Abstract
Objective/Background
This study aimed to investigate the antioxidant activity and phenolic compound profiles of flowers and leaves of
Methods
Qualitative and quantitative analyses were conducted using a range of in vitro assays to assess antioxidant capacities, including total phenolic and flavonoid contents, ferric reducing antioxidant power (FRAP), DPPH and ABTS radical scavenging activities, and SOD-like enzyme assays. High-performance liquid chromatography coupled with high-resolution mass spectrometry (HPLC-HRMS) was employed to identify and quantify phenolic compounds in both flowers and leaves.
Results
Although the leaves showed numerically higher total polyphenol content and FRAP values, and the flowers exhibited greater mean values of flavonoids and radical scavenging capacity, these differences were not statistically significant (p > .05). SOD-like activities were similar for both tissues. 74 phenolic compounds were identified in leaves and 64 in flowers, with isoflavones and hydroxycinnamic acids abundant in leaves and hyperoside, hirsutrin, rutin, and myricetin predominant in flowers.
Conclusion
The distinct antioxidant profiles and metabolite compositions of
Introduction
Recently, the research for natural antioxidant sources has gained considerable attention due to their potential benefits in food, pharmaceutical, and health-related industries. 1 Among these antioxidants, polyphenols and flavonoids derived from plants are recognized for their capacity to scavenge reactive oxygen species (ROS), thus contributing to the prevention of oxidative stress-related diseases. This has led to an increasing interest in identifying and characterizing bioactive compounds of edible and medicinal plants.
Notably, this study is restricted to in vitro assays, and there is an absence of in vivo or clinical validation to support the practical application of the observed antioxidant activities. Consequently, the interpretation and extrapolation of these findings to medical, pharmacological, or industrial applications should be approached with caution. There remain critical gaps, including bioavailability, metabolism, and physiological relevance, which must be addressed through comprehensive in vivo studies, cell-based experiments, and clinical trials.
Therefore, while the current results contribute foundational knowledge on the antioxidant properties of
Material and Methods
Material
Flower and leaf of
Antioxidative Properties
Flower and leaf of
Identification of Phenolic Compounds
Referring to the method of Jung and Park (2009), 0.1 g freeze-dried sample was weighed. 5 And 5 ml of 80% methanol aqueous solution was used to extract these samples with sonication for 1 h and filtered with a 0.2 μm membrane filter. An Ultimate 3000 RSLC UHPLC system (Thermo Fisher Scientific Inc., MA, USA) was exposed to the phenolic compounds profiling of H. syriacus for the study. The system consisted of an autosampler, a column oven, an ultra- high pressure solvent delivery pump, and an automatic degasser. Chromatographic separations of samples were performed using a Cortecs C18 column (150 × 2.1 mm, 1.6 μm, Waters Co., MA, USA). The column temperature was set to 45 °C. The injection volume was 5 μl with a flow rate of 0.3 ml/min. The mobile phases (0.1% formic acid in HPLC grade water, Solvent A; 0.1% formic acid in acetonitrile, Solvent B) was eluted with the linear gradient programmed as follows: (i) 0–1 min, 5% B; (ii) 1–10 min, from 5% to 30% B; (iii) 10–20 min, from 30% to 100% B; (iv) 20–24 min, 100% B; (v) 24–25 min 5% B. MS and MS/MS detection were conducted on a TripleTOF 5600+ (AB SCIEX, Concord) operating in a positive and negative electrospray ionization mode.
Quantification of Phenolic Compounds
The mass scan was conducted in full scan and information- dependent acquisition (IDA) mode with a mass range set at 100–2000 m/z according to the method of Jung and Park (2009). 5 Mass spectrometry conditions were as follows: ion spray voltage of 5.5 kV in positive mode and 4.5 kV in negative mode; ion source gas-1 and gas-2 at 50 psi; curtain gas pressure at 35 psi; collision gas pressure at 20 psi; source temperature at 500 °C; collision energy of 35 ± 15 eV in positive mode and −35 ± 15 eV in negative mode. Phenolic compounds were identified based on their accurate mass (m/z) and molecular ion fragmentation patterns using Scafford 2,2,1, and compared to mass spectral libraries such as the NIST Library 2017 and the MassBank of North America (MoNA) library database.
Quantification was performed using external standard calibration curves constructed from serial dilutions of pure phenolic standards covering an appropriate concentration range. Calibration curves demonstrated excellent linearity with correlation coefficients (R2) exceeding 0.99. Method validation included assessments of precision, accuracy, and repeatability, with relative standard deviations (%RSD) below a satisfactory threshold. Limits of detection (LOD) and limits of quantification (LOQ) were determined based on signal-to-noise ratios of 3 and 10, respectively, ensuring reliable quantification of phenolic compounds at the reported concentrations. Quality control measures included regular analysis of standard solutions and blank samples to confirm instrument performance and data reliability.
Statistical Analysis
An independent sample t-test was conducted to verify the difference in mean between the two groups, and for this, the data were analyzed using the SPSS statistical program. That is, after confirming the normality of each group, the homogeneity of variance was examined through an equal variance test. After that, the average difference between the two groups was analyzed using the t-test function provided by SPSS (Version 28, NY, USA), and the validity of the research hypothesis was verified through this. To address the issue of multiple comparisons due to testing dozens of metabolites separately, the p-values obtained from each t-test were adjusted for false discovery rate (FDR) using the Benjamini-Hochberg procedure. Statistical significance was determined based on the FDR-adjusted q-values, ensuring control over type I error inflation.
Results
Antioxidant Capacity
Table 1 shows the antioxidant capacity of flower and leaf of
Comparison of Antioxidant Capacity in Different Units of

Values are presented as mean ± standard deviation (n = 3). Statistical significance between flowers and leaves was evaluated by independent t-tests; p-values are indicated for each parameter.
Polyphenols are recognized as major antioxidant components in plants, contributing to protection against environmental stress and affecting physiological functions. 6 The marginally higher polyphenol content in leaves may relate to specific physiological and structural characteristics of these plant parts. 7 This study provides organ-specific data on metabolite accumulation and antioxidant capacity, but contributes as baseline research, not as direct support for functional or health food development. Although leaves showed numerically higher polyphenol content and reducing antioxidant activity, these differences did not reach statistical significance, highlighting the need for caution in interpreting physiological efficacy or potential food formulation benefits. 1 Furthermore, while polyphenol content correlated with antioxidant assays, these results are limited to in vitro analyses.
It should be emphasized that the lack of in vivo or cell-based validation represents a limitation of this study. Therefore, further studies including animal models and clinical evaluations are necessary to substantiate the antioxidant properties of
Flavonoid biosynthesis may vary according to the physiological roles of specific plant organs.
4
For instance, flowers may accumulate secondary metabolites such as anthocyanins and flavonol glycosides that potentially function in UV protection and interaction with environmental mediators, which could be related to enhanced expression of quercetin derivatives. Conversely, leaves predominantly accumulate polyphenols, likely as photosynthetic by-products contributing to neutralization of reactive oxygen species generated during photosynthesis. Thus, flowers and leaves of
Flavonoids, known antioxidants capable of scavenging radicals via electron donation by hydroxyl groups (–OH), were found in these plant parts; however, these in vitro measurements must be interpreted cautiously. 8 While the data suggest antioxidant compounds and activities are present, any conclusions about synergistic effects or activation of antioxidant pathways such as metal ion chelation and enhancing reducing power in vivo remain speculative. 9 Moreover, in vivo bioavailability, metabolism, and physiological relevance were not assessed in this study. Therefore, future in vivo and cell-based studies are necessary to validate these preliminary findings and to better understand their potential implications for food systems or functional food material development.
The phenolic hydroxyl groups (-OH) of flavonoids react with ABTS radicals to form stabilized compounds, and components such as quercetin and myricetin have been reported as major contributors to this activity. Leaves showed relatively lower ABTS activity despite their high polyphenol content in prior experiments, possibly due to a greater involvement of alternative antioxidant mechanisms such as metal ion chelation or SOD-like activity.
9
These results suggest that flower-leaf composites might exert synergistic antioxidant effects compared to individual parts, indicating potential for development of health-functional foods that activate multiple antioxidant pathways by combining flowers and leaves. However, as this study was limited to in vitro assays and the differences were not statistically significant, further validation through in vivo and cell-based studies is essential to establish the practical applicability of these findings. Additionally, the leaves of
However, the statistical analysis indicated that these differences were not significant, and thus the observed variations should be interpreted cautiously. These data are based solely on in vitro assays, which may not fully represent antioxidant effects in vivo. The potential of
While these findings provide preliminary evidence supporting the antioxidant potential of
Identification of Phenolic Compounds
Phenolic compounds present in

Chromatogram obtained from HPLC-HRMS analysis of
Distribution of Identified Qualitative Metabolites in Different Units of

This table presents the quantitative distribution of metabolites identified in different plant parts (flowers and leaves) of
Significant at *p < .05.
Among the phenolic compounds contained in the flower, Hyperoside was found to be the largest proportion, belonging to a group of flavonols with diverse health benefits reported in previous studies. 14 Hyperoside is widely distributed in fruits and herbs of various plants such as Hypericaceae, Rosaceae, and Leguminosae, and has been reported to exhibit properties including antioxidant, antimicrobial, anti-inflammatory, anti-cancer, antiviral, and cardiobrovascular protection activities in the literature. 15 However, since this study did not directly evaluate these functional effects, the corresponding effects cannot be determined from the results of this study. Similarly, Hirsutrin was found at a high proportion, and has been reported elsewhere to exhibit antioxidant, anti-cancer, and anti-hepatitis B effects.16,17 However, these effects remain to be confirmed in this study.
Myricetin and Quercetin, along with their isomers and Quercetagetin 3-O-glucoside, were also abundant. These flavonoids have been reported to possess anti-inflammatory, anti-cancer, and antioxidant properties in previous works, with potential mechanisms including reduction of oxidative stress and modulation of cellular pathways.18–20 Nevertheless, the present study did not investigate these mechanistic effects.
Leaves showed similar flavonol subclass profiles, with other phenolic subclasses such as hydroxycinnamic acids and isoflavonoids predominant, as documented in the literature with potential roles in skin protection, metabolism, and hormone regulation. 21 Esculin, identified in leaves, is recognized for pharmaceutical roles as anticoagulant and antioxidant in prior studies, but such functional activities were not examined here. 22
Overall, while these phenolic compounds have been documented to possess varied bioactivities in the literature, the present study provides analytical data limited to compositional profiling and in vitro antioxidant potential. Confirmation of functional mechanisms or disease prevention claims requires dedicated follow-up studies including cellular, animal, and clinical experiments.
Figure 2 clearly shows the log2(FC) and –log10(p-value) values for each compound, indicating that major detected metabolites exhibit statistically significant differences in abundance. In the graph, red-colored points represent metabolites significantly more abundant in flowers, while blue-colored points indicate those more abundant in leaves. Notably, metabolites such as Gallocatechin, Myricetin 3-O-malonylglucoside, Epicatechin, and Gossypetin 3-O-glucoside show high levels and statistical significance in flowers, suggesting flowers are specialized for accumulation of functional metabolites in flavonoid and polyphenol classes. In contrast, metabolites such as Hydroxytyrosol 1-O-glucoside, Neodiosmin, Kaempferol 3-O-rutinoside, and Genistein are more highly accumulated in leaves, confirming leaves as major sites for isoflavones and specific flavonoids accumulation. Point sizes correspond to p-values, with larger points indicating greater statistical significance. Metabolites at far left or right extremes with large points exhibit the most pronounced tissue differences. These findings indicate that flowers and leaves possess unique metabolic profiles, suggesting tissue-specific secondary metabolite biosynthetic pathways.
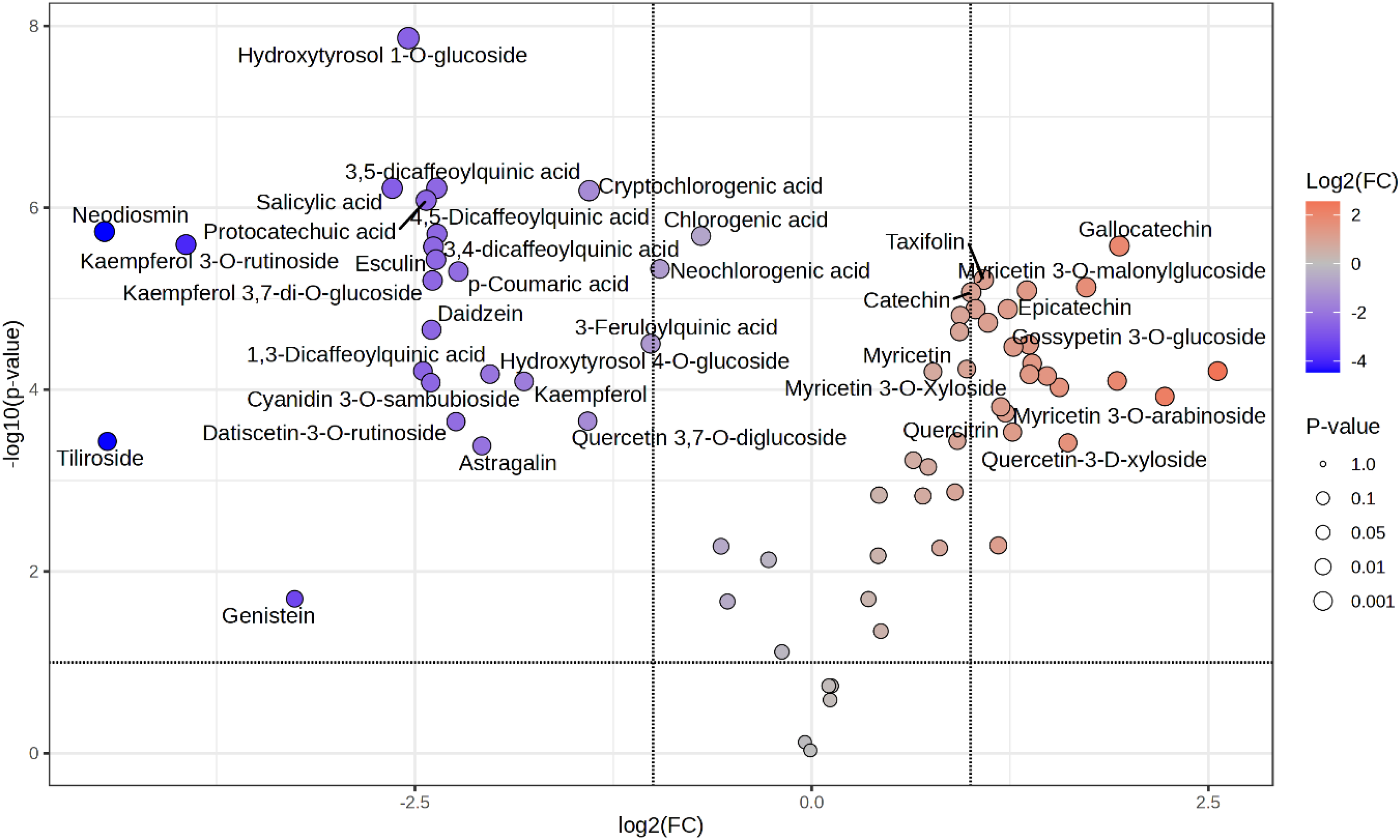
Volcano plot showing the differentially accumulated metabolites between flower and leaf of
Figure 3 consistently demonstrates metabolite distribution patterns in both flowers and leaves. Hierarchical clustering further supports distinct separation between flower and leaf samples, and among metabolites by accumulation patterns. Figure 4 shows clear separation of flowers and leaves reflecting distinct metabolite distributions observed earlier. Flower and leaf samples form clearly separated clusters on opposite plot sides, indicating distinct tissue metabolite compositions. Flower samples are on the left, leaf samples on the right, with metabolites specifically accumulated in each. Notably, Hyperoside, Hirsutrin, Rutin, and Myricetin orient toward flowers, suggesting high accumulation of these antioxidant, anti-inflammatory, and anticancer flavonoids and polyphenols in flowers. Quercetin, Chlorogenic acid, Daidzein, Genistein, and Glycitein distribute toward leaves, indicating higher abundance or exclusive detection there. Isoflavones Daidzein, Genistein, and Glycitein confirm leaves as main site for these compounds.

Hierarchical clustering heatmap illustrating differential metabolite profiles between flower and leaf of

PCA biplot of metabolite profiles in flower and leaf of
It is important to note that these results are based on in vitro analytical data and may not fully represent biological activity in vivo. The distinct metabolic profiles in different plant parts suggest potential for differential functional applications. These findings can contribute to reproducibility and consistency in industrial applications, particularly in food development where product efficacy and marketing strategies require scientific reliability and competitiveness. However, further in vivo and cell-based studies are necessary to validate the bioactivity and practical health benefits of these phenolic compounds in
Quantification of Phenolic Compounds
Quantitative analysis was conducted to determine the content of phenolic compounds identified by qualitative analysis in
Distribution of Identified Quantitative Metabolites in Different of

Data represent mean concentrations ± standard deviations for each metabolite detected by HPLC-HRMS analysis. Differences between tissues were statistically evaluated, and significant values are indicated. This table provides a comprehensive profile of tissue-specific metabolite distribution contributing to the phytochemical characterization of
ND means not detected.
Significant at *p < 0.05.
Taken together, these findings indicate that flowers and leaves of
In particular, the high levels of Hyperoside, Hirsutrin, Rutin, and Myricetin in the flower provide important biochemical data supporting the physiological and pharmacological potential suggested in literature; however, it should be noted that these biological activities, including antioxidant, anti-inflammatory, anticancer, neuroprotective, and cardiovascular protective effects, are reported from previous studies and have not been directly evaluated in this work. 23
For example, Hyperoside has been reported in other studies to exert strong antioxidant activity by scavenging ROS and suppressing pro-inflammatory cytokines, possibly contributing to inflammatory regulation.24,25 Similarly, Hirsutrin is known for antioxidant and anti-inflammatory properties and potential anticancer activities, while Rutin and Myricetin are associated with cardiovascular health, glucose metabolism, neuroprotection, and anticancer effects. 26 However, these mechanistic and disease-related effects remain to be validated in vivo and in clinical contexts.
Our study is limited to in vitro analyses of antioxidant activity and phenolic content; therefore, the translational relevance of these findings to medical, pharmacological, or industrial applications requires further validation through animal and clinical studies.
Moreover, the higher amounts of Quercetin and Chlorogenic acid and the exclusive presence of Daidzein, Genistein, and Glycitein in the leaf indicate tissue-specific metabolic profiles and suggest potential for functional differentiation. 27 Isoflavones found exclusively in the leaf are known phytoestrogens with diverse biological activities reported in other studies, including antioxidant, anticancer, anti-inflammatory, anti-osteoporotic, cardiovascular protective, and menopausal symptom-alleviating effects. The potential hormone-like effects and health applications, however, should be confirmed through dedicated experimental investigations.
Consequently, while the distinct biochemical profiles of
Conclusion
This study revealed distinct part-specific differences in antioxidant activity and phenolic compound composition of
Footnotes
Acknowledgements
The authors declare no acknowledgements.
Ethical Approval
Ethical approval is not applicable for this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The authors confirm that the data supporting of this study are available within the article. Additional raw data and datasets can be provided by the author upon reasonable request.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
