study, we explored the neuroprotective effect of hyperoside on rat pheochromoma (PC12) cells. Specifically, the antioxidant effect and mechanism of hyperoside in hydrogen peroxide (H2O2)-induced cellular cytotoxicity were investigated. Our results showed that hyperoside could significantly increase the survival rate of rat PC12 cells when exposed to H2O2. In addition, hyperoside regulated the expression of genes and proteins in the corresponding pathways by up-regulating the phosphatidylinositol-3-kinase (PI3K), protein kinase B (Akt), and light chain 3β (LC3B) pathways and down-regulating the nuclear factor-ᴋ-gene binding (NF-κB), Bcl2-associated X (Bax), cysteinyl aspartate specific proteinase 3 (Caspase 3), and P62 pathways, thereby inhibiting cell apoptosis. Therefore, hyperoside can effectively inhibit H2O2-induced oxidative stress damage by regulating inflammation, autophagy, and apoptosis-related pathways.
Neurodegenerative diseases are caused by progressive neuronal death in the brain and spinal cord, leading to damage to the nervous system resulting in cognitive and motor dysfunction. 1,2 Several studies have shown that oxidative stress is closely related to the pathogenesis of neurodegenerative diseases. 3 -5 In vivo studies show that the imbalance of oxidative and antioxidant systems is the cause of oxidative stress. Oxidative stress produces a large number of free radicals. Neurons are very sensitive to free radicals and can be easily damaged. The causes of oxidative stress also include protein misfolding caused by excessive reactive oxygen species (ROS), activation of glial cells, and mitochondrial dysfunction leading to apoptosis. 6 -8 Therefore, the improvement of or reduction in oxidative stress injury is closely related to the management of neurodegenerative diseases.
Hyperoside {quercetin 3-O-β-D-galactoside (Figure 1)}, is a flavanol glycoside found in Acanthopanax senticosus (Rupr. & Maxim.) Harms (Araliaceae) . 9 This compound is effective in alleviating oxidative injury, inhibiting apoptosis, and reducing inflammation. 10 Studies report that hyperoside can protect rat cardiomyocytes from apoptosis induced by ischemia-reperfusion injury. 11 It has also been shown that hyperoside can reduce H2O2-induced osteoblast MC3T3-E1 phosphorylation Jun terminal kinase (JNK) and lower P38 levels by inhibiting mitogen-activated protein kinase (MAPK) signaling; it reduces H2O2-induced cell apoptosis and apoptosis-related proteins. 12 However, there are only a few studies that have explored the protective mechanism of hyperoside in alleviating the oxidative stress injury of neurons. In this study, PC12 rat pheochromocytoma cells, a sympathetic nervous system tumor-cell line widely used to study neuronal injury-related models and highly similar to dopaminergic neurons in structure and function, were selected to establish an H2O2-induced oxidative-injury model. Herein, we explored the mechanism of hyperoside in protecting PC12 cells from damage owing to oxidative stress.
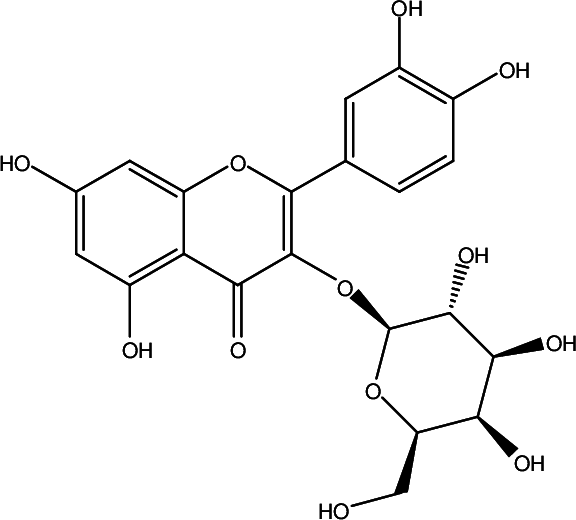
Structure of hyperoside.
Materials and Methods
Reagents and Drugs
Hyperoside was purchased from Shanghai Yuanye Biological Technology Co., Ltd. Dulbecco’s modified Eagle medium (DMEM) and MTT were obtained from Beijing Solarbio Sciences & Technology Co., Ltd., fetal bovine serum (FBS) from Genimi Biological Products Co., Ltd, anti-phospho-PI3K P85 antibody (Y458)/p55/(Y199), anti-PI3K antibody (19H8), anti-phospho Akt antibody (S473) (D9E), anti-Akt antibody (ab179463)(EPR167), anti-phospho NF-κB P65 antibody (bs-17502R), and anti-NF-κB P65 antibody (bs-20160R) from Cell Signaling Technology Inc., anti-Bax antibody (50599, 2-lg), anti-caspase 3 antibody (bs-0081R), anti-LC3B-specific antibody (16396, 1-AP), and anti-P62 antibody (18420, 1-AP) from Proteintech, and β-actin (GB11001) from Servicebio.
Cell Culture and Modeling
PC12 cell line (Institute of Basic Medicine, Chinese Academy of Medical Sciences.) was cultured in DMEM supplemented with 10% FBS, 100 U/mL penicillin, and 100 µg/mL streptomycin, in an incubator with 5% CO2 at 37 °C. PC12 cells were inoculated in a 96-well-plate at a density of 1.25 × 104 cells/well for 24 hours. PC12 cells were treated with 10% H2O2 diluted to different modeling concentrations (700, 600, 500, 400, 300, 200, 100, and 0 μmol/L) with DMEM. After 12 hours, 5 mg/mL of MTT (Beijing Mengyimei Biological Technology Co., Ltd) solution was added to each well and allowed to react for 4 hours, after which the absorbance was detected at 490 nm. The corresponding survival rate was calculated and H2O2 at half lethal concentration was used for further experiments. We selected hyperoside concentrations of 50, 25, and 12.5 µmol/L to pretreat PC12 cells for 12 hours, followed by treatment with 400 µmol/L H2O2 for 12 hours.
Cell Viability Assays
An MTT assay was used to determine the relative viability of PC12 cells after pretreatment with different concentrations of hyperoside. Cells were seeded in a 96-well plate at a density of 1.25 × 104 cells/well with complete medium. After pretreatment with different concentrations of hyperoside (50, 25, and 12.5 μmol/L) for 12 hours based on groupings, PC12 cells were then exposed to H2O2 for 12 hours as described in Section 2.2. Then, 20 µL MTT solution was added (final concentration 5 mg/mL). Subsequently, the supernatant was removed and the formazan crystals were dissolved in dimethyl sulfoxide. The absorbance of the samples was measured at 490 nm.
Reverse Transcription PCR (RT-PCR)
Highly differentiated PC12 cells were seeded in 6 cm cell culture plates at a concentration of 5 × 105 cells/mL. After 24 hours of seeding, cells in the treatment group were treated with 50 μmol/L hyperoside for 12 hours, while control cells were treated with DMEM. All groups, except the control, were treated with 400 μmol/L H2O2 for 12 hours and total RNA was isolated from the cells using TRIzol reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. HiFi-MMLV cDNA kit (Cwbio, Beijing, China) was used to determine the composition of cDNA conforming to the specification. Primer sequences employed in RT-Qpcr are listed in the following:PI3K(Forward: 5’-ACACGGGGGCATTCAAAGAT-3’,Reverse:5’-GTCGTTGTGCCTGTCACCTA-3’),Akt1(Forward:5’-AGCATGGAGTGTGTGGACAG-3’,Reverse:5’-TACAGATGATCCATGCGGGG-3’),β-actint(F:5’-ACTCTGTGTGGATTGGTGGC-3’,R:5’-ACTCTGTGTGGATTGGTGGC-3’),Bax(F:5’-GCCTCCTTTCCTACTTCGGG-3’,R:5’-TTCCCCGTTCCCCATTCATC-3’),Caspase3(F:5’-TCTACCGCACCCGGTTACTA-3’,R:5’-TCAAATTCCGTGGCCACCTT-3’),NF-ƘB(F:5’-GGAGATGGCCCACTGCTATC-3’,R:5’-GCACCTTGGGATGCGTTTTT-3’),LC3B(F:5’-TCCTGAACCCCAGCCATTTC-3’,R:5’-GGCATGGACCAGAGAAGTCC-3’),P62(F:5’-GCCGCCTAAATGGAGTCTGA-3’,R:5’-ACAGTGCGAAACTGACAATGG-3’). The expression levels of PI3K, Akt, NF-κB, Bax, Caspase 3, LC3B, and P62 were determined using an SYBR V R Green PCR Kit (Takara). RT-PCR was conducted using an Applied Biosystems 7500 real-time PCR instrument (ABI, Foster, CA, USA).
Western Blot (WB) Analysis
PC12 cells were seeded in 6 cm cell culture plates at a concentration of 5 × 105 cells/mL. After 24 hours of seeding, cells were treated with 50 µM hyperoside for 12 hours in the treatment group, while control cells were treated with DMEM. Next, cells were treated with 400 μmol/L H2O2 for 12 hours except for the control group. The total soluble proteins were extracted from cultured PC12 cells using RIPA buffer (Beyotime Institute of Biotechnology, Nantong, China) containing the protease inhibitor, PMSF. Protein concentrations were determined using a Pierce BCA protein assay (Thermo Fisher Scientific, USA). Equal quantities of protein samples were separated using SDS-PAGE and then transferred to PVDF membranes (EMD Millipore, USA). Membranes were blocked with 5% skim milk for 3 hours. Subsequently, the proteins were incubated with the following corresponding primary antibodies overnight at 4 °C: anti-β-actin (1:1000, GB11001, Servicebio), P-PI3K [1:500, Y458, Cell Signaling Technology, Inc (CST)], PI3K [1:250, 19H8, Inc (CST)], Akt [1:2000, ab179463, Inc (CST)], P-Akt [1:1000, s473, Inc (CST)], NF-κB [1:1000, bs-20160R, Inc (CST)], P-NF-κB [1:1000, bs-17502R, Inc (CST)], Bax (1:1000, 50599‐2-lg, Proteintech), Caspase 3 (bs-0081R, Bioss), P62 (1:1000, 18420‐1-AP, Proteintech), and LC3B (1:1000, 16396‐1-AP, Proteintech). After washing with TBST, membranes were incubated with HRP-linked secondary antibody (Zsbio, Beijing, China; 1:2000) for 2 hours. Lastly, ECL ultra-sensitive luminescence solution and a Western blotting detection kit were used to visualize the protein bands. Images were captured using a BIO-RAD gel imaging system and the grayscale was measured using Image J software.
Statistical Analysis
All experiments were performed at least three times and the results are expressed as means ± standard deviation (‾X ± SD). One-way analysis of variance was used to examine the differences between treatment groups. All statistical analyses were performed using GraphPad Prism (GraphPad Software, Inc., La Jolla, CA). A difference was considered statistically significant at # P < 0.05 or *P < 0.05.
Results
Protective Effect of Hyperoside on PC12 Cell Viability After H2O2 Treatment
PC12 cells were treated with different doses of hyperoside (0, 400 μmol/L). It was found that hyperoside had no significant effects on cell viability at doses ranging from 0 to 50 μmol/L (Figure 2(A)). As seen in Figure 2(B), hydrogen peroxide significantly decreased the viability of PC12 cells at doses of 200, 300, 400, 500, 600, and 700 μmol/L. Considering that cell viability was inhibited by 50% at 400 μmol/L H2O2, this dose was used to induce the oxidative stress injury in PC12 cells. Therefore, we selected hyperoside concentrations of 50, 25, and 12.5 µmol/L to pretreat PC12 cells for 12 hours, followed by treatment with 400 µmol/L H2O2 for 12 hours. The cell viability was found to be significantly higher than that of the model group (Figure 2(C)).

Protective effect of hyperoside on PC12 cell viability after H2O2 treatment. (
Increased PI3K and Akt mRNA and Its Phosphorylated Protein Expression in Hyperoside-Treated Cells
As shown in Figure 3, compared to the control group, RT-PCR and WB analysis showed that the expression of PI3K, Akt mRNA, and related proteins in the model group (400 μmol/L H2O2) was significantly down-regulated. After 12 hours of treatment of cells with hyperoside (50 μmol/L), the mRNA expression of PI3K and Akt was significantly higher than that of the cells induced by H2O2 (Figure 3(A) and (B)). As mRNA expression increased, the protein expression levels of P-PI3K and P-Akt also increased significantly (Figure 3(C), (D) and (E)). Our results showed that hyperoside may regulate oxidative stress damage by regulating the PI3K/Akt signaling pathway and significantly promote the expression of PI3K and Akt genes and corresponding proteins after oxidative damage.
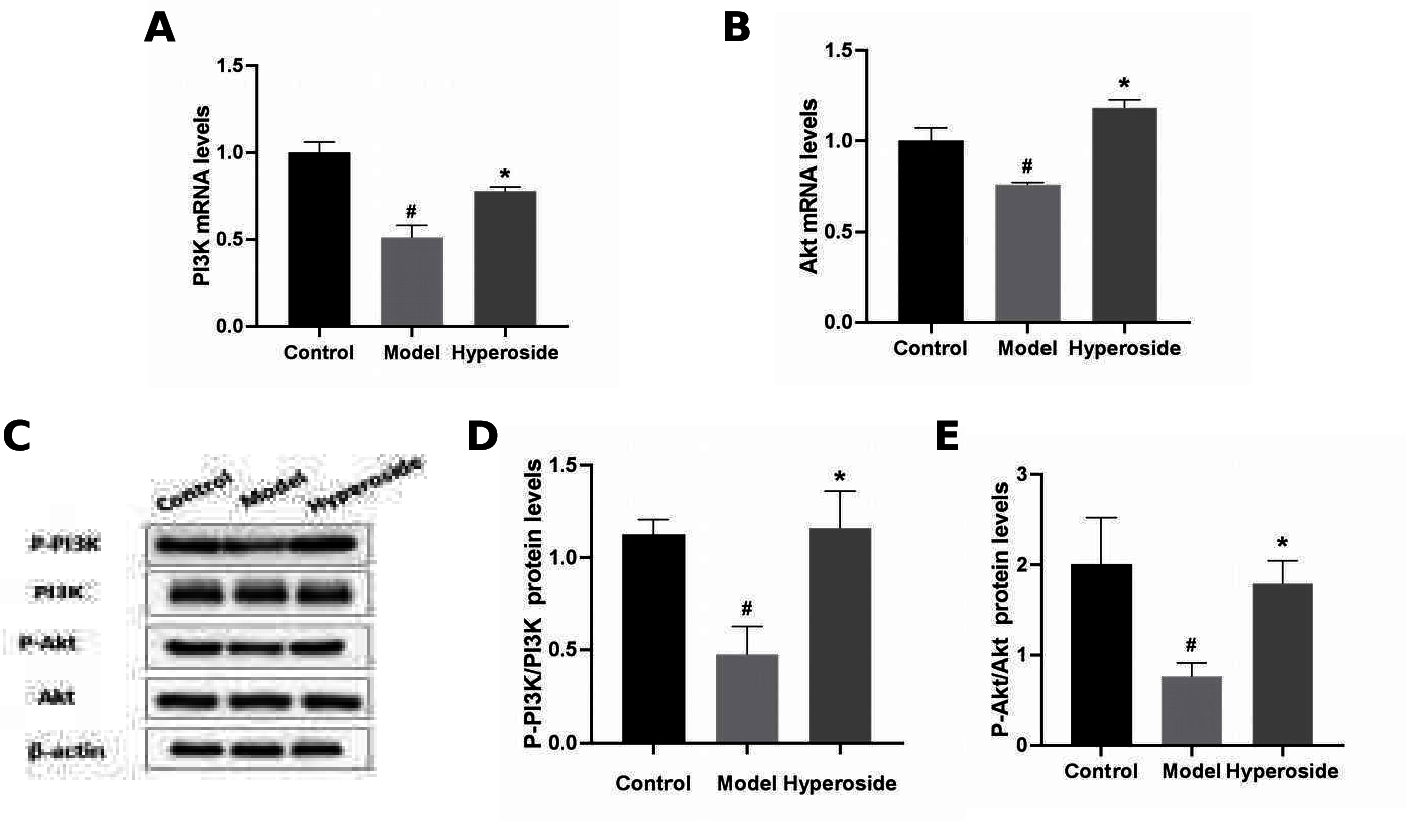
Increased PI3K and Akt mRNA and phosphorylated protein expression in hyperoside-treated cells. (
Changes in NF-κB/Bax/Caspase 3 mRNA and phosphorylated protein expression in hyperoside-treated cells
As shown in Figure 4, compared to the control group, RT-PCR and WB analysis showed that the expression of NF-κB, Bax, Caspase 3 mRNA, and related proteins in the model group (400 µmol/L H2O2) was significantly up-regulated. After the cells were treated with hyperoside (50 μmol/L) for 12 hours, the mRNA expressions of NF-κB, Bax, and Caspase 3 were significantly lower than in those treated with H2O2 alone (Figure 4(A), (B) and (C)). While mRNA expression decreased, the expression levels of NF-κB, Bax, and Caspase 3 proteins also decreased significantly (Figure 4(D), (E), (F) and (G)). These results indicate that hyperoside may regulate oxidative stress damage by regulating the NF-κB/Bax/Caspase 3 signaling pathway and significantly promote the expression of NF-κB/Bax/Caspase 3 genes and related proteins after oxidative damage, thereby reducing oxidative stress damage.

Effected NF-κB/Bax/Caspase 3 mRNA and phosphorylated protein expression in hyperoside-treated cells. (
Changes in P62/LC3B mRNA and Phosphorylated Protein Expression in Hyperoside-Treated Cells
As shown in Figure 5, compared to the control group, RT-PCR and WB analysis showed that the expression of P62 mRNA and related proteins in the model group (400 µmol/L H2O2) increased significantly, while that of LC3B mRNA and related proteins decreased significantly. After 12 hours of treatment with hyperoside (50 μmol/L), the mRNA expression of P62 was significantly lower than that of H2O2-induced cells (Figure 5(A)). While mRNA expression decreased, the expression level of P62 protein was also significantly decreased (Figure 5(C) and D); the mRNA expression of LC3B was significantly higher than that of H2O2-induced cells (Figure 5(B)). While mRNA expression increased, the expression level of P62 protein also increased significantly (Figure 5(C) and (E)). The results indicate that hyperoside may regulate oxidative stress damage by regulating the P62/LC3B signaling pathway, significantly promoting the expression of LC3B genes and corresponding proteins after oxidative damage, and reducing the expression of P62 genes and corresponding proteins.

Effected P62/LC3B mRNA and phosphorylated protein expression in hyperoside-treated cells. (
Discussion
In our previous studies, we found that hyperoside, isolated from Acanthopanax senticosus, has significant anti-oxidative stress damage. It has been reported to alleviate the effects of oxidative stress damage, to varying degrees, in the treatment of inflammatory apoptosis. 12,13 H2O2 acts as an inducer of oxidative stress in vitro and can induce apoptosis in many different cell types, but H2O2 can be inhibited by antioxidants. 14 Therefore, this study explored the role of hyperoside as an antioxidant and studied its protective mechanism in H2O2-treated PC12 cells. An MTT assay was used to study the proliferation of PC12 cells treated with hyperoside and our findings revealed that this flavonoid could effectively reduce the oxidative stress damage induced by H2O2 in these cells, consistent with the results reported in the literature. 15
The PI3K/Akt signaling pathway is mediated by enzyme-linked receptors. Phosphatidylinositol-3-kinase (PI3K) and its downstream molecule, serine/threonine protein kinase B (protein kinase, PKB, alias Akt), constitute this pathway. This pathway plays an important role in protecting against oxidative stress by suppressing cell apoptosis and facilitating cell survival. 16 Under certain circumstances, it can effectively inhibit neuronal apoptosis by activating the PI3K/Akt signaling pathway. Studies have shown that deuterium-depleted water (DDW) can inhibit H2O2-induced apoptosis in PC12 cells, reduce the generation of ROS, and increase the activities of catalase (CAT), copper, zinc SOD, and SOD. DDW may protect differentiated PC12 cells from the oxidative stimulation induced by H2O2 through the PI3K/Akt signaling pathway. 17 Studies have also shown that luteolin inhibits H2O2-induced cell apoptosis by decreasing ROS levels and activating the PI3K/Akt pathway. 18 Puerarin may play a neuroprotective role by activating the PI3K/Akt signaling pathway. 19 Fucoidan can inhibit the apoptosis of PC12 cells induced by H2O2 by increasing the ratio of Bcl-2/Bax, reducing the expression of Caspase 3, and enhancing Akt phosphorylation. 20 A number of studies have shown that antioxidants participate in the PI3K/Akt pathway, which eventually protects PC12 cells from oxidative stress induced by H2O2. Therefore, in this study, to explore whether the PI3K/Akt signaling pathway was involved in hyperoside exerting its antioxidant effect on PC12 cells, we evaluated the expression levels of the flavonoid on the mRNA and key proteins involved in the PI3K/Akt signaling pathway. Therefore, in this study, in order to explore whether the PI3K/Akt signaling pathway is involved in the antioxidant effect of homoserin on PC12 cells, we evaluated the expression levels of flavonoids on mRNA and key proteins in the PI3K/Akt signaling pathway. Our results show that the expression of PI3K/Akt mRNA and P-PI3K/P-Akt protein after hyperoside treatment was significantly normalized. It indicates that the mechanism of homoserin reducing oxidative stress damage and inhibiting cell apoptosis may be through the PI3K/Akt signaling pathway.
As a transcription factor regulating the redox response, after activation, NF-κB can regulate gene expression in the early response of cell death induced by oxidative stress. 21 NF-κB, as a signal element, controls the expression of the downstream inflammatory factor, nitric oxide synthase (iNOS). 22 iNOS regulates cell apoptosis by synthesizing NO to influence the activation of NF-κB. 23 Bax, as one of the apoptotic factors, can accelerate apoptosis. Caspase 3 protein, as a vital apoptotic executive protein in the middle and lower reaches of the Caspase cascade, 24 exists and plays an important role in apoptosis. NF-κB is a downstream factor of the Akt signaling pathway, and activated Akt can promote the expression of NF-κB protein. At the same time, NF-κB, as a key intermediate product of apoptosis induced by oxidative stress, also mediates Akt phosphorylation and gene expression. Therefore, activation of the PI3K/Akt and NF-κB pathways plays a functional role in bringing about anti-apoptotic effects. H2O2-induced oxidative stress injury increases NF-ĸB and Caspase 3 protein expression, which also results in an increase in apoptosis. 25,26 Studies report that H2O2 toxicity can increase the levels of NF-κB, Caspase 3, and the pro-apoptotic protein, Bax, while 6-(2-hydroxyethyl)-adenosine (HEA) treatment reduces NF-κB. The levels of Caspase 3 and Bax indicate that HEA can regulate the above pathways to reduce H2O2-induced cell apoptosis. 27 Numerous studies show that we can protect oxidative stress damage induced by H2O2 by reducing the activity of ROS and Caspase 3 in cells and by down-regulating the NF-κB pathway, thereby inhibiting cell apoptosis and inflammation. 28,29 Therefore, to elucidate further the anti-oxidant mechanism of hyperoside, we evaluated its effect on mRNA and protein expression of the NF-κB/Bax/Caspase 3 pathways. Our results showed hyperoside could reduce the expression level of NF-κB/Bax/Caspase 3 gene by regulating the NF-κB/Bax/Caspase 3 signaling pathway to decrease oxidative stress injury and protein expression levels; the decrease of its protein expression level indicates that the mechanism of hyperoside to reduce stress injury may be related to the signal pathway related to the regulation of inflammation and apoptosis by NF-KB/Bax/Caspase.
In addition to participating in the mediation of apoptosis-related pathways, the PI3K/Akt pathway can also regulate autophagy to eliminate protein aggregation in neurodegenerative diseases and regulate the autophagy proteins related to neurodegeneration. When autophagy occurs, LC3B, an important autophagy marker of the LC3 family, is recruited to autophagy. On the phage membrane, the multifunctional selective autophagy adaptor protein, P62, aggregates on the mitochondrial membrane through voltage-dependent anion channel protein 1 and specifically interacts with the ubiquitin-binding protein. Next, it binds to the LC3B protein and is selectively degraded in the autophagosome. 25 Studies show that andrographolide activates autophagy and Nrf-2 mediated P62 signaling pathway-related autophagy genes and proteins (Beclin-1 and LC3), while reducing the expression of P-tau and P21 proteins, thereby protecting amyloid-induced and autophagy-related death in PC12 cells. 30 Studies have confirmed that the increase of P62 is closely linked to the unusual progress of autophagy flux and selectivity in the coaction environment stimulated by oxidative stress. In order to explore the protective action of hyperoside in H2O2-induced damage through autophagy-related pathways, our study evaluated the expression levels of this flavonoid on the mRNA and protein expression levels in the P62/LC3B pathway. The results showed that inhibition of autophagy after oxidative damage caused an increase in the expression and accumulation of the protein, P62, and the expression of LC3B was inhibited. After the interposition of hyperoside, the expression of P62 protein was significantly reduced, the expression of LC3B protein was activated, and the expression was significantly increased. It further indicates that the protective effect of hyperoside against H2O2 damage is achieved through autophagy-related pathways.
In summary, our results showed that hyperoside significantly up-regulated the gene and protein expression levels of PI3K, Akt, and LC3B. At the same time, the gene and protein expression levels of NF-κB, Bax, Caspase 3, and P62 showed a significant down-regulation trend, indicating that hyperoside could treat oxidative stress injury through the PI3K/Akt, NF-κB/Bax/Caspase 3, and P62/LC3B pathways .The above data indicate that hyperoside provides protection against oxidative stress induced by H2O2 in nerve cells through a variety of mechanisms. The protective effect of hyperoside in H2O2-induced injury was mainly achieved by regulating the pathways related to inflammation, autophagy, and apoptosis. Oxidative stress and apoptosis have a mutual regulatory effect on the activation and development of autophagy. 31,32 Oxidative stress, inflammation, apoptosis, and autophagy are inter-related and jointly regulate cell survival and growth. Hyperoside may be a promising therapeutic drug for the treatment of neurological diseases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China [Grant number 81960701]; the Natural Science Foundation of Jiangxi Province [Grant number 20171ACB20039]; the Jiangxi Province talent Project [grant numbers (2016) 332]; Nanchang innovative talent team [Grant number (2018) 274]; Nanchang Key Laboratory of active ingredients of traditional Chinese medicine and natural medicine [Grant number 2019-NCZDSY-011].
