Abstract
Four 1,4-naphthoquinone dithioglucoside derivatives based on natural polyhydroxy-1,4-naphthoquinones were synthesized. These thioglucosides were screened for their antiradical and antiviral activity in vitro. Antiradical activity of tested compounds was determined by the 2,2-diphenyl-1-picrylhydrazyl radical scavenging assay. The anti-herpes simplex virus type 1 (anti-HSV-1) activity of thioglucosides was analyzed by the cytopathic effect inhibition assay and mode of antiviral action was determined by the addition of the tested compounds to uninfected cells, to the virus prior to infection, or to herpes-infected cells. Most effective inhibition of HSV-1 replication was observed at pretreatment of virus by the compounds (direct virucidal effect). The dithioglucoside conjugate with the single β-OH group and lipophilic ethyl substituent in naphthoquinone core showed the greatest antiviral activity.
Herpes simplex virus type 1 (HSV-1) is a ubiquitous human pathogen, with a worldwide prevalence rate of approximately 90%. 1 Usually HSV-1 infection is acquired within the first 2 decades of life and is characterized by latent infection in human peripheral nervous system. Approximately in 25% of infected individuals the periodic viral reactivations are followed by such common clinical manifestation as facial herpes, also known as herpes labialis or cold sores. However, HSV-1 can also cause such severe diseases as recurrent keratitis, encephalitis, and systemic disease in neonates and immunocompromised patients. 2 The most effective drugs for the selective treatment of HSV-1 are acyclovir and its nucleoside analogs targeting the virus DNA polymerase, but their prolonged use for the treatment of the disease can lead to the formation of drug-resistant strains. 3,4
The problem of drug resistance poses a current and future threat that should be addressed through the development of new effective antivirals. 5,6 It is well known that oxidative stress plays an important role in the pathogenesis of viral infections 7,8 promoting virus replication in infected cells, decreasing cell proliferation, and inducing cell apoptosis. 9
It was established that the sea urchin pigment echinochrome (
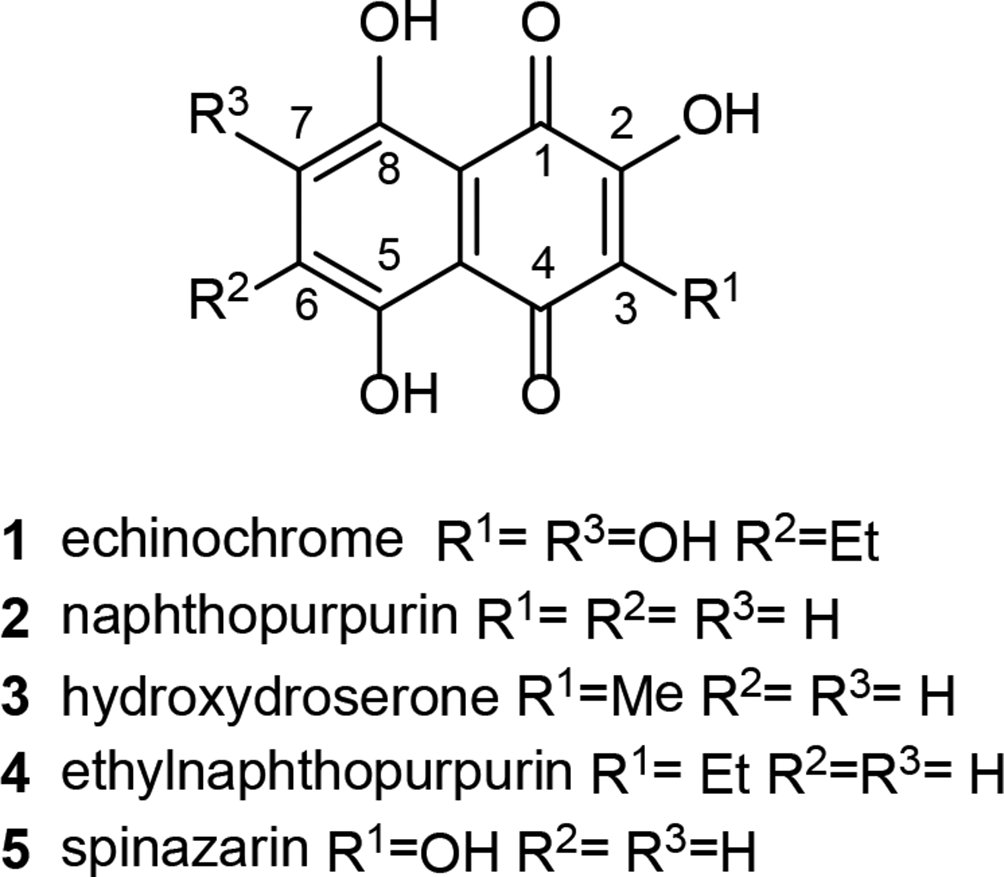
Natural hydroxylated 1,4-naphthoquinones.
Echinochrome
The aim of our study was the synthesis of new thioglucoside derivatives of polyhydroxynaphthoquinones structurally similar to echinochrome and evaluation of their antiradical and anti-HSV-1 effects in vitro.
Conjugation of Natural Naphthoquinones with Thioglucose
Conjugation of natural naphthoquinones

Preparation of thioglucosides of 1,4-naphthoquinones 11 to
Dithioglucoside
Antiradical Activity of Compounds
For the best understanding of the features of structure-activity relationship of synthesized quinone compounds, we evaluated the antiradical activity of diglucosides
Antioxidant Activities in DPPH Assay of Thioglucosides 15 to

DPPH, 2,2-diphenyl-1-picrylhydrazyl.
aEach value is presented as the mean ± standard deviation (
*Statistically significant differences between echinochrome and other compound (
Our studies revealed that thioglucosides
Cytotoxicity and Antiviral Activity of Compounds
Cytotoxicity assay was carried out to determine the concentration range of compounds for the subsequent study of its anti-HSV-1 activity in the nontoxic range for Vero cells. Echinochrome was used as reference compound. Acyclovir was used as standard antiviral for HSV-1. Based on the results of methylthiazolyltetrazolium bromide (MTT) assay the 50% cytotoxic concentrations (CC50) were calculated for all tested compounds (Table 2). The results showed that the conjugation of 1,4-naphthoquinones with thioglucose led to the formation of low toxic substances
Cytotoxic Effect (CC50), Anti-HSV-1 Activity (IC50), and Selectivity Index (SI) of Thioglucosides 15 to

The results are given as mean ± standard deviation (
*Statistically significant differences between echinochrome and other compounds (
The HSV-antiviral activity of glucosides
In conclusion, 4 hydroxy- and alkyl dithioglucoside derivatives
Experimental
General
Melting points (uncorrected) were measured with a Boetius apparatus. IR spectra were recorded on Bruker Vector-22 FT-IR spectrophotometer. 1H NMR spectra were recorded on Bruker AVANCE-500 and Bruker AVANCE-700 at frequencies 500 and 700 MHz for 1H spectra and 125 and 176 MHz for 13C spectra, respectively. 2D NMR experiments {1H−1H} COSY, {1H−13C} HMBC-qs, and {1H−13C} HSQC were used where necessary in assigning NMR spectra. Spin-spin coupling constants (
Reagents
DPPH and ascorbic acid were purchased from Alfa Aesar, Germany. Tetra-
Preparation of 14
Dichloronaphthoquinone
6,7-Di(tetra-O -Acetyl-β-D-Glucopyranosyl-1-Thio)-5,8-Dihydroxy-Naphthalene-1,4-Dione (14)
MP: 235°C to 238°C.
IR
1H NMR (500 MHz, CDCl3): 2.00 (6H, s, 2 × OAc), 2.01 (6H, s, 2 × OAc), 2.02 (6H, s, 2 × OAc), 2.11 (6H, s, 2 × OAc), 3.64 (2H, ddd, 2 × H-5′,
13C NMR (125 MHz, CDCl3): 20.52 (2 × OCO
HRMS (ESI):
Preparation of 18
To partially dissolved diacetylthioglucoside
6,7-Di(β-D-Glucopyranosyl-1-Thio)-2,3,5,8-Tetrahydroxynaphtha-Lene-1,4-Dione (18)
MP: >360°C.
IR
1H NMR (500 MHz, DMSO-
13C NMR (DMSO-
HRMS (ESI):
DPPH Radical Scavenging Assay
The determination of DPPH antiradical activity of thioglucosides was established according to the published method
33
with adaptation to colored quinones. Solutions of pure compounds in EtOH at different concentrations (0, 200 µM final concentrations) were prepared and adjusted to 2 mL total volume with 0.7 mL of DPPH-EtOH solution (6 mg/50 mL; 0.1 mM final concentration). The absorbance at 517 nm was determined after 20 minutes, and the percent free radical inhibition was calculated as follows: Inhibition (%) =100 – [(
Virus, Cell Culture, and Dissolution of Tested Compounds
The DNA-containing herpes virus (HSV-1, strain L2) was obtained from the State Collection of Viruses (Moscow, Russia). HSV-1 was grown in African green monkey kidney (Vero) cells using Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% fetal bovine serum (FBS), 100 U/mL gentamicin, and glutamine. Viral titers were determined by CPE assay and expressed as the 50% tissue culture infectious dose (TCID50/mL). The titer of HSV-1 was 106.2 TCID50/mL. Tested compounds
Determination of the Cytotoxic Activity by MTT Assay
The cytotoxicity of the tested compounds was estimated by MTT assay in Vero cell lines. 34 A monolayer of cells (1 × 104 cells/well) grown in 96-well plates was treated with different concentrations of tested compounds (from 0 to 1000 µM) and cultured at 37°C in a CO2-incubator for 3 days; untreated cells were used as controls. Cell viability was assayed using МТТ reagent. Optical density (OD) was measured at 540 nm using an ELISA microplate reader (Labsystems Multiskan RC, Vantaa, Finland) with a reference absorbance at 620 nm. The viability of the cells was calculated as (ODt)/(ODc) × 100%, where ODt and ODc correspond to the absorbance of treated and control cells, respectively. Cytotoxicity was expressed as CC50 of the tested compound that reduced the viability of treated cells by 50% compared with control cells. Experiments were performed in triplicate and repeated 3 times.
Determination of the Antiviral Activity
The anti-HSV-1 activity of tested compounds was evaluated by inhibition of virus-induced CPE using MTT test. Vero cells monolayers on 96-well microplates (1 × 104 cells/well) were infected with 100 TCID50/mL of HSV-1 and tested compounds in the concentrations from 30 to 1000 µM were used for experiments. Untreated cells were used as a cells control, and cells treated only with a virus were used as a virus control. The plates were kept at 37°C in CO2 incubator for 3 days until 80% to 90% CPE was observed in virus control compared with cells control. To investigate the mode of anti-HVS-1 action of tested compounds the following procedures were performed.
Co-treatment of Cells with Virus and Compound
Cell monolayer was simultaneously treated with virus and tested compound at a ratio 1:1 (v/v) in triplicate. After 1 hour adsorption at 37°C, cells were washed and maintenance medium was added followed by incubation for 3 days.
Pretreatment of Virus with Compounds
The virus was mixed with tested compound at a ratio 1:1 (v/v), incubated for 1 hour at 37°C, then applied to monolayer of cells in triplicate. After 1 hour adsorption at 37°C, cells were washed and maintenance medium was added followed by incubation for 3 days.
Pretreatment of Cells with Compounds
Monolayer of cells was pretreated with tested compounds in triplicate and incubated at 37°C for 1 hour. Thereafter, the cells were washed and infected with virus at 37°C for 1 hour. Unabsorbed virus was removed by washing and cells were incubated under maintenance medium for 3 days.
Treatment of Infected Cells
Monolayer of cells was infected with the virus at 37°C for 1 hour, then washed, and treated with tested compounds in triplicate for 3 days.
MTT test was used for assessing the antiviral activity of compounds, and the viral inhibition rate (IR, %) was calculated according to the formula, 35 IR = (ODtv − ODcv) / (ODcd − ODcv) × 100, where ODtv represents the OD of cells infected with virus and treated with the test compound; ODcv corresponds to the OD of the untreated virus-infected cells, and ODcd is OD of control (untreated and noninfected) cells. The concentration of compound that reduced the virus-induced CPE by 50% was determined as IC50. SI was calculated as the ratio of CC50 to IC50. Experiments were repeated 3 times.
Statistical Analysis
CC50 and IC50 were calculated by regression analysis of the dose-response curve. Statistical processing of the data was performed using Statistica 10.0 software. The results are given as mean ± standard deviation. The differences between parameters of control and experimental groups were estimated using the Wilcoxon test and were considered significant at
Footnotes
Acknowledgments
The authors acknowledge Dr R.S. Popov, Dr V.P. Glazunov, and Dr V.A. Denisenko for MS, IR, and NMR measurements.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
