Abstract
Exposure to ionizing radiation (IR) during procedures such as abdominal and pelvic radiotherapy results in severe intestinal injury. In the current study, we demonstrated that a lethal dose of IR triggered intestinal epithelial cell apoptosis and pyroptosis via Caspase-3 cleavage. The ginseng active ingredients, ginsenosides (GS) and ginseng phenolic acids (GPA), significantly inhibited IR-induced apoptosis in IEC-6 cells. GS and GPA also reduced Caspase-3 and GSDME cleavage and HMGB1 and LDH release, thus inhibiting pyroptosis after IR. Moreover, GS- or GPA-treated IEC-6 cells showed reduced reactive oxygen species and DNA damage accumulation. These results suggest that IR induces apoptosis and GSDME-dependent pyroptosis through Caspase-3 activation, which can be relieved by GS and GPA, thus supporting the potential use of GS and GPA in the prevention and treatment of IR-induced intestinal injury.
Introduction
Radiotherapy remains a mainstay of cancer therapeutic modality in addition to surgery and chemotherapy.1,2 However, the rapidly renewing small intestine is particularly sensitive to ionizing radiation. Radiation exposure during procedures such as abdominal and pelvic radiotherapy results in acute gastrointestinal (GI) injury and the occurrence of GI syndrome,3,4 which has become a major clinical issue. Previous studies have indicated that intestinal epithelial cell apoptosis plays an important role in acute radiation toxicity. 5 Pyroptosis, which is secondary necrosis after apoptosis, is an inflammatory form of programmed cell death that depends on activated caspases and can be divided into Caspase-1-, Caspase-11-, Caspase-8-, or Caspase-3-dependent cell death. 6 Recent studies have shown that radiotherapy and some chemotherapy compounds induce pyroptosis in tumor cells through gasdermin E (GSDME) cleavage caused by Caspase-3 cleavage.7,8 Activation of the Bcl-2/Bax-Caspase-9-Caspase-3-GSDME signaling pathway is essential for ionizing radiation (IR)-mediated GSDME-dependent pyroptosis. 9 In addition, several studies have reported that intracellular reactive oxygen species (ROS) generation induced by various stimuli can regulate pyroptosis.10–12 IR also induces DNA damage and rapidly causes ROS release, 13 which is known as radiation enteritis and may disrupt homeostasis, activate proinflammatory factors, and lead to cell apoptosis/pyroptosis. 14 Amifostine, which is the only FDA-approved drug, has high side effects. 15 Therefore, it is necessary to find novel protective agents against radiation. In recent years, compounds derived from natural herbaceous plants have attracted much attention due to their low toxicity. 16
Ginseng, the rhizome of Panax ginseng C.A. Meyer, is widely used in traditional Chinese medicine for the prevention and treatment of metabolic and aging-related diseases. 17 Ginseng, which has low toxicity, has antioxidant, anti-inflammatory and DNA damage relief effects by improving the antioxidant capacity of free radicals.18–21 The main pharmacological components of ginseng, including ginsenosides (GS), ginseng phenolic acids (GPA) and ginseng polysaccharides (GPS), have been reported to reduce radiation-induced cell apoptosis and increase intestinal crypt survival. 22 GS relieves aging-related oxidative damage in mice with accelerated aging and inhibits cisplatin-induced DNA fragmentation in rats. 23 GPS has also been shown to protect DNA from oxidative damage by activating Nrf2. 24 Although the pharmacological and biological effects of ginseng extracts and their active compounds have been extensively studied, their distinct functions and the underlying mechanisms by which they alleviate intestinal radiation damage remain unclear. In this study, we demonstrated the protective effects of GS, GPA, and GPS against radiation injury by blocking apoptosis and pyroptosis in intestinal epithelial cells.
Results
IR-Induced Apoptosis in IEC-6 Cells
IEC-6 cells were irradiated with or without 15 Gy of IR (treatment protocol as shown in Figure 1A), and apoptotic indicators were detected. Cell viability was measured by cell counting kit-8 (CCK-8) assays and showed a 60% decrease in cell number after irradiation (Figure 1B). Western blot analysis showed that Caspase-3 cleavage increased after irradiation, peaked at 4 hours, and then decreased (Figure 1C and D), and immunofluorescence assays were consistent, with the positive rate reaching 30% at 4 hours after IR (Figure 1E and F). Flow cytometry showed a significant increase in apoptotic cells at 4 hours after irradiation (Figure 1G and H). These results suggested that IR rapidly induced apoptosis in IEC-6 cells.
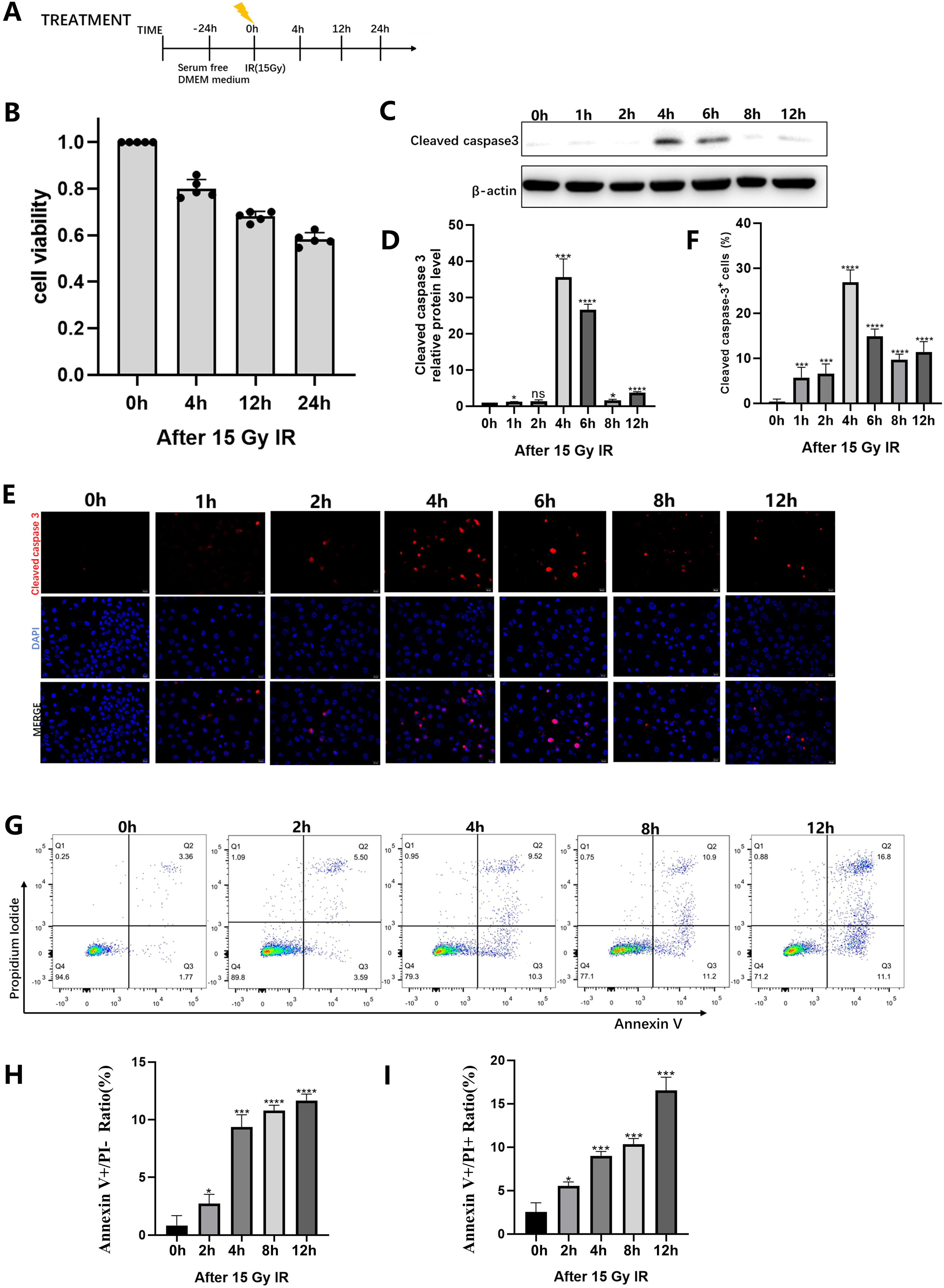
IR-induced apoptosis of IEC-6 cells. IEC-6 cells were detected at specific time points after 15 Gy radiation. (A) Schematics of radiation dosing and analyses at indicated time points. (B) IEC-6 cell viability was evaluated by cell counting kit-8 assay. (C) Representative immunoblot analysis of cleaved caspase-3 expression levels and quantification of relative expression (D). (E) Fluorescence microscopy images showing Cleaved Caspase3 staining. Scale bar, 20 µm, and quantification of Cleaved Caspase-3 expression (F). (G) Flow cytometry assay of Annexin V—FITC and PI. The percentage of Annexin V—FITC positive cells, presumably apoptosis of cells (H) and the percentage of double-positive cells, presumably proptosis of cells (I). Results are presented as the mean ± SD of three independent experiments. * P < .05, **P < .01, ***P < .001, and ****P < .0001 versus control. IR, ionizing radiation; ns, not significant; CTRL/NC/0 hours, control.
IR-Induced Pyroptosis in IEC-6 Cells via Caspase-3/GSDME
The results showed that 15% of IEC-6 cells were Annexin V and PI double positive at 12 hours after irradiation (Figure 1I), which suggested necrosis-like cell death. 25 LDH and HMGB1 are generally used to detect the occurrence of pyroptosis. The release of LDH is closely related to the occurrence of pyroptosis, and HMGB1 regulates pyroptosis through ROS/ERK1/2/caspase-3/GSDME signaling pathway.26,27 LDH release and HMGB1 expression were significantly increased at 24 hours after irradiation (Figure 2A and B). Caspase-1/4/5/11 activate GSDMD to trigger pyroptosis. 28 Although Caspase-1 and GSDMD were expressed in IEC-6 cells, IR did not induce the cleavage of GSDMD or Caspase-1 (Figure 2C), which suggested that GSDMD was not involved in IR-induced LDH release and HMGB1 expression. GSDME could promote pyroptosis through N-terminal domain release induced by cleaved caspase-3. 7 Radiation significantly induced the release of the GSDME N-terminal domain at 12 hours (Figure 2D). Current studies have shown that the activation of GSDME or GSDMD was involved in pyroptosis induced by various agents and pathways. In our experiments, we observed IR-induced cleavage of GSDME, but not GSDMD, in IEC-6 cells, which indicated that radiation-induced pyroptosis in intestinal epithelial cells is associated with GSDME activation. However, the activation of GSDME was dramatically blocked by the caspase-3-specific inhibitor Z-DEVD-FMK (Figure 2E). Inhibition of Caspase-3 reduced cell death, as indicated by PI staining (Figure 2F). Microscopic observations revealed membrane lysis and large bubbles emerging from the plasma membrane in IR-treated IEC-6 cells, which are morphological features that are distinct from apoptosis but similar to pyroptosis, and plasma membrane expansion was reduced by inhibitor treatment (Figure 2G). The release of LDH and HMGB1 induced by radiation was also reduced by inhibitor treatment. These results demonstrated that IR triggered Caspase-3-mediated cleavage of GSDME and pyroptosis.

IR-induced pyroptosis of IEC-6 cells via Caspase-3/GSDME. IEC-6 cells were detected at indicated time points after 15 Gy radiation. (A) LDH release levels. Each column represented the mean value of three biological replicates, and error bars indicated SD. (B) Representative immunoblot analysis of HMGB1 expression levels (C) Representative immunoblot analysis of pro-Caspase-1, cleaved caspase-1, GSDMD/GSDMD-N expression levels. (D) Representative immunoblot analysis of Cleaved Caspase-3 and GSDME/GSDME-N. (E) Expression levels of Cleaved Caspase-3, Cleaved PARP, GSDME, and GSDME-N in IEC-6 cells treated with or without Caspase-3 inhibitor (Z-DEVD-FMK) 12 hours after 15 Gy IR. (F) Fluorescent microscopy images showing PI staining in IEC-6 cells treated with or without Caspase-3 inhibitor (Z-DEVD-FMK) 12 hours after 15 Gy IR. Scale bar, 100 µm. (G) Phase-contrast microscope imaging of IEC-6 cells treated with or without Caspase-3 inhibitor (Z-DEVD-FMK) 12 hours after 15 Gy IR. Pyroptotic cell morphology was pinpointed by red arrows. Scale bar, 50 µm. (H) LDH release in the IEC-6 cells treated with or without Caspase-3 inhibitor (Z-DEVD-FMK) 12 hours after 15 Gy IR. Each line represented the mean value of five biological replicates, and error bars indicated SD. (I) Expression levels of HMGB1 in IEC-6 cells treated with or without Caspase-3 inhibitor (Z-DEVD-FMK) 12 hours after 15 Gy IR. Results are represented as the mean ± SD of at least three independent experiments. *P < .05, **P < .01, and ***P < .001. C-casp3, Cleaved Caspase-3; GSDME, gasdermin E; GSDME-N, gasdermin E-N-terminal; LDH, lactate dehydrogenase; PI, propidium iodide; ns, not significant; NC, control; IR, ionizing radiation.
Ginseng Extract Alleviates the Apoptosis and Pyroptosis Induced by IR
IEC-6 cells were irradiated with or without 15 Gy of radiation (Figure 3A). To assess the potential of crude ginseng extract in enhancing IEC-6 cell viability and mitigating cell death following IR, various concentrations of the extract (0, 10, 100, 300, 1000 µg/mL) were administered to cells 24 hours before IR, and CCK-8 assay was performed to measure cell viability. The findings indicated that both 1 µg/mL and 10 µg/mL of ginseng extract significantly boosted cell viability, and 10 µg/mL concentration treatment exhibited more increase. Conversely, treatment with 100, 300, and 1000 µg/mL of ginseng extract notably reduced cell viability in comparison to the control group, possibly due to adverse effects. As a result, a 10 µg/mL concentration of ginseng extract was preferred for further assays. (Figure 3B). Ginseng extract treatment increased the colony formation of irradiated IEC-6 cells (P < .01, Figure 3C). Western blotting was used to detect the expression of the apoptosis-related protein cleaved caspase-3 and the pyroptosis-related protein GSDME. The expression levels of cleaved caspase-3 and GSDME in ginseng extract- and IR-treated cells were significantly lower than those in the radiation-alone group (Figure 3D and E). PI and SYTOX Green fluorescence staining were used to detect apoptosis and pyroptosis, respectively. The fluorescence microscopy results showed that cells treated with ginseng extract combined with IR had a lower positive rate than those treated with IR alone (Figure 3F and G).

Ginseng extract alleviates apoptosis and pyroptosis in irradiated IEC-6 cells. IEC-6 cells were treated with ginseng extract followed by radiation and detections at indicated time points. (A) Schematics of ginseng extract and radiation dosing and analyses at indicated timepoints. (B) Cell viability was evaluated by cell counting kit-8 assay. (C) IEC-6 cells were treated with 10 µg/mL Ginseng for 24 hours, then 24 hours after IR, and cell proliferation was evaluated by colony-formation assays. (D) Immunoblot analysis of Cleaved Caspase-3 expression levels 4 hours after IR. (E) Expression levels of GSDME and GSDME-N 12 hours after IR. Fluorescent microscopy images showing PI (F) and Sytox Green (G) staining 12 hours after IR (15Gy). Scale bar, 100 µm. Results are represented as the mean ± SD of at least three independent experiments. *P < .05, **P < .01, and ***P < .001. C-casp3, Cleaved Caspase-3; GSDME, gasdermin E; GSDME-N, gasdermin E-N-terminal; PI, propidium iodide; ns, not significant; NC, control; IR, ionizing radiation.
GS and GPA Alleviated Apoptosis in IEC-6 Cells Induced by IR
The results showed that ginseng could alleviate radiation-induced apoptosis and pyroptosis and protect intestinal epithelial cells. Next, we detected whether the main active compounds of ginseng, GS, GPA, and GPS, possess protective effects against IR-induced damage to IEC6 cells. Cells were treated with different concentrations of GS, GPA, or GPS (0, 10, 100, 300, 1000 µg/mL) for 4, 12, or 24 hours after IR (Figure 4A), and cell viability was detected by CCK-8 assay. We found that the decrease of cell viability induced by IR in IEC6 cells was not significantly rescued by either low or high concentrations of GPS, whereas low concentrations of both GS and GPA (10 µg/mL) significantly increased IEC6 cell viability. Therefore, 10 µg/mL of compounds was used in all following assays (Figure 4B). Since previous studies have shown that cell apoptosis is significant at 4 hours after radiation, we examined IEC-6 cell apoptosis at 4 hours after radiation. Flow cytometry showed that at 4 hours after radiation, GS or GPA treatment reduced the population of Annexin V-positive and PI-negative (apoptotic cells) cells (Figure 4C). TUNEL and PI staining showed that the combination of GS or GPA plus IR reduced PI fluorescence compared with IR alone (Figure 4D and E). Additionally, as shown in the WB results, GS and GPA increased the Bcl-2/Bax ratio and attenuated the cleavage of Caspase-9, Caspase-3, PARP1, and GSDME in IR-treated IEC-6 cells (Figure 4F). Similar results were observed in cleaved caspase-3 staining at 4 hours after IR (Figure 4G). However, the low concentrations of GPS did not show a significant amelioration of IR-induced apoptosis in IEC6 cells. Taken together, these data indicated that GS and GPA alleviated apoptosis induced by radiation in IEC-6 cells.

GS and GPA alleviate the apoptosis of IEC-6 cells induced by radiation. IEC-6 cells were treated with GS, GPA or GS followed by radiation and detections at indicated time points. (A) Schematics of GS, GPA or GS and radiation dosing and analyses at indicated time points. (B) IEC-6 cells were treated with the indicated concentrations of GPS, GPA or GS for 24 hours, then cell viability was evaluated by cell counting kit-8 assay. (C) Flow cytometry assay of Annexin V-FITC and PI 4 hours after IR. The percentage of Annexin V - FITC positive cells, presumably apoptosis of cells. Fluorescent microscopy images showing TUNEL (D) and PI (E) staining. Scale bar, 50 µm. (F) Representative immunoblot analysis of Bcl-2, Bax, Caspase-9, Cleaved Caspase-9, Cleaved Caspase-3, PARP, Cleaved PARP expression levels 4 hours after IR. (G) Fluorescent microscopy images showing Cleaved Caspase-3 staining 4 hours after IR. Scale bar, 50 µm. Results are represented as the mean ± SD of at least three independent experiments. * P < .05, **P < .01 and ***P < .001. GPS, Ginseng polysaccharides; GPA, Ginseng phenolic acids; GS, Ginsenosides; C-casp3, Cleaved Caspase-3; GSDME, gasdermin E; GSDME-N, gasdermin E-N terminal; PI, propidium iodide; ns, not significant; NC, control; IR, ionizing radiation.
GS and GPA Relieved IR-Induced Pyroptosis in IEC-6 Cells via Caspase-3/GSDME
The results suggested that pyroptosis was most obvious at 12 hours after IR. Flow cytometry showed that the number of pyroptotic cells increased significantly at 12 hours after IR, and treatment with GS and GPA reduced the population of Annexin V and PI double-positive cells (Figure 5A). Flow cytometry showed that GS or GPA treatment reduced the population of Annexin V and Sytox Green double-positive cells (Figure 5B). Immunofluorescence staining with Sytox Green indicated the same result (Figure 5C). GS or GPA treatment reduced IR-induced plasma membrane ballooning, LDH release and HMGB1 expression (Figure 5D-F). Western blot analysis showed that the expression levels of cleaved caspase-3 and GSDME-N in IEC-6 cells treated with GS or GPA were significantly lower than those in IEC-6 cells treated with IR alone at 12 hours after irradiation (Figure 5G). Immunofluorescence staining showed that cleaved caspase-3-positive cells in IEC-6 cells treated with GS or GPA combined with IR were significantly lower than those treated with IR alone at 12 hours after IR (Figure 5H). Moreover, compared with the fluorescence staining of cleaved caspase-3 at 4 hours after radiation (Figure 4H), the number of cleaved caspase-3-positive cells decreased at 12 hours after IR, which confirmed that Caspase-3 and other indicators of apoptosis were mainly affected at 4 hours after IR. However, 10 µg/mL GPS did not show a significant amelioration of IR-induced pyroptosis in IEC6 cells. These data suggested that GS and GPA relieved IR-induced pyroptosis in IEC-6 cells via the Caspase-3/GSDME pathway.

GS and GPA relieve radiation-induced pyroptosis of IEC-6 cells via Caspase3/GSDME. IEC-6 cells were treated with GS, GPA or GS for 24 hours followed by 15 Gy radiation and detections at indicated time points. (A)Representative flow cytometry scatter plots, flow cytometry assay of Annexin V-FITC and PI 12 hours after IR. (B) Representative flow cytometry scatter plots, flow cytometry assay of Annexin V and Sytox Green 12 hours after IR. (C) Fluorescent microscopy images showing Sytox Green staining 12 hours after IR. Scale bar, 50 µm. (D) Phase-contrast images 12 hours after IR. Pyroptotic cell morphology was pinpointed by red arrows. (E) Release of LDH 12 hours after IR. Each line represented the mean value of five biological replicates, and error bars indicated SD. Representative immunoblot analysis of HMGB1 (F); Cleaved Caspase-3, GSDME and GSDME-N (G) expression levels 12 hours after IR (15 Gy). (H) Fluorescent microscopy images showing Cleaved Caspase-3 staining 12 hours after IR. Scale bar, 50 µm. Results are represented as the mean ± SD of at least three independent experiments. *P < .05, **P < .01, and ***P < .001. GPS, Ginseng polysaccharides; GPA, Ginseng phenolic acids; GS, Ginsenosides; C-casp3, Cleaved Caspase-3; GSDME, gasdermin E; GSDME-N, gasdermin E-N terminal; PI, propidium iodide; ns, not significant; NC, control; IR, ionizing radiation.
GS and GPA Reduced Radiation-Induced ROS Production and DNA Damage in IEC-6 Cells
Activation of ROS can induce multiple cell death modes, such as apoptosis, necroptosis, and ferroptosis. 29 Intracellular ROS production induced by various stimuli also regulates pyroptosis.10–12 Considering that ionizing radiation injury is closely related to ROS production, we detected the level of ROS in irradiated cells. Fluorescence staining showed that intracellular ROS levels increased significantly after radiation, but the ROS level decreased in the cell pretreated with GS or GPA (Figure 6A). The superoxide dismutases (SOD) are considered as the first line of ROS scavengers, so the SOD expression was measured in compounds-treated cells 12 hours post-IR (Figure 6B). The result indicated that GPA and GS significantly increased SOD levels in radiated cells. Previous studies have shown that apoptosis and pyroptosis caused by radiation-induced DNA damage are related to p-H2A.X. 30 In this study, it was shown that p-H2A.X was significantly increased after IR but decreased after GS and GPA treatment (Figure 6C and D). Although 10 µg/mL GPS can partially alleviate the release of ROS and DNA damage induced by IR in IEC6 cells, the effect is not significant. These results suggested that ROS accumulation and DNA damage induced by IR in IEC-6 cells were alleviated by GS and GPA.

GS and GPA alleviated radiation-induced mitochondrial damage and ROS production in IEC-6 cells. IEC-6 cells were treated with the GPS, GPA, or GS for 24 hours before 15 Gy IR and analyzed 12 hours after IR. (A) Cellular ROS levels were detected by Reactive Oxygen Species Assay Kit. Scale bar, 50 µm. (B) Cellular SOD expression level. (C) Fluorescent microscopy images showing p-H2A.X (red) and DAPI (blue) staining. Scale bar, 20 µm. (D) Representative immunoblot analysis of p-H2A.X and quantification of relative expression level. Results are represented as the mean ± SD of at least three independent experiments. *P < .05, **P < .01, and ***P < .001. GPS, ginseng polysaccharides; GPA, ginseng phenolic acids; GS, ginsenosides; ns, not significant; NC, control; IR, ionizing radiation; SOD, superoxide dismutase.
GS and GPA Promoted the Proliferation of IEC-6 Cells
EdU staining showed that GS and GPA promoted IEC-6 cell proliferation (Figure 7A). Cell cycle analysis by flow cytometry showed that there were more IEC-6 cells in S-phase and G2/M phase in response to GS or GPA (Figure 7B). Moreover, GS or GPA treatment significantly increased colony formation compared with the control group, with or without radiation (Figure 7C). Immunofluorescence staining of p-H3 showed that GS or GPA increased the ratio of p-H3-positive cells in both the irradiated group and nonirradiated group compared with the control (Figure 7D). These results suggested that GS and GPA were involved in cell cycle regulation to promote proliferation and regeneration after radiation injury.

Effects of GS and GPA on IEC-6 cells proliferation and regeneration. IEC-6 cells were treated by GPS, GPA, and GS for 24 hours, and then followed with or without 15 Gy radiation. Analyses were processed at indicated time points. (A) IEC-6 cells were treated with GPS, GPA, and GS for 24 hours, and then cell proliferation was assessed by EdU staining. Scale bar, 50 µm. (B) IEC-6 cells were treated with GPA and GS for 24 hours followed by cell cycle assays, representative flow cytometry results were recorded (left panel) and cell cycle distribution was quantified (right panel). (C) Cell proliferation was evaluated using the colony-formation assays 1 week after compound treatment. (D) Fluorescent microscopy images of p-H3 staining 12 hours after IR. Scale bar, 20 µm. Results are represented as the mean ± SD of at least three independent experiments. *P < .05, **P < .01, and ***P < .001. NC, control. GPS, ginseng polysaccharides; GPA, ginseng phenolic acids; GS, ginsenosides; ns, not significant; NC, control; IR, ionizing radiation.
Discussion
GI injury has always been a serious problem associated with abdominal radiotherapy due to rapid intestinal renewal and high sensitivity. 31 A recent report showed that GPA and GS reduced the frequency of apoptosis in the intestinal crypts of irradiated mice, 22 and inhibited DNA damage in the kidneys of rats induced by cisplatin treatment. 32 Here, we confirmed that radiation-induced intestinal epithelial cell apoptosis, which was inhibited by GPA and GS. More importantly, we demonstrated that radiation also induced pyroptosis through the Caspase-3/GSDME pathway, while pyroptosis was attenuated by GPA and GS.
In this study, the temporal effects of radiation-induced apoptosis and pyroptosis on IEC-6 cells were shown. Apoptosis and pyroptosis peaked at 4 hours and 12 hours after IR, respectively. Furthermore, we found that at 12 hours after 15 Gy radiation, apoptosis and pyroptosis were mainly induced through the Caspase-3/GSDME pathway. The mitochondrial pathway of cell death is mediated by Bcl-2 family proteins, which are a group of anti- and proapoptotic proteins that regulate the passage of small molecules, and Bax is a member of the Bcl-2 family of apoptotic regulatory proteins. In cells undergoing apoptosis, proapoptotic Bcl-2 family members such as Bax destroy mitochondria and cause the release of other proapoptotic proteins, leading to caspase release and cell death.33,34 Activated Caspase-9 cleaves pro-Caspase-3 and converts it to active Caspase-3, which then forms a positive feedback loop to cleave Caspase-9.35,36 Bax is expressed in cells at the base of intestinal epithelial crypts. 37 In this study, we found that GS and GPA reduced the expression of Bax and the cleavage of Caspase-9, Caspase-3, and PARP in IEC-6 cells after irradiation. These results suggest that the mitochondrial/Caspase-3 pathway is involved in the protective effects of GS and GPA against radiation-induced apoptosis.
IR-induced LDH release and cell swelling suggested the occurrence of pyroptosis or necrosis. In addition, the results showed that the level of cleaved caspase-3 protein was positively correlated with GSDME. These results suggest that caspase-3 may play a key role in IR-induced pyroptosis. Notably, LDH, GSDME, and HMGB1 levels were decreased in IEC-6 cells treated with a caspase-3 inhibitor after the same radiation treatment. These results indicate the involvement of the Caspase-3/GSDME pathway in radiation-induced pyroptosis. Next, we tested whether treatment with GS, GPA, and GPS, which are the three main active components of ginseng, could attenuate radiation-induced pyroptosis. We demonstrated that the number of dead cells decreased after the addition of GS and GPA. Moreover, the release of LDH and the expression of Caspase-3, GSDME, and HMGB1 were decreased after the addition of GS and GPA, and the same results were observed by fluorescence staining. The results showed that GS and GPA could attenuate radiation-induced pyroptosis.
Recent studies have shown that increased ROS and mitochondrial and DNA damage can mediate pyroptosis through the ROS-Caspase-3-GSDME axis.7,12,38 Excessive ROS are produced after radiation, and they act as cellular mediators of radiation-induced intestinal injury.39–42 Antioxidant enzymes, including SOD, scavenge ROS from tissues. However, excess ROS production triggers a state of oxidative stress. A previous study showed that increased ROS induced Caspase-3/GSDME pathway-dependent pyroptosis in lobaplatin-treated colon cancer cells. 43 Another key study showed that GM increased ROS production and induced apoptosis in HepG2 cells. 44 These results indicated that the increased activation of ROS is closely related to the occurrence of pyroptosis and apoptosis in cells. GS and GPA were confirmed in this study to abrogate the generation of ROS and exerted antioxidant effects in vitro. The occurrence of cell cycle arrest and cell death depends on the radiation dose and subsequent DNA repair events. We found that cell DNA damage and cell cycle arrest were alleviated by GS or GPA treatment after radiation. Consistent with previous results, GS and GPA increased cell viability and proliferation. In conclusion, GS and GPA, the main components of ginseng, can alleviate intestinal epithelial cell injury by alleviating GSDME-dependent pyroptosis and Caspase-3 cleavage-induced apoptosis and promoting the proliferation and turnover of intestinal epithelial cells.
However, this study has some limitations. First, the beneficial effects of GS and GPA on intestinal epithelial damage caused by radiation need to be further studied. Future studies will include validating the protective effects of GS and GPA and their mechanisms in different intestinal cell lines. In addition, GSDME expression should be suppressed to demonstrate that the cell death observed in IEC-6 cells is due to GSDME-mediated pyroptosis and not to other types of cell death. Although IR increases ROS production by inducing mitochondrial damage, the mechanism by which GS and GPA alleviate mitochondrial damage has not been fully elucidated.
To further elucidate the underlying mechanisms of the pharmacological effects, isolation and characteristics of the monomeric components of ginseng saponins and phenolic acids will be performed in further research. The Studies should investigate the effects of GS and GPA on the relevant regulatory mechanisms of mitochondrial damage induced by ionizing radiation. Moreover, we hypothesized that the increases in ROS and DNA damage induced by IR induced not only apoptosis but also cell death through the pyroptosis pathway, and pyroptosis in the acute phase after ionizing radiation was mediated by the Bcl-2/Bax–Caspase-9-Caspase-3-GSDME pathway. This hypothesis needs to be verified in the future.
The results of the present study suggested that GS and GPA may be potential candidates for the treatment of radiation enteritis by alleviating the activation of caspase-3/GSDME-induced pyroptosis. These findings provide novel insight into the ROS/caspase-3/GSDME-dependent pyroptosis mechanism in intestinal radiation injury and the potential druggability of GS and GPA.
Materials and Methods
GS, GPA, and GPS Isolation
GS, GPA, and GPS isolated from P. ginseng C.A. Meyer were gently gifted and deposited by Traditional Chinese Medicine Biotechnology Innovation Center in Jilin Province, Beihua University. The isolation methods and identification were described.45–48 In summary, the plant material of P. ginseng C.A. Meyer was collected from Fusong country of Jilin Province, China. The dried ginseng whiskers were extracted with water and precipitated with ethanol. The supernatant was collected to remove ethanol and dried to obtain ginseng extract. Ginseng extract was added to D101 macroporous resin for adsorption, and then eluted with water, 30% ethanol, and 60% ethanol in turn. After removing ethanol, the eluted sample was dried to obtain a series of samples, which were GPS, GPA, and GS.
Cell Lines, Reagents, and Radiation
IEC-6 cell lines (normal rat small intestinal crypt epithelial cells) were obtained from ATCC and tested and identified without murine pathogens. Anti-β-Actin (4970), anti-Cleaved Caspase-3 (#9664), anti-p-H2A.X (#60566), and anti-Caspase-1 (#24232) antibodies were purchased from Cell Signaling Technology. Anti-Bcl-2 (bf9103) antibody was purchased from Affinity Biosciences. Anti-Bax (50599-2-lg), anti-Caspase-9 (66169-1-lg), anti-HMGB1 (10829-1-AP), anti-PARP (66520-1-lg), and anti-Phospho-Histone H3 (66863-1-Ig) antibodies were purchased from Proteintech. Anti-GSDME (ab215191), GSDMD (ab219800) antibodies were purchased from Abcam. The inhibitor Z-DEVD-FMK was purchased from MedChemExpress. The CCK-8 (C0037, BeyoClick™), propidium iodide (PI; ST511); the Beyotime ROS Assay Kit (S0033), Crystal Violet staining solution (C0121), Cell apoptosis detection kit with Annexin V-mCherry and Sytox Green (C1070) were bought from Beyotime Biotechnology. Mounting Medium With DAPI-Aqueous, Fluoroshield (ab104139) was purchased from Abcam. Annexin V-FITC/PI Apoptosis Detection Kit (KGA108), Sytox Green Dead nucleic acid stain (KGA261), and Cell Cycle Detection Kit (KGA512) were bought from Keygen Biotech. Cell-Light™ EdU Apollo In Vitro Kit was bought from RiboBio. The IEC-6 cells were irradiated at 15 Gy using a PXi X-RAD 225XL orthovoltage x-ray generator operating at 225 kV 13.3 mA 30 cm SSD.
Cell Culture and Treatment
Cells were cultured in DMEM (Gibco) medium supplemented with a two-antibiotic combination of 10% FBS (Gibco) and 1% penicillin/streptomycin (VICMED), at 37 °C with 5% CO2. IEC-6 cells were pretreated with 0, 1, 10, 100, 300, 1000 µg/mL Ginseng, GPS, GPA, or GS at 37 ˚C for 24 hours, then with or without 15 Gy radiation, following which different assays were performed. IEC-6 cells were pretreated with or without 10 µg/mL Ginseng, GPS, GPA, or GS for 24 hours before treatment with 15 Gy radiation, following which different assays were performed after 2, 4, 6, 8, 12, and 24 hours.
Cell Viability and Proliferation Assay
CCK-8 assay and EdU staining assay were processed according to the manual books. For colony formation assay, 1000 cells were seeded in 6-well plates. Twenty-four hours treatment of GPS, GPA, or GS was followed by radiation treatment. Then the cells were stained with 1% crystal violet for 10 minutes and counted manually. Details on the CCK-8, EdU staining and Colony formation assay are provided in the Supplemental Methods.
Flow Cytometry
Annexin V-FITC/PI detection and Annexin V-mCherry/Sytox Green detection were processed according to the manual books of the kit. For cell cycle detection, 1 million cells were collected and fixed. PI/RNase A staining solution was added and analyzed by BD FACS Canto II (Becton, Dickinson and Company). Details on flow cytometry are provided in the Supplemental Methods.
Immunofluorescence Microscopy
Prior to immunostaining, the cells were fixed in 4% paraformaldehyde and blocked by 2% BSA. Immunofluorescence staining was observed by an Olympus IX83 fluorescence microscope. Details information can be found in the Supplemental Methods.
Detection of ROS
IEC-6 cells were pretreated with GS and GPA for 24 hours before 15 Gy IR treatment. ROS detection was performed 12 hours after the IR exposure. DCFH-DA was diluted in serum-free medium at a ratio of 1:1000 and the final concentration was 10 µmol/L. After removing the culture medium, 1 mL diluted DCFH-DA was added, and the cells were incubated in a cell incubator at 37 °C for 20 minutes. The cells were then washed three times using serum-free cell culture medium to remove residual DCFH-DA. ROS detection was performed within 30 minutes after DCFH-DA treatment.
Detection of SOD
IEC-6 cells were pretreated with GS and GPA for 24 hours before 15 Gy IR. The SOD detection was performed 12 hours after the IR exposure according to the SOD Detection Kit manufacturer's instruction (A001; Nanjing Jiancheng Bioengineering Institute). Detailed information can be found in the Supplemental Methods.
Western Blot Analysis
Total protein extracts were subjected to BCA protein assay and SDS-PAGE gel electrophoresis, followed by Western blotting. Details information can be found in the Supplemental Methods.
Statistical Analysis
Each experiment was repeated at least three times independently. An unpaired t-test was used for comparison between each two groups. P < 0.05 was considered statistically significant. All analyses were performed using GraphPad Prism 8 (GraphPad Prism).
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231189954 - Supplemental material for Ginsenosides and Ginseng Phenolic Acids Relieve Ionizing Radiation-Induced Pyroptosis and Apoptosis in Intestinal Epithelial Cells
Supplemental material, sj-docx-1-npx-10.1177_1934578X231189954 for Ginsenosides and Ginseng Phenolic Acids Relieve Ionizing Radiation-Induced Pyroptosis and Apoptosis in Intestinal Epithelial Cells by Kai Shi, Fei Chen, Xiang Li, Chen Liang, Haiming Sun, Tao Yang, Junnian Zheng, Bo Ma and Liang Wei in Natural Product Communications
Footnotes
Acknowledgments
We thank members in the Wei Labs for helpful discussions.
Authors’ Note
Kai Shi and Fei Chen contributed equally to this work.
Author Contributions
L.W., B.M., and H.M.S contributed to the conception of the study. C.L., T.Y., and L.W. contributed to the experimental design. K.S., F.C., and X.L. performed the experiments. K.S., F.C., and X.L. performed the data analyses. L.W. helped perform the analysis with constructive discussions and paper modification. K.S. and L.W. wrote the manuscript. L.W., J.N.Z., and B.M. supervised all experiments.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Jiangsu Provincial Key Medical Discipline, the Project of Invigorating Health Care through Science, Technology and Education, the Natural Science Foundation of the Jiangsu Higher Education Institutions of China, (grant numbers ZDXKA2016014, 22KJA320009).
Supplemental Material
Supplemental material for this article is available online.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
