Abstract
Objectives
We aimed to investigate the antitumor effects of isoforsythiaside (IFY), a novel natural small compound, on osteosarcoma (OS) and breast cancer (BC), and to explore whether IFY exerts its activity by directly targeting the formin homology 2 domain (FH2) of dishevelled-associated activator of morphogenesis 1 (DAAM1).
Methods
Public data from The Human Protein Atlas (HPA) were analyzed for DAAM1 expression. Molecular docking (PyMOL) and microscale thermophoresis (MST) were used to confirm the direct binding of IFY to the FH2 domain of DAAM1 in vitro. Cell Counting Kit-8 (CCK8) assay, wound healing assay, Boyden chamber assay, Western blotting and microfilament assembly (immunofluorescence) were assessed in OS and BC cell lines. Antitumor efficacy was further evaluated in a 4T1 mouse breast cancer xenograft model.
Results
DAAM1 was highly expressed in OS and BC tissues and cells. IFY directly bound the FH2 domain of DAAM1. IFY (10-15 μmol/L) significantly reduced cell viability, migration, invasion, microfilament assembly, and DAAM1 protein expression levels in OS and BC cell lines. In vivo, IFY (10 mg/kg, i.p., q.2d) markedly suppressed tumor growth in BC-bearing mice.
Conclusions
IFY is a novel DAAM1 inhibitor that directly targets the FH2 domain and effectively suppresses the viability, motility, and microfilament assembly of OS and BC cells and inhibits tumor growth, suggesting its potential as a promising therapeutic candidate targeting the Wnt/PCP pathway.
Introduction
Tumors are complex diseases that pose a threat to human life and health. Their pathogenesis is intricate and exhibits heterogeneity. Osteosarcoma (OS) is the most prevalent primary malignant tumor of bone in the pediatric population and young adults.1–4 Breast cancer (BC),5,6 one of the most common cancers among women worldwide, exhibits diverse molecular subtypes and a high propensity for recurrence and metastasis.7,8 Current clinical management of OS and BC relies on a combination of surgery and systemic pharmacotherapy.9,10 For OS, standard chemotherapeutic agents, including doxorubicin, cisplatin, methotrexate, and ifosfamide, are commonly administered in regimens like the MAP (methotrexate-doxorubicin-cisplatin) protocol.11–14 For BC, treatment varies by molecular subtype, such as hormone receptor-positive cases treated with endocrine therapies like tamoxifen or aromatase inhibitors, human epidermal growth factor receptor 2-positive (HER2+) cases managed with trastuzumab or pertuzumab, and triple-negative BC primarily treated with taxanes or anthracyclines.15–17 However, these drugs are associated with varying degrees of drug resistance and significant side effects, such as cardiotoxicity, nephrotoxicity, and myelosuppression.18–20 Therefore, there is an urgent need to develop novel drugs to improve treatment outcomes for these malignancies.
Dishevelled-associated activator of morphogenesis 1 (DAAM1) belongs to the formin family and is closely associated with the Wnt signal transduction and the regulation of cytoskeletal dynamics.21–23 Evolutionary studies have revealed that the protein DAAM1 possesses an N-terminal GTPase binding domain (GBD) and formin homology (FH) 1 and 2 domains, which nucleate and polymerize actin.24–26 DAAM1 drives microfilament reorganization and malignant phenotypes (proliferation, migration, and invasion) in BC, ovarian cancer, and lung cancer, functions that require intact formin homology 2 (FH2) domain activity.27–31 We have reported that Wnt5a promotes OS cell migration via receptor tyrosine kinase-like orphan receptor 1/2 (ROR1/2), and then activates DAAM1 and Ras homolog family member A (RhoA). 32 The natural small molecules targeting DAAM1 may be developed as novel candidate drugs to inhibit the proliferation and metastasis of OS and BC.
We have used a program SYBYL-X 1.3 to predict the potential natural small molecules targeting FH2 domain of DAAM1 and found nearly 20 candidate molecules. 28 Isoforsythiaside (IFY), a novel phenylethanoid glycoside isolated from the dried fruit of Forsythia suspensa, displays antioxidant effect.33–35 We validate direct binding of IFY to DAAM1 using Microscale Thermophoresis (MST) and demonstrate that IFY potently suppresses the viability, migration, and invasion of OS and BC cells. Unlike conventional chemotherapeutic agents that frequently induce severe systemic toxicity and drug resistance, IFY selectively targets the FH2 domain of DAAM1, thereby inhibiting tumor proliferation and metastasis while exhibiting minimal effects on normal cells and favorable preliminary biosafety in vivo. This unique mechanism positions IFY as a first-in-class DAAM1 inhibitor and offers a promising strategy to overcome the limitations of current therapies for these aggressive malignancies.
Materials and Methods
Cell Culture and in Vitro Assays
Cell Lines and Culture
Human OS cell lines U2OS (Cat. No. SCSP-5030), 143B (Cat. No. TCHu264), MG63 (Cat. No. TCHu124), human BC cell lines MCF-7 (Cat. No. SCSP-531), MDA-MB-231 (Cat. No. TCHu227), human cervical cancer cell line HeLa (Cat. No. TCHu187) and mouse BC cell line 4T1 (Cat. No. TCM32) were purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). These cells were cultured in Dulbecco's modified Eagle's medium (Cat. No. 6125052, high glucose, DMEM, Gibco™) supplemented with 10% (V/V) foetal bovine serum (FBS) (Cat. No. A5669701, Gibco™) and 1% penicillin/streptomycin (Cat. No. 15140122, Gibco™) at 37 °C with 5% CO2 (Cat. No. 3111, Thermo Scientific™). All human cell lines were authenticated using short tandem repeat profiling and all assays were conducted with mycoplasma-free.27,36
Western Blotting (WB) Analysis
U2OS, 143B, MG63, MCF-7, MDA-MB-231 and HeLa cells washed with PBS (Cat. No. 10010023, Gibco™) and then lysed with RIPA Lysis Buffer (Cat. No. P0013C, Beyotime) with the necessary protease inhibitors. Cell lysates were boiled at 100 °C for 10minutes. The protein extracts were separated by 10% SDS-PAGE and then transferred into Nitrocellulose membrane (Cat. No. HATF00010, Merck, Germany). Anti-DAAM1 (1:1000 dilution, Cat. No. sc-100942, Santa Cruz, USA) and anti-GAPDH (1:2000 dilution, Proteintech, China) antibodies were used. The target proteins were exposed with highly sensitive ECL luminescent liquid (Cat. No. 180-5001, Tanon, China) and in a Fully automated chemiluminescence image analysis system (Tanon, China).27,28,37
Immunofluorescence (IF)
Glass slides (14 mm) (Cat. No. BS-14-RC, Biosharp, China) were placed in 24-well plates (Cat. No. 11310, Labselect, China), washed with PBS 3 times, and sterilized with ultraviolet light for 1 h. U2OS, 143B, MG63, MCF-7, MDA-MB-231 and 4T1 cells were seeded on glass slides and cultured for 24 h. After the rinse with PBS twice, cells were fixed with 4% paraformaldehyde (Cat. No. BL539A, Biosharp, China) for 20 min. The slides were rinsed with PBS 3 times, and then penetrated with blocking buffer at room temperature (RT) for 40 min. Next, cells were incubated with TRITC-labeled phalloidin (Cat. No. BL1189A, Biosharp, China) for 45 min and rinsed with PBS for 3 times in the dark at RT. The cells were incubated with DAPI Fluoromount-G (Cat. No. 010-20, SouthernBiotech, Alabama, USA). Fluorescent images were captured by confocal microscopy (Cat. No. LSM710, Zeiss, Oberkochen, Germany) and analyzed by ZEM software.27,37
Wound Healing Assay
U2OS, 143B, MG63, MCF-7, MDA-MB-231 and 4T1 cells were seeded in 24-well plates (Cat. No. 11310, Labselect, China). After the cells reached a confluence, sterile pipette tips were used to scratch cells. The cells were rinsed with PBS twice and taken a photo with an inverted microscope (Mshot M152-N, Guangzhou, China). The wounded cells were cultured in DMEM with FBS and allowed to migrate for 23 h. The migratory distances of the wounded cells were measured and calculated by ImageJ at 100× magnification.27,38
Bacterial Protein Expression and Purification
GST-FH2 plasmids (GenScript, China) were transformed into E. coli BL21 (Cat. No. D1009S, Beyotime). The bacteria were cultured with antibiotics and induced by IPTG (Cat. No. 367931, MCE) at 16 °C for 13 h. The bacteria were lysed and centrifuged to obtain the supernatant. GST-tag Purification Resin (Cat. No. P2251, Beyotime, China) were equilibrated with lysis buffer and mixed with the supernatant and induced at 4 °C for 2-3 h. The liquid was transferred to the purification column and eluted 5 times. Finally, the purified proteins were verified by Western blotting. 28
Microscale Thermophoresis (MST)
FH2 domain of DAAM1 was labeled with MonolithTM RED-NHS protein labeling kit (Cat. No. MO-L011, AmineReactive). Pretest were performed to the protein to check fluorescence intensity. The protein was mixed with isoforsythiaside (IFY) (Cat. No. 1357910-26-9, Yuanye, China) after the fluorescence intensity was above 200. The mixture was extracted by capillary (Cat. No. MO-K022, Nanotemper, Germany) and detected on the machine (Monolith NT.115, Nanotemper, Germany).27,28
Cell Counting Kit-8 (CCK-8) Assay
U2OS, 143B, MG63 and MCF-7 cells were digested with protease and inoculated in 96-well plates. When the cells reached ∼80% confluence, the cells were treated with IFY (0, 1, 5, 10 and 15 μmol/L) for 24 h at 37 °C, 5% CO2 and then incubated with CCK-8 (Cat. No. C0037, Beyotime) for 1 h. Finally, optical density at 450 nm was measured with a microplate reader (Cat. No. ELx800, Bio Tek, USA).27,38
Invasion Assay (Boyden Chamber Assay)
The upper chambers of Boyden chambers (Cat. No. 725321, NEST, China) were coated with 3.3 μL of Matrigel (Cat. No. 356234, Corning, USA) diluted at a ratio of 1:30 (v/v) in serum-free DMEM medium and incubated at 37 °C for 1 h to allow gelation in the invasion assay. U2OS, 143B, MG63, MCF-7, MDA-MB-231 and 4T1 cells (1 × 105 cells/well) were inoculated into the upper chamber of Boyden chamber treated with 10 μmol/L IFY and serum-free medium, and the lower chamber was filled with medium containing 20% FBS. After 23 h cell invasion, the chambers were washed with PBS and then fixed with 4% paraformaldehyde for 20 min. The cells in the upper chambers were removed with a cotton swab. The lower chambers were washed with PBS and dyed with crystal violet solution for 30 min. The chambers were washed with clean water and air-dried. The invasive cells were examined under a bright field microscope.27,37,38
Animal Models
Female Balb/c mice (6 weeks old) obtained from the Jiangsu Laboratory Animal Center were housed in specific pathogen-free facilities at 22 °C ± 2 °C under 12-h light/dark cycles. The tumor-bearing mice model was established by subcutaneous injection of approximately 5 × 106 4T1 mouse BC cells. Tumor size was measured by vernier calipers, and tumor volume was calculated using the formula V = (length × width2)/2. When the average tumor size reached about 50 mm3, the tumor-bearing mice were randomly divided into a control group and an IFY-treated group. Mice in the IFY therapy group were intraperitoneally injected with IFY (10 mg/kg, Cat. No. 1357910-26-9, Yuanye, China) once every two days and lasted for 2 weeks, while mice in the control group were subcutaneously injected with the same volume of PBS. Mice were euthanized using carbon dioxide (Euthanex Chamber). The tumors were removed from the unconscious animals, which were subsequently documented and weighed.28,38,39 The reporting of this animal study conforms to the ARRIVE 2.0 guidelines. 40
The Human Protein Atlas Database
The protein expression levels of DAAM1 in human tumor tissues were analyzed and visualized using The Human Protein Atlas (THPA, https://www.proteinatlas.org/). The THPA database characterizes protein expression across 23 cancer tissues and 83 cell lines. For mRNA quantification, it employed RNA-seq with normalized transcripts per million (nTPM) as the statistical normalization method. At the protein level, tumor tissue proteomics data were analyzed using normalized relative protein expression (nRPX). All images and annotations within the database were freely accessible for download.
Statistical Analysis
GraphPad Prism 10 software was used to analyze the experimental data. The statistical difference was analyzed using one-way ANOVA or Student's t test. All data were expressed as mean ± SD. P < .05 (*), P < .01 (**), P < .001 (***), P < .0001 (****) show that there are significant differences.
Result
High Expression Levels of DAAM1 are Determined in Bone Cancer and Breast Cancer Tissues and Cells
To determine the relative protein expression of DAAM1 across multiple cancer types, we analyzed the data derived from THPA using nRPX. DAAM1 showed the highest expression levels in bone cancer, head and neck cancer, and BC (Figure 1A). We further analyzed the RNA and protein expression levels of DAAM1 across 62 BC cell lines and 21 bone cancer cell lines. RNA expression levels (normalized Transcripts Per Million, nTPM) and protein expression (represented by circle size) were also obtained from THPA. We found that DAAM1 was highly expressed in U2OS, 143B, MG63 OS cells, and MCF-7, MDA-MB-231 BC cells (Figure 1B-C).

THPA analysis reveals significantly elevated DAAM1 expression in osteosarcoma and breast cancer tissues and cells. (A) Relative protein expression of DAAM1 across multiple cancer types. Data are derived from The Human Protein Atlas (THPA) using normalized relative protein expression (nRPX). DAAM1 shows the highest expression levels in bone cancer, head and neck cancer, and breast cancer (BC). (B-C) RNA and protein expression levels of DAAM1 across 62 BC cell lines and 21 bone cancer cell lines. RNA expression levels (normalized Transcripts Per Million, nTPM) and protein expression (represented by circle size) are obtained from THPA.
Isoforsythiaside (IFY) Directly Targets FH2 Domain of DAAM1
To accurately identify potential chemical compounds targeting DAAM1, we utilized a program SYBYL-X 1.3 to perform small molecule compound-protein docking and found that IFY could target FH2 domain of DAAM1 (reported in the Master's thesis, Xinqian Yu, Chinese version, 2022). This binding was also confirmed and visualized by PyMOL (PyMOL 3.1), a user-sponsored molecular visualization system on an open-source foundation (Figure 2A). IFY bound to Trp 615, Ser 616, Lys 842, and Arg 829 amino acid residues of DAAM1 (Figure 2A).

Isoforsythiaside (IFY) directly targets FH2 domain of DAAM1. (A) The precise amino acid residues of DAAM1 targeted by isoforsythiaside (IFY) are predicted and visualized by PyMOL. DAAM1 is represented by purple helixes. IFY is represented by green, red and blue bars. (B) The binding of IFY and FH2 domain of DAAM1 is confirmed by MST technology. (C) DAAM1 expression levels in tumor cell lines are assessed by Western blotting. HeLa is a cervical cancer cell line. U2OS, 143B, and MG63 are osteosarcoma cell lines. MCF-7 and MDA-MB-231 are breast cancer cell lines. The bands with high molecular weight (upper bands, ∼122 kDa) are the target bands of DAAM1. GAPDH is the internal control. The grayscale values of the ratios of DAAM1 to GAPDH are calculated and presented in the right panel. n = 3.
MST is a highly sensitive, immobilization-free technique that measures molecular interactions in solution under near-native conditions using minimal sample volume (<5 μL). It is particularly suitable for detecting interactions involving small molecules and proteins with dissociation constants ranging from pM to mM, making it an ideal choice for validating the direct binding of natural compounds. To validate the interaction between IFY and FH2 in vitro, we labeled FH2 protein (100 nmol/L) with fluorescent reagent and mixed with a concentration gradient of IFY (from 16 nmol/L to 500 μmol/L) to detect the binding of protein-molecule compound using MST technology. MST showed a signal-to-noise ratio (S/N) of 16.7, which exceeded the threshold of S/N 5.0 (Figure 2B). Therefore, these data fully demonstrate IFY directly binds to FH2 domain of DAAM1. After that, we determined the expression of DAAM1 in certain cancer cell lines by Western blotting. The results revealed that DAAM1 expression was markedly higher in U2OS, 143B, MG63, MCF-7 and MDA-MB-231 cells compared with HeLa cells (Figure 2C).
IFY Downregulates DAAM1 Expression and the Viability of OS and BC Cells
To determine the role of IFY on DAAM1 expression of U2OS, 143B, MG63 and MCF-7 cells, we performed CCK-8 and Western blotting. In CCK-8 assay, cells were treated with 0, 1, 5, 10, 15 μmol/L IFY for 24 h. Under the treatment with 10 and 15 μmol/L IFY, cell viability was significantly reduced (Figure 3A-D). Then, we treated these cells with 10 μmol/L IFY and measured DAAM1 expression by Western blotting. DAAM1 expression was significantly downregulated in IFY-treated U2OS, 143B, MG63, MCF-7, MDA-MB-231, and 4T1 cells (Figure 3E-J). These results demonstrate that IFY downregulates DAAM1 expression and inhibits cell viability in OS and BC cells.

IFY downregulates the expression of DAAM1 in OS and BC cells. (A-D) The viability of U2OS, 143B, MG63, MCF-7 cells are measured by CCK-8 assays. Cells are treated with 0, 1, 5, 10, 15 μmol/L isoforsythiaside (IFY) for 24 h and then subjected to CCK-8 assays. n = 6. (E-J) U2OS, 143B, MG63, MCF-7, MDA-MB-231, 4T1 cells are treated with 10 μmol/L IFY for 24 h and then lysed for Western blotting. GAPDH is the internal control. The grayscale values of the ratios of DAAM1 to GAPDH are calculated and presented in the right panels n = 4.
IFY Retards the Migration and Invasion of OS and BC Cells
We assessed the motility of OS and BC cells treated with 10 μmol/L IFY for 23 h by using a wound healing assay. Compared with the control group, IFY treatment significantly reduced the migration rates of U2OS, 143B, MG63, MCF-7, MDA-MB-231, and 4T1 cells (Figure 4A-F). The retardant effect of migration on OS and BC cells was also confirmed by Boyden chamber assays (Invasion assays). IFY treatment significantly reduced the number of migrated U2OS, 143B, MCF-7, MDA-MB-231, and 4T1 cells compared with the control group (Figure 5A-E). Compared to the control group, the invasive rates of U2OS, 143B, MCF-7, MDA-MB-231, and 4T1 cells in the IFY-treated group were significantly reduced (Figure 5A-E). These results indicate that IFY has an inhibitory effect on cell motility of OS and BC cells.

IFY retards the migration of OS and BC cells. (A-F) U2OS, 143B, MG63, MCF-7, MDA-MB-231, 4T1 cells are cultured in 24-well plates and scratched by a pipette tip. Then, cells are treated with 10 μmol/L isoforsythiaside (IFY) and allowed to migrate for 23 h. The migratory distances are measured by ImageJ. bar = 50 μm. Magnification, ×100. n = 6.

IFY inhibits the migration and invasion of OS and BC cells. (A-E) For migration assays, U2OS, 143B, MCF-7, MDA-MB-231, 4T1 cells are seeded on the upper sides of Boyden chambers and allowed to migrate in media with or without 10 μmol/L isoforsythiaside (IFY) for 23 h. For invasion assays, these cells are seeded on the upper sides of Boyden chambers pre-coated with matrigel and allowed to invade in media with or without 10 μmol/L IFY for 23 h. The numbers of migratory and invasive cells are counted after crystal violet staining. bar = 50 μm. Magnification, ×200. n = 6.
IFY Attenuates the Microfilament Assembly of OS and BC Cells
Next, we examined the impact of IFY on microfilament organization in OS and BC cells. U2OS, 143B, MG63, MCF-7, MDA-MB-231, and 4T1 cells were treated with IFY for 24 h, followed by staining with rhodamine-conjugated phalloidin and DAPI (4′,6-diamidino-2-phenylindole). Under a fluorescence microscope, microfilaments in the control group exhibited thick, straight, and well-organized fibrous structures, often extending several tens of micrometers (Figure 6A-F). In contrast, IFY-treated cells displayed disrupted actin architecture, characterized by a higher prevalence of diffuse or fragmented filaments. Furthermore, the fluorescence intensity of microfilaments were significantly reduced compared to the control group (Figure 6A-F). The fluorescent staining of IFY-treated groups showed a reduction of the number and intensity of filamentous actin (F-actin) (Figure 6A-F). Thus, IFY may inhibit the microfilament assembly of OS and BC cells via targeting DAAM1.

IFY attenuates the microfilament assembly of OS and BC cells. (A-E) U2OS, 143B, MG63, MCF-7, MDA-MB-231, 4T1 cells are seeded on coverslips and treated with 10 μmol/L isoforsythiaside (IFY) for 24 h, then stained with phalloidin (red) and DAPI (blue). The number of microfilaments is counted under a fluorescence microscope. The fluorescent intensity of microfilaments is measured by ImageJ. bar = 10 μm. Magnification, ×630. n = 6.
IFY Inhibits the Growth of BC in Mice
To determine the effect of IFY on tumor growth, Balb/c mice were subcutaneously implanted with 4T1 mouse BC cells. Four days after implantation, tumor-bearing mice were randomly divided into PBS-treated group (Ctrl) and IFY-treated group. Mice in the IFY-treated group were intraperitoneal injected with 10 mg/kg IFY once every two days and lasted for 2 weeks (Figure 7A). Tumor-bearing mice treated with IFY exhibited a significant decrease in both tumor weight and volume compared to Ctrl group (Figure 7B-F). These results demonstrate that IFY indeed inhibits the tumor growth of BC-bearing mice.

IFY inhibits the growth of BC in vivo. (A) The schematic diagram of tumor-bearing mouse modeling. Balb/c mice are subcutaneously injected with 4T1 mouse BC cells. Isoforsythiaside (IFY, 10 mg/kg) is administered once every two days (i.p., q.2d.), which has been continued for 2 weeks. The mice are executed after 21 days of injection of 4T1 cells. n = 6. (B) The representative image of tumor-bearing mice. Red arrows indicate the subcutaneous tumors. (C-D) The tumors dissected out from the tumor-bearing mice. The length and width of the tumors are measured using vernier calipers. (E-F) The weights and volumes of the harvested tumors are compared between with and without IFY treatment. n = 6.
Discussion
DAAM1, a formin-like protein in the Wnt/PCP signaling pathway, plays an essential role in F-actin polymerization and has emerged as an important promoter of cell migration in multiple malignancies.36,41–43 In our preliminary studies, we identified ~20 small molecules that potentially target the FH2 domain of DAAM1 using Sybyl software, including etoposide, lovastatin, and IFY. Yu et al employed molecular docking to confirm that etoposide binds specific FH2 residues (Gln 775, Arg 786, Glu 789, Glu 894, Glu 895, Thr 898), disrupting DAAM1-mediated actin polymerization and suppressing proliferation and migration in small cell lung cancer (SCLC) and BC cells. 28 Wan et al reported that statin targets the FH2 domain of diaphanous-related formin 1 (DIAPH1) (Ile 923, Lys 926, Leu 927), another formin protein, primarily inhibiting DIAPH1-driven actin elongation to modulate tumor immune evasion. 27 With an S/N ratio of 16.7, the MST data provide robust evidence of the direct IFY binding to the FH2 domain of DAAM1. Sybyl analysis reveals that the FH2 domain of DAAM1 residues Trp 615, Ser 616, Lys 842, and Arg 829 are targeted, revealing distinct binding sites compared with etoposide and lovastatin. The discovery of these small molecule compounds offers multiple options for targeting DAAM1 and inhibiting tumor growth and metastasis.
Etoposide is a derivative of podophyllotoxin and belongs to cell cycle-specific anti-tumor drugs. It mainly acts on the cell DNA, hindering DNA repair, thereby exerting its anti-tumor effect. Etoposide is clinically approved primarily for SCLC, malignant lymphoma, malignant germ cell tumors, leukemia. Its newly discovered interaction with DAAM1 offers novel insights into its antitumor mechanism. However, dose-dependent toxicity and emerging resistance underscore the need for structural modification or combination therapies. Lovastatin, primarily a hypercholesterolemia treatment, shows immunotherapeutic potential by inhibiting DIAPH1 to curb tumor immune evasion, 27 yet its anticancer efficacy and long-term safety await clinical validation. IFY, a novel phenylethanoid glycoside, exhibits potent antioxidant, antibacterial, anti-Alzheimer's, and anti-renal fibrosis activities.33,35,44,45 Here, we demonstrate potent in vitro suppression of DAAM1-dependent tumor migration. However, its pharmacokinetic properties, toxicological profile, and systematic in vivo efficacy remain uncharacterized. Knockdown of DAAM1 with siRNA or shRNA inhibit migration in various tumor cells, including ovarian cancer, BC, glioblastoma, melanoma,36,42,46,47 and OS cells. 32
In vivo experiments demonstrated that IFY significantly suppresses tumor growth in a BC xenograft model. An OS xenograft model was not established in this study, because human-derived OS cell lines (eg, U2OS and 143B) require immunodeficient (nude or SCID) mice, which lack a functional immune system and thus preclude investigation of tumor-immune interactions. Moreover, the in vivo experiments were performed with relatively small sample sizes (n = 6 per group), which may limit statistical robustness. Wan et al demonstrated that DIAPH1 inhibition by lovastatin treatment enhances antitumor immunity in immunocompetent C57BL/6 mice, indicating a vital role of formin proteins in immune-regulatory. 27 Thus, immunocompetent models (eg, C57BL/6) are better suited to explore DAAM1's oncogenic mechanisms and therapeutic potential, closely mimicking clinical tumor microenvironments and informing immune-related therapies.
Although our findings establish IFY as a highly promising DAAM1-targeted candidate, considerable preclinical work remains essential prior to clinical translation. This includes comprehensive pharmacokinetic and toxicological profiling, validation in larger cohorts, testing in immunocompetent and spontaneous metastatic models, and thorough evaluation of potential off-target effects.
While virtual docking and MST identify the FH2 domain of DAAM1 as the primary, high-affinity target of IFY (Kd in the low micromolar range), additional mechanisms may also contribute. Furthermore, the strong antioxidant properties of IFY could partially attenuate reactive oxygen species (ROS)-driven migratory signaling, representing a DAAM1-independent complementary mechanism.33,44 Proteomic profiling are ongoing to definitively map the full interactome of IFY and exclude significant off-target contributions at therapeutically relevant concentrations.
We did not establish BC metastasis models in the present study owing to technical and biological constraints. Tail-vein injection, a standard metastasis model, often causes pulmonary embolism, leading to false-positive lung metastases and obscuring true metastatic mechanisms. 48 Left ventricular injection, which better recapitulates systemic metastasis (eg, to bone or brain), is technically demanding and beyond our laboratory's current capabilities. Future studies could leverage highly metastatic BC cell lines (eg, MDA-MB-231) or improved injection techniques to investigate DAAM1's role in BC metastasis.
Conclusion
This study demonstrates that IFY directly targeting the FH2 domain of DAAM1 and potently suppresses the viability, migration, and invasion of BC and OS cells by downregulating DAAM1 expression and disrupting F-actin assembly (Figure 8). By the blockage of the microfilament polymerization and cell motility, IFY acts as a robust antimetastatic agent in both BC and OS models and holds considerable promise as a novel therapeutic candidate for the prevention and treatment of tumorigenesis and metastasis in these aggressive malignancies. Ongoing studies are evaluating its pharmacokinetics broader anticancer spectrum and potential advancement into phase I clinical trials.
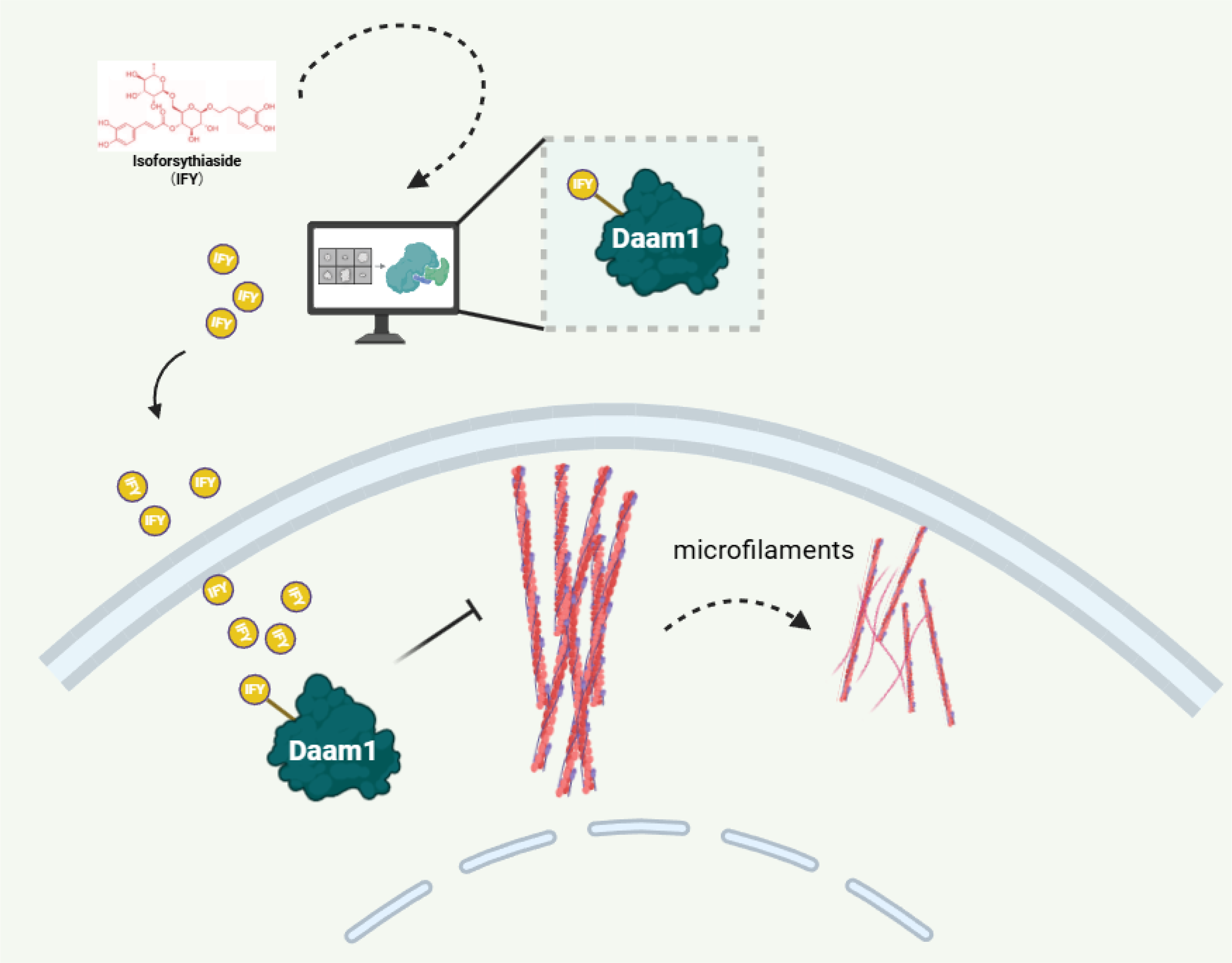
The graphical summary of this study. Isoforsythiaside is a novel inhibitor directly targeting DAAM1. Isoforsythiaside suppresses the viability, motility, and microfilament assembly of tumor cells.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261418401 - Supplemental material for Isoforsythiaside Impedes the Viability, Motility and Microfilament Assembly in Osteosarcoma and Breast Cancer Cells via Targeting DAAM1
Supplemental material, sj-docx-1-npx-10.1177_1934578X261418401 for Isoforsythiaside Impedes the Viability, Motility and Microfilament Assembly in Osteosarcoma and Breast Cancer Cells via Targeting DAAM1 by Ji Zhou, Haocen Guo, Mengyun Wan, Ningyi Xue, Ting Yan, Yichao Zhu, Fan Hu and Ailiang Zhang in Natural Product Communications
Footnotes
Acknowledgements
We thank Ms. Xinqian Yu (Master's thesis, Nanjing Medical University, 2022) for performing SYBYL-X 1.3 virtual screening that first identified isoforsythiaside as a potential DAAM1 ligand.
Ethical Statement
This study was approved by the Institutional Animal Care and Use Committee of Nanjing Medical University (No. IACUC-2412077).
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contribution Statements
Ji Zhou: Investigation, Formal analysis, Writing - Original Draft
Haocen Guo: Investigation, Software, Writing - Original Draft
Mengyun Wan: Methodology, Data Curation
Ningyi Xue: Formal analysis
Ting Yan: Validation
Yichao Zhu: Conceptualization, Writing - Review & Editing
Fan Hu: Supervision, Writing - Review & Editing
Ailiang Zhang: Conceptualization, Funding acquisition, Writing - Review & Editing
Funding
This study is supported by Changzhou High-Level Medical Talents Training Project (2022062), the Major Program of Science and Technology Project of Changzhou Health Commission (ZD202208), the Changzhou Science and Technology Plan Project (CJ20253081).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The datasets generated during and/or analyzed during the current study will be available from the corresponding author upon reasonable request.
Statement of Human and Animal Rights
There are no human subjects in this article. The reporting of this animal study conforms to the ARRIVE 2.0 guidelines.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
