Abstract
Sweroside is a natural iridoid glycoside found abundantly in a diverse range of medicinal plants, many of which are valued for their traditional therapeutic uses. This review offers an in-depth overview of its distribution, pharmacokinetic profile, and pharmacological activities. So far, sweroside has been reported from 46 plant species within 19 genera, belonging to seven plant families—several of which are commonly used in traditional Chinese medicine, where sweroside functions as a key bioactive marker. Pharmacokinetic studies revealed that sweroside is rapidly metabolized in the liver, has a relatively short duration of action, and is primarily eliminated via the kidneys in its metabolite form. A wide range of in vitro and animal studies have shown that sweroside exhibits multiple therapeutic effects, including bone-protective, liver-protective, heart-protective, brain-protective, anti-inflammatory, anti-infective, and dermatological benefits. This review focuses on the latest research exploring its mechanisms of action across various disease-specific signaling pathways. The broad therapeutic range of sweroside further supports its potential as a drug candidate, although improving its pharmacokinetic properties and validating its safety through clinical studies will be critical for future development.
Introduction
Plants are a rich source of natural compounds with extensive structural and functional diversity, making them highly valuable for modern medicinal applications. For decades, they have been the primary source of therapeutic agents across the globe. 1 Plants generate a vast array of low molecular weight organic compounds, including both primary and secondary metabolites, which encompass sugars, nucleic acids, amino acids, and proteins. These substances are typically synthesized in response to various environmental stresses, often originating from primary metabolites via the shikimic acid pathway. 2 While primary metabolites are essential for the plant's growth and development, secondary metabolites play crucial roles in adaptive responses such as pathogen defense, secondary growth, flowering, and enhancing protective mechanisms. 3 These secondary compounds, generally under 3000 Da in molecular mass, are derived from primary metabolism during plant development. The chemical composition of plant metabolites can vary significantly among different species. Notably, the distinction between primary and secondary metabolites is often ambiguous, as the majority of bioactive compounds in plant-derived natural products fall under the category of secondary metabolites. 4 Secondary metabolites can be categorized into various groups based on their chemical structures, including nitrogen-containing compounds, phenylpropanoids, benzenoids, flavonoids, terpenes, and glycosides. These classes of compounds are widely investigated for their therapeutic potential and medicinal benefits.5,6 Among them, glycosides represent a prominent group of secondary metabolites, characterized by the presence of sugar moieties covalently bonded to non-sugar aglycone parts. Due to their diverse bioactivities and structural complexity, glycosides hold substantial commercial importance in both the pharmaceutical and food industries. Numerous studies have highlighted the significant medicinal properties of plant-derived glycosides, recognizing them as powerful bioactive agents. 7
Iridoid and secoiridoid glycosides are commonly found across various plant families and are known for exhibiting a broad spectrum of biological activities. Sweroside (C16H22O9; molecular weight 358.344) is an iridoid glycoside and part of a large class of natural glycosides, distinguished by the attachment of a glucose unit at the C-1 position of its pyran ring (Figure 1). This compound is abundantly present in Swertia species and is associated with numerous pharmacological and biological effects. 8 Sweroside has long been used in traditional Chinese medicine for its effectiveness in reducing internal heat, detoxifying the body, and treating acute gastritis. 9 Contemporary pharmacological research has demonstrated that sweroside possesses a wide range of therapeutic properties, including anti-osteoporotic, hepatoprotective, cardioprotective, antiproliferative, vasorelaxant, anti-infective, and anti-inflammatory activities.10–13 Pharmacokinetic studies have shown that this bioactive compound distributes primarily to highly perfused organs and is capable of crossing the blood-brain barrier, supporting its potential neuroprotective role. 8

Chemical structure of sweroside.
Although sweroside has been the focus of numerous studies, a comprehensive review compiling and evaluating all existing evidence is currently lacking in the literature. Therefore, this study systematically summarizes and critically examines published research on sweroside, covering its plant sources, pharmacokinetics, pharmacological properties, molecular targets, and mechanisms of action at the cellular level. This review aims to offer a thorough understanding of sweroside as a promising therapeutic agent and to serve as a foundation for future research.
Plant Source and Distribution of Sweroside
Sweroside is distributed across a broad range of plant parts, such as the entire plant, aerial parts, leaves, flower tops or buds, fruits, stem bark, flowers, rhizomes, roots, and shoots. Notably, the compound is most commonly extracted from the underground components—particularly rhizomes and roots—followed by the aerial portions and the whole plant (Table 1). So far, 46 plant species (from 19 genera) across seven different families have been identified as natural sources of sweroside. These include 30 species from the Gentianaceae family, 8 from Caprifoliaceae, 3 from Apocynaceae, 2 from Cornaceae, and one species each from Nymphaeaceae, Nyssaceae, and Rubiaceae (Figure 2). A variety of analytical techniques have been employed to detect sweroside, including high-performance liquid chromatography (HPLC), LC-MS/MS, ultra-high-performance liquid chromatography (UHPLC), UPLC-TOF-MS, UPLC-qTOF-MS/MS, LC-ESI-TOF-MS, and nuclear magnetic resonance (NMR).

Occurrence of sweroside in (A) the total 19 genera and (B) 7 families. The number of species from each genus and family is provided in brackets.
Natural Occurrence and Distribution of Sweroside in Plants.

Insight into Plasma Pharmacokinetics, Distribution, Metabolism, and Excretion of Sweroside
A thorough understanding of pharmacokinetics forms the foundation for rational drug development and safe therapeutic application. Although sweroside exhibits diverse pharmacological properties, its clinical translation depends largely on how it is absorbed, distributed, metabolized, and eliminated in vivo. Hence, this section reviews available studies on the pharmacokinetic behavior of sweroside, providing insight into the factors influencing its bioavailability and systemic disposition.
Sweroside rapidly reaches peak plasma concentrations and declines just as quickly, indicating its short-acting nature. Despite its low oral bioavailability, it is widely distributed in highly perfused organs and is capable of crossing the blood-brain barrier. In the body, sweroside undergoes extensive metabolism, and the majority of it is excreted in the form of metabolites, primarily via renal elimination (Figure 3).8,67

Pharmacokinetic characteristics of sweroside.
Luo et al investigated the pharmacokinetic profile of sweroside in Sprague-Dawley rats. Following intravenous administration at a dose of 1 mg/kg and oral administration at 500 mg/kg, the Cmax values were reported as 1.18 ± 0.54 µg/mL and 0.32 ± 0.02 µg/mL, respectively. The corresponding half-lives (T1/2) were 23.27 ± 2.06 min for the intravenous route and 64.34 ± 3.21 min for the oral route. Due to its high-water solubility, sweroside was found to be rapidly excreted via the renal tubules, leading to a swift decline in plasma concentration after intravenous administration. Similarly, after oral dosing, the compound was quickly eliminated from the body. The oral bioavailability of Sweroside at 500 mg/kg was approximately 0.31%, likely due to extensive first-pass metabolism and biotransformation by intestinal microflora. 68 Sheng et al conducted a similar pharmacokinetic study in Sprague-Dawley rats, observing consistent trends with previous findings. Following intragastric (i.g.) administration, plasma concentrations of sweroside rose rapidly, reaching Cmax values of 272.5 ± 45.4 ng/mL at 5 mg/kg, 496.5 ± 81.2 ng/mL at 10 mg/kg, and 712.8 ± 123.5 ng/mL at 15 mg/kg within 60 min. After this peak, concentrations declined sharply over the next 210 min. The average half-life (T1/2) following i.g. dosing was 74.5 ± 12.3 min. In the intravenous (i.v.) group, plasma levels of sweroside dropped rapidly at first and then stabilized around 120 min post-administration, with a shorter T1/2 of 33.9 ± 5.2 min compared to the i.g. route. The study also confirmed that sweroside had a short residence time in vivo for both routes. The absolute oral bioavailability was estimated at 11.90 ± 1.33%, indicating limited systemic absorption. Overall, the pharmacokinetic profile of sweroside suggested that it reaches peak plasma levels quickly but is also eliminated rapidly, pointing to a fast-acting yet short-lived pharmacological effect. 8
Sweroside primarily localizes in organs with a high blood supply. Tissue distribution analysis revealed that sweroside predominantly accumulated in the kidney, liver, spleen, and lungs, with smaller amounts detected in the heart and brain, indicating its capacity to cross the blood-brain barrier. Therefore, tissue perfusion appears to play a critical role in determining its distribution. The significant presence of sweroside in the liver supports previous studies suggesting its hepatoprotective effects. 8
Sweroside undergoes extensive metabolism in rats. In a study conducted by Han et al, a total of 11 metabolites were identified in rat urine following a single oral dose, using UHPLC/Q-TOF-MS. These included four direct metabolites and seven aglycone-related derivatives. The proposed metabolic pathways involved reduction, N-heterocyclization, N-acetylation, and glucuronidation following deglycosylation. 69 Nearly a decade later, Sun et al conducted a follow-up investigation and identified 18 metabolites resulting from the complex biotransformation of sweroside. These included processes such as glycosylation, sulfonation, glucuronidation, and deglycosylation, among others. Additionally, N-heterocyclization metabolites were suggested to be produced via the catalytic action of intestinal microbiota. 67
Sweroside was primarily eliminated in the form of its metabolites. Due to its high-water solubility, it was efficiently excreted through the renal tubules and predominantly eliminated via the urine. A small portion of the unchanged compound was also excreted in bile.8,67 In a separate investigation, a substantial amount of unconjugated sweroside was detected in the rat bile duct 30 min post-administration, suggesting that biliary excretion constitutes a significant elimination pathway for the compound. 68
Pharmacology of Sweroside
Sweroside exhibits a broad spectrum of biological activities, including therapeutic potential in osteoporosis, cardiovascular diseases, inflammatory and liver disorders, cancer, diabetic complications, and infectious diseases. Additionally, it demonstrates antioxidant properties and holds promise in dermatological and cosmetic applications. The following subsections will provide a detailed discussion of these pharmacological effects along with their underlying mechanisms of action.
Protective Effects Against Osteoporosis and Osteoarthritis
Osteoporosis is a skeletal disorder characterized by a disrupted balance between bone resorption and bone formation. 70 Globally, it impacts more than 200 million women, with postmenopausal osteoporosis being the most prevalent form due to estrogen deficiency. The decline in estrogen accelerates bone turnover, favoring bone degradation over formation, ultimately resulting in weakened, fragile bones. 71 Sweroside showed anti-osteoporosis through a multitargeted role by promoting osteoblast differentiation and inhibiting osteoclastogenesis (Figure 4). Sweroside was shown to enhance cell proliferation, alkaline phosphatase (ALP) activity, and osteocalcin secretion in MG-63 cells and rat osteoblasts. It also mitigated dexamethasone-induced apoptosis in cells. 24 In a separate study by Wu et al, although sweroside did not significantly influence cell proliferation in MC3T3-E1 cells, it markedly increased ALP activity and mineralization. The compound also elevated the expression of estrogen receptor-α (ER-α) and G protein-coupled receptor 30 (GPR30), while activating phosphorylated p38 kinase (p-p38). These findings suggest that sweroside may exert its osteogenic effects in MC3T3-E1 cells through interactions with ER-α and GPR30, leading to activation of the p38 signaling pathway. 72 Moreover, sweroside was shown to protect against osteoporosis in both cell and animal models. In bone marrow-derived macrophages, it inhibited RANKL-induced osteoclast formation and bone resorption in a dose- and time-dependent manner. It blocked key signaling events, including IκB-α degradation, p65 activation, and MAPK phosphorylation (JNK, ERK, and p38), thereby preventing osteoclast differentiation. Sweroside also reduced the expression of osteoclast-related proteins (CTSK, c-Fos, NFATc1, integrin β3) and prevented NFATc1 from entering the nucleus. In vivo, it significantly reduced bone loss and alleviated ovariectomy-induced osteoporosis in mice. 73 Sweroside increased both bone mineral content (BMC) and bone mineral density (BMD), as well as significantly reduced bone marrow fat cells in the femur. It also promoted bone matrix production in SaOS-2 cells and enhanced BMP-2 and RUNX2 expression in LV and SaOS-2 cells. Osteogenic markers regulated by BMP-2/RUNX2, including ALP, OPN, and BSP-1, were also elevated. These results indicate that sweroside supports bone formation by activating the BMP-2/RUNX2 signaling pathway. 74 In in vivo studies using an ovariectomized mouse model, sweroside was found to alleviate osteoporosis, as indicated by improved trabecular thickness, bone mineral density (BMD), and trabecular number in the treatment group. Sweroside enhanced the expression of key osteogenic markers, including OCN, RUNX2, and OSX. It also significantly elevated serum P1NP levels and reduced CTX-1 levels, indicating increased bone formation and decreased bone resorption. Additionally, activation of the mTORC1 signaling pathway was observed. In vitro experiments using bone marrow mesenchymal stem cells further confirmed that sweroside activated mTORC1 signaling and upregulated the osteogenic markers RUNX2, OSX, and OCN, supporting the in vivo findings. 75 In addition to its anti-osteoporotic effects, sweroside has shown potential in managing osteoarthritis, a condition where inflammation is a key pathogenic factor. In an in vitro study conducted by Zhang et al using rat chondrocytes, sweroside was found to be non-toxic and significantly inhibited IL-1β-induced production of nitric oxide (NO) and prostaglandin E2 (PGE2), as well as the expression of matrix-degrading enzymes including MMP-1, MMP-3, MMP-13, and ADAMTS-5. Furthermore, sweroside suppressed IL-1β-induced activation of the NF-κB pathway, degradation of IκB, and phosphorylation of S6K1 and S6. These results suggest that its protective effects may involve the inhibition of NF-κB and mTORC1 signaling pathways. 76

The anti-osteoporotic effects of sweroside. Sweroside enhances osteogenic signaling by upregulating BMP-2, which activates RUNX2, leading to increased expression of osteogenic markers, thereby facilitating bone matrix formation. Sweroside also activates the mTORC1 pathway, further elevating RUNX2, OSX, and OCN levels, promoting osteoblast differentiation and mineralization. It also binds to estrogen receptor-α (ER-α) and GPR30, activating p38 MAPK, which supports osteoblastogenesis. In parallel, sweroside exerts anti-osteoclastic effects by inhibiting the RANKL pathway. It reduces IκB-α degradation and blocks NF-κB (p65) activation, suppresses phosphorylation of JNK, ERK, and p38 MAPKs, and inhibits NFATc1 nuclear translocation. These actions collectively downregulate osteoclast-associated genes, including CTSK, c-Fos, and integrin β3.
Cardioprotective Effects
Sweroside showed cardioprotective effects in several in vitro and in vivo cardiotoxicity models (Figure 5). Sweroside demonstrated cardioprotective effects against aconitine-induced cardiac toxicity. In H9c2 cells, it suppressed the upregulation of mRNA expression of NaV1.5, RyR2, and DHPR induced by aconitine, while restoring the reduced expression of SERCA, thereby preventing sustained intracellular Ca²+ accumulation and calcium overload. Sweroside also stabilized mitochondrial membrane potential, which had been disrupted by aconitine, and downregulated autophagy-related gene expressions, including Beclin-1, Caspase-3, and LC3-II. Furthermore, in an in vivo rat model, sweroside (50 mg/kg) effectively alleviated aconitine-induced arrhythmias. 77 In a study by Li et al, sweroside was shown to protect against myocardial ischemia–reperfusion (I/R) injury in vitro. In H9c2 cardiomyoblast cells, pretreatment with sweroside significantly improved cell viability and reduced the release of injury markers CK-MB and LDH, infarct size, and cardiac dysfunction in a dose-dependent manner. Sweroside also suppressed oxidative stress while enhancing the enzymatic activities of superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px). Furthermore, it inhibited pyroptosis by preventing pore formation in the cell membrane, reducing caspase-1 and IL-1β activity, and downregulating NLRP3, ASC, cleaved caspase-1, and IL-1β expression. These cardioprotective effects may be partly mediated through the Nrf2 signaling pathway. 78 Sweroside was also shown to be effective in treating pressure overload-induced heart failure. It exerted cardioprotective effects by suppressing ROS-mediated activation of the NF-κB/NLRP3 inflammasome pathway in Ang II-treated cardiomyocytes through direct binding to CaMKIIδ. In cardiomyocytes from TAC/Ang II-induced mice, sweroside significantly inhibited the CaMKIIδ/NF-κB/NLRP3 signaling cascade. However, when CaMKIIδ was overexpressed using AAV-CaMKIIδ in the myocardium, this inhibitory effect of sweroside was abolished, indicating the central role of CaMKIIδ in mediating sweroside's action. These outcomes helped reduce myocardial remodeling and improve contractile function by the eighth week after the initiation of the inflammatory response. 79 In vitro experiments using human umbilical vein endothelial cells (HUVECs) showed that exposure to H2O2 led to a significant decrease in cell proliferation, FOXO1 expression, and nitric oxide levels, while apoptosis and levels of malondialdehyde, VCAM-1, ICAM-1, and E-selectin were markedly increased. Sweroside was able to ameliorate H2O2-induced endothelial dysfunction, primarily through mechanisms involving FOXO1. In a hypertensive rat model, sweroside dose-dependently improved vascular function. Compared to the sham group, hypertensive rats had higher blood pressure, apoptosis rates, and inflammatory markers, along with reduced FOXO1 expression and nitric oxide levels. Sweroside treatment alleviated these abnormalities, suggesting a beneficial role in managing hypertension-induced vascular injury. 80 Sweroside provided effective protection against ischemia/reperfusion injury by regulating excitation–contraction coupling. In the I/R model, infarct size and LVEDP increased, while LVDP and LVESP declined, with a rising trend in + dp/dtmax and a drop in –dp/dtmax. Sweroside treatment reversed these changes. In NRCMs, it improved decreased cell viability, heart rate, and contraction amplitude, and corrected prolonged contraction and relaxation parameters. Additionally, sweroside significantly upregulated key cardiac genes involved in contractile function, including Cacnb2, Cox6a2, Tnnc1, Tnni3, Tnnt2, Actc1, Myh6, Myl2, and Myl4. 12 Exosomes enriched with sweroside from mesenchymal stem cells reduced sepsis-induced heart damage in rats by controlling oxidative stress and apoptosis. They lowered oxidative injury caused by LPS, as indicated by changes in MDA, SOD, and GSH-Px levels. Sweroside-exosomes also reduced inflammation, protected mitochondria, and improved heart function. They blocked LPS-triggered NF-κB activation and decreased levels of TNF-α, IL-1β, and IL-6, along with inhibiting p65 phosphorylation and nuclear entry. Additionally, they improved survival rates and reversed LPS-related cardiac impairments, including reduced HR, FS%, EF%, and LVPWs, and elevated LVIDs. 81

Cardioprotective mechanisms of sweroside in cardiac injury models. In aconitine-treated H9c2 cells, sweroside downregulates NaV1.5, RyR2, and DHPR mRNA expression, restores SERCA levels, prevents calcium overload, stabilizes mitochondrial membrane potential, and reduces autophagy-related gene expression. In myocardial ischemia-reperfusion (I/R) injury in H9c2 cells, sweroside activates the Nrf2 signaling pathway, enhancing cell viability and reducing oxidative stress, pyroptosis, cardiac dysfunction, infarct size, and release of injury markers. In Angiotensin II (Ang II)-treated cardiomyocytes, sweroside improves cardiac function by inhibiting CaMKIIδ and NF-κB nuclear translocation and suppressing NLRP3 inflammasome assembly and pro-inflammatory mediators. In H2O2-exposed HUVECs, sweroside enhances cell proliferation, FOXO1 expression, and nitric oxide levels, while reducing apoptosis, malondialdehyde content, and expression of inflammatory adhesion molecules. In ischemia/reperfusion injury models, sweroside improves cell viability, heart rate, and contractility, corrects abnormal contraction/relaxation parameters, and upregulates key genes associated with cardiac contractile function.
Hepatoprotective Effects
In multiple experimental models of liver toxicity, both cellular and animal-based, sweroside exhibited significant hepatoprotective effects (Figure 6). Sweroside, at a concentration of 25 µg, demonstrated protective effects against aconitine-induced cytotoxicity (100 nM) in HepG2 cells, suggesting its potential hepatoprotective activity. 82 Boateng et al conducted an in vitro study demonstrating that pre-treatment with sweroside (20 µM) provided hepatoprotective effects against fatty acid-induced toxicity. Cells exposed to arachidonic acid following sweroside treatment exhibited improved viability, as observed in both HepG2 and THLE-2 cell lines. Additionally, HepG2 cells pre-treated with sweroside displayed enhanced basal respiration and ATP production despite arachidonic acid exposure, suggesting that sweroside mitigated mitochondrial dysfunction and restored hepatic ATP synthesis impaired by fatty acid accumulation. Sweroside also showed significant reactive oxygen species (ROS) scavenging activity under these conditions. 11 Moreover, Sweroside exhibited hepatoprotective activity in a D-galactosamine/lipopolysaccharide-induced mouse model of liver injury at doses of 25 and 50 mg/kg, as indicated by lowered serum ALT levels. While both doses reduced ALT levels compared to the disease control group, a statistically significant decrease was observed only at the 25 mg/kg dose. 58 Gong et al demonstrated that sweroside alleviated liver injury and fibrosis induced by carbon tetrachloride (CCl4). Treatment with sweroside significantly lowered elevated serum levels of AST, ALT, and TBA caused by CCl4 exposure. Additionally, sweroside mitigated pathological changes such as inflammatory cell infiltration, central vein wall thickening, and extensive fibrous tissue formation. These findings support the hepatoprotective effects of sweroside. Sweroside treatment in mice resulted in reduced collagen fiber deposition, lower hydroxyproline content, and decreased α-SMA expression, confirming its anti-fibrotic effects. Mechanistically, sweroside activated the FXR–miR-29a signaling pathway, as demonstrated by significantly increased expression of FXR and miR-29a, along with downregulation of fibrotic genes (Col1a1, Col1a2, Col3a1, and Timp1) and reduced TIMP1 protein levels. In LX-2 cells, sweroside markedly upregulated miR-29a while suppressing COL1A1 and TIMP1 expression, and it inhibited hepatic stellate cell (HSC) proliferation. Importantly, these effects were reversed by transfection with a miR-29a inhibitor, and additional experiments confirmed that sweroside-induced miR-29a expression is mediated through FXR activation. Together, these findings establish that the anti-fibrotic action of sweroside is exerted via the FXR–miR-29a axis. 83 In another study, YANG et al demonstrated that sweroside administered orally at a dose of 120 mg/kg/day effectively alleviated α-naphthylisothiocyanate (ANIT)-induced cholestatic liver injury in mice. The treatment significantly reduced serum levels of ALP, TBIL, DBIL, and TBA, and mitigated pathological liver damage, including hepatocellular necrosis and inflammatory infiltration. Additionally, sweroside preserved bile acid balance by lowering the hepatic accumulation of bile acids such as β-MCA, CA, and TCA induced by ANIT. Gene expression analysis suggested that the hepatoprotective effects of sweroside are associated with modulation of genes involved in bile acid metabolism, transport, and inflammatory pathways. 84 In a study by Ding et al, sweroside was shown to counteract hepatic steatosis in mice with NAFLD induced by a high-fat diet. In HFD, it helped reduce insulin resistance, improve lipid profiles, and decrease liver damage markers ALT and AST. The treatment also suppressed the HFD-triggered rise in inflammatory cytokines, TNF-α, and IL-6. In NAFLD mice, sweroside reduced body weight, liver size, and expression of lipogenic genes like FAS and SREBP1c. It promoted fat breakdown by increasing PPARα and CPT1a levels. Sweroside also decreased liver inflammation by lowering TNF-α and IL-6. It lessened liver fat buildup, as seen by reduced lipid droplet accumulation, and activated autophagy through the AMPK/mTOR pathway, contributing to its protective effects. 13 Another study investigated the protective effects of sweroside in a high-fat diet-induced model of non-alcoholic fatty liver disease (NAFLD). Sweroside treatment effectively inhibited HFD-induced weight gain and improved glucose regulation and insulin sensitivity in diet-induced obese (DIO) mice. Additionally, it reduced hepatic steatosis, dyslipidemia, and liver inflammation triggered by the high-fat diet. Sweroside also modulated the expression of genes related to lipid metabolism and inflammatory pathways altered by HFD. 63 Non-alcoholic steatohepatitis (NASH), a progressive form of non-alcoholic fatty liver disease, was addressed in a study where sweroside functioned as an NLRP3 inflammasome inhibitor in a mouse model fed a methionine–choline-deficient diet. Sweroside suppressed NLRP3 activation in primary macrophages, as evidenced by reduced production of IL-1β and caspase-1. Additionally, it significantly lowered serum ALT and AST levels, reduced hepatic immune cell infiltration, decreased liver triglyceride content, and attenuated fibrosis. The compound also inhibited mitochondrial DNA synthesis in liver tissue, thereby further contributing to NLRP3 inflammasome suppression. 85

Hepatoprotective effects of sweroside. In arachidonic acid-induced hepatotoxicity (HepG2 cells), sweroside improves cell viability and enhances mitochondrial function by increasing ATP production and basal respiration, while reducing reactive oxygen species (ROS). In carbon tetrachloride (CCl4)-induced liver injury in mice, sweroside reduces serum levels of AST, ALT, and TBA, and decreases inflammatory cell infiltration, central vein wall thickening, and fibrous tissue development. In a model of D-galactosamine/lipopolysaccharide-induced hepatotoxicity, it significantly lowers serum ALT levels. In α-naphthylisothiocyanate-induced cholestatic liver injury, sweroside attenuates hepatocellular necrosis and inflammatory infiltration, while reducing indicators of liver damage. In mice with non-alcoholic steatohepatitis (NASH), sweroside inhibits NLRP3 inflammasome activation, reduces liver inflammation and fibrosis, decreases AST and ALT levels, and restores mitochondrial DNA synthesis. In a high-fat diet-induced hepatic steatosis model, sweroside improves insulin sensitivity, lowers AST and ALT levels, reduces TNF-α and IL-6 expression, and modulates lipid metabolism via FASN and SREBP1c, leading to reductions in body weight, liver size, and fat accumulation.
Anticancer Effects
Sweroside was found to be effective against glioblastoma, leukemia, and prostate cancer (Figure 7). Sweroside effectively inhibited U251 glioblastoma cell growth (IC50 = 10 µM) while showing low toxicity to normal astrocytes (IC50 = 100 µM). It induced apoptosis by upregulating caspase-3, caspase-9, and Bax, and caused G0/G1 cell cycle arrest. Additionally, sweroside dose-dependently blocked the JNK/p38 MAPK pathway in glioblastoma cells. 86 Sweroside exhibited selective antiproliferative effects on various human leukemia cell lines, including K562 (IC50 = 156.83 µM), U937 (IC50 = 67.92 µM), HL-60 (IC50 = 62.58 µM), NB4 (IC50 = 160.73 µM), and THP-1 (IC50 = 318.60 µM), while showing minimal cytotoxicity to normal human cells. In HL-60 cells, sweroside significantly inhibited colony formation and induced cell cycle arrest in the S and G2/M phases in a dose-dependent manner. It also downregulated Cyclin D1, CDK4, and CDC2, while upregulating p53 and p21. Apoptosis was induced via the caspase-dependent pathway, as shown by increased levels of cleaved caspase-9, caspase-3, and PARP. Cytoplasmic cytochrome c levels were elevated, whereas mitochondrial cytochrome c was reduced following sweroside treatment. Moreover, sweroside decreased Bcl-2 expression and increased Bax protein levels. Apoptosis was also observed in primary h74uman leukemia cells, and sweroside significantly suppressed HL-60 tumor growth in a xenograft mouse model by promoting apoptosis. 87 In PC-3 prostate cancer cells, sweroside-loaded nanoparticles reduced cell proliferation and triggered apoptosis, accompanied by elevated intracellular ROS levels and upregulation of pro-apoptotic proteins. It also inhibited cell invasion and migration, while suppressing cancer stem cell-like properties such as sphere and colony formation, and the expression of stem cell markers CD133 and CD44. Additionally, it inhibited β-catenin transcription by reducing TCF/LEF transcriptional activity in β-catenin-overexpressing cells. This was associated with the downregulation of Wnt/β-catenin downstream target genes, including c-myc, Cyclin D1, Survivin, and MMP-7. 88
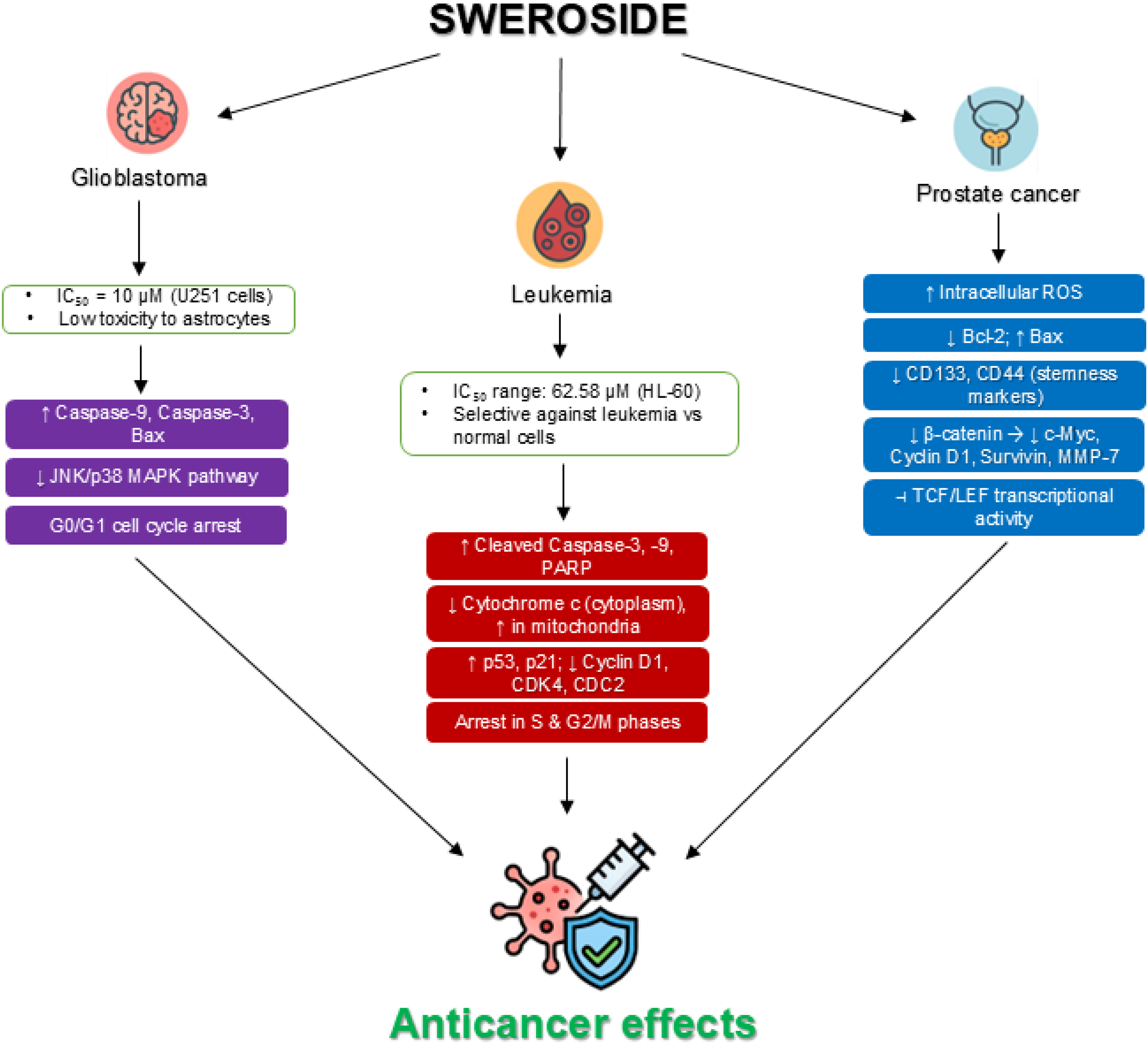
Anticancer properties of sweroside. Sweroside has demonstrated notable anticancer effects across multiple cancer cell lines. In glioblastoma (U251) cells, it promotes apoptosis by enhancing the expression of caspase-3, caspase-9, and Bax, while inducing G0/G1 phase arrest. Additionally, it inhibits the JNK/p38 MAPK signaling pathway in a dose-dependent manner. In leukemia (HL-60) cells, sweroside significantly suppresses colony formation and induces cell cycle arrest in the S and G2/M phases. This is associated with the downregulation of Cyclin D1, CDK4, and CDC2, and the upregulation of tumor suppressors p53 and p21. Apoptosis is mediated through a caspase-dependent mechanism, as indicated by increased levels of cleaved caspase-9, caspase-3, and PARP, along with elevated cytosolic cytochrome c and decreased mitochondrial cytochrome c. Moreover, sweroside lowers the expression of the anti-apoptotic protein Bcl-2 while boosting Bax expression. In PC-3 prostate cancer cells, sweroside-loaded nanoparticles inhibit cell proliferation and trigger apoptosis, characterized by elevated reactive oxygen species (ROS) levels and upregulation of pro-apoptotic markers. The treatment also reduces cell invasion and migration and downregulates cancer stem cell markers CD133 and CD44. Additionally, it disrupts the Wnt/β-catenin signaling pathway by inhibiting TCF/LEF-mediated transcription of β-catenin.
Anti-Inflammatory Effects
Jiang et al evaluated the in vitro anti-inflammatory potential of sweroside at concentrations of 1, 10, and 100 µmol/L. In HEK 293 cells, NF-κB luciferase reporter assay revealed significant inhibition at 10 and 100 µmol/L, whereas the 1 µmol/L dose showed no marked effect. The same concentration range was tested for IL-6 and IL-8 secretion in TNF-α-stimulated BEAS-2B cells. Both 10 and 100 µmol/L doses significantly reduced IL-6 levels, but IL-8 suppression was only observed at 100 µmol/L. Sweroside at 1 µmol/L did not produce significant changes in either cytokine. 44 In LPS-stimulated RAW264.7 macrophages, sweroside treatment significantly reduced PGE2 and ROS levels in a dose-dependent manner using concentrations of 20, 40, and 80 µM. Notably, PGE2 levels declined significantly at 40 and 80 µM, while a significant decrease in ROS was observed only at the highest dose. No evident apoptosis occurred with sweroside treatment, but a dose-dependent increase in the percentage of cells in the G0/G1 phase was noted, indicating cell cycle arrest. Additionally, sweroside upregulated SIRT1 expression and suppressed both total and nuclear NF-κB expression. Consequently, the downstream inflammatory mediators iNOS and COX-2, as well as the cytokines TNF-α, IL-1β, and IL-6, were downregulated, whereas the anti-inflammatory cytokine IL-10 was enhanced. Sweroside also elevated the expression of FOXO1 and its nuclear form (N-FOXO1), along with its downstream targets P27 and MnSOD. Following nicotinamide administration, the FOXO1-regulated pathway was partially reversed, suggesting that sweroside's anti-proliferative effects are mediated via SIRT1–FOXO1 signaling. 89 Ryu et al investigated the in vivo anti-inflammatory activity of sweroside at doses of 0.1 and 1 mg/kg. At both concentrations, sweroside significantly suppressed ear swelling in mouse models induced by croton oil and arachidonic acid. Additionally, these doses demonstrated notable analgesic effects, as shown by a reduced number of writhing responses following acetic acid injection. 45 The anti-inflammatory properties of sweroside were further examined using an LPS-induced acute lung injury mouse model. Sweroside treatment reduced the lung wet-to-dry weight ratio and suppressed myeloperoxidase activity. Histological analysis revealed that sweroside alleviated lung tissue damage and decreased the infiltration of inflammatory cells, including neutrophils and macrophages. Furthermore, it significantly lowered inflammatory cytokine levels, inhibited activation of the NF-κB signaling pathway, and enhanced SIRT1 expression. 9
Antidiabetic Effects
In terms of antidiabetic potential, sweroside demonstrated inhibitory activity against both α-amylase and α-glucosidase, with values of 0.10 mmol ACAE/g and 1.54 mmol ACAE/g, respectively. 10 Moreover, Sweroside markedly downregulated the mRNA expression of Pck1 in HL1C hepatoma cells. Additionally, it led to a modest increase in the phosphorylation levels of Akt at Ser473 and Erk1/2. These changes suggest that sweroside may exert insulin-like activity by modulating key components of the insulin signaling pathway. 65 Sweroside protected mouse podocytes from high glucose-induced damage and apoptosis by activating the Akt/BAD pathway and increasing levels of p-Akt and BAD. In diabetic nephropathy (DN) mice, sweroside reduced albuminuria, glomerular enlargement, and mesangial expansion. It also lowered profibrotic gene expression, preserved nephrin and podocin, and decreased podocyte injury. Additionally, sweroside reduced desmin and HIF1α expression while enhancing nephrin and podocin levels, helping to relieve the pathological features of diabetic nephropathy. 90 A separate investigation by Ma et al examined the renoprotective role of sweroside in an in vitro model of diabetic nephropathy, where HK-2 cells were exposed to high glucose to simulate renal injury. Sweroside treatment improved cell viability, suppressed the release of inflammatory markers such as TNF-α, IL-1β, and VCAM-1, reduced ROS levels, and inhibited epithelial–mesenchymal transition (EMT). These protective effects appear to be linked to SIRT1-mediated deacetylation of NF-κB p65, suggesting a possible mechanism for sweroside's activity under high glucose stress. 91
Neuroprotective Effects
Sweroside exhibited in vitro inhibitory activity against butyrylcholinesterase (BChE), with a value of 6.03 ± 0.44 mg GALAE/g, while showing no inhibitory activity against acetylcholinesterase (AChE). 10 Moreover, Sweroside improved anxiety and memory behaviors in scopolamine-treated zebrafish, as shown in the NTT, Y-maze, and NOR tests. It inhibited AChE activity, supporting enhanced cholinergic function. Additionally, sweroside reduced oxidative stress by lowering protein carbonylation and boosting SOD, CAT, and GPX activity in the zebrafish brain. 54
Antioxidant Effects
Sweroside did not exhibit DPPH radical scavenging activity; however, it showed moderate ABTS scavenging capacity, measured at 0.34 mg TE/g. In antioxidant capacity assays, sweroside demonstrated values of 21.14 mg TE/g in CUPRAC and 12.32 mg TE/g in FRAP. It was also active in the phosphomolybdenum assay, with a total antioxidant capacity of 0.75 mmol TE/g. 10 In a separate investigation, sweroside inhibited fMLP-induced superoxide production in neutrophils in a concentration-dependent manner. 36
Anti-Infective Effects
Sweroside displayed broad-spectrum antimicrobial activity at a minimum inhibitory concentration (MIC) of 1.0 mg mL–¹, effectively inhibiting several Gram-positive bacteria—Bacillus cereus, B. pumilus, B. subtilis, Micrococcus kristinae, and Staphylococcus aureus—as well as Gram-negative species including Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, and Enterobacter cloacae. 53 In addition, sweroside showed notable nematocidal activity against the plant-parasitic nematode Meloidogyne incognita. 15
Dermatoprotective and Cosmetic Effects
Sweroside has demonstrated skin-whitening effects both in cell culture and animal models. It inhibited tyrosinase activity, yielding 55.06 ± 1.85 mg kojic-acid equivalents per gram, 10 and strongly suppressed melanogenesis in melan-a cells at 300 µM without causing cytotoxicity. The compound also reduced the protein levels of tyrosinase, TRP-1, and TRP-2 in these cells, while dose-dependently activating the Akt and ERK pathways. In vivo, sweroside lowered overall pigmentation and tyrosinase activity in zebrafish, confirming its depigmenting potential. 47 In CCD-986Sk human dermal fibroblasts, sweroside reduced UVB-induced reactive oxygen species (ROS) production and enhanced pro-collagen expression. Additionally, it suppressed the expression of matrix metalloproteinase-1 (MMP-1). 51 These findings suggest that, beyond its depigmenting effects, sweroside also exhibits anti-aging properties. Another study further supported sweroside's ability to stimulate collagen production, showing that it enhanced cell morphology and increased collagen granule formation by eightfold in chicken embryonic fibroblasts. 31
A study by Su et al demonstrated that sweroside is effective in treating psoriasis, a chronic autoimmune skin condition. In a mouse model, sweroside alleviated imiquimod-induced psoriasis-like skin lesions, reduced inflammatory responses, and improved skin barrier function. Its anti-inflammatory action was attributed to modulation of the NLRP3/Caspase-1 pathway, leading to decreased IL-1β production, as well as inhibition of NF-κB signaling. In vitro, sweroside also reduced inflammation in HaCaT cells stimulated with TNF-α and IL-17A by suppressing the expression of NLRP3, Caspase-1, and IL-1β. 92
Conclusion
The demand for phytopharmaceuticals has grown significantly over time, supported by their long-standing use and established safety and efficacy. In the face of increasing drug resistance and adverse side effects associated with conventional therapies, the search for new therapeutic agents remains critical to enhance treatment outcomes. Sweroside has attracted considerable attention due to its wide range of documented biological and pharmacological activities, including anti-osteoporotic, hepatoprotective, cardioprotective, anti-inflammatory, antioxidant, anticancer, and cosmetic benefits. Despite this progress, its advancement into clinical applications is still at an early stage. To facilitate this transition, several key steps must be taken. First, comprehensive toxicological evaluations are essential to uncover both overt and subtle side effects. Second, findings from small animal studies need to be validated in larger animal models and eventually in human trials. Third, the issue of sweroside's low bioavailability must be addressed—potential solutions include advanced drug delivery systems or the administration of its aglycone form, which is often the pharmacologically active component. Finally, more comprehensive studies and clinical evaluations are crucial to confirm its exact intracellular mechanisms and therapeutic targets.
To conclude, sweroside shows considerable promise as a safe and multifunctional bioactive molecule for future therapeutic use. Currently, there is a lack of clinical evidence to validate the use of sweroside in human subjects. Consequently, future research should prioritize investigating its molecular mechanisms, pharmacokinetic behavior, and tissue distribution within the human body to facilitate the development of a more effective therapeutic agent with minimal side effects.
Footnotes
Acknowledgements
None.
ORCID iDs
Author Contribution Statement
Md. Liakot Ali: Conceptualization, Methodology, Writing – original draft, Writing – review & editing. Sajeda Akter: Writing – original draft. Sadia Hosna Rony: Writing – original draft. Md. Jahirul Islam Mamun: Writing – original draft. Md. Hossain Rasel: Writing – original draft. Mohammed Kamrul Hossain: Writing – original draft. Nawreen Monir Proma: Supervision, Project administration, Writing – review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data that support the findings of this study are available on the manuscript.
