Abstract
Objective
As the first of its kind, this research investigates the influence of ontogeny and seasonality on yield and composition of essential oils (EOs) from Corymbia citriodora grown in a homogeneous setting, offering new perspectives on their chemodiversity and antifungal properties against Botryosphaeria dothidea.
Methods
Leaves of C. citriodora from different seasons and leaf stages were hydro-distilled to extract EOs. Chemical analysis of EOs was done using GC-MS and GC-FID. The EOs from the fresh leaves (FL) and abscission leaves (AL) were tested for their antifungal activity using the microatmosphere method against B. dothidea.
Results
EO yield varied seasonally in FL, being higher in summer (1.8-2.25% w/w) compared to winter (1.0-1.75% w/w) and significantly lower (0.8-0.82% w/w) in AL. EO from FL was transparent and had a strong lemon fragrance. Whereas EO from AL was pale yellow color and weak aroma. The refractive index (RIs) values of EOs ranges from 1.405 to 1.539. Citronellal was a dominant compound (50.35-82.01%), and it higher levels found in FL. Seasonal and ontogenetic variations influenced the EO composition. Citronellol content (1.10% seasonally, 1.50% across leaf types) and isopulegol content (2.55% seasonally, 2.70% across leaf types) remained relatively less varied across the seasons and developmental stages. Fresh leaf EOs showed strong antifungal activity against B. dothidea, with 100% inhibition at all concentrations (15 µL, 30 µL and 60 µL). However, abscission leaf EOs showed partial inhibition (5.6%) at 15 µL and complete growth inhibition was observed at 30 and 60 µL.
Conclusion
Essential oil yield was influenced by leaf type and season. Fresh summer leaves producing citronellal-rich oils which showed highest antifungal activity. Notably, the strong antifungal activity of EO from fresh leaves of C. citriodora further reinforces its potential as natural antifungal agents against B. dothidea.
This is a visual representation of the abstract.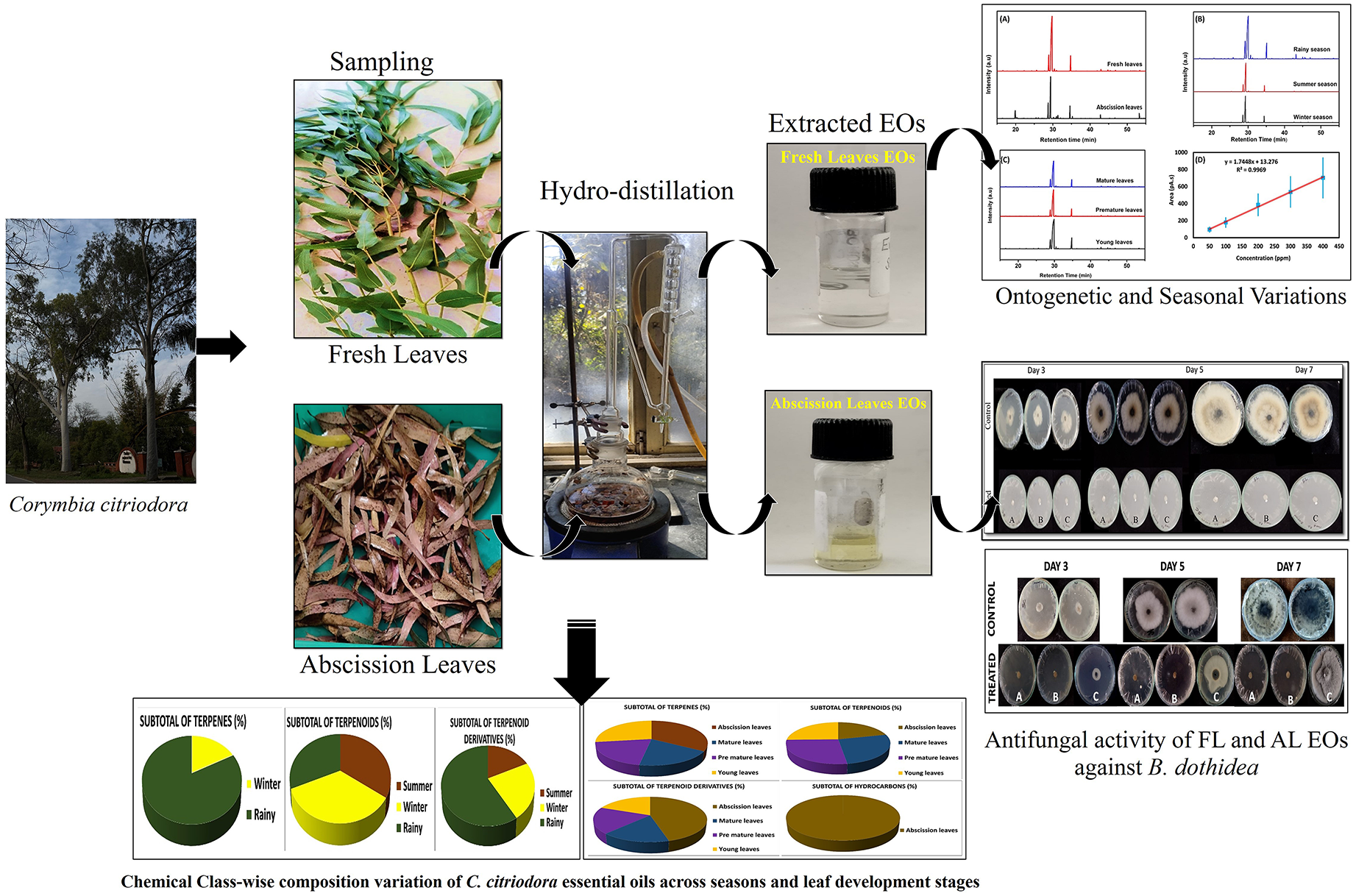
Keywords
Introduction
Corymbia citriodora (Hook.) is a native Australian tree that is widely known as lemon scented Eucalyptus. This tree can reach heights of up to 50 meters and has been valued for its wood and essential oil. It is commercially planted in India and has adapted well to the country's agroclimatic conditions. 1 C. citriodora has a high carbon sequestration capability. 2 A significant characteristic ie, high density of C. citriodora wood has a direct impact on pulp manufacturing, influencing the entire process from wood chipping to the quality of the finished pulp. 3 Due to its high durability and strength, wood is used in making furniture, poles, fences, houses, paper and fuel, while its essential oil (EO) holds significant value in the chemical and pharmaceutical industries. 4 C. citriodora is the leading source of Eucalyptus EOs production in the world trade. Commercially, EOs are produced in the countries such as China, India and Brazil, 4 and are mostly used for perfumes, medicines as pain reliever, 5 antifungal, 6 allelopathic, 7 insect repellent agent. 8
EOs of C. citriodora contains mainly oxygenated monoterpenoids (∼91.8%), monoterpene hydrocarbons (∼0.76%), sesquiterpene hydrocarbons (∼1.51%), oxygenated sesquiterpenes (∼0.29%), and non-terpenoid (∼0.91%) class of compounds. 9 Its (EO's) major constituents are citronellal (46.2-86%), followed by citronellol (4.5-11.86%) and isopulegol (∼4.66%), while the remaining compounds occur in minor amounts (<2%).9,10 A characteristic aromatic and medicinal property of C. citriodora EOs is due to its unique chemical composition with abundant presence of citronellal. EO yield and composition have been shown to vary depending on the method of extraction, 11 part of plant used, 12 location of plant, 13 light conditions, 14 the time of plant material collection, 13 or the hybridization of the plant with another related tree species. 15 Recently a comparative study on the morphological traits, EO composition and yield of C. citriodora growing in two different environmental conditions has been reported. 16
However, several factors such as environmental conditions, season, soil characteristics, and plant, have been observed to exert a notable influence on leaf and EOs yield.17–19 These influences are also strongly affected by the environment, climatic conditions, geographic location, growth stages, harvesting time and extraction methods.19–21 Therefore, identifying the ontogenetic and seasonal variations in the content and composition of high valued essential oil yielding plants becomes an important aspect. In view of the great commercial demand and wide applications, it is justifiable to carry out studies on the influences of the ontogenetic and seasonal variation in EO composition and yield, marking its importance to farmers and producers of essential oils. In order to optimizing the practical applications of C. citriodora EO in disease control, it requires to assess its antifungal activity. Further it addresses a growing need for natural alternatives to synthetic fungicides which can be used in disease control. Eucalyptus is widely planted to meet the global demand for wood-based paper and pulp. However, diseases caused by members of the Botryosphaeriaceae family pose a significant threat to the productivity of woody species, including Eucalyptus. The recent appearance of leaf blight symptoms on Eucalyptus plantations in northern India, attributed to Botryosphaeria dothidea, highlights the urgent need to strengthen current management strategies. 22 Essential oils offer a promising alternative, as they provide safer and multi-target antifungal activity in situations where limited chemical control options and resistance development are major concern.
Therefore, this study aims to assess the EO yield and composition of C. citriodora leaves as a function of the ontogenetic and seasonal variation. Further, in view of utilization, the extracted EO was evaluated for antifungal activity against Botryosphaeria dothidea, a foliar pathogen of Eucalyptus in northern India.
Materials and Method
Plant Materials and Chemicals
All chemicals such as standard, reagent, solvent of GC, analytical and laboratory grade were purchased from Sigma – Aldrich Pvt. Ltd and Himedia Pvt Ltd Leaves of C. citriodora were collected from two distinct locations grown in homogenous environment ie, ICFRE-FRI Campus (30°20′41.61286″(N) Longitude:(E)78°0′45.42297″) and FRI Deemed to university campus (30°20′24.43795″(N) Longitude: (E) 77°59′42.06785″) at Forest Research Institute, Dehradun. Total four trees and three samples per types were considered during the experiments. Fresh leaves from different parts of the same branch were collected from top, middle, and base part of the branch. Leaves were collected in three different seasons ie, in the months of January-February 2024 (winter season), April-June 2024 (summer season) and July-August 2024 (monsoon season). Young, pre mature, mature, and abscission leaves of C. citriodora were collected during summer season (Table 1). The plant specimens were identified and authenticated (Herbarium Reg. No. 175075) by the Systematic Botany Division, Forest Research Institute, Dehradun and the voucher specimen was submitted to the DD herbarium (Figure S1).
Seasonal Variation and Essential Oil Yield from Corymbia citriodora Leaves.
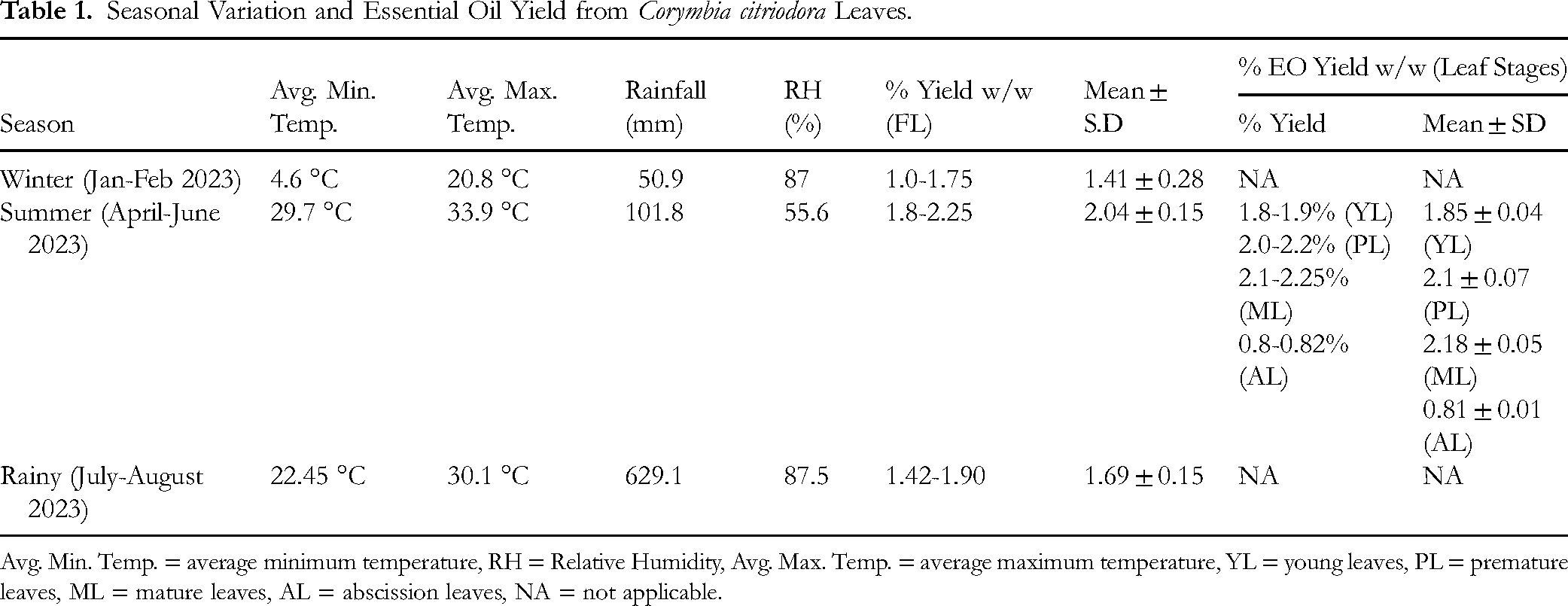
Avg. Min. Temp. = average minimum temperature, RH = Relative Humidity, Avg. Max. Temp. = average maximum temperature, YL = young leaves, PL = premature leaves, ML = mature leaves, AL = abscission leaves, NA = not applicable.
Sample Preparation and Isolation of EOs
All collected fresh (young, pre mature, mature) and abscission leaves from C. citriodora were cleaned and processed into small pieces before distillation. Processed sample (250 g) was hydro distilled using distilled water by a Clevenger apparatus for 4 h to 4.5 h, until no further oil was recovered. After completion of extraction, EO was separated with the help of separating funnel and collected in storage vial. This process was performed for all types of samples in triplicate. Yield of EOs was calculated with respect to the weight of the leaves. All isolated EOs were stored in amber colour glass vials and kept in refrigerator at −18 °C until analysis.
Characterization of Essential Oil
Refractive Index (RI) Determination
RI of the EOs was measured by Decibel refractometer (model R-4, Year 2008) at room temperature (ie, 24-26 °C). All the samples were performed in five times and reported as average value and standard deviation.
Gas Chromatography-Mass Spectrometry (GC-MS) Analysis
The GC-MS analysis was carried out in Agilent 7890 B GC equipped with Agilent 5977A mass spectrometer using a 30 m × 250 µm × 0.25 µm DB 5MS capillary column. Essential oil samples were diluted in GC grade DCM and prepare a 1000 ppm solution for GC-MS analysis. Each sample (1 µL) was injected in the split less mode with the column temperature programmed for 40 °C for 4 min and afterward increased to 130 °C at 2 °C/min, subsequently increased to 220 °C at 10 °C/min and finally held at this last temperature for 5 min. The injector and detector were set at 250 °C and 280 °C respectively. The carrier gas was He with a flow rate of 1 mL min−1. Mass spectra were acquired over 40-450 amu range at 3.5 scan/second with MS source and MS quad temperature at 230 °C and 180 °C respectively. The transfer line was set at 280 °C. The identification of the EO composition was performed by their retention indices (RI) using n-alkanes (C8-C21) for analysis (Figure S2), peak matching library search, as well as published mass spectra. The data generated from GC-MS analysis were expressed as relative peak areas of the total ion chromatogram.
Gas Chromatography-Flame Ionization Detector (GC-FID) Analysis
In order to estimation of the volatile compounds in the EOs, GC-FID analysis was carried out in Agilent 8890 GC system using the capillary column ie, a 30 m × 250 µm × 0.25 µm DB 5MS capillary column. EO samples were diluted in GC grade dichloromethane (DCM) and prepared a 1000 ppm solution for GC-FID analysis. Each sample was injected (1 µL) in the split less mode with the column temperature programmed for 40 °C for 4 min and afterward increased to 130 °C at 2 °C/min, subsequently increased to 220 °C at 10 °C/min and finally held at this last temperature for 5 min. The injector and detector were set at 250 °C and 280 °C respectively. The carrier gas was N2 with a flow rate of 1 mL/min.
The estimation of the compounds were carried out by using a constituent ie, ±-citronellal as a standard. During analysis, a series of standard solution of ±-citronellal were performed at 1000 ppm, 800 ppm, 400 ppm and 200 ppm. From the values of concentration and peak area a calibration curve (amount vs peak area) was prepared (Figure S3). The calibration curve shows significant linearity with correlation r = 0.994.
Fungicidal Activity
The antifungal activity of essential oils (EOs) derived from the fresh leaves (FL) and abscission leaves (AL) of C. citriodora was evaluated against Botryosphaeria dothidea, a foliar pathogen of Eucalyptus prevalent in northern India (GenBank accession numbers: ITS = OR539471, tef1 = OR545806, tub2 = OR545808). 18 The pure culture of B. dothidea (NTCC 1305) was obtained from the National Type Culture Collection (NTCC) of the Forest Research Institute, Dehradun, India. The fungus was cultured on freshly prepared Potato Dextrose Agar (PDA) medium and incubated at 25 °C for 7 days.
The microatmosphere method was used to assess the antifungal activity of essential oils (EOs) at three concentrations: 60 µL, 30 µL, and 15 µL.
23
Initially 5-mm-diameter mycelial discs obtained from a 7-day-old culture of B. dothidea (NTCC 1305) were placed at the center of each Petri dish. Following the inversion of the plates, sterilized filter paper disc treated with 60 µL, 30 µL, or 15 µL of each EO was placed in the middle of the inverted lid. The equivalent procedure was undertaken to prepare control plates, but no EO was added. After being parafilm-sealed, the plates were incubated at 25 °C until the control plates exhibited full fungal growth. Following the incubation period, the radial mycelial growth in each plate was measured. All experiments were conducted in triplicate using a completely randomized design. The antifungal efficacy of the EOs was expressed as the percentage inhibition of mycelial growth, calculated using the following formula:
Statistical Analysis
Total four trees and 3-5 samples per types were considered during the experiments. Statistical analysis was carried out using R software. Data were analyzed using one-way ANOVA followed by Tukey's HSD post-hoc test to assess pairwise differences among seasons and leaf developmental stages, with significance set at P < .001. Mean and standard deviation were calculated for the compounds to assess central tendency and variability across different leaf types and seasonal conditions. In addition to it, CRD (Completely Randomized Design) was employed with laboratory experiment while conducting the antifungal activity.
Results
Physicochemical Properties, Ontogenetic and Seasonal Variations in Essential Oils (EOs)
The EOs of C. citriodora, extracted from fresh leaves (FL) ie, mature, premature and young, were transparent, whereas the EO extracted from abscission leaves (AL) acquired a pale yellowish (Figure S4). Moreover, essential oils extracted from fresh leaves (pre mature and mature) exhibited a more pronounced fresh lemon-like aroma compared to those from abscission and young leaves. The total yield of EO from the leaves of C. citriodora along with the weather data of the experimental area have been shown in Table 1. A significant increase in yield was observed in the summer season compared to the winter season. The EO yield from FL collected in summer (SL) and winter season (WL) is 1.8-2.25% (w/w) and 1.0-1.75% (w/w) respectively.
The EO yield (w/w) from FL and AL during summer season ranged between 1.8-2.25% (w/w) and 0.8-0.82% (w/w) respectively. Notably EO yield was low (1.0 to 1.75% w/w) during winter season when the temperature, rainfall and relative humidity (RH) were 4.6 °C to 20.8 °C, 50.9 mm and 87% respectively. Similarly, maximum EO yield (up to 2.25%) was observed under the lowest RH (55%), highest temperature (29.7 °C to 33.9 °C) and average rain-fall (101.8 mm) environmental conditions. The average refractive index (RIs) for C. citriodora EO of AL and all three types of FL were 1.405 ± 0.0015 and 1.539 ± 0.0011 respectively.
During summer season the EO yield was maximum, therefore further the yield of EOs from FL of C. citriodora was studied at different developmental stages such as young (at top of the branch), premature (at middle of the branch) and mature (at base of the branch). Total 1.8-1.9% (w/w) EO content was observed in the leaves grown at the top of the branch which were found at their young stage. On the other hand, EO content of leaves at middle (premature) and bottom (mature) of the branches were almost similar, which was ranging from 2.0% to 2.25% (w/w). ANOVA confirmed that both season and leaf developmental stage had a significant effect on EO yield (Tables 2 and 3). Post-hoc comparisons showed that yield peaked in summer and in premature leaves, whereas the lowest yield values were consistently observed in winter and abscission leaves (Tables S1 and S2).
One-way ANOVA for the Effect of Season on Essential oil Yield of Corymbia citriodora.

***P < .001, **P < .01, *P < .05, P < .1.
One-way ANOVA for the Effect of Leaf Stages on Essential oil Yield of Corymbia citriodora.

***P < .001, **P < .01, *P < .05, P < 0.
Ontogenetic and Seasonal Variations in EO Composition
Chemical constituent of EO were identified for their name, class, retention time (Rt), molecular formula (Mf), molecular mass (Mm), mass fragmentation (m/z) values, similarity index (SI) and linear retention indices (LRI) values as shown in Table 4. A total of 24 compounds which belongs to the terpenes, terpenoids, terpenoid derivatives and hydrocarbons classes have been identified.
Chemical Characterization of Essential Oils from Corymbia citriodora.

Figure 1 presents the GC chromatograms of C. citriodora EOs. Panel (A) compares the essential oil profiles of fresh versus abscission leaves, revealing differences in chemical composition. Panel (B) illustrates the seasonal variations in composition, while panel (C) demonstrates the impact of developmental stages on the essential oil profile. Panel (D) shows the calibration curve, confirming the linear response of the mass spectrometry detector. The ontogenetic and seasonal variations in the composition of essential oils (EOs) from C. citriodora grown in a homogeneous environment was summarized in Table 5. The data highlight how different developmental stages and seasonal changes influence the relative abundance of key compounds in the essential oil. Notable shifts in the chemical profile were observed, indicating the effects of ontogenetic and seasonal factors on EO composition.

GC-MS Chromatogram of C. citriodora Essential oil: (A) Fresh Versus Abscission Leaves, (B) Seasonal Variations, and (C) Developmental Stage Differences. (D) Calibration Curve Demonstrating the Linear Response of the Mass Spectrometry Detector (Error Bars Indicate Standard Deviation (Scaled ×40 for Clarity).
Ontogenetic and Seasonal Variation in Composition of EOs of C. citriodora Grown in Homogeneous Environment.

As shown in Table 5, different class of the compounds were detected in varied content ie, terpenes (0 to 1.49%), terpenoids (72.39 to 92.20%), terpenoid derivatives (0.81 to 2.71%) and hydrocarbons (8.51%). The three predominant constituents such as citronellal (50.35 to 82.01%), isopulegol (7.64 to 14.35%), and citronellol (7.16 to 10.54%) were detected in the EOs. Substantial amount of distinct class of compound ie, hydrocarbon p-cymene (∼8.51%) along with eucalyptol (∼1.16%) were also observed in abscission leaves.
Moreover, trace amount of p-Methane-3, 8-diol (PMD ∼0.32%) was detected in the fresh leaves of rainy season. The seasonal variation study revealed that citronellal (∼74.37% mean) is the dominant chemical with a moderate variation ie, standard deviation (SD) of 6.80%(Table 6). Citronellal showed significant differences between summer and rainy seasons, as well as between winter and rainy seasons (Table 7 and Table S3). Similarly, isopulegol exhibited a mean of 11.51 ± 2.55% and varied significantly between the summer-rainy, winter-rainy, and winter-summer (Table 8 and Table S4). Citronellol showed minor seasonal changes (8.94 ± 1.10%) (Table 6) with significant differences detected by Tukey's HSD test between rainy-summer, winter-summer, and rainy-winter seasons (Table 9 and Table S5).
Effect of Seasonal and Leaf Type Variations on the Content of Major Compounds of Corymbia citriodora Essential Oils (Mean ± SD, %).

ND = Not detected; *Detected only in abscission leaves.
One-way ANOVA for the Effect of Season on Citronellal Content of Corymbia citriodora.

***P < .001, **P < .01, *P < .05, P < .1.
One-way ANOVA for the Effect of Season on Isopulegol Content of Corymbia citriodora.

***P < .001, **P < .01, *P < .05, P < .1.
One-way ANOVA for the Effect of Season on Citronellol Content of Corymbia citriodora.

***P < .001, **P < .01, *P < .05, P < .1.
The analysis of volatile compounds in EOs of mature, pre-mature, and young leaves revealed a diverse array of terpenes, terpenoid and its derivatives. Major compound identified ie, citronellal emerged as the highest concentration of 82.01%, 80.99%, and 74.52% in pre-mature, mature, and young leaves, respectively. The Table 5 illustrate that as leaves mature, there were noteworthy changes in the content of several compounds. Citronellal content declined from 82.01% in pre-mature leaves to 74.52% in young leaves, a decrease of 7.49% points, corresponding to an approximate 9% relative reduction.
The EO composition of C. citriodora exhibited significant ontogenetic and seasonal variability in the relative abundance of major chemical classes (Table 5, Figures 2 and 3). Whereas, the pre-mature leaves contained the highest content of terpenoids (∼92.2%), followed by mature (∼90.51%) and young leaves (∼86.65%). In contrast, abscission leaves shows a relatively lower terpenoid content (∼72.39%) along with hydrocarbon content (∼8.51%). The terpenes were present in trace to small amount across all leaf stages, ranging from 0.66% to 1.15%, while terpenoid derivatives varied slightly between 0.98% and 2.71%. Seasonally, EO from leaves harvested during the summer shown the highest total content of terpenoids (∼89.66%), followed by winter (∼88.37%) and rainy seasons (∼81.55%). Significant increase in the content of terpenes (∼1.49%) and terpenoid derivatives (∼2.61%) was observed in the rainy season in compared to other seasons. Exceptional observation was that the hydrocarbons (p-cymene) not detected in FL EOs.

Class-wise Composition of C. citriodora Essential Oils Across Seasons Showing Variation in Terpenes, and Terpenoids, and Terpenoids Derivatives.

Class-wise Composition of C. citriodora Essential Oils Across Different Leaf Stages Showing Variation in Terpenes, Terpenoids, Terpenoids Derivatives, and Hydrocarbons.
Antifungal Activity of Essential Oils
In this study, the EOs extracted from fresh (FL) and abscised (AL) leaves of C. citriodora exhibited significant antifungal activity against Botryosphaeria dothidea (NTCC 1305). The FL oils were highly effective, showing complete inhibition (100%) of mycelial growth at all tested concentrations (60 µL, 30 µL, and even 15 µL), with no visible fungal development throughout the 7-day incubation period (Figure 4). AL oils displayed concentration-dependent effects: at 60 µL and 30 µL 100% suppression while at 15 µL only 5.6% inhibition was recorded. This marginal activity was not sufficient to restrict colony development, as fungal growth nearly covered the entire dish by day 7 (85 mm) as compare to the untreated control (Figure 5). In the fallen leaf control set, mycelial expansion was consistent, ranging from 19-25 mm at day 3 to over 90 mm by day 7. Such as small difference at 15 µL, although measurable, does not indicate any meaningful antifungal effect. In the fresh leaf control set, colony diameters increased rapidly from 33-40 mm at day 3 to 53-60 mm at day 5, and further to 88-90 mm by day 7, completely covering the 90 mm Petri dishes within one week. Notably, these results clearly demonstrate the consistent and rapid growth capacity of B. dothidea in the absence of EOs which is further validating the experimental results. Thus relative to untreated controls, FL oils underscored their strong antifungal activity against B. dothidea at all tested concentrations.

Evaluating the Antifungal Efficacy of the EOs Extracted from Fresh Leaves at Three Different Concentrations 60 µl (A), 30 µl (B) and 15 µl (C) Against Botryosphaeria dothidea Using Micro Atmosphere Method.

Evaluating the Antifungal Efficacy of the EOs Extracted from Abscission Leaves at Three Different Concentrations 60 µl (A), 30 µl (B) and 15 µl (C) Against Botryosphaeria dothidea Using Micro Atmosphere Method.
Discussion
Physicochemical Properties, Ontogenetic and Seasonal Variations in Essential Oils (EOs)
The pale yellow colour of the EO from AL could be due to the presence of aromatic and oxygenated constituent in the aged and denatured leaves. The pattern of EO accumulation in C. citriodora leaves showed that the dynamics of the EO content. The significant yield increase in leaves collected in summer season appears to be metabolically regulated by different weather conditions. These findings were consistent with a previous study on E. tereticornis and E. citriodora that found significant seasonal variability in EO yield.24,25 The variation in % oil yield from E. tereticornis was observed as 0.95% (w/w) in summer, 0.65% (w/w) in winter, and 0.79% (w/w) in rainy season. Variability in climatic factors, such as temperature, rain fall, and precipitation, may have a direct impact on oil yield.
Furthermore, the physiological state of the plant itself is crucial. The essential oil accumulation in C. citriodora leaves shows a dynamic regulation across seasons, which is likely metabolically controlled. The observed reduction in EO content of abscised leaves (AL) may be attributed to the non-metabolic state of senescent tissue and the natural volatilization of compounds during prolonged exposure to environmental factors such as heat, light, oxygen, and air. Similar observations were reported where oils from decaying leaves of E. citriodora exhibited lower yields and compositional shifts compared to fresh foliage, largely due to the loss of more volatile monoterpenes during decomposition. 26
Ontogenetic and Seasonal Variations in EO Composition
The essential oil composition of C. citriodora exhibited significant ontogenetic and seasonal variations, as revealed by GC-FID or GC-MS analysis. The highest content of citronellal has been found in fresh leaves, whereas significant drop in citronellal content was observed in abscission leaves. At the same time isopulegol content was increased slightly in AL compare to FL. These finding were consistent with who also reported a noticeable increase in isopulegol (∼13.35%) in the decaying leaf oil of Eucalyptus tereticornis, whereas isopulegol was absent in the fresh leaf oil. 27 The content of few compounds such as p-cymene and eucalyptol in AL essential oil was absent or minimal in FL. This suggests that leaf composition changes during senescence. Only summer abscission leaves contain p-cymene, suggesting a possible function in stress response or leaf senescence. These patterns demonstrate how the type of leaf affects the content of essential oils. Concentration of major constituent citronellal was observed to be maximum in the summer season followed by the winter and rainy season. On the other hand, the concentration of other two predominant compounds ie, isopulegol and citronellol became maximum during rainy season.
C. citriodora EO was reported to have 1%-2% p-methane 3,8-diol (PMD), when refined to increase its PMD content (up to 70%), is widely used as potential insect repellents. 28 But, surprisingly in this study trace amount of PMD (∼0.32%) was detected that to in the FL of rainy season only. Citronellal is a precursor of PMD, and this seasonal occurrence may suggest environmental or physiological factors favoring its formation. Further investigation is required to elucidate the mechanisms and conditions that lead to PMD formation in the C. citriodora. Citronellal compound showed moderate variation likely driven by environmental conditions. Whereas isopulegol and citronellol exhibited minimal seasonal changes, indicating their relative biochemical stability. Moreover, absence of p-cymene in FL, confirms its function in leaf senescence. 29
The exclusive presence of above discussed compounds in the young leaf stage suggests that specific volatiles compounds were synthesized during early leaf development. These compounds likely playing a role in leaf protection and interactions with the environment during this critical growth phase.30–32 Citronellal consistently emerged as the most abundant compound across all leaf stages, with slightly higher concentrations in mature and pre-mature leaves in compare to young leaves. The highest levels of citronellal in oils from 3- and 5-year-old trees, with values of 82.6% and 82.5% respectively was estimated. However, as tree aged, there was a significant decrease in citronellal yield (55.5%). This decline in citronellal content with age was comparable to the observed decrease in citronellal levels in abscission leaves in the current study.
Citronellal yield may be influenced by the age or condition of the foliage, which reinforces the notion that senescence and leaf maturity were significant factors in altering the chemical composition of essential oils. The high citronellal content likely plays an ecological role by either attracting or deterring herbivores. The cultivation of C. citriodora under sub-tropical conditions shows that, despite variation in environmental condition or seasonal variations, the overall EO yield quite good and comparable with the commercial cultivation.13,25,33 However for highest EO yield, the leaves should be harvested during summer season. Further, harvesting of pre mature and mature leaves should be carried out during summer for maximizing EO and citronellal yield.
The findings demonstrate that the effect of the both ontogeny and season variation in the particular classes of compounds of C. citriodora EOs. The high terpenoid content (%) observed in pre- mature and summer-harvested leaves. The specified developmental stage and the summer season represent the most biosynthetically active phase and the optimal conditions, respectively, for terpenoid production. Whereas, AL showed reduced terpenoid levels which indicative of compositional oxidation during senescence. Similarly, the elevated levels of terpenes and terpenoid derivatives during the rainy season may reflect physiological adaptations to increased humidity or heightened biotic stress.
Antifungal Activity of Essential Oils
The findings suggested that the antifungal compounds in FL EOs remain potent enough to inhibit fungal growth even at 15 µl (minimal) concentrations. Whereas, the reduced efficacy of AL EOs at 15 µL indicates a decline in the concentration or potency of antifungal compounds particularly citronellal (∼50.35%). These results shows that the antifungal activity of essential oils is primarily attributed to the concentration and composition of specified bioactive compounds.34,35 The potent antimicrobial activity of C. citriodora due to citronellal was reported earlier. 36 Being consistent with previous studies, the MIC values of C. citriodora EO ranged from 0.11 mg/mL to 0.52 mg/mL against various fungal species including Aspergillus niger (ATCC 6275), A. flavus (ATCC 9643), A. ochraceus (ATCC 12066), Candida albicans (ATCC 12066), Penicillium ochrochloron (ATCC 48663) and P. funiculosum (ATCC 56755). 37 Further these findings support the strong antifungal properties of the essential oils from C. citriodora.
The enhanced antifungal activity of essential oil from fresh leaves compared to abscission leaves appears to be primarily associated with the higher content of citronellal in fresh leaves relative to abscission leaves. Additionally, minor terpenoids such as β-pinene, β-myrcene, geraniol, and geranyl acetate were detected in fresh leaves but were absent in abscission leaves, and may contribute synergistically to the antifungal effect. The reduced activity of AL EOs may also be attributed to the degradation or reduction in content of these bioactive compounds during leaf senescence, as suggested by studies on other plant species. 17 It was observed that fresh C. citriodora leaf essential oils exhibited strong antifungal activity against B. dothidea. While the efficacy of oils from abscised leaves decreased, likely due to the degradation of bioactive compounds. Despite all valuable insights, current study holds restricted geographic sampling, and inter- annual variation. Further, antifungal efficacy was evaluated only in a lab conditions, which could be tested in field conditions and mode of action to be investigated.
Conclusion
This study examined the yield, composition, and antifungal properties of C. citriodora essential oils (EOs). The results show that EO yield and citronellal content were highest in fresh leaves harvested during the summer. The composition of the oils varied with leaf development, and citronellal being the dominant compound. EOs from fresh leaves was most effective in inhibiting the growth of B. dothidea, while EOs from abscission leaves showed poor antifungal activity. These findings are valuable for optimizing the harvesting period to maximize EO yield and its quality in terms of bioactive compounds. C. citriodora essential oil shows potential as a natural antifungal agent for agricultural use. The mode of action, in vivo activities, formulation strategies, and long-term efficacy of C. citriodora EO in agricultural applications should be investigated in future research.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251385775 - Supplemental material for Ontogenetic and Seasonal Variations in Corymbia citriodora Essential Oils: A Study of Chemodiversity and Antifungal Effects Against Botryosphaeria dothidea
Supplemental material, sj-docx-1-npx-10.1177_1934578X251385775 for Ontogenetic and Seasonal Variations in Corymbia citriodora Essential Oils: A Study of Chemodiversity and Antifungal Effects Against Botryosphaeria dothidea by Shikha Chandola, Surendra Singh Bisht, Shailesh Pandey, Rajendra K. Meena, Chaitanya Lakhera and Rakesh Kumar Bachheti in Natural Product Communications
Footnotes
Acknowledgments
The authors are thankful to the Director of ICFRE-FRI for providing the necessary laboratory facilities. The authors also acknowledge Smt. Baishakhi Sarkhel, Technical Assistant, Chemistry & Bioprospecting Division, ICFRE-FRI, Dehradun, for recording the GC-MS of essential oils and for her assistance.
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contribution Statement
Surendra Singh Bisht conceptualized and designed the study, coordinated the work, and provided overall supervision. Shikha Chandola was responsible for drafting the manuscript analyzing the biological experiment data. Chaitanya Lakhera conducted the chemical and biological experiments. Shailesh Pandey contributed to the biological data analysis and provided related supervision. Rajendra K. Meena assisted in statistical analysis. Rakesh Kumar Bachheti assisted in data analysis and interpretation. All authors reviewed and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
