Abstract
Herpes simplex virus type 1 (HSV-1) infection is one of the most common oral diseases, clinically presenting as gingivostomatitis and cold sores. Current treatments primarily rely on nucleoside antiviral drugs, which often suffer from low bioavailability and a tendency to induce drug resistance. Therefore, preventing HSV-1 infection and developing new antiviral agents are of great importance. Wood vinegar, a liquid obtained from the pyrolysis and condensation of wood such as tree branches, plant stems, or fruit trees, has been reported to possess various biological activities, including antiviral and anti-inflammatory effects. This study aims to investigate the cytotoxicity, anti-HSV-1 activity, and chemical composition of two types of wood vinegar derived from Vitis vinifera and Casuarina equisetifolia using the CCK-8 assay, cytopathic effect (CPE) analysis, and gas chromatography–mass spectrometry (GC-MS). Results showed that both wood vinegars exhibited certain toxicity toward Vero and L929 cells, but no toxicity was observed in Vero cells at a 64-fold dilution. In vitro antiviral experiments demonstrated that both preparations possessed good anti-HSV-1 activity, though their preventive effects against HSV-1 infection require further investigation. GC-MS analysis revealed that the main chemical components of the two wood vinegars were acids, aldehydes, and phenolic compounds, with higher levels of trans,trans-2,4-Heptadienal, N,N-Dimethylaniline, and sorbic acid, among others. In summary, this study provides a new theoretical foundation and research direction for the potential application of wood vinegar in the prevention and treatment of herpes simplex virus infections.
Introduction
Herpes simplex virus type 1 (HSV-1) is a double-stranded DNA virus in the herpesviridae family. According to the World Health Organization, approximately 67% of the global population is infected with HSV-1, a figure that does not include asymptomatic carriers. 1 Clinical manifestations of HSV-1 infection range from mild recurrent skin lesions around the mouth and face (eg, cold sores) to more severe conditions such as herpes keratitis and potentially fatal herpes encephalitis. Following initial infection, the virus often remains latent in the body, leading to a lifelong risk of recurrence or subclinical shedding. 2 HSV-1 can be detected in saliva and oral mucosa. Infected individuals frequently shed oral mucosal epithelial cells unknowingly, releasing the virus in the process. Such asymptomatic shedding, along with visible lesions, represents a significant transmission route.3–9 Most primary infections result from direct contact with infected bodily fluids (eg, saliva or fluid from active lesions) or through close personal interactions. This includes sharing utensils, cups, or food, kissing and other face-to-face contact, children sharing chewing toys, as well as using personal items such as toothbrushes or lip balm.3,10–12 Given its often-hidden nature, intimate modes of transmission, and widespread prevalence, preventing HSV-1 spread and developing new antiviral treatments remain important public health challenges.
Currently, clinical treatment of HSV-1 primarily relies on nucleoside antiviral drugs, but long-term use often fails to completely eliminate the virus and may lead to drug resistance. Although several candidate vaccines have entered preclinical and clinical studies, no vaccine has yet been approved for the prevention or treatment of HSV-1. 13 In recent years, natural products have emerged as a promising direction in anti-HSV-1 drug research, owing to their wide availability, low toxicity, and reduced tendency to induce drug resistance. 14 For example, aristolochic acid, nano-formulated rhubarb, and aqueous extracts of Phellodendron amurense have all demonstrated significant anti-HSV-1 activity.15–17 Therefore, screening and developing new anti-HSV-1 drugs from natural sources holds considerable value.
Vitis vinifera is a plant rich in bioactive compounds, 18 with extracts from its various parts exhibiting diverse pharmacological effects. Studies have shown that different grape extracts can display varying antiviral activities. For example, Matias et al 19 found that extracts from Portuguese white wine inhibited the replication of adenovirus-5. Carla Zannella et al 20 demonstrated that grape leaf extracts exhibited antiviral effects against both SARS-CoV-2 and HSV-1. Other studies have reported that grape seed extract (GSE) shows potential anti-HCV activity. 21 On the other hand, Casuarina equisetifolia, an evergreen tree of the Casuarinaceae family, is often used medicinally through its young branches, leaves, and bark. Cyclic diarylheptanoids isolated from its roots have been confirmed to possess anti-influenza A virus (IAV) activity. 22
Wood vinegar is a liquid product obtained from the pyrolysis and condensation of wood or plant residues. It is acidic and contains a complex variety of chemical components, primarily including acids, alcohols, phenols, and neutral compounds.23–25 Its chemical composition is mainly influenced by the type of raw material, 26 and also varies with pyrolysis temperature and heating rate.27,28 As a natural and environmentally friendly product, wood vinegar is rapidly degraded in the environment, exhibits high safety for humans and animals, and poses no environmental pollution risk, leading to its increasing recognition and application. 29
Currently, wood vinegar has demonstrated a wide range of uses in various fields, such as increasing collagen content in eggs,
30
enhancing fruit yield and nutritional quality,
31
improving soil properties and promoting plant photosynthesis,
32
as well as enhancing intestinal morphology and nutrient absorption in chickens.
33
It also holds significant application value in animal husbandry
34
and the food processing industry.
35
Moreover, studies have shown that wood vinegar possesses multiple biological activities, including anti-inflammatory, antibacterial, and antioxidant effects. For example, wood vinegar from Griffith's ash can suppress lipopolysaccharide-activated inflammatory responses in macrophages
36
; Pyroligneous extract of
However, research on the antiviral effects of wood vinegar remains relatively limited. Currently, it is known that wood vinegar derived from Japanese larch significantly inhibits encephalomyocarditis virus, 39 but there are no reports on the anti-HSV-1 activity of wood vinegars from grapevine or casuarina. Therefore, this study aims to preliminarily investigate the inhibitory activity of these two types of wood vinegar against HSV-1 and analyze their chemical compositions, with the goal of providing experimental evidence for developing novel natural product-based strategies against HSV-1.
Materials and Methods
Cell Lines and Virus Strains
African green monkey kidney cells (Vero cells) and HSV-1 virus were provided by the National Institute for Viral Disease Control and Prevention, China CDC.
NCTC clone 929 [L cell, L-929] (CL-0137) was obtained from Procell Life Science & Technology Co., Ltd in Wuhan.
Vero cells and L929 cells were cultured in DMEM medium (Gibco, USA) supplemented with 10% FBS and 1% penicillin-streptomycin.
Preparation of Wood Vinegar
The Vitis vinifera and Casuarina equisetifolia samples, along with the equipment for wood vinegar preparation, were collected and provided by staff from Tangshan Xinyanuo Company in China. Healthy branches from both plant species were selected as raw materials. These were first cleaned to remove surface impurities, and any rotten or moldy parts were discarded. The branches were then crushed into pieces approximately 3–5 cm in length using a branch shredder. Branches with a diameter of ≤2 cm were used directly without further crushing.
The processed material was dried in an 80 °C oven for 24–48 h. During this period, moisture content was periodically tested using a moisture meter until it reached 18%–22%. The dried material was then loaded into a continuous pyrolysis furnace and sealed to maintain oxygen levels below 2%, ensuring pyrolysis proceeded under oxygen-limited or anaerobic conditions.
The pyrolysis process consisted of three main stages:
Heating and drying stage (room temperature to 150 °C): Free water and some bound water were evaporated, accompanied by the release of white steam. Pre-carbonization stage (150-275 °C): Light yellow smoke appeared. Main carbonization stage (275-400 °C): A large volume of mixed steam (smoke) was released, from which wood vinegar was collected. To ensure product quality, the heating rate was carefully controlled between 280–350 °C to avoid excessive tar formation and deterioration of wood vinegar quality.
The hot vapor generated during the main carbonization stage was directed through a pipeline into a multi-stage condensation system. Cooled to room temperature with circulating water, it condensed into crude wood vinegar. This crude liquid appeared dark brown or reddish-brown, was turbid, and had a strong smoky odor, making it unsuitable for direct use. Further refining was therefore required.
The refining process included the following steps:
The crude wood vinegar was left to settle for 6 months, yielding the raw wood vinegar solution. The raw wood vinegar was subjected to first-stage atmospheric distillation to obtain purified wood vinegar. The purified wood vinegar was filtered through PP cotton and then underwent a second atmospheric distillation, resulting in refined wood vinegar. The refined wood vinegar was filtered through a carbon fiber filter to produce the two final experimental samples: Vitis vinifera wood vinegar (referred to as Wood Vinegar No. 1 or WV-1) and Casuarina equisetifolia wood vinegar (referred to as Wood Vinegar No. 2 or WV-2). (Image 1)

Wood vinegar preparation process.
Preparation of HSV-1 Suspension
The HSV-1 stock was thawed in a 37 °C water bath (Thermo Scientific, USA) and diluted 10-fold with DMEM (Gibco, USA) containing 2% FBS (ie, cell maintenance medium). When Vero cells reached approximately 90%–100% confluency, the diluted HSV-1 solution was inoculated into a T25 flask (Costar, USA) containing the Vero cells and allowed to adsorb for 2 h, with gentle shaking every 15 min.
After adsorption, 10 mL of fresh maintenance medium was added to the flask, and the culture was continued in a 37 °C, 5% CO2 incubator (Thermo Scientific, USA). The cells were observed under a microscope until more than 95% exhibited clear cytopathic effects—such as swelling or rounding—indicating advanced infection.
The infected cells were then subjected to repeated freeze-thaw cycles. The lysate was centrifuged at 3000 rpm for 10 min using a high-speed centrifuge (Eppendorf, Germany) to remove cellular debris. The supernatant was aliquoted in small volumes into sterile centrifuge tubes (Costar, USA) and stored in an −80 °C ultra-low temperature freezer (Thermo Scientific, USA).
Virus Virulence Assay
Vero cells were seeded in a 96-well plate (Costar, USA) and incubated until a confluent monolayer formed the next day. Ten sterile centrifuge tubes were labeled and arranged in order. To each tube, 900 µL of cell maintenance medium (DMEM containing 2% FBS) was added. Then, 100 µL of fully dissolved HSV-1 stock solution was pipetted into the first tube, which was labeled as 101 dilution. From this tube, 100 µL of the virus mixture was transferred to the next tube to prepare a 102 dilution. This serial dilution procedure was repeated sequentially to obtain dilution gradients of 103, 104, 105, 106, 107, 108, 109, and 1010.
The cell culture medium (DMEM containing 10% FBS and 1% penicillin-streptomycin) in the 96-well plate was removed. Each dilution of HSV-1 was added to four replicate wells (100 µL per well). Additionally, four wells were assigned as the uninfected cell control group and received 100 µL of maintenance medium per well. The plate was then placed in a 37 °C, 5% CO2 incubator (Thermo Scientific, USA) for further cultivation.
Cytopathic effects (CPE) were observed daily under a microscope and scored as follows: “++++": 75%–100% of cells affected; “+++": 50%–75% of cells affected; “++": 25%–50% of cells affected; “+": 1%–25% of cells affected. After 3–4 days, when CPE progression had stabilized in all wells, the 50% tissue culture infectious dose (TCID50) was calculated using the Reed–Muench method. The formulas applied were: Distance ratio = (CPE ≥ 50% group infection rate) / (CPE ≥ 50% group infection rate – CPE ≤ 50% group infection rate); TCID50 (per 100 µL) = log(dilution of CPE ≥ 50% group) + distance ratio.
Cytotoxicity Assay of Wood Vinegar
Vero cells were seeded in a 96-well plate (Costar, USA) and cultured until a confluent monolayer formed the next day. Ten sterilized centrifuge tubes were arranged and labeled sequentially. Each tube was filled with 500 µL of cell maintenance medium (DMEM containing 2% FBS). Then, 500 µL of Vitis vinifera wood vinegar was added to one tube, labeled as concentration 2-fold dilution(2D). From this tube, 500 µL of the mixture was transferred to another tube to prepare the 4-fold dilution(4D). This serial dilution process was repeated to obtain concentration gradients of 8D, 16D, 32D, 64D, 128D, 256D, 512D, and 1024D. The same dilution method was applied to Casuarina equisetifolia wood vinegar.
After removing the culture medium, 100 µL of each diluted wood vinegar solution was added to the 96-well plate, with four replicate wells per concentration. A cell control group was set up with four replicate wells containing only 100 µL of maintenance medium (no wood vinegar). Another four wells were left without any reagents as blank controls. The plate was then incubated at 37 °C with 5% CO2 (Thermo Scientific, USA).
After 72 h, the liquid in the plate was discarded under light-protected conditions. The wells were rinsed with PBS, followed by the addition of 100 µL CCK-8 assay solution (Gibco, USA) per well. The plate was returned to the incubator for 1–2 h. Absorbance was measured at 450 nm using a microplate reader (Thermo Scientific, USA), and cell viability at each wood vinegar concentration was calculated as follows: Cell viability (%) = [(A450 of treatment group – A450 of blank) / (A450 of control group – A450 of blank)] × 100%. The same experimental procedure was used to evaluate the cytotoxicity of Wood Vinegar No. 1 and No. 2 on L929 cells.
Anti-HSV-1 Activity of Wood Vinegar
Direct Inactivation Effect
Vero cells were seeded in 96-well plates (Costar, USA) and allowed to form confluent monolayers by the following day. Vitis vinifera wood vinegar (WV-1) and Casuarina equisetifolia wood vinegar (WV-2) were diluted with cell maintenance medium (DMEM containing 2% FBS) using the same method described in Section 1.5 to generate concentration gradients of 1D, 2D, 4D, 8D, 16D, 32D, and 64D. Equal volumes of each diluted wood vinegar solution and the original HSV-1 stock were mixed in sterile centrifuge tubes, with three replicates per concentration. The mixtures were incubated at 37 °C in a 5% CO2 incubator (Thermo Scientific, USA) for 15 min. Simultaneously, a virus control group was prepared by mixing the cell maintenance medium with the HSV-1 stock in equal volume for 15 min, while a drug control group was prepared by mixing the cell maintenance medium with the diluted wood vinegar solution for the same duration. After 15 min, the experimental, virus control, and drug control groups were diluted 64-fold with cell maintenance medium (DMEM containing 2% FBS) to ensure that the wood vinegar concentration in all tubes was reduced to a safe level. These diluted mixtures were then added to the 96-well plates. Viral titers were measured for the experimental and virus control groups, and CPE values were observed for the drug control group.
Inhibition After HSV - 1 Infection of Cells
Vero cells were seeded in 96 - well plates and T25 flasks (Costar, USA) and incubated until a confluent monolayer formed the next day. WV-1 and WV-2 were diluted with cell maintenance medium (DMEM containing 2% FBS) following the same method as in section 1.6.1 to create a concentration gradient from 1D to 64D.
Two milliliters of the original HSV - 1 stock was added to the T25 flasks. Once cell rounding, brightness, or fusion was observed, the original medium was removed, and 10 mL of diluted wood vinegar solution at each concentration was added. Each concentration was tested in triplicate, and the flasks were incubated for 24 h at 37 °C with 5% CO2 (Thermo Scientific, USA). A virus control was prepared by adding cell maintenance medium to HSV-1 - infected T25 flasks, and a drug control was prepared by adding diluted wood vinegar solution to uninfected T25 flasks.
After 24 h, the experimental, virus control, and drug control flasks underwent repeated freeze - thaw cycles. The supernatants were collected after centrifugation at 3000 rpm for 10 min (Eppendorf, Germany) to remove cell debris, and were then diluted 64-fold with cell maintenance medium to ensure all wood vinegar concentrations were at safe levels. The diluted supernatants from the experimental groups were added to 96 - well plates containing Vero cells, with each concentration tested in triplicate and each replicate plated in quadruplicate (100 µL per well). Cytopathic effect (CPE) was observed under a microscope, and the positive well rate was calculated for each concentration. Positive and negative controls were similarly assessed. (A well was considered positive if >50% of cells showed CPE. Positive well rate (%) = number of positive wells / total wells × 100%).
Inhibitory Effect After Pre-Treating Cells with Wood Vinegar
Vero cells were seeded into a 96-well plate (Costar, USA) and cultured overnight to form a confluent monolayer. WV-1 and WV-2 were diluted with cell maintenance medium (DMEM containing 2% FBS) at a safe concentration (64-fold) to prepare concentration gradients of 64D, 128D, 256D, and 512D, following the same method described in section 1.6.1.
The original medium in the 96-well plate was removed, and the diluted wood vinegar solutions at various concentrations were added (100 μL per well, with four replicates per concentration). In parallel, cell maintenance medium (DMEM with 2% FBS) was added to wells designated as the virus control group, while wood vinegar diluent was added to HSV-1-free wells as the drug control group. The plate was incubated at 37 °C with 5% CO2 (Thermo Scientific, USA) for 1 h.
After 1 h, 100 TCID50 of HSV-1 suspension (100 μL/well) was added to both the experimental group and the virus control group, followed by further incubation for 2 h. Subsequently, the HSV-1 adsorption solution was removed, and each well was replenished with 100 μL of cell maintenance medium. The plate was returned to the incubator for continued culture.
When cytopathic effects in the virus control group reached at least 75%, the absorbance of each well was measured at 450 nm using a microplate reader (Thermo Scientific, USA). Cell viability was calculated according to the following formula: Cell viability (%) = [(A450 of treatment group – A450 of blank) / (A450 of control group – A450 of blank)] × 100%.
pH Measurement of Wood Vinegar
Vitis vinifera wood vinegar (WV-1) and Casuarina equisetifolia wood vinegar were diluted to concentration gradients from 1D to 64D using cell maintenance medium (DMEM with 2% FBS), following the method described in Section 1.6.1. The pH of each diluted wood vinegar solution and the cell maintenance medium was tested using precision pH test strips with ranges of 1.4–3.0, 3.8–5.4, and 5.5–9.0. After 30 s of contact with the reagent, the test strips were removed and compared with the color chart to determine the pH values.
GC-MS Analysis of Wood Vinegar
The components of wood vinegar were subjected to GC-MS analysis on an Agilent system consisting of a model 7890B gas chromatographer and a model 5977A mass selective detector (MSD, electron energy, 70 eV). The carrier gas was helium (99.99%) with a flow rate of 0.8 mL/min. The injector and detector temperatures were respectively set at 250 °C and 250 °C. Spectra were obtained over a scan range of 50 to 550 amu at 2 scans/s. The GC program was set as follows: the initial temperature was 60 °C and held for 10 min, then increased by 2 °C/min to 80 °C and held for 5 min, then raised by 2 °C/min to 110 °C and held 6 min, then raised by 2 °C/min to 120 °C and held for 5 min, and finally raised by 2 °C/min to 180 °C and held at 180 °C for 2 min. The wood vinegar (1.0 μL) was injected automatically while maintaining a solvent delay of 4 min. Interpretation of the mass spectrum was made by comparing the peak distribution against the database of National Institute Standard and Technology (NIST MS 14.0, Gaithersburg, MD, USA). Relative percentages of the chemical compositions were calculated based on the GC peak areas without correction.
Statistical Analysis
Graphs were generated and all data were analyzed using GraphPad Prism 8. Comparisons between two groups were performed using t - tests. A
Experimental Results
Preparation of Wood Vinegar
The Vitis vinifera wood vinegar (WV-1) we obtained was clear and acidic (pH 3.0), while the Casuarina equisetifolia wood vinegar (WV-2) appeared light yellow and also acidic (pH 2.5). After diluting both with cell maintenance medium (DMEM containing 2% FBS), corresponding color changes were observed (Image 2A for WV-1, Image 2B for WV-2).

Images A and B display the original Vitis vinifera wood vinegar and Casuarina equisetifolia wood vinegar, as well as the wood vinegar diluted in a two-fold gradient using cell maintenance medium (DMEM with 2% FBS). From left to right, the tubes contain: original solution, 2-fold dilution, 4-fold dilution, 8-fold dilution, and 16-fold dilution.
HSV-1 Virulence Assay
The viral titer of HSV-1 was determined to be 6.56 TCID50/100 µL.
Cytotoxicity Assay of Wood Vinegar
CCK-8 assay results showed that the Vitis vinifera wood vinegar (WV-1) (Figure 1A) exhibited significantly different cytotoxic effects on Vero and L929 cells (

A: Cytotoxicity of Vitis vinifera wood vinegar on Vero and L929 cells. B: Cytotoxicity of Casuarina equisetifolia wood vinegar on Vero and L929 cells.
For the Casuarina equisetifolia wood vinegar (WV-2) (Figure 1B), both Vero and L929 cells demonstrated near-complete viability at a 64-fold dilution, with survival rates of (1.02 ± 0.08) × 100% and (0.98 ± 0.03) × 100%, respectively. To ensure experimental consistency, Vero cells were selected for subsequent studies.
Anti-HSV-1 Activity of Wood Vinegar
According to the direct inactivation results (Figure 2AB), viral titers measured by endpoint dilution showed that both Vitis vinifera wood vinegar (WV-1) at 1- to 4-fold dilutions and Casuarina equisetifolia wood vinegar (WV-2) at 1- to 8-fold dilutions exhibited significant direct inactivation effects against HSV-1 (
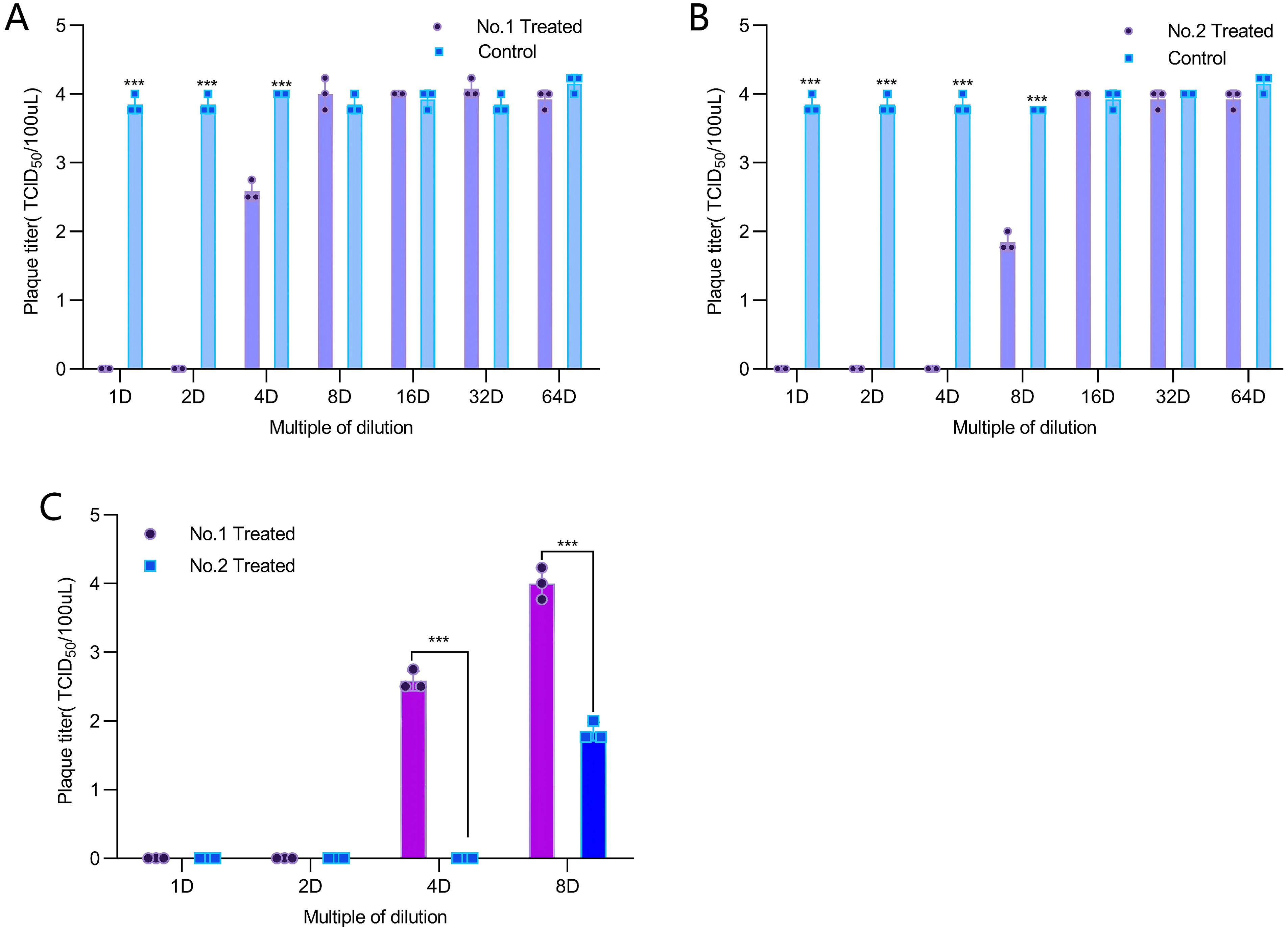
After mixing wood vinegar with HSV-1 for 15 min, the virus titer was measured to evaluate the direct inactivation effects of Vitis vinifera wood vinegar (A) and Casuarina equisetifolia wood vinegar (B) on HSV-1. Based on the virus titers detected across the 1- to 8-fold dilution series, the direct antiviral activities of the two wood vinegar formulations were comparatively evaluated (C).
Under microscopic observation, wells with more than 50% cytopathic effect (CPE) were recorded as positive, otherwise as negative (Image 3). The CPE assay results (Figure 3AB) showed that at 1- to 4-fold dilutions of both wood vinegars, the CPE positive rate in the experimental groups was 0%. When diluted to 8- to 64-fold, the CPE positive rate reached (83.33 ± 14.43)% or even 100%, indicating that both wood vinegars at 1- to 4-fold dilutions significantly inhibited HSV-1 infection in cells.

Observation under a microscope: wells with cytopathic effects in over 50% of cells are recorded as positive wells (AB), while those with effects in less than 50% of cells are recorded as negative wells (CD). The inhibitory effects of Vitis vinifera wood vinegar and Casuarina equisetifolia wood vinegar on HSV-1 infected Vero cells are evaluated by calculating the positive well rate.
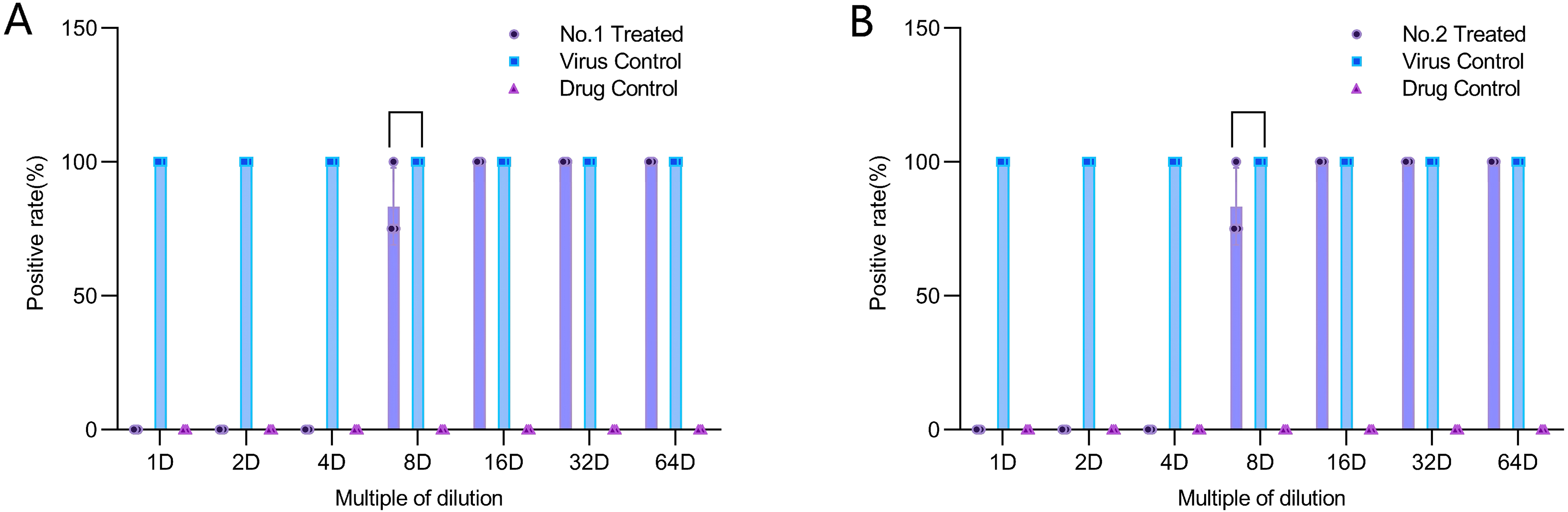
Vero cells were infected with HSV-1, followed by the addition of wood vinegar at different concentrations. Positive well rates were observed under a microscope and calculated to evaluate the inhibitory effects of Vitis vinifera wood vinegar (A) and Casuarina equisetifolia wood vinegar (B) on HSV-1 infection in Vero cells.
The CCK-8 assay results (Figure 4AB) showed that pretreatment of cells with WV-1 and WV-2 at the tested concentrations did not significantly inhibit HSV-1 (
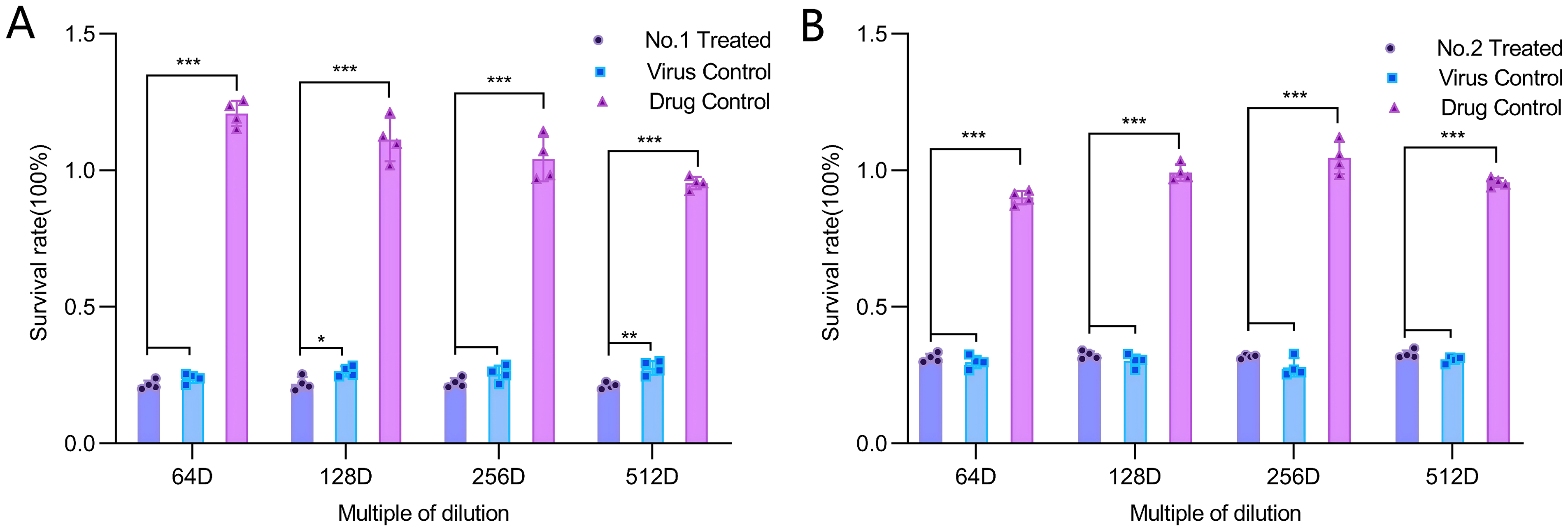
Vero cells were pretreated for 1 h with wood vinegar diluted to safe concentrations (64-, 128-, 256-, and 512-fold dilutions), followed by HSV-1 infection. Cell viability was measured using the CCK-8 assay to evaluate the protective effect of Vitis vinifera wood vinegar (A) and Casuarina equisetifolia wood vinegar (B) against viral infection.
pH Measurement of Wood Vinegar
The results (Figure 5) indicate that both Vitis vinifera wood vinegar (WV-1) and Casuarina equisetifolia wood vinegar (WV-2) are acidic. The WV-1 approaches neutrality when diluted 8-fold, whereas the WV-2 requires dilution 32-fold to reach a near-neutral pH.

pH measurement of Vitis vinifera wood vinegar (WV-1) and Casuarina equisetifolia wood vinegar (WV-2).
GC-MS Analysis of Wood Vinegar
High-resolution mass spectrometry of Vitis vinifera wood vinegar samples (Table 1) identified numerous compounds, with many showing high-confidence matches (Checked = True). The most abundant compound was trans,trans-2,4-heptadienal, with a peak area of 9.21 × 109, a retention time of 4.606 min, and a molecular formula of C7H10O. 3-Hydroxy-2-methylpyridine and N,N-dimethylaniline were also present at relatively high levels, with peak areas of 4.56 × 109 and 3.74 × 109, respectively. The identified compounds mainly included aromatic amines (eg, o-toluidine), nitrogen-containing heterocycles (eg, isoquinoline), phenolic aldehydes (eg, 5-hydroxyindole), and some fatty acid derivatives (eg, sorbic acid). Mass spectral database matching indicated that most high-abundance compounds had high-confidence matches (eg, mzCloud score of 83.1 for N,N,4-trimethylaniline), supporting the reliability of the identifications. Some compounds were verified across multiple databases, further confirming their presence.
Major Metabolites Identified in *Vitis vinifera* Extract by GC-MS Analysis, Ranked by Relative Abundance (Peak Area).

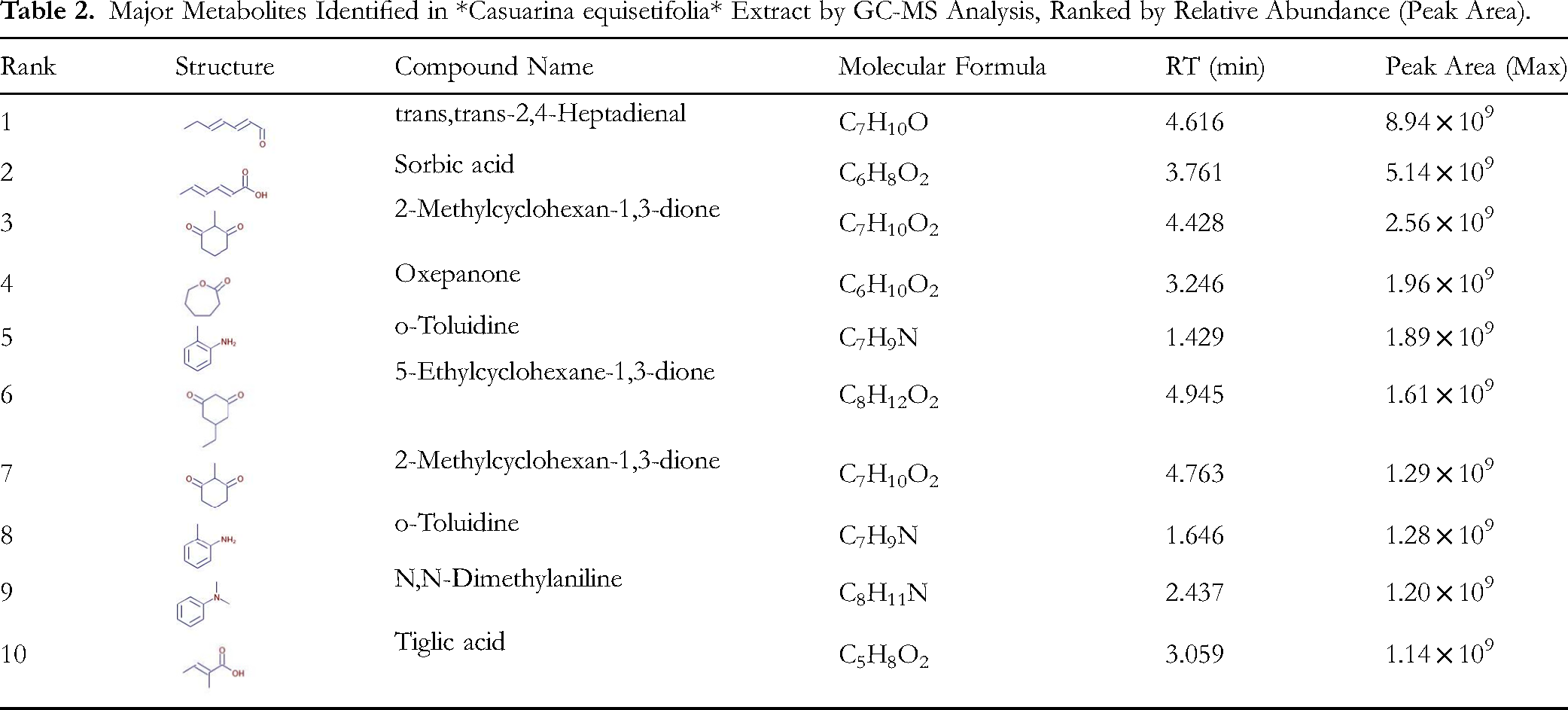
A systematic analysis was also conducted on Casuarina equisetifolia wood vinegar samples (Table 2). Here, trans,trans-2,4-heptadienal was the most abundant component, with a peak area of 8.94 × 109. Another notable compound, sorbic acid, was detected at multiple retention times and showed considerable overall abundance. Additionally, cyclic diketones such as 2-methylcyclohexane-1,3-dione and 5-ethylcyclohexane-1,3-dione were frequently identified. Cross-database verification revealed approximately 30% of the compounds had high-confidence matches. Overall, the extract exhibits a complex composition, rich in aldehydes, organic acids, ketones, and some aromatic constituents, providing a solid basis for further investigation into its chemical profile and bioactivity.
Major Metabolites Identified in *Casuarina equisetifolia* Extract by GC-MS Analysis, Ranked by Relative Abundance (Peak Area).
Discussion
During dental procedures, healthcare workers are directly exposed to respiratory aerosols containing microorganisms due to close contact with patients. These aerosols originate from both the patient's normal respiratory activities (such as breathing, talking, and coughing) and the droplets generated by dental equipment (eg, high - speed handpieces and ultrasonic scalers) during intraoral operations.40,41 As a highly diverse microecosystem, the oral cavity harbors a wide range of microbes including bacteria, archaea, and fungi, as well as human viruses such as HSV-1. These microorganisms often exist in the form of biofilms or saliva and can spread via aerosols during dental treatments, posing a risk of nosocomial infection. 42
To evaluate the potential of wood vinegar in infection control, this study assessed its cytotoxicity using Vero cells—a common model for HSV-1 research43,44 — and L929 cells, a standard cell line for oral toxicity evaluation.45,46 The results showed that wood vinegar exhibits concentration - dependent toxicity toward both cell types, with a safe and non - toxic concentration achieved at a 64-fold dilution. Notably, this toxicity threshold overlaps with the effective concentration observed in subsequent antiviral experiments. In the direct inactivation assay in vitro, wood vinegar significantly inhibited HSV-1 activity after only 15 min of exposure. Although Vitis vinifera wood vinegar (WV-1) (1- to 4-fold dilutions) and Casuarina equisetifolia wood vinegar (WV-2) (1- to 8-fold dilutions) showed cytotoxicity at effective concentrations, studies on similar disinfectants derived from Casuarina equisetifolia wood vinegar—such as the product from Tangshan Xinyanuo Company—indicate no acute oral toxicity or skin irritation. 47 This suggests that such formulations are feasible for use as surface or environmental disinfectants, capable of blocking HSV-1 transmission through localized disinfection.
However, Vitis vinifera wood vinegar exhibits higher biological risk, showing mild toxicity to L929 cells even at a 64-fold dilution. Although it demonstrates significant antiviral effects in vitro, its narrow safety window limits its potential for application. This difference highlights important distinctions in the toxicological profiles of wood vinegars from different plant sources and underscores the need to strictly examine the impact of raw material origin and preparation processes on biosafety during practical formulation development. Therefore, when selecting wood vinegar raw materials, priority should be given to systematically verified varieties with low toxicity and high efficacy (such as Casuarina equisetifolia wood vinegar). In subsequent research, purification process optimization and compound formulation design should be employed to further broaden its safe application scope.
Asymptomatic and invisible intraoral HSV-1 shedding poses a significant public health concern for viral transmission. Studies have shown that oral shedding of HSV-1 is not limited to periods of visible lesions but can also occur in the absence of symptoms, thereby increasing the risk of interpersonal transmission. 3 For instance, newborns may contract HSV-1 through contact with adult oral secretions, while genital HSV-1 infections mostly originate from oral-genital contact, particularly receptive oral sex.48,49
Based on the findings above, in the HSV-1-infected cell model, both types of wood vinegar diluted 1–4 fold significantly inhibited viral activity. This suggests that adding wood vinegar to oral mouthwash could directly inhibit HSV-1 activity, block its spread within oral tissues, and reduce interpersonal transmission risks. Notably, Tangshan Xinyanuo Company has developed a mucosal antimicrobial agent primarily composed of Casuarina equisetifolia wood vinegar, which laboratory tests confirmed can effectively inactivate the H1N1 influenza virus (Test Report No. 2019FM15560R01). This provides a precedent for applying wood vinegar in antiviral oral care products. Additionally, market research shows a clear consumer preference for natural-ingredient mouthwashes, 50 yet such products remain scarce in the market. Combining experimentally validated wood vinegar with this market demand could not only fill the gap for natural antiviral mouthwashes but also offer an innovative solution to curb oral pathogen transmission.
We also evaluated the potential of wood vinegar in preventing HSV-1 infection. Safe concentrations (64-, 128-, 256-, and 512-fold dilutions) were tested for pre-infection preventive effects. Results showed no significant viral inhibition at these concentrations, possibly due to insufficient active ingredients at safe dilution levels. Further tests revealed that at 64-fold dilution, both wood vinegars had a pH of 8.0, indicating a weakly alkaline environment. This suggests that the antiviral activity of wood vinegar may rely on an acidic setting or specific acidic components, which are ineffective under alkaline conditions.
Although the aforementioned experiments did not observe significant preventive effects, the potential of wood vinegar as an antiviral agent in vivo cannot be completely ruled out. If considered for in vivo applications, cytotoxicity must be carefully addressed. As noted by Chai et al, 51 wood vinegar can be pretreated—for example, by removing harmful substances or enriching active components—to improve its biocompatibility and application value, thereby enhancing its antiviral performance. Follow-up research could focus on optimizing and modifying the composition of wood vinegar, with further exploration of its true antiviral potential in vivo through animal experiments.
In this study, mass spectrometry analysis was performed on both WV-1 and WV-2, revealing distinct chemical species primarily consisting of acids, aldehydes, and phenols. These findings are consistent with those reported in the literature.47,52
Phenolic compounds have therapeutic uses, such as bactericides, preservatives, and disinfectants, 53 and are effective against various microorganisms, including several viruses. Grape leaf extracts contain multiple phenolic compounds that inhibit HSV-1 by blocking viral surface enrichment proteins. 20 Phenolic derivatives from GSE exhibit virucidal activity against the enveloped virus HSV-2, likely by inhibiting viral entry into host cells and reducing infectivity. 54
Both wood vinegars were found to contain high levels of acidic compounds, particularly sorbic acid, which can reduce infectivity by decomposing the viral envelope.
55
Other acidic compounds have also been identified with notable antiviral activity. For example, Masateru Ono et al isolated new resin glycosides (jalapins) from the leaves, stems, and roots of
Aldehydes are widely used in industry, medicine, and consumer products. They are toxic to cells, potentially causing DNA damage and protein adduct formation, 58 which may be related to the cytotoxicity of wood vinegars. Some aldehydes also exhibit antiviral activity. For instance, cinnamaldehyde can inhibit porcine reproductive and respiratory syndrome virus (PRRSV) by blocking viral binding, entry, replication, and release. 59 The salicylaldehyde thiosemicarbazone scaffold inhibits influenza virus PA endonuclease. 60 Treatment with an iron complex of salicylaldehyde isonicotinoyl hydrazone (Fe-SIH) in hepatitis C virus (HCV)-infected cells significantly reduced the expression of viral proteins (core and nonstructural proteins) and RNA. 61
The Vitis vinifera wood vinegar and Casuarina equisetifolia wood vinegar are rich in various compounds, most of which have demonstrated notable antiviral activity as discussed above. Therefore, their anti-HSV-1 activity may not arise from a single compound but rather from the synergistic or combined effects of multiple components.
Conclusion
Plants serve as important sources of natural products with high medical relevance. 62 This study demonstrates that wood vinegars from Vitis vinifera and Casuarina equisetifolia exhibit significant anti-HSV-1 activity in vitro. However, their cytotoxicity also shows concentration dependence, and the safety windows vary markedly depending on the plant source, highlighting the importance of raw material selection and process optimization. Composition analysis suggests that the antiviral effect may stem from the synergistic action of multiple active components, such as acids and phenols. Although preventive application shows limited efficacy, wood vinegar, as a naturally derived disinfecting ingredient, holds potential for developing surface or environmental disinfection products—including oral rinses—offering a new approach to controlling viral aerosol transmission in dental settings. Future research should focus on component purification and formulation optimization to enhance its safety and applicability.
Footnotes
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
Author contributions statement: Zhao Yiyang is the first author, Gu Jianqi is the second author, and Wu Wenhui is the corresponding author.
Funding
The Finance Department of Hebei Province under Grant [ZF2024022]. Study on the Therapeutic Effect and Mechanism of Plant-derived Wood Vinegar Mucosal Antimicrobial Agent on Oral Ulcers [ZD2022144].
Conflict of Interest
We declare that we do not have any commercial or associative interest that represents a conflict of interest in connection with the work submitted.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
