Abstract
Objective
The anti-tumor effects of 3-O-α-D-furanoribosyl-damar-24-ene-3β, 6α, 12β, 20S-tetraol (RPPT) in vitro were explored in the present study.
Methods
This study initially employed the CCK-8 proliferation inhibition assay to systematically evaluate RPPT's anticancer activity against three distinct tumor cell lineages and its cytotoxic effects on primary normal cell lines. Subsequently, multiparameter flow cytometry was utilized to characterize RPPT's functional attributes, quantitatively analyzing its apoptotic induction efficacy and dose-dependent cell cycle arrest patterns. Furthermore, Transwell migration assays were conducted to determine the derivative's capacity to inhibit tumor cell motility.
Results
In vitro cytotoxicity assays demonstrate that RPPT exhibits significantly enhanced selective cytotoxic activity against tumor cell lines compared to its natural precursor compound protopanaxatriol. Under same concentration conditions, this derivative demonstrates equivalent biological efficacy to the reference compound Rg3, characterized by dose-dependent inhibitory characteristics. The experimental data reveal that RPPT effectively suppresses tumor cell proliferation through concentration-dependent mechanisms, while maintaining no statistically significant cytotoxic effects on normal cell lines within the tested concentration ranges. Subsequent flow cytometry analysis revealed that RPPT suppress the cell cycle progression by blocking cell entry into the G0/G1 phase, and promoting cell apoptosis. Furthermore, Western blot analysis showed that RPPT could regulate the modulated the equilibrium between anti-apoptotic Bcl-2 and pro-apoptotic Bax, coupled with Caspase-9 activation, indicating that RPPT might facilitate tumor cells apoptosis by activating the apoptotic signaling pathway. Moreover, RPPT could efficiently inhibit the invasion and metastasis.
Conclusions
In summary, the studies showed that the new derivative RPPT has a strong growth inhibition effect on tumor cells, which expands new sources for subsequent development of potential anti-tumor drugs with more efficacy.
Introduction
At present, cancer has become a major disease threatening human life and health, and its morbidity and mortality are rising rapidly in the world. 1 In the process of tumor treatment, many anti-tumor drugs, due to the lack of selectivity, 2 kill cancer cells but also cause great damage to normal cells, easy to produce serious toxic side effects, thus affecting the therapeutic effect.3–5 Compared with traditional chemotherapeutic drugs, the natural products have obvious curative effect, fewer side effects and low toxicity to normal human cells. 6
Ginsenosides, the primary bioactive chemical components of ginseng, are recognized for their potential antitumor biological activity.7,8 Notably, naturally occurring ginsenosides exhibiting significant antitumor activity—such as Rg3, Rh2, and compound K (CK)—are predominantly conjugated to six-carbon sugars (eg, glucose) via glycosidic bonds to form glycosides, whereas they seldom form glycosides with five-carbon sugars, especially ribose, which exhibits broad pharmacological activities. The D-ribose, a natural pentose, plays an indispensable role in life activities, not only directly used as a drug for treating diseases, but also as an intermediate for synthesizing other drugs. 9
The structurally novel ocotillol-type ginsenoside RPDQ (12-riboside-pseudoginsengenin DQ) was chemically synthesized via a glycosylation protocol in our prior research, employing PDQ as the aglycone component and D-ribose as the glycosyl donor under optimized reaction conditions. 10 Notably, our study demonstrated that the derivative RPDQ of PDQ, exhibits more potent anti-tumor efficacy compared to PDQ. On this basis, we conducted a glycosylation reaction to modify the chemical structure of protopanaxatriol (PPT) and successfully synthesized a ginsenoside derivative with D-ribose as the glycosylated side chain, which was named RPPT. 11 The objective of the current investigation was to corroborate the inhibitory impact of the novel derivative RPPT on tumor cells, as well as to conduct a comparative analysis with the parent compound PPT, in order to ascertain the potency and prospective pharmacological efficacy of the former. The structure of PPT and RPPT is shown in Figure 1.

Chemical structures of PPT and RPPT.
Materials and Methods
Materials
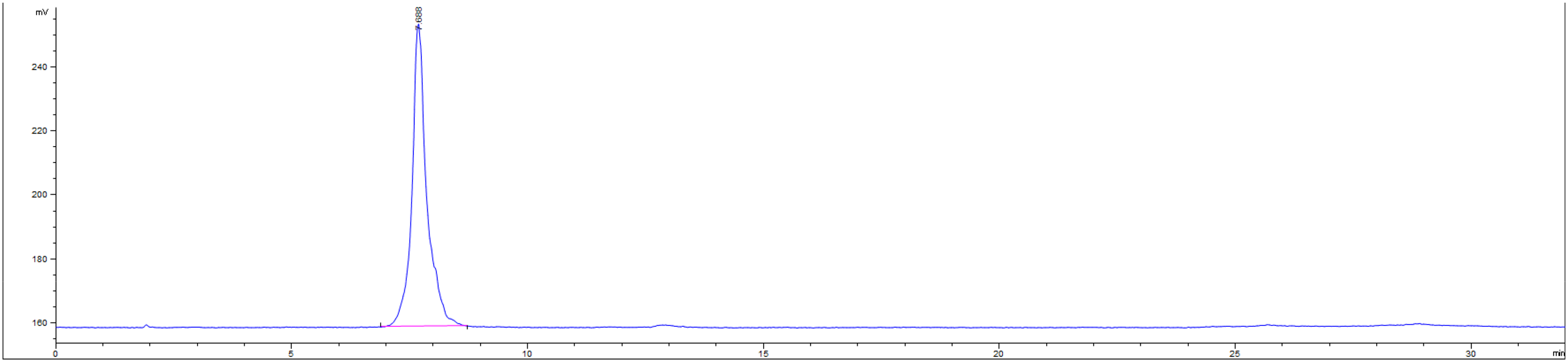
The protopanaxatriol and Rg3 were obtained from National Institutes for Food and Drug Control. The PPT derivative (RPPT) was prepared in the laboratory, and its purity was ≥98% as determined by HPLC analysis using an Agilent 1260 Infinity system (Agilent Technologies, USA). Chromatographic conditions: Column, Agilent ZORBAX SB-C18 (4.6 × 250 mm, 5 μm); mobile phase A (water) and B (acetonitrile); gradient elution (0-5 min: 80% B → 75% B; 5-10 min: 75% B → 65% B; 10-28 min: 65% B → 50% B; 28-50 min: 50% B constant); flow rate, 1.0 mL/min; column temperature, 30 °C. The retention time of RPPT was 7.6 min. Three human cancer cell lines (HepG2, A549, and MCF-7) and three normal cells (L-02, BEAS-2B and HBL-100) were purchased from the Fenghui Biology (Hunan, China).
Synthesis of Compounds
As shown in Scheme 1,using pyridine as the solvent, D-ribose underwent an acylation reaction with benzoyl chloride to afford compound 1. A bromination reaction was performed using 33% acetic acid-hydrobromic acid; subsequently, under acetone-water conditions, silver carbonate was added to selectively hydrolyze the protecting group at the C1 position, exposing the hydroxyl group to yield compound 2. After dissolving it in dichloromethane, DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) and trichloroacetonitrile were added for the reaction to obtain compound 3. Using TMSOTf (trimethylsilyl trifluoromethanesulfonate) as a catalyst, a glycosylation reaction with PPT was conducted to yield compound 4. Finally, deprotection afforded RPPT.

Synthesis of RPPT.
Cell Culture
The human hepatocellular carcinoma (HepG2), lung adenocarcinoma (A549), and breast adenocarcinoma (MCF-7) cell lines were cultured in Medium supplemented with 10% (v/v) heat-inactivated fetal bovine serum (FBS) and 1% (v/v) penicillin-streptomycin solution. All cultures were maintained under standard cell culture conditions (37 °C, 5% CO2 humidified atmosphere with 95% air) in tissue culture-treated flasks.
Cell Viability Assay
To assess the growth-suppressing properties of synthetic PPT derivatives, a standardized CCK-8 colorimetric assay was implemented. Cellular suspensions were precisely aliquoted into 96-well microtiter plates (7000 cells/well) and cultured conventionally. Subsequently, the cells were treated with the derivative RPPT at the concentration gradients of 0, 10, 20 and 40 μg/mL, respectively. Following compound exposure, CCK-8 detection solution was aliquoted into each well using an automated dispenser. The microplates were then maintained under physiological conditions (37°C, 5% CO2) for 60 min to facilitate formazan crystal formation. Absorbance values at 450 nm spectral channel were quantified using a microplate spectrophotometer (Biotek Instruments, Inc., USA) for quantitative analysis
Flow Cytometry Analysis
Cell cycle dynamics and apoptotic responses in RPPT-treated malignancies were systematically profiled using a Flow cytometry (Beckman Coulter, CA). For the analysis of tumor cell apoptosis: Cells in the logarithmic growth phase were trypsinized into a single-cell suspension, seeded in 6 cm dishes at a density adjusted based on cell size and morphology according to experimental requirements. After treatment according to grouping, cells from each group were collected by centrifugation. The cells were washed twice with pre-cooled PBS at 4°C, then resuspended in 500 μL of binding buffer to adjust the concentration to 1 × 107 cells/mL. A 100 μL aliquot of the cell suspension was transferred to a 5 mL flow cytometer tube, followed by the addition of 5 μL of Annexin V-FITC and thorough mixing. Subsequently, 5 μL of propidium iodide (PI) was added and thoroughly mixed.Post-mixing and light-protected incubation (RT, 15 min), apoptotic indices were determined by flow cytometry.
For cell cycle analysis, calculate the required volume of staining working solution based on the number of samples, with 500 μL of working solution allocated per sample for incubation. Immediately before use, prepare the staining working solution by mixing RNase Aand PI working solutions in a 1:9 volume ratio. Wash the cells once with PBS, centrifuge at 2000 rpm for 5 min, collect the cell pellet, adjust the cell concentration to 1 × 106 cells/mL, and transfer 1 mL of the single-cell suspension. After centrifugation of the prepared single-cell suspension, discard the supernatant. Fix the cells by adding 500 μL of ice-cold 70% (v/v) ethanol for 2 h to overnight, followed by storage at 4°C. Prior to staining, remove the fixative by washing the cells with PBS. Add 500 μL of the pre-prepared PI/RNase Astaining working solution, and incubate at room temperature in the dark for 30-60 min. Subsequently, perform cell cycle analysis.
Transwell Assay
Following assembly in 24-well plates, the Transwell system was prepared through differential medium loading: serum-depleted medium in upper chambers versus 500 μL complete medium (10% FBS) in lower chambers. The cell suspension (adjusted to the desired density) was then seeded into the upper chamber and incubated in a cell culture incubator for 48 h. After removal, fixed migrated cells in chilled 4% PFA for structural preservation, then performed histochemical staining with crystal violet solution for 10 min. Subsequently, the migrated/invasive cells were imaged and counted using a Nikon DS-Fi3 inverted fluorescence microscope (magnification, ×100, Tokyo, Japan).
Western Blot Assay
HepG2, A549 and MCF-7 cell lines at the exponentially proliferating cells and conducted 6-well plate seeding (optimal density) with 12-h pre-incubation prior to experimental interventions. Following 24-h RPPT exposure at graded concentrations, harvested tumor cells underwent protein extraction via lysis buffer treatment. Electrophoretic separation using SDS-PAGE was conducted, succeeded by nitrocellulose membrane electroblotting. Polyvinylidene difluoride (PVDF) membranes were initially blocked with 5% skim milk powder (dissolved in TBST) at room temperature for 1 h to prevent nonspecific binding. Subsequently, membranes were incubated overnight at 4 °C with apoptosis-related primary antibodies (Caspase-9/Bcl-2/Bax, diluted 1:1000 in TBST) on a rocking platform. After three sequential TBST washes (5 min per cycle), membranes were probed with HRP-conjugated goat anti-rabbit/mouse secondary antibodies (1:5000 dilution in TBST) under dark-adapted conditions at room temperature for 1 h. Protein bands were visualized using the Tanon 4800 Gel Imaging System (Tanon Scientific Instrument Co., Ltd, Shanghai, China), followed by grayscale densitometry analysis with ImageJ software (v1.53k).
Statistical Analysis
Quantitative data obtained from three independent experimental repetitions were expressed as mean values ± standard deviation (SD). Statistical processing was performed using GraphPad Prism 9.0 software, applying one-way analysis of variance (ANOVA) with Tukey's honestly significant difference (HSD) post hoc test for multiple group comparisons. The criterion for statistical significance was defined as p < 0.05.
Results
Synthesis of RPPT
The synthetic process of RPPT is shown in Scheme 1. The HPLC chromatogram of RPPT is shown in Figure 2.
Effects on Cell Proliferation
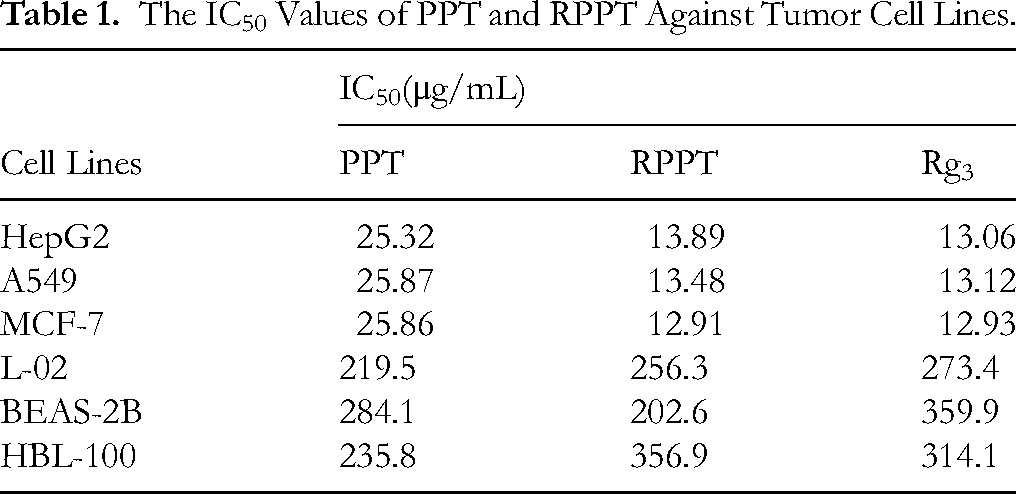
In order to evaluate the effect of RPPT on tumor cell survival, the cytotoxicity of RPPT was determined by CCK-8 assay, and the cytotoxicity of RPPT was verified using hepatoma cell line (HepG2), non-small cell lung cancer cell line (A549) and human breast cancer cell line (MCF-7) as models. The results were presented in the form of half-inhibitory concentration (IC50) values using PPT and Rg3 as positive controls. As shown in Figure 3, the RPPT treatment group showed a dose-dependent growth inhibition effect on each of the tumor cell lines tested compared to the control group. Specifically, with the increase of drug concentration, the survival vitality of tumor cells decreased in turn. According to the results, we can conclude that RPPT can significantly inhibit the proliferation activity of the above tumor cells. Notably, as can be seen from Table 1, compared with the substrate PPT, the new compound RPPT has a better inhibitory effect on tumor cells, but no significant inhibitory activity on normal cells. Furthermore, Table 1 reveals that the IC50 values of RPPT for HepG2, A549, and MCF-7 cells were 13.89 μg/mL, 13.48 μg/mL, and 12.91 μg/mL, respectively. In contrast, the IC50 values of Rg3 for these three cell lines were 13.06 μg/mL, 13.12 μg/mL, and 12.93 μg/mL, respectively. These results suggest that the inhibitory effect of RPPT on cell proliferation is comparable to that of Rg3.

Inhibitory effects of RPPT, PPT and Rg3 at different concentrations (0, 10, 20, and 40 μg/mL) on proliferation of HepG2, A549, and MCF-7 cells. All results are shown as means ± SD of three independent experiments. ***p < 0.001 versus PPT group.
The IC50 Values of PPT and RPPT Against Tumor Cell Lines.
Effects on Cell Apoptosis
In order to investigate whether RPPT can induce apoptosis of HepG2, A549 and MCF-7 tumor cells, Annexin V-FITC/PI assay was used to quantitatively analyze the apoptosis levels of these three tumor cells treated with RPPT. The results showed that compared with the blank control group, the number of apoptotic cells increased significantly after RPPT treatment, and this trend was positively correlated with drug concentration. In particular, under different drug concentrations, the proportion of early apoptotic cells exceeded that of late apoptotic cells, which indicates that the compound mainly acts to promote the early apoptotic process. Upon exposure to 40 μg/mL RPPT, the apoptosis rates of HepG2, A549, and MCF-7 cells were observed to be 32.47%, 28.91%, and 23.81%, respectively, compared to 4.81%, 6.14%, and 5.57% in the untreated control group. These observations suggest that RPPT induces tumor cell apoptosis in a concentration-dependent manner (Figure 4(A)). However, when cells were treated with an equivalent concentration of PPT, the apoptosis rates were recorded as 25.72%, 19.36%, and 18.25%. This indicates that, at the same concentration, RPPT exhibits a more pronounced effect on promoting tumor cell apoptosis than PPT. Furthermore, when compared to Rg3, RPPT's capacity to induce tumor cell apoptosis is comparable to that of Rg3 under identical treatment conditions. Of particular interest is the observation that RPPT slightly surpasses Rg3 in inducing apoptosis in HepG2 cells.
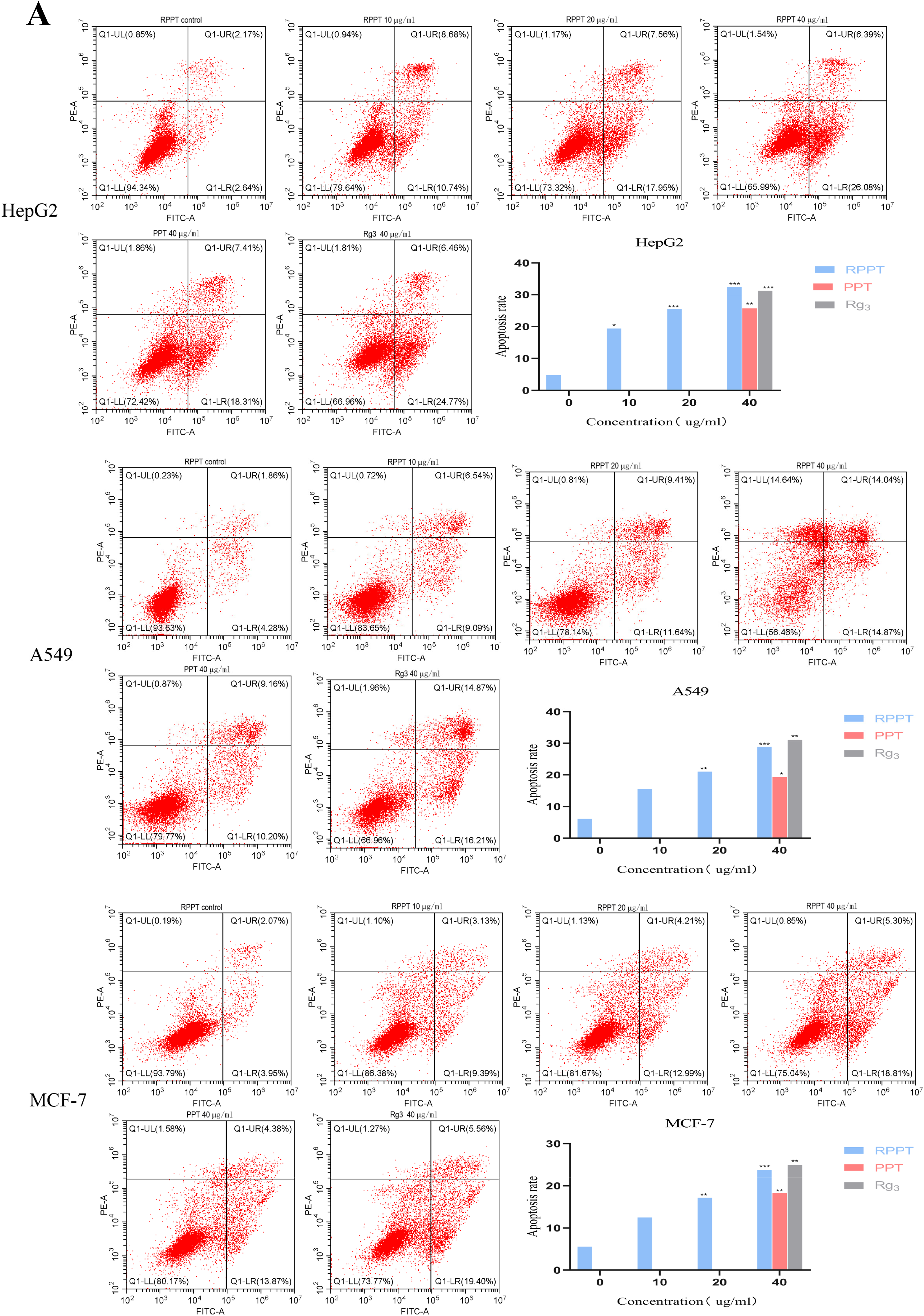
Effects of RPPT on HepG2, A549, and MCF-7 apoptosis. (A) The apoptosis rates of HepG2, A549, and MCF-7 cells were detected by flow cytometry with different concentrations of RPPT (0, 10, 20, and 40 μg/mL). (B) Effects of RPPT on the expression of Bax, Bcl-2 and caspase-9. Cells were treated with RPPT for 24 h. All results are shown as means ± SD of three independent experiments. *p < 0.05, **p < 0.01, ***p < 0.001 versus control group.
Subsequently, in order to further explore the molecular mechanism of RPPT inducing tumor cell apoptosis, we quantitatively analyzed the expression levels of apoptosis-related proteins Bax, Bcl-2 and Caspase-9 by Western Blot. As shown in Figure 4(B), under the conditions of different concentrations of RPPT, the expression levels of Bax and Caspase-9 were significantly up-regulated, while the expression of Bcl-2 was significantly down-regulated, and this regulatory effect was obviously dose-dependent. The above experimental results confirmed that RPPT may activate the mitochondria-dependent apoptosis signaling pathway by regulating the expression levels of Bax, Bcl-2 and Caspase-9, thereby inducing programmed death of tumor cells
Effects on Cell Cycle
Flow cytometric analysis was conducted to systematically evaluate the dose-dependent effects of RPPT on cell cycle progression in HepG2, A549, and MCF-7 carcinoma cells (Figure 5). Treatment with RPPT at 40 μg/mL induced a marked G0/G1 phase arrest across all tested models, with HepG2 cells showing a 2.76-fold increase in G0/G1 population (28.85% control vs 79.74% treated), while A549 and MCF-7 cells exhibited 2.27-fold (35.84% vs 81.37%) and 2.04-fold (29.00% vs 79.18%) increases, respectively. Concurrently, S-phase and G2/M-phase populations demonstrated concentration-dependent reduction, establishing a dose-response relationship. These findings collectively indicate that RPPT exerts potent antiproliferative effects through G1 phase arrest, accompanied by suppression of DNA synthesis machinery activation
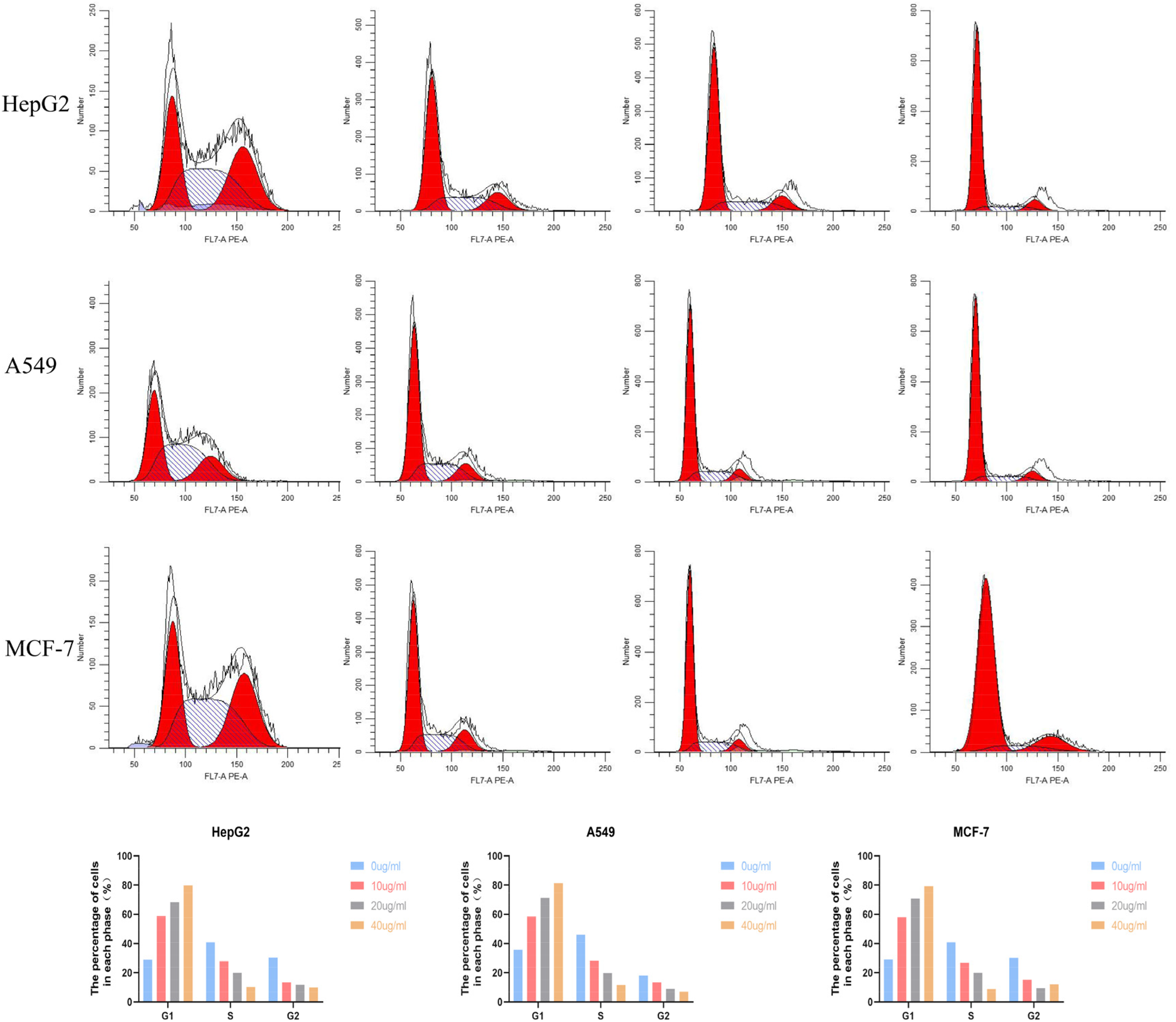
RPPT induced G1 phase arrest of HepG2 cells, A549 cells and MCF-7 cells. Propidium iodide staining and flow cytometry analysis were used to evaluate cell cycle progression. Column diagram shows the percentage of cell cycles at different dose concentrations. All results are shown as means ± SD of three independent experiments.
Effects on the Metastasis and Invasion
Cell migration and invasion represent fundamental biological activities that are crucial in numerous physiological and pathological processes. The capacity of tumor cells to migrate, invade, and metastasize significantly influences patient clinical outcomes. Tumor cells constantly metastasize and spread, often causing the cancer to recur repeatedly, making it difficult to cure. To explore the impact of RPPT on tumor cell migration, Transwell migration assays were conducted using HepG2, A549, and MCF-7 cells treated with varying concentrations of RPPT (0, 10, 20, and 40 µg/mL). As shown in Figure 6A and B, the findings revealed that the RPPT-treated group exhibited a marked reduction in the number of tumor cells traversing the Transwell chamber compared with the untreated control group. With the increase of dose concentration, there was a notable decrease in the invasive and migratory capabilities of the tumor cells, suggesting that RPPT's inhibitory effect on tumor metastasis and invasion is dose-dependent. In addition, it was observed that RPPT demonstrates a stronger ability to inhibit tumor cells migration than PPT at the same concentration (Figure 6C).

Effect of RPPT on migration of HepG2 cells, A549 cells and MCF-7 cells. (A) Representative image of the effect of different concentrations of RPPT and 40 μg/mL of PPT on tumor cell migration. (B) Quantitative evaluation of the number of migrating cells at different dose concentrations. (C) Quantitative evaluation of the effects of RPPT and PPT on the number of migrating cells at the same concentration. All data are representative of three independent experiments. ***p < 0.001 versus the control group, ###p< 0.001 versus PPT group.
Discussion
In this study, the aim was to evaluate the antitumor activity of a new ginsenoside derivative in order to find a new compound with potential excellent pharmacodynamic activity. It is well known that ginsenoside Rg3 has good preventive and anti-tumor effects, and the “Shenyi capsule” made from ginsenoside Rg3 monomer exhibited therapeutic potentiation through combinatorial administration with chemotherapy, leading to improved the therapeutic effect of tumors.12–14 PPT is the RPPT precursor compound, and there are many literature reports on the anti-tumor activity of PPT.15–17 Therefore, in the study of anti-tumor activity of RPPT, PPT and Rg3 were selected as reference indicators.
Ginsenosides demonstrate anticancer properties through pleiotropic mechanisms, including proliferation inhibition, 18 cell cycle arrest, 19 apoptotic induction, 20 and suppression of invasion/metastasis. 21 The uncontrolled proliferation and differentiation of tumor cells is one of the important causes of tumor formation.22,23 Therefore, inhibiting tumor cell proliferation is an important goal to evaluating the effects of drugs to exert therapeutic tumors. This study established a holistic mechanistic evaluation system through an integrative methodology encompassing proliferation inhibition, cell cycle modulation, and metastatic suppression, thereby systematically assessing the therapeutic potential of RPPT in tumor biology.
In the current study, the cytotoxic activity of RPPT was evaluated using the CCK-8 method, and it was found that its ability to inhibit the proliferation of HepG2, A549, and MCF-7 tumor cells in vitro was positively correlated with concentration. Compared with PPT, its IC50 value is lower, indicating that RPPT has stronger toxicity to tumor cells. We also studied the cytotoxic effect of Rg3 on three kinds of tumor cells, and the data showed that the IC50 value of RPPT was very close to that of Rg3, further indicating that RPPT had a better cytotoxic effect on tumor cells than PPT. Furthermore, we examined the cytotoxicity of RPPT against three normal cells, L-02, BEAS-2B and HBL-100, and found that it is less cytotoxic to normal cells than to tumor cells.
Apoptosis is a genetically regulated, programmed cell death process, and its changes are an important mechanism to prevent the emergence of tumors.24,25 Therefore, inhibiting cell proliferation and promoting apoptosis are effective strategies in tumor therapy26,27 Flow cytometry analysis of HepG2, A549 and MCF-7 cells showed different degrees of apoptosis of the three tumor cells, and their apoptosis rates were positively correlated with RPPT administration concentration, indicating that RPPT has the potential to induce apoptosis and inhibit the proliferation of cancer cells. In addition, RPPT induced apoptosis at a rate comparable to Rg3 and higher than PPT, indicating that RPTP have a favorable pharmacological effect on tumor inhibition. Bax and Bcl-2 are representative apoptosis-promoting and inhibiting apoptosis proteins of the Bcl-2 family, respectively, functioning mainly at the mitochondrial membrane of the cells, and the balance between them determines the degree of apoptosis. 28 In cells, excessive expression of Bax can activate the apoptotic process, correspondingly, excessive production of Bcl-2 leads to uncontrolled cell proliferation. Caspase-9 is an important apoptotic effector enzyme in the apoptotic cascade in the Caspase family of proteins, which plays a key role in the mitochondria-mediated apoptotic pathway. 29 Therefore, next, this study further explored the mechanism of RPPT-induced apoptosis. Western blot analysis demonstrated that RPPT treatment significantly upregulated the expression of the anti-apoptotic protein Bcl-2 and the pro-apoptotic protein Bax in a dose-dependent manner. Concurrently, RPPT enhanced the protein expression of caspase-9, indicating its role in initiating the mitochondrial apoptotic pathway. These findings suggest that RPPT modulates the Bcl-2/Bax apoptotic regulatory axis and activates caspase-9-mediated downstream effector cascades, thereby inhibiting tumor cell proliferation and inducing apoptosis. Flow cytometric analysis revealed that RPPT treatment induced G1-phase cell cycle arrest in all tested tumor cell lines, characterized by a marked increase in the G1-phase population and a concomitant decrease in the S-phase fraction. This G1-phase block effectively disrupts DNA replication and mitotic progression, leading to proliferation inhibition. Collectively, these results highlight RPPT's dual regulatory mechanisms targeting both apoptotic signaling and cell cycle progression as a strategy for tumor growth suppression.
A remarkable feature of tumor is that tumor cells are metastatic nature, 30 and when the proliferation of tumor cells in a certain area reaches a certain degree, it will metastasize to other areas. Studies have found that more than 90% of cancer deaths are caused by metastases. 31 Therefore, inhibiting tumor cell metastasis holds critical significance in cancer therapy. Studies have demonstrated that RPPT significantly reduces the viability and invasive capacity of A549 and SK-MES-1 cells, exhibiting effects comparable to cisplatin. 32 Transwell migration assays were performed to analyze and compare the migratory ability of tumor cells following treatment with PPT and RPPT. Our findings revealed that RPPT suppressed tumor cell invasion and migration in a concentration-dependent manner. Notably, at a dose of 40 μg/mL, the number of RPPT-treated cells migrating through the Transwell chambers was significantly reduced, indicating that RPPT effectively inhibits tumor cell migration. Furthermore, RPPT-treated cells exhibited fewer migratory events compared to the PPT group, suggesting that RPPT possesses superior inhibitory activity against cell migration.
Despite systematic evaluation of the pharmacological activity of the novel compound RPPT against tumor cells through mechanisms involving proliferative suppression, cell cycle regulation, and metastatic inhibition, these in vitro-based assessments remain insufficient to fully reflect the complexity of the in vivo microenvironment. Future research should establish animal model experiments for validation, aiming to comprehensively evaluate the actual therapeutic efficacy and safety profiles of this new compound.
Conclusion
Our research indicates that the new compound RPPT exhibits significant anti-tumor activity against tumor cells, and it shows better anti-tumor efficacy compared to the parent compound PPT. However, it should be noted that the current assessment of anti-tumor activity is limited to in vitro experiments, and the conclusions are preliminary. Therefore, the anti-tumor activity of RPPT still needs to be further explored and analyzed through in vivo verification and mechanism studies using more physiological feature-based experimental models.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251411177 - Supplemental material for Evaluation of Anti-Tumor Activity of a Novel Ginsenoside Derivative RPPT in Vitro
Supplemental material, sj-docx-1-npx-10.1177_1934578X251411177 for Evaluation of Anti-Tumor Activity of a Novel Ginsenoside Derivative RPPT in Vitro by Wei Zhang, Zhidong Qiu, Junge Li and Zhenzhou Wang in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X251411177 - Supplemental material for Evaluation of Anti-Tumor Activity of a Novel Ginsenoside Derivative RPPT in Vitro
Supplemental material, sj-docx-2-npx-10.1177_1934578X251411177 for Evaluation of Anti-Tumor Activity of a Novel Ginsenoside Derivative RPPT in Vitro by Wei Zhang, Zhidong Qiu, Junge Li and Zhenzhou Wang in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X251411177 - Supplemental material for Evaluation of Anti-Tumor Activity of a Novel Ginsenoside Derivative RPPT in Vitro
Supplemental material, sj-docx-3-npx-10.1177_1934578X251411177 for Evaluation of Anti-Tumor Activity of a Novel Ginsenoside Derivative RPPT in Vitro by Wei Zhang, Zhidong Qiu, Junge Li and Zhenzhou Wang in Natural Product Communications
Supplemental Material
sj-docx-4-npx-10.1177_1934578X251411177 - Supplemental material for Evaluation of Anti-Tumor Activity of a Novel Ginsenoside Derivative RPPT in Vitro
Supplemental material, sj-docx-4-npx-10.1177_1934578X251411177 for Evaluation of Anti-Tumor Activity of a Novel Ginsenoside Derivative RPPT in Vitro by Wei Zhang, Zhidong Qiu, Junge Li and Zhenzhou Wang in Natural Product Communications
Footnotes
Acknowledgements
Special thanks to the support of the Key Research and Development Program of Jilin Province.
Contributors' Statement
Data collection: Junge Li, Zhidong Qiu; design of the study: Wei Zhang, Junge Li, Zhidong Qiu; statistical analysis: Wei Zhang, Junge Li; analysis and interpretation of the data:Wei Zhang, Zhenzhou Wang; drafting the manuscript: Wei Zhang; critical revision of the manuscript: Wei Zhang, Zhidong Qiu, Zhenzhou Wang.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Research and Development Program of Jilin Province (Grant number 20210204149YY).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
