Abstract
Objective
Honey from stingless bees has strong antioxidant qualities due the presence of phytochemical substances, especially phenolic and flavonoid compounds. This study aimed to determine phenolic, flavonoid content, color intensity and antioxidant activity of stingless bee honey (SBH).
Methodology
Fifty-seven SBH samples were collected from four regional states of Ethiopia representing various geographical origins. The total phenolic content (TPC) was determined using 25 to 75 mg gallic acid/100 g of honey. The total flavonoid content (TFC) was determined by adding 5 mL of a honey solution to 5 mL of 2% aluminum chloride (AlCl3) in a standard procedure using colorimetric methods. The free radical scavenging activity of honey samples was estimated by using 2,2-diphenyl-1-picrylhydrazyl (DPPH).
Results
The SBH's average phytochemical composition and antioxidant activity were as follows: TPC (42.3 mg GAE/100 g), TFC (54.6 mg QE/100 g) and DPPH activity (112.1 mg of ascorbic acid equivalent/100 g). The phytochemical composition and antioxidant activity of SBH were: TPC (73.8 mg GAE/100 g), TFC (86.6 mg QE/100 g) and DPPH activity (165.8 mg of ascorbic acid equivalent/100 g) respectively. The present results showed that the phenolic and flavonoid contents and antioxidant activity of SBH varied significantly among the sampling areas. Phenolic and flavonoid compounds were the most abundant in dark amber honeys, however the amounts varied widely. Additionally, dark amber honeys showed higher antioxidant qualities. The statistical analysis showed a positive relationship between color intensity, phenolic and flavonoid content and antioxidant activity.
Conclusion
Particularly, M. beccarii honey exhibited remarkable quantities of phenolic and flavonoid contents and antioxidant activity. SBH generated from the two stingless bee species, has promising phenolic and flavonoid content and antioxidant qualities. Further study on the physiology of the active ingredients on the Ethiopian SBH is recommended to discover the benefit for human therapy.
Introduction
Ethiopia hosts at least six identified species of stingless bees, including the widespread ground- nesting species Meliponula beccarii, alongside Liotrigona bottegoi, Liotrigona baleensis sp.nov., Hypotrigona gribodoi, Hypotrigona ruspolii and Plebeina armata. 1 It has a significant ecological, economic, and cultural benefit to the farming community. 2 Because of their great value and pollination services, stingless bees are considered as beneficial bee species for the societal economy and natural environments. 3 These bees are important for rural livelihoods of Ethiopia and they have become recently studied for their honey's physicochemical properties, through destructive harvesting practices and habitat loss that pose threats to their populations. 4 Meliponula beccarii (M. beccarii) is among which, and are widely distributed from midland to highland of Ethiopia,5–7 but Liotrigona bottegoi (L. bottegoi) is commonly found in the lowland geographic zones of the country. 8
SBH comprises about 200 compounds, despite the fact that water and sugar are its fundamental components. The specific organoleptic properties of honey are hypothesized to be influenced by trace levels of organic acids, proteins, amino acids, minerals, vitamins, and phenolic compounds. 9 According to study, SBH includes unique phenolic and flavonoid compounds that appears to be key to their antibacterial and antioxidant capabilities. 10 Many factors directly influence the chemical composition of each kind of honey, including the location and ambient conditions of the nectar supply, the honey's floral origin, the processing and storage conditions, and the exact species of stingless bees that produce it. 11
SBH contains biological activity such as antioxidants and antibacterial characteristics due to polyphenols. 10 Which are a key group of phytochemicals from the secondary metabolism of plants that consist of phenolic acid, flavonoids, and phenolic. The phenolic, and flavonoid contents, and the antioxidant activity of SBH are used to differentiate honey of different botanical and geographic origins. 12 Furthermore, nectar comes from a variety of plants and the seasons vary throughout the year, and influences on the honey's color variations. 13 Hydrogen peroxide and other non-peroxide components of the honey's chemical composition are closely linked to these therapeutic benefits. 14 Flavonoids, phenolic and other naturally occurring compounds with antioxidant properties are among the many non-peroxide components that give honey help combat free radicals and protect the cells from oxidative damage. 10 Research found the associations between honey's color and antioxidant activity, as well as its contents of phenolic and flavonoids that determine the floral origin. 15
SBH has gained international recognition for its exceptional medical properties, establishing it as a natural therapeutic agent with potential applications in modern medicine. 16 Unlike regular honey, it contains bioactive components such as phenolics, flavonoids, organic acids, vitamins, and antimicrobial peptides, all of which have strong antioxidant, anti-inflammatory, antibacterial, wound-healing, and immune-modulating properties. 17 Its capacity to reduce dangerous bacteria, fungi, and viruses makes it useful in the creation of natural antibacterial compositions, especially as antibiotic resistance spreads globally. 18 Honey is often used in traditional medicine to treat lung infections, gastrointestinal issues, skin injuries, eye infections, and throat infections. 19 Because of SBH anti-glycemic and cytoprotective properties, scientists in several countries are investigating its significance in the treatment of chronic diseases such as diabetes, ulcers, and cardiovascular disorders. Its application in the nutraceutical, pharmaceutical, and cosmetic industries emphasizes its global commercial and therapeutic importance. 20 As a result, SBH is a promising natural treatment that is becoming more widely used in both traditional and modern healthcare systems.
This study revealed that the phenolic, flavonoid content, and antioxidant activity of SBH variation significantly between honey samples collected from different geographical zones or stingless bee species. Nonetheless, research on Ethiopian stingless bees’ phenolic, flavonoid, and antioxidant activity of SBH from various geographic regions is relatively limited and has given little attention so far. As a result, the purpose of our research is to assess the TPC, TFC, and DPPH activity of SBH produced by M. beccarii and L. bottegoi collected from four different regions of Ethiopia.
Material and Methods
Study Area and Description
The study was conducted in four regional states of Ethiopia, located in Eastern Africa. Honey samples were collected from fifty-seven locations (Figure 1) representing four regional states with nine zones. The study was conducted from four zones of Oromia Regional State (West Shoa, West Arsi, Iluababora, Jimma, and Bale), two zones of the South West Ethiopia Peoples’ Region (Keffa and Bench-Sheko), one zone of South Ethiopia Region (Gedeo) and one zone of Sidama Region (Sidama zone) the study four region located in Ethiopia where the data collected between geographical coordinates of 6.1°-9.1°N latitudes and 35.3°- 38.4°E longitude. Its altitude ranges from 1129 to 2453 meters above sea level and is subdivided into three ecological zones: lowland, midland and highland. The study regions have a bimodal rainfall pattern with major and minor rainy seasons mostly lasting from July to September and March to May, respectively.21,22 Average total annual rainfall ranges from 1100 to 1900 mm and the mean daily temperature ranges from 13.5 to 21.5 °C. 23

Map of Ethiopia showing the locations of sampled regions and the geo-locations of the nests that were sampled (shown as Red-colored circles). The geo-locations of some nesting sites are superimposed on top of the others.
The study regions have natural vegetation composed of extensive Afromontane forests, montane grasslands, woodland and savanna in lowland zones. The vegetation type of these regions includes natural coffee forest, coffee, protected forest, coffee farm, cultivated crops, grass land, grazing land, natural forest, agricultural crops and scattered shrubs, riverine forests, bamboo forests and wooded savanna. 24
The geological features of the four study regions lies largely on the Ethiopian highlands’ southern flank, the elevated Ethiopian plateau and the margin of the main Ethiopian rift. Bedrock is dominated by tertiary–quaternary volcanic sequences (basalts, ignimbrites, rhyolites, tuffs) with assorted intrusive and reworked sediments in places, 24 dominated by Oligocene–Miocene volcanic (flood basalts) sequences overlain in places by later sediments and deeply weathered surface.
The dominant land forms of the study regions are Afromontane highlands, dissected plateaus, volcanic cones, rift escarpments and broad lowland plains, dissected plateaus and rolling hills, steep escarpments and valleys. The major soil types of the four regions are highly weathered tropical soils, Nitisols, Acrisols, and Ferralsols and deeply weathered uplands, Alluvial Fluvisols in valley bottoms, organic soils in wetlands. 23 Volcanic and deeply weathered soils dominate the highlands, nitisols and the well-drained red tropical soils, with localized Andosols where volcanic ash is fresh; valley bottoms have alluvial soils and seasonally waterlogged soils (Vertisols). 25
The study was conducted based on the availability of native stingless bees (M. beccarii and L. bottegoi) and the market value for the honey they produced were the primary factors for selecting the sample zones. The presence of SBH markets and nearby stingless bee hunters also played a role in the selection of the district and peasant groups inside the zone. However, the stingless bee species (M. beccarii) are present at Holeta and Gedo sub sites of the Holeta Bee Research Center, they were included subjectively. Similarly, sampling for L. bottegoi was done from colonies managed at the backyard of local beekeepers of the Bench-Sheko zone. Various vegetation types, including farmlands, grazing fields, natural forests, plantations, home gardens, natural coffee forests, and crop fields side refuges, were among the sample locations.
SBH Sample Collection
Fifty-seven samples of SBH were directly collected from colonies of M. beccarii, modern pot hive (n = 6), M. beccarii, wild nest (n = 46) and L. bottegoi, traditional log hive (n = 5). Honey samples were harvested between October 2022 and the end June 2023 from the different agro-ecology of Ethiopia during active seasons. The samples were included from wild and modern nests following our previous method. 26 Every SBH sample collection area in Ethiopia has two main honey harvesting seasons: a major season, which lasts from October to November and is the most significant harvesting time of the year, and a secondary season, which lasts from April to June and is reliant on the availability of nectar from secondary flowering plants. Fresh and pure honey samples were harvested directly from sealed honey pots using pipette filler and transferred to 50 mL falcon tube (Figure 2). The samples were collected in clean, airtight glass containers to protect them from oxygen and light exposure. The samples were transported to the laboratory by insulated ice boxes with cooling packs to maintain a stable low temperature (4-10 °C) to avoid heating and mechanical agitation. Upon arrival, the honey was stored in the refrigerator at 4 °C until the laboratory analysis was conducted.

SBH harvesting and sample collection process. Nest ecology (A), Guard bees on entrance (B), Nest excavation (C), Nest with honey (D), Honey harvesting process (E), Collected honey (F).
Phenolic, Flavonoid Content and Antioxidant Activity Determination
Total Phenolic Content (TPC)
The TPC of SHB is determined using colorimetric Folin-Ciocalteu assay for TPC. 27 Briefly, each a sample diluting 5 g of each honey sample with 50 µL of distilled water, 25 mL of 0.2 N Follin-Ciocalteu was added and then it was filtered through Whatman No.1 filter paper. The mixture was left for 2 h to settle in dark condition. Distilled water was used as blank to standardize the spectrophotometer (PerkinElmer Lambda 950 UV/VIS/NIR) (Figure 3) and TPC was determined at 760 nm. 28 The TPC was calculated using a linear equation derived from the standard gallic acid calibration curve and expressed as (mg GAE/100 g) of honey weight.

Determination of proces of TPC, TFC and DPPH activity. Prepared honey extract (A), SBH dilution (B), Methanolic extaction of honey (C), Absorbance reading by spectrophotometer (D).
Total Flavonoid Content (TFC)
The TFC of SBH was determine using a colorimetric methods with aluminum chloride (AlCl3), a spectophotometer, and a standard curve (quercetin). 29 Combining 0.5 g of honey with 5 mL of 50% methanol, then rinsing the liquid. Five milliliters of the honey solution and five milliliters of 2% aluminum chloride (AlCl3) were then mixed and left to stand for 30 min at room temperature. TFC was quantified by absorbance at 420 nm using a spectrophotometer (PerkinElmer Lambda 950 UV/VIS/NIR). 28 The mg of quercetin equivalent (QE)/100 g of honey values was to fit in the standard curve to calculate the TFC.
Antioxidant Activity of SBH
Free Radical Scavenging Activity
The radical scavenging activity of SBH is determined using 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay, which measure antioxidant capacity. These assay was employed with minor adjustments, to evaluate the honey samples’ scavenging capabilities in accordance with a previous report.
30
100 mL of methanol were used to dissolve 24 mg of DPPH in order to make a stock solution. Using 400 mg of the honey sample, different amounts of methanol (3, 6, 9, 12, and 15 mL) were made from the stock solution individually. It was subsequently mixed with 0.75 mL of honey in methanol and 1.5 mL of DPPH solution. The mixture was allowed to keep at room temperature for 15 min and then kept in a dark spot for 30 min. A spectrophotometer set to 517 nm was used to measure the absorbance in relation to a blank solution containing water. The formula below was used to determine the sample's antiradical activity.
Color Analysis
The color intensity of SBH is determined using a Pfund color grader (Koehler equipment, New York), 31 and prepare a diluted and filtered honey sample and place it in the sample container of the Pfund grader for reading. Lightest, Pfund is 0 mm long, and darkest, Pfund is 140 mm. The honey sample is compared to a calibrated amber glass wedge that acts as a scale at predetermined locations along the wedge, which is set inside a glass wedge-shaped trough. Before the color was assessed, 100 g of honey were heated at 50 °C and centrifuged for 5 min at 4000 rpm to eliminate any air bubbles. 32 The liquid sample was placed in the sample container of the Pfund grader for reading.
Statistical Analysis
The results were presented as mean ± standard deviation from the triplicate analysis. The data of TPC, TFC and DPPH activity were analyzed with one way analysis of variance (ANOVA) using SPSS version 24 software. Significant differences between the means were identified with Tukey's honestly significant difference (HSD) test. 33 At 95% confidence level, deviations were deemed statistically significant (p < 0.05). The correlations between the factors being evaluated were ascertained using Pearson's correlation coefficient.
Results
Total Phenolic Content (TPC)
The TPC for SBH analyzed in present study were ranged from 28.7 to 73.8 mg of gallic acid equivalents (GAE)/100 g of honey. The average TPC in the honey sample was 42.3 ± 1.5 mg GAE/100 g of honey. There were notable (p < 0.05) variations among the honey samples taken from the different study areas. According to Table 1, honey from the Bale zone had the lowest TPC (28.7 ± 1.3) values, while honey from the West Arsi zone had the highest TPC (73.8 ± 1.2).
Phytochemical and Radical Scavenging Activity of SBH Samples Collected from Ethiopian (N = 57).

Similar superscript letters within the column represent statistical difference (p < 0.05), the standard deviation is denoted by SD, and there are N samples. TPC, TFC, and RSA stand for total phenol content (mg of gallic acid equivalents (GAE)/100 g of honey), total flavonoid content (mg of quercetin equivalent (QE)/100 g of honey), and radical scavenging activity (mg of ascorbic acid equivalent/100 g of honey).
Total Flavonoid Content (TFC)
The TFC for SBH analyzed in present study were ranged from 34.1 to 86.6 mg of quercetin equivalent (QE)/100 g of honey
Antioxidant Activity
The average radical scavenging activity of the SBH sample for 2, 2-diphenyl-2-picrylhydrazyl (DPPH) was found to be 112.1 ± 1.9 mg of ascorbic acid equivalent/100 g of honey. In terms of DPPH activity, there was a significant variation (p < 0.05) among the honey samples collected from the study areas. The honey from the West Shoa zone had the lowest DPPH scavenging activity (88.7 ± 1.1), whereas the West Arsi zone honey had the highest (165.8 ± 2.3).
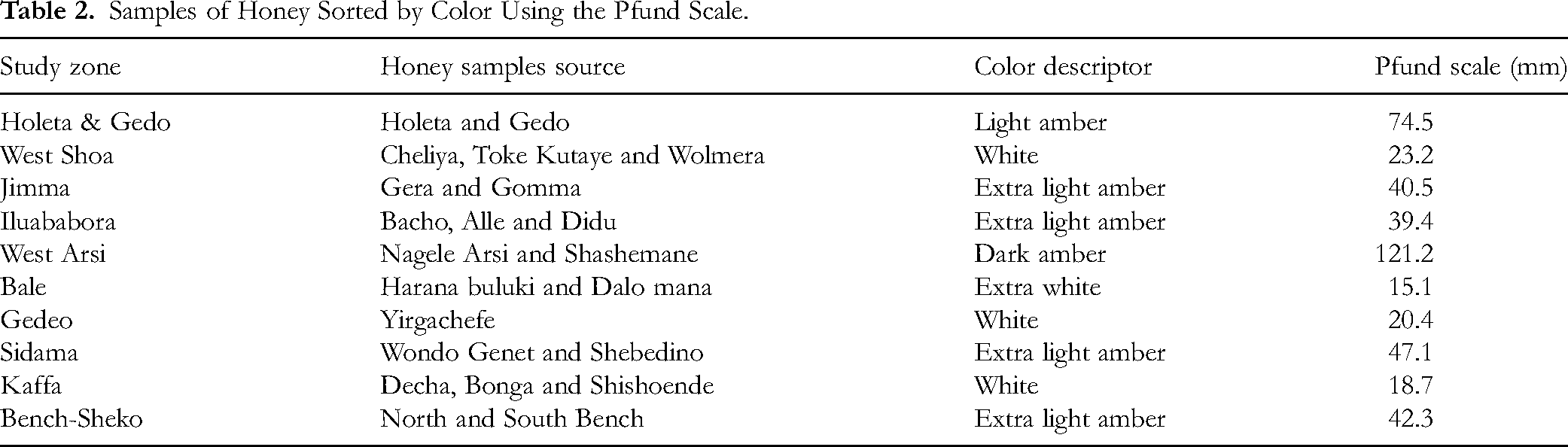
Honey Color Intensity
Five types of honey colors were identified in this study, which are extra white (10%), white (30%), extra light amber (40%), light amber (10%), and dark amber (10%) (Table 2). Our study result indicated that SBH's color is impacted by its geographic origins; the honey from the West Arsi zone is dark amber, while the honey from the Bale zone is extra white and ranges in type from 15.1 mm to 121.2 mm Pfund (Table 2). SBH samples from Jimma, Iluababora, Sidama, and Bench-sheko zones were also light amber in color, while samples from Holeta and Gedo sites were light amber. The SBH sample from the Bale zone was the only SBH that classified as extra white, while the SBH from the West Shoa and Gedeo zones were determined as white.
Samples of Honey Sorted by Color Using the Pfund Scale.
Correlation of Phytochemical, Antioxidant and Color
The correlation matrix was created to look into any relationships between the various parameters being studied (Table 3). Positive associations were found between TFC, color, TPC, and antioxidant activity. The highest phenolic, flavonoid, and antioxidant activity of the SBH is indicated by the honey samples’ deeper color. A statistical analysis of samples from the West Arsi zones, Sidama, Holeta, Gedo, and Bench-Sheko sites has shown a relationship between antioxidant activity, color, phenolic content, and flavonoid content concentration (Table 2). Higher concentrations of antioxidant activity, color, and phenolic and flavonoid content were found in the honey sample from the West Arsi zone. Color characteristics and phenolic content were more strongly correlated (p < 0.01, 0.945) (Table 3). Higher concentrations of TPC and TFC are generally associated with darker honey colors. This is because honey's color is influenced by phenolic and flavonoids compound, which are often found in larger proportions in darker honeys (Figure 4).

Relationship between total phenolic content and SBH color.
Quantitative Measurement of SBH Phytochemicals, Antioxidant Properties, and Color Analysis Using Pearson's Correlation Coefficients.

A relationship between color, antioxidant activity (DPPH), total phenol content (TPC), and total flavonoid content (TFC) is demonstrated by Pearson's coefficient. Considerable differences are indicated by *p < 0.05 and ** p < 0.01.
Discussion
Our current investigation on Ethiopian SBH validates its significant phenolic, flavonoid, and antioxidant activity. Honey includes a high concentration of phenolic and flavonoid chemicals, which are responsible for its antioxidant and medicinal qualities such as antibacterial and wound healing. SBH contains more phenolic and flavonoid chemicals than honey bee honey, resulting in stronger antioxidant activity. 6 The diverse foraging patterns of bees, as well as the compositions of their hives, determine their unique profile. SBH's phenolic and flavonoid composition includes a high concentration of polyphenols. 34 The group of chemicals that comprises phenolic acid flavonoids. The phenolic and flavonoid concentration and composition differ significantly depending on the floral source, geographical origin, and stingless bee species. 35 SBH is a powerful antioxidant due to its antioxidant action and high concentration of phenolic compounds. 36 Antioxidant activity is primarily measured by assays like DPPH (radical scavenging) and FRAP (ferric-reducing antioxidant power), which demonstrate the ability to eliminate free radicals. 10 Various investigations have found that SBH has significantly higher antioxidant activity than honey bee honey.37,38 SBH's antioxidant capacity is significantly related to its TPC, 10 and honey with higher phenolic component content consistently has better antioxidant activity.
According to study report, 39 antioxidant activity has been associated with a number of medicinal qualities, such as anti-inflammatory, anti-microbial, anti-cancer, and anti-obesity capabilities. Antioxidants are known to contribute to oxidative damage and are associated with a range of illnesses. Additionally, antioxidants can regulate cellular responses (inflammation, survival, proliferation, and death) in a variety of illnesses and modify signal transduction pathways that are impacted by free radicals under oxidative stress. 17 This implies that their actions extend beyond simply scavenging free radicals.
According to our current finding SBH collected from West Arsi zone have higher TPC and TFC, while honey collected from Bale zone have lower than the other study zones. The variation of this based botanical origin, phenolic and flavonoid content of stingless bees forage from local flowering plants, shrubs, herbs and trees and each plant contributes its own secondary metabolites. 40 The most abundant stingless bee forage plants, Croton macrostachyus, Vernonia amygdalina, Eucalyptus spp., Terminalia brownii, Trifolium steudneri and Acacia brevispica rich in polyphenols and flavonoids in study areas.
According to study report the higher phenolic and flavonoid content of SBH honey from West Arsi zone, different dominant nectar and pollen plants visited by the bees and the West Arsi zone the agro-ecosystems have many herbaceous and agricultural crops for forage plants including Eucalyptus camaldulensis, Vernonia auriculifera, Schefflera abyssinica, Croton macrostachys, Bidens ghedoensis, Hyposthus trifolium and Guizotia scabra that are kmnown sources of nectar and pollen with measurable phenolic, a multifloral mixture with higher phenolic content of SBH. Bale zone vegetation includes afro-alpine belts, forest species and endemic montane flora with different nectar phenolic and flavonoid content.
According to study report higher phenolic and flavonoid content of SBH have stronger antioxidant and antimicrobial activity. 6 This makes SBH from diverse forest flora of our study area kaffa, Bench-Sheko and Sidama forest in Ethiopia more valued for medicinal use than monofloral honey. The phytochemical and antioxidant activity of SBH are known to be mostly dependent on bioactive compounds such as TPC, TFC, and DPPH. 41 Because SBH contains a high concentration of phenolic components such gallic acid and flavonoids, it is recognized for its antioxidant qualities. 42 These chemicals increase honey's antioxidant activity by scavenging free radicals. 43 According to study report, SBH exhibits considerable phenolic content-related antioxidant activity and notable DPPH radical scavenging activity. 6
According different study results indicated that altitude and local climate significantly influence SBH phenolic content due to the plants at those altitudes makes different amount and types of polyphenols. 44 From our present finding variation in altitude can be makes variation in phenolic concentration in one zone verse another study zone. The floral diversity and flowering season can cause variation in TPC of honey across study zones. The diverse and overlapping flowering calendar can produce multifloral honeys which higher phenolic content. 45 Comparatively from our finding West Arsi, Holeta and Gedo, Sidama and Bench-Sheko zones have a long season with many herbaceous nectar plants that increases both phenolic and observed antioxidant activity versus areas where fewer nectar species dominant at sampling times.
SBH in Ethiopia has a high phenolic and flavonoid concentration that varies based on geographical region, honey color, and hive type. The phenolic and flavonoid content contributes to honey's antioxidant capacity, and some study indicates that modern hives may produce honey with higher levels of these compounds than wild nests. 26 Similar to our finding the TPC ranging from 21.3 to 89.7 mg GAE/100 g and but lower TFC than our finding, which ranging from 19.2 to 41.9 mg QE/100 g) SBH sample from three zones of Oromia region. 46 Inconsistent to our finding the higher average TPC (273 mg GAE/100 g) and lower average TFC (41 mg QE/100 g) SBH sample collected from market in Western Oromia. 47
Comparatively, the TPC differs between West Arsi and Bale zone SBH due to botanical and geographical variations. 48 West Arsi's higher phenolic content likely results from different flowers sources and nectar collection unique to the region's specific plant life and environment. The distinct ecological factors and plant diversity in West Arsi provide a richer source of polyphenols for the bees compared to the Bale zone, leading to a higher phenolic concentrations in the honey produced there. 49
According previous study report, 7 the higher TFC in West Arsi SBH compared to other study zones is likely due to the specific flora available in West Arsi zone, which provides a richer source of flavonoid-producing nectar and resins for the stingless bees. Factors like geographical origin, climate, and stingless bee foraging habits significantly influence honey's chemical composition, including flavonoid content. 50
The TPC and TFC in SBH from West Arsi are likely higher than in other study zones due to a combination of factors, including a greater diversity of plants visited by the bees, access to a broader range of plant resins and nectar from these diverse flora, 51 and the specific preferences of the stingless bee species in that region for specific high-polyphenol plants. These botanical and geographical characteristics have a direct impact on the types and quantities of polyphenolic chemicals that stingless bees collect, as phenolic content is highly associated with botanical origin. 52
According previous report, 53 the SBH's TPC and TFC is strongly linked to its botanical origin, with specific plant species in our present study zones contributing to variations in these antioxidant compounds. Different honey types and geographic locations exhibit different phenolic and flavonoid levels, with a study identifying dominant plants like Acacia etbaica, Acacia mellifera, Coffee arabica, Cordia african, Croton macrostachyus, Eucalyptus globulus, Guizotia scabra. Schefflera abyssinica, Phaseolus vulgaris, Terminalia brownie and Vernonia amygdalina as sources of these compounds. 54 Honey aroma and antioxidant potential are attributed to flavonoids, which are a low molecular weight molecule. Different floral sources may provide honey with varying flavonoid types and contents. 52 A direct comparison is limited due to the variations in the researched stingless bee species and the geographical characteristics of the study regions, even though various studies looked at the honey samples from African stingless bees.
The average DPPH scavenging activity of SBH ranged from (88.7 to 165.8 mg of ascorbic acid equivalent/100 g) with mean value of (112.1 mg of ascorbic acid equivalent/100 g). The high DPPH activity of SBH in study area is attributed to its high content of beneficial plant secondary metabolites like phenol and flavonoid, which are potent antioxidant absorbed by stingless bees from floral sources. 55 The specific botanical origin and plant diversity in different study locations, combined with the unique characteristic of stingless bees, lead to a honey with a high antioxidant profile, distinguishing it from Apis mellifera honey. 56 SBH from West Arsi is anticipated to have increased DPPH activity due to unique floral sources and regional environmental conditions that influence the honey's high quantities of phenolic compounds and flavonoids. These chemicals have a clear correlation with antioxidant capacity, making West Arsi honey an excellent source of free radical scavenging activity, as assessed by the DPPH assay.
Several studies have found a link between strong DPPH activity in Ethiopian SBH and its different botanical origins, as the forged nectar contains various antioxidant chemicals such as flavonoids and phenolic derivatives. 46 Different plant species have distinct antioxidant profiles, and a diverse flora in a region results in honeys with higher and more diversified antioxidant capabilities. 30 The stingless bee species, botanical sources, and geographic region all significantly affect honey's ability to scavenge DPPH radicals. 57 According to a study by Combarros-Fuertes et al, 58 the antioxidant activity of honey samples was associated with their phenolic and flavonoid contents, suggesting that the bioactive components in honey samples are crucial for antioxidant activity.
The types and concentrations of phenolic chemicals each of which has a unique capacity for reduction and scavenging may account for the variances in antioxidant activity across the honey samples. 52 The type and quantity of phenolic compounds and hence the antioxidant activity of honey, 59 may be influenced by a variety of factors including botanical origin geographic location harvest season and environmental conditions the antioxidant qualities of honey, 60 may be impacted by handling procedures processing methods and storage conditions.
The color of SBH is intimately tied to its floral source, as different plant species contribute varying amounts of colors, minerals, and beneficial compounds to the nectar. 42 Flowers have strong phenolic acids, flavonoids, and minerals, such as Eucalyptus globulus, Vernonia amygdalina, and Coffee arabica, create darker honey, whereas nectar from lighter floral sources, such as Guizotia scabra and Acacia abyssinica, generate amber honey. Dark-colored SBH has more antioxidants and minerals, resulting in a deeper flavor and higher medicinal value. 61 Lighter honey, on the other hand, contains less phenolic components and has a milder smell, making it ideal for use as table honey. 52 As a result, the botanical origin not only dictates the color of the honey but also reflects its nutritional and medicinal value.
The pigments presented in honey is an indication for honey color, which is affected by the composition of the nectar, the honey's botanical origin, the acquisition procedure, temperature, and storage duration. 35 From nearly colorless white to extra light amber, the hues progress through light amber and dark amber characteristics. The physical characteristic that a customer notices right away and that may have an impact on their decision to buy the honey. 62 The current findings are consistent with the earlier study by Mendoza-Bacilio et al, 15 in which they discovered a favorable relationship between color, antioxidant activity, phenolic content, and flavonoid concentration.
Depending on the location, the techniques employed for gathering, and the flowers utilized, SBH can range in hue from extremely white to dark amber. The color is often linked to the specific plants that the bees visited for nectar, and darker colors may indicate a higher concentration of specific minerals and antioxidants. 12 The varieties of nectar used in honey production 63 and the mineral presence 64 have a significant impact on the color of honey. Honey color has also been observed to be influenced by pollen kinds, phenolic compounds, and Hydroxyethylfurfural (HMF). 65
The color of the SBH in this study was classified as extra white (10%), white (30%), extra light amber (40%), light amber (10%), and dark amber (10%), according to USDA color specifications for extracted honey. 66 This explains that SBH is made up of several colors, could be caused by a variety of things, including the production process, exposure to light, heat, storage duration, and enzymatic processes. Similar to our study finding, the color of the M. beccarii honey harvested from different zones of Oromia region were classified as amber and water white. 31 According to 31 the total honey samples, 50% were classified as light amber, 21.5% as extra light amber, and 7% as water white. The honey's color selection is typically determined by consumer preferences, which are regarded as indicators of market acceptability and quality appreciation. The color of a honey sample is greatly influenced by the type of nectar in honey production and the mineral present. 67 Pollen types, phenolic compound and HMF have also been reported to influence the color of honey. 52 Our current investigation revealed that the darker honey had higher levels of flavonoids, phenolic, and antioxidant activity than the lighter honey, which is in line with results from a previous study. 15
Darker SBH has a higher phenolic content due to multiple interrelated botanical and physiological mechanisms. 52 Dark honeys are often made from the nectar, pollen, and resins of medicinal plants, which naturally contain higher levels of phenolics to protect against UV radiation, herbivory, and microbial attack. 68 Stingless bees additionally incorporate propolis and pollen into the honey during processing, which are high in flavonoids, phenolic acids, and tannins, contributing to the deeper color and increased bioactivity. 69
Furthermore, enzymatic processes in the bee digestive system, particularly glucose oxidase and polyphenol oxidase, convert precursor chemicals into more complex polyphenols, which enhance color and antioxidant activity. 70 During honey maturity, oxidation and Maillard reactions produce melanoidins, dark-colored chemicals that increase phenolic concentrations. 71 As a result, the increased phenolic content of darker SBH is mechanistically related to plant source composition, bee physiology, and chemical changes during honey processing and storage.
This investigation primarily focused on the phenolic content, flavonoid content, and antioxidant activity of SBH. While this study concentrated on these bioactive substances, it did not assess the physicochemical components or in-vitro antimicrobial properties against foodborne pathogens. Although these findings provide a foundation for understanding the phytochemical and antioxidant profile of SBH, further research is required to evaluate physicochemical and antimicrobial properties. Such studies are necessary to confirm chemical variations based on botanical and geographical origins and to expand upon the current findings.
Conclusion
This study's findings demonstrated significant quantities of phenolic, flavonoids content, and antioxidant activity in SBH, emphasize its unique bioactive and therapeutic potential. These natural antioxidants have anti-inflammatory, antibacterial, immune-boosting, and disease-preventive properties, suggesting that SBH could be used as a medical food to promote public health in Ethiopia. SBH also underlines the significance of protecting stingless bee species and promoting meliponiculture as a sustainable economic option, particularly in rural areas. Furthermore, the growing global demand for high-value natural health products indicates enormous prospects for the marketing and branding of Ethiopian SBH in local and international markets. Therefore, improving quality standards, strengthening value-chain development, and promoting research-based production practices could help Ethiopian SBH establish itself as a high-quality natural product with significant health, economic, and ecological benefits.
Footnotes
Acknowledgments
This study was funded by the Department of Microbial Science and Genetics, the Holeta Bee Research Center, the Oromia Agricultural Research Institute, and the Addis Ababa University seventh round Thematic Research Fund [RD/LT076, 2019]. The authors also thank the National Agricultural Biotechnology Research Center, the Addis Ababa University Center for Food Science and Nutrition, and the Ethiopian Institute of Agricultural Research for their laboratory facilities and resources.
Ethics Approval and Consent to Participate
The authors also declare that the research was conducted without any human and /or animal experimentation and so no ethical approval was required. This article does not contain any studies that could require an ethical statement. The authors declare no conflict of ethics. This research study complies with research and publishing ethics. The scientific and legal responsibility for manuscripts published in Journal of Natural Product Communications belongs to the author (s).
Funding
Addis Ababa University 7th Round Thematic Research Fund [RD/LT076/2019].
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability and Materials
All data supporting the findings of this study are available within the paper.

