Abstract
Betanin is a phytocompound whose effect in steatohepatitis has not yet been tested. Betanin was extracted from the fruits of Hylocereus ocamponis, and its effects were evaluated in a mice model for non-alcoholic fatty liver disease. Six-week-old male BALB/c mice fed with a high-fat diet received 9.6 mg of betanin per day during 40 days. Body, liver, and epididymal fat pad weights and the levels of blood serum total cholesterol, triglycerides, high-density lipoproteins, alanine aminotransferase, blood nitrogen urea, creatinine, and total antioxidant capacity were measured. Hepatosteatosis and inflammatory infiltration were categorized, and the relative cell area of hepatocytes was determined. Betanin inhibited the inflammatory infiltration of the liver (P = 4.000 × 10−6) and the necrosis of hepatocytes (P = 9.634 × 10−7); it also produced a predominance of microvesicular steatosis (P = 9.634 × 10−7), decreased epididymal fat pad weight (P = 8.250 × 10−4), and increased blood serum total cholesterol (P = 0.011). Betanin is a promising compound for fatty liver, steatohepatitis, and chronic liver disease.
Introduction
Non-alcoholic fatty liver disease is the most common cause of chronic liver diseases worldwide, and, in the United States, it is responsible for 75.1% of such cases. 1 -4 Non-alcoholic fatty liver disease starts with the development of hepatic steatosis, which can be the only histological finding, or it can be accompanied by the presence of inflammatory infiltrate—non-alcoholic steatohepatitis. 1,2,5 One of the factors considered responsible for the progression of hepatic steatosis to steatohepatitis is the increased production of reactive oxygen species by the peroxidation of lipids and by the disruption of mitochondria 2,6,7 ; in relation to this, adult patients with non-alcoholic steatohepatitis treated with vitamin E showed higher rates of improvement and a decrease in hepatic steatosis and lobular inflammation, which putatively, was produced by the antioxidant effect of this vitamin. 8,9 Therefore, the use of antioxidants in the treatment of liver disease is seen as a potential therapeutic approach; many of them have already been tested, producing no consistent or meaningful results, like those seen with vitamin E. 10 With respect to this, the effect of betalains in hepatic steatosis or non-alcoholic steatohepatitis has not been evaluated to date. Betalains are a group of phytopigments contained in most plants from the order Caryophyllales, such as Beta vulgaris (beetroot), which is widely consumed in Europe; betalains possess a high antioxidant capacity and are commonly used in the food industry. 11 -23 Betalains have been shown to be able to bind low-density lipoproteins ex vivo and in vivo, 7,24 to inhibit or to reduce the oxidation of low-density lipoproteins in vitro and ex vivo, 7,23 -25 to efficiently scavenge hypochlorous acid, and to exert an inhibitory effect in the chlorination activity of human myeloperoxidase in vitro. 26 Furthermore, the capacity to induce quinone reductases in vitro, 27 to generate an increase in caspase-3 activation, to produce a reduction in reactive oxygen species levels in isolated polymorphonuclear leukocytes, and to downregulate and produce a decrease in the secretion of proinflammatory cytokines in isolated macrophages has also been found for betalains. 28,29 In addition, the decrease in liver weight and hepatic steatosis found in tumor necrosis factor alpha (TNF-α) KO mice and in wild-type mice with increased quinone reductase activity, 30,31 respectively, suggests that a reduction in hepatic steatosis could also occur with the administration of betalains. 27,29 With regard to the aforementioned properties, we assessed the effects of the most common betalain, betanin (betanidin-5-O-glucoside), 32,33 in the steatosis and inflammatory infiltration of liver produced in mice chronically fed a high-fat diet. 32,33
Materials and Methods
Betanin Extraction and Purification
Fresh red-purple pitaya fruits (Hylocereus ocamponis) from a plantation in Jalisco, Mexico, were washed and allowed to air-dry for 5 minutes. Afterward, they were peeled by hand and the fruit flesh was used immediately after for extraction, as reported by Butera et al, 23 and then purified according to Stintzing et al. 34 Betanin was stored in the dark at −80°C until use.
Administration of Betanin and Dissection
Six-week-old male BALB/c mice were randomly assigned into 2 groups: high-fat diet (n = 13) and betanin (n = 12). For 24 weeks, they were fed a high-fat rodent diet (15.5% kcal/kcal protein; 41.9% kcal/kcal carbohydrates; 42.6% kcal/kcal fat) and purified water ad libitum. Animals were kept in a 12 hour/12 hour light-dark cycle, at 25°C and 70% humidity. All animals received humane care in compliance with the criteria outlined in the Guide for the Care and Use of Laboratory Animals prepared by the National Academy of Sciences and published by the National Institutes of Health, USA. During the last 40 days of the experiment, mice from the betanin group were given purified water mixed with betanin; each individual received a calculated dose of betanin of 9.6 mg per mouse per day. At the end of the experiment, all individuals were left to fast overnight (purified water was not suspended); afterward, cervical dislocation followed by decapitation was performed. All livers and epididymal fat pads were extracted and blood collected for further analysis. Mice were individually weighed during the first and last days of the experiment; final thoracic and abdominal circumferences were also measured and percentages of weight gain were obtained.
Blood Serum Collection and Measurement of Levels of Blood Serum Lipids, Alanine Aminotransferase, Blood Urea Nitrogen, and Creatinine
Using BD Vacutainer Serum Collection Tubes (BD Vacutainer, NJ, USA), blood was collected from a neck wound and allowed to clot at room temperature for 25 minutes; afterward, centrifugation at 2000 r/min for 15 minutes at 4°C was performed to remove the clot. For the determination of blood serum lipids, alanine aminotransferase, urea, and creatinine, samples were analyzed in a Roche cobas c 111 analyzer (Roche Diagnostics Ltd., Rotkreuz, Switzerland). Blood urea nitrogen was calculated by dividing serum urea by 2.14.
Measurement of Weights and Histological Analysis of Livers
Livers and epididymal fat pads were weighed on an analytical balance (Sartorius, Göttingen, Germany). Means were obtained for each group. Afterward, livers were fixed in 10% neutral buffered formaldehyde (Sigma-Aldrich, MO, USA) and left overnight at room temperature. Afterward, livers were embedded in paraffin wax and sectioned transversally with a thickness between 3 and 5 µm. Hematoxylin and eosin staining was performed, and the tissue sections were observed under an optical microscope (Zeiss, Oberkochen, Germany). All samples were coded and rated blind. Grading of hepatic steatosis was reported based on the percentage of hepatocytes affected as low (<33%), moderate (33%-66%), or severe (>66%). 35 The type of steatosis was graded as only macrovesicular, only microvesicular, or mixed, according to the size of lipid droplets found. Inflammatory infiltrate was defined as the presence of polymorphonuclear and mononuclear leukocytes in hepatic parenchyma; it was graded according to its intensity as low (isolated individual inflammatory cells), moderate (isolated clusters of inflammatory cells), or severe (diffuse inflammatory infiltrate).
Determination of Relative Cell Area of Hepatocytes
Using 20× images from livers of both groups, individual areas of 100 hepatocytes from all oxygenation regions of a lobule were measured using ImageJ software. 36 Means were obtained; values normalized to controls and results expressed as percentage of cell area.
Measurement of Blood Serum Total Antioxidant Capacity
Blood serum total antioxidant capacity (TAC) was measured using TAC Assay Kits (Abcam, Cambridge, UK). Blood serum (0.1 µL) was mixed with 99.9 µL of double-distilled water and added to a well plate. Then, 100 µL of Cu2+ working solution was added, followed by incubation in the dark for 1.5 hours at room temperature. The plate was read at 570 nm (BIORAD, CA, USA). Four samples from each group were analyzed. All samples were made in duplicate. Results were expressed in trolox equivalents.
Statistical Analysis
All data, except histological findings, represent means; ±standard errors of means are indicated. One-way analysis of variance, Kruskal-Wallis, and 2-tailed Fisher’s exact tests were used when appropriate. Differences in P value lower than 0.05 were considered statistically significant. Statistical analysis was performed using SPSS software (version 22, IBM, NY, USA).
Results and Discussion
The consumption of betanin produced no significant differences in body weight, liver weight, or thoracoabdominal circumferences. However, betanin significantly reduced epididymal fat pad weight (P = 8.250 × 10−4) and epididymal fat pad/body weight ratio (P = 9.610 × 10−4) (Table 1). In addition, betanin increased blood serum total cholesterol levels (19.8%, P = 0.011), but generated no relevant variations in the levels of triglycerides, high-density lipoproteins, alanine aminotransferase, blood urea nitrogen, or creatinine (Table 2). With regard to histopathological characteristics, all mice developed hepatic steatosis; however, betanin produced a predominance of microvesicular steatosis in all treated mice (P = 9.634 × 10−7). Moreover, inflammatory infiltrate in hepatic parenchyma and isolated necrotic hepatocytes was found in all controls but was absent in betanin-treated mice (P = 4.000 × 10−6 and P = 9.634 × 10−7, respectively) (Table 3). Furthermore, all individuals presented occasional hepatocellular ballooning degeneration (Figure 1). No significant difference was found in the relative cell area of hepatocytes or the blood serum TAC.

Representative histological findings in livers from mice chronically fed a high-fat diet. Pictures show 20× hematoxylin and eosin stained liver sections. (a) Control and (b) betanin. All individuals from both groups presented occasional hepatocellular ballooning degeneration and a mixed macrovesicular and microvesicular steatosis; in the group given betanin, microvesicular steatosis (double arrow) was the predominant type. Neither inflammatory infiltrate nor necrotic hepatocytes were found in livers from the betanin group. Single black and single outlined arrows indicate inflammatory infiltrate and necrotic hepatocytes, respectively.
Body, Liver, and Epididymal Fat Pad Weights and Thoracic and Abdominal Circumferences After the Chronic Consumption of a High-Fat Diet.

SEM, standard error of the mean; F.a.c., final abdominal circumference; F.t.c., final thoracic circumference.
a P < 0.001.
Concentrations of Blood Serum Lipids, Alanine Aminotransferase, Blood Urea Nitrogen, and Creatinine After the Consumption of a High-Fat Diet for 24 Weeks.
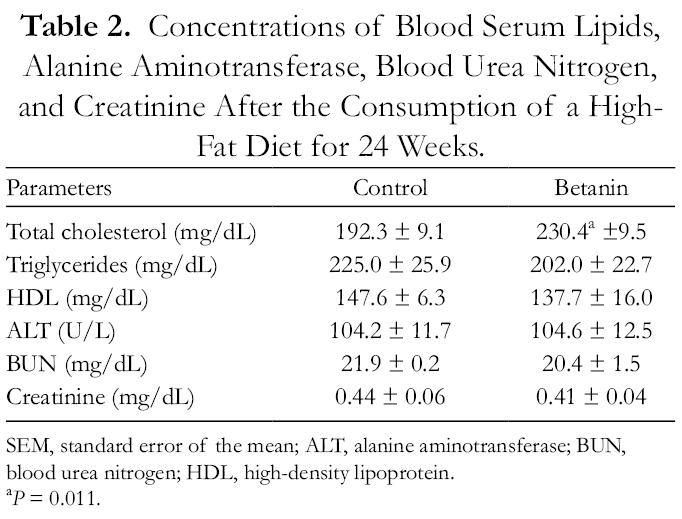
SEM, standard error of the mean; ALT, alanine aminotransferase; BUN, blood urea nitrogen; HDL, high-density lipoprotein.
a P = 0.011.
Histological Findings in Livers After the Chronic Consumption of a High-Fat Diet.

a P < 0.00001.
In the present study, we describe the capacity of a betalain to modulate inflammatory cells and necrosis in vivo, to modify steatosis, and to generate significant effects in steatohepatitis. This finding is supported by the results of previous in vitro studies showing the activation of caspase-3 and the downregulation of TNF-α, COX-2, interleukin (IL)-1β, IL-6, and inducible nitric oxide synthase in isolated inflammatory cells treated with a betalain. 28,29 The mechanism of the anti-inflammatory effect we found is unknown; however, since TNF-α KO mice fed a high-fat diet present a reduction in markers of systemic inflammation and macrophage infiltration in adipose tissue, 30 and since the release of proinflammatory molecules is related to the pathophysiology of steatohepatitis, it suggests that a reduction in the secretion of inflammatory cytokines by M1 Kupffer cells may be implicated. 9 Additionally, the absence of necrotic hepatocytes found in betanin-treated mice is concordant with the lack of significant changes observed in the concentrations of blood serum alanine aminotransferase, blood urea nitrogen, and creatinine; this implies that betanin causes no meaningful damage to hepatocytes, if any, and that it produces an antinecrotic effect; the mechanism of the latter may be associated either with a reduction in the generation of reactive oxygen species by inflammatory cells, 28 with increased protection against deoxyribonucleic acid damage or with a possible decrease in the expression of TNF-α. 28,29 We ignore the physiopathological impact in hepatosteatosis and steatohepatitis of the increase observed in serum total cholesterol. In addition, changes were expected in weights, thoracoabdominal circumferences, and hepatosteatosis through the mediated increase of NAD(P)H:quinone oxidoreductase 1 27,31,37 ; therefore, the relevant reduction in weight found in epididymal fat pad indicates that betanin effectively produces an effect on body fat deposits, albeit with a differential topographical pattern. With respect to this, the absence of meaningful changes in body weight, degree of steatosis, and relative cell area of hepatocytes and hepatocellular ballooning, together with the significant predominance of microvesicular steatosis in betanin-treated mice, suggest that the lipid-modulatory effect observed is not limited to body fat deposits or a net change in the accumulation of lipids, that it is histologically differential, and that it may also modulate the vacuolization of lipids and produce the accumulation of specific fatty acids. Moreover, the predominance of microvesicular steatosis detected implies a conjoined reduction in the progression of hepatic steatosis. 38 Additionally, the lack of significant changes in the blood serum TAC levels of betanin-treated mice, together with the absence of inflammatory infiltrate in livers from this group, suggests that this absence is not dependent on long-term changes in TAC; measurements of blood TAC after shorter time-lapses post-ingestion of betanin will determine if or not this compound exerts significant changes in TAC and if the regulation of the levels of reactive oxygen species is needed for the production of any of the effects found; moreover, it is known that betalains can be internalized into cells, 39 and therefore, a relation between a potential intracellular increase in TAC with the antisteatohepatitic and antinecrotic effects found cannot yet be discarded. Further studies will determine the molecular mechanisms behind the effects found and will clarify if betanin is directly responsible for them or if they are produced by one of its metabolites; the latter, since the relative low levels of elimination of betalains in urine of less than 1% of the ingested amount and the drastic decrease in their plasmatic concentration after 3 hours of ingestion suggest that free betalains are rapidly retired from blood, rapidly excreted by other pathways, or intensely metabolized to derivatives not identified as betalains. 40,41 Further studies will determine the molecular mechanisms behind the effects observed and their reproducibility in humans.
Conclusions
Our findings show that betanin is a potential therapeutic agent for hepatosteatosis, steatohepatitis, and diseases with similar physiopathological characteristics. Betanin inhibits the inflammatory infiltration of liver, the necrosis of hepatocytes, and produces a predominance of microvesicular steatosis. Betanin is a promising compound for fatty liver, steatohepatitis, and chronic liver disease.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
