Abstract
Abutilon theophrasti Medic.((A. theophrasti, Malvaceae) has been recognized for its ethnopharmacological values for a long time. Various studies of A. theophrasti have demonstrated that different extracts from this species exhibit a wide range of pharmacological activities, including antioxidant, antitumor, antibacterial, and anti-inflammatory analgesia activities. Until now, more than 100 kinds of phytochemical compounds have been isolated and identified in various parts of A. theophrasti including flavonoids, fatty acid, essential oil, and others. In the present review, we have been trying to summarize an up-to-date research on the traditional uses, phytochemistry, pharmacology, and pharmacokinetics of A. theophrasti. Consequently, a significant and broad overview on the current knowledge of A. theophrasti is provided to explore its clinical therapeutic potential.
Introduction
Abutilon theophrasti Medic. (A. theophrasti), widely distributed in China, India, Japan, Europe, North America and other regions, belongs to Malvaceae. According to the records in the Chinese Materia, the roots, seeds, and leaves of A. theophrasti can be used for medicinal purposes. 1 Modern pharmacological research shows that A. theophrasti may contribute to prevent and/or alleviate certain types of cancers and chronic diseases, such as diabetes, asthma, ulcer, and lung injury2–4 As a traditional Chinese medicine (TCM), A. theophrasti was widely used in many prescriptions, such as Niaoshitong Pills and Paishi Granules5,6 Phytochemical research revealed that more than 100 compounds have been separated and identified from A. theophrasti, containing flavonoids, fatty acid, essential oil, coumarins, alkaloids, and others.7,8
So far, no comprehensive review of A. theophrasti has been published. With the Chinese and English words of “Abutilon theophrasti Medic.,” “active ingredient,” “pharmacology,” “pharmacokinetics,” and “identified” as keywords, we combined and searched the related information in Web of Science, Google Scholar, China National Knowledge Internet, PubMed, and Elsevier from 2000 to 2023. The present work aims to summarize an updated and systematical assessment of traditional uses, phytochemistry, pharmacology, and pharmacokinetics of A. theophrasti to provide a better understanding about the health benefits of A. theophrasti.
Traditional Uses and Botanical Description
The cultivation and utilization of A. theophrasti has a long history in China. It was first recorded in The Book of Songs and Rites of the Zhou. It was used as raw material for clothing, with a history of more than 2600 years. According to The Dictionary of Chinese Herbal Medicine, there are two species of Abutilon Miller plants that are used for medicinal purposes: Abutilon theophrasti Medic. and Abutilon indicum (Linn.) Sweet.9,10 According to The Dictionary of Chinese Herbal Medicine, the roots, seeds, and leaves of A. theophrasti can be used for medicinal purposes. Whole grass tastes bitter with neutral in nature. It can detoxify and dispel wind-cold, treating dysentery, otitis media, tinnitus, deafness, joint soreness, and so on. Carbuncle gangrene can be treated with fresh hemp leaves and honey at a dose of 15-50 g. If it is swollen and headless, take fresh leaves and brown sugar to pound it externally, take a seed fruit inside, and take it twice a day. A. theophrasti roots can detoxify, inducing diuresis for treating stranguria, mainly treating dribbling urination; A. theophrasti roots should be collected after the beginning of winter, the stems and leaves should be removed, washed, and dried for later use; fried soup should be taken in the soup, and the dose is generally 30-60 g. 9 A. theophrasti seeds were originally published in Tang Bencao. TCM believes that A. theophrasti seeds have the effect of clearing heat and dampness, detoxification, and removing nebula for improving eyesight. It treats erythral and leukal dysentery, gonorrhea, astringent pain, carbuncle swelling, and opacity of the cornea, scrofula. “Tang Bencao” records: “The main red, white, cold and hot dysentery, drink it loosely.”11–13 In addition, according to Fujian Folk Herbal Medicine, A. theophrasti leaves have the effect of treating carbuncle and deep-rooted carbuncle. 9 According to Xinjiang Chinese Herbal Medicine, the seeds of A. theophrasti are sweet, nontoxic, the roots of A. theophrasti are bitter and neutral in nature. Both of them have the effect of anti-inflammatory, diuresis, and lactagogue. 14
In China, the suitable population for A. theophrasti is dysentery, carbuncle, and gonorrhea. However, the taste of A. theophrasti is bitter, which is easy to cut the stomach and hurt the saliva. Therefore, people with weak spleen and stomach should use it carefully to avoid aggravating the original disease. At present, the common proprietary Chinese medicines containing A. theophrasti include Zhenbao pills, tonic fitness pills, Dangshi tablets, Paishi granules, and Niaoshitong Pills. For example, Niaoshitong Pills are clinically used in patients with qi stagnation and damp obstruction of urinary tract stones and shock wave lithotripsy, the prescription contains A. theophrasti seeds, which mainly play a role in clearing heat and promoting dampness. 5 Paishi granules can effectively promote the excretion of stones, reduce the irritation of stones to the ureter and kidneys, and relieve clinical manifestations such as pain in the waist and abdomen, hematuria, and frequent urination. A. theophrasti seeds in the prescription play the role of clearing away heat, promoting dampness, detoxifying, and eliminating carbuncles in the prescription, which is conducive to alleviating abdominal pain caused by stones. 6
A. theophrasti is an annual subshrubby herbs, with 1-2 meters high. The stem and branches are covered with pubescence. Leaves are alternate and cordate, 5-10 cm long, apex becomes tapered, base appears cordate, edge of the leaves are serrated, both of the surfaces are covered with densely stellate tomentum; the petioles are 3-12 cm long, covered with stellate fine tomentum; stipules are caducous. Flowers grow solitarily in the axillay. Peduncles are 1-13 cm long, with pubescence all over and buds at the tips. Calyx is cup-shaped and puberulent, with five lobes, ovate shape and 6 mm long. Flowers are yellow. Petals present an obovate shape and 1 cm long. Stamens are smooth, with 15-20 carpels, 1-1.5 cm long, flat at the top, with spreading 2hairy awns, arranged in a wheel shape, and covered with densely puberulent. Capsule is hemispherical, ca. 2 cm in diameter, ca. 1.2 cm long, schizocarps H15-20, with shag-hairs and two long awns at the top. Seeds are kidney-shaped, brown and covered with stellate pubescence. The flowering phase of A. theophrasti was from July to August. The morphological characteristics of A. theophrasti is displayed in Figure 1.
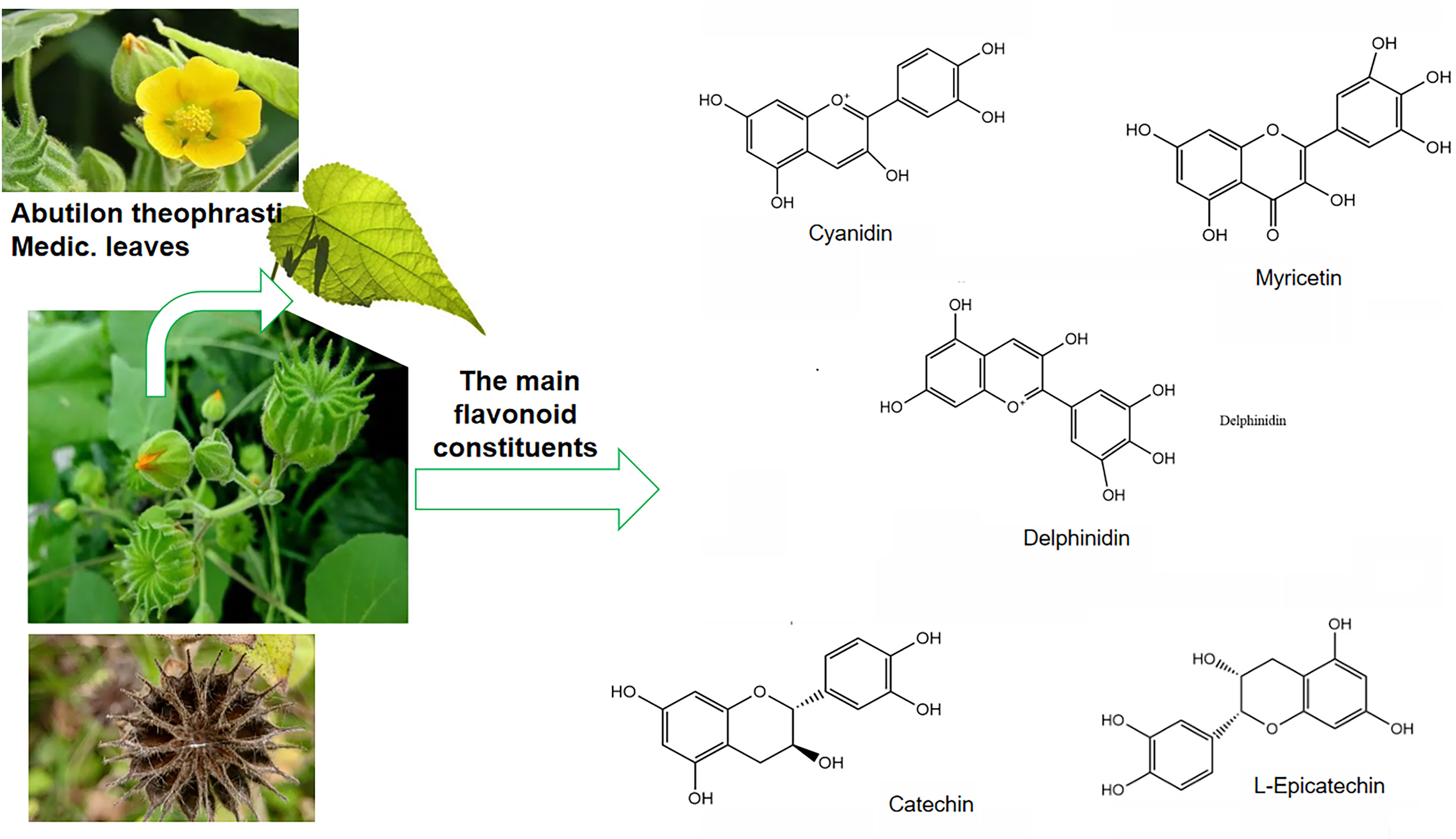
The leaves of A. theophrasti and its five main flavonoid constituents.
Chemical Compositions
Until now, scholars all over the world mainly isolated and identified more than 100 chemical components from A. theophrasti extract, including flavonoids, fatty acid, and essential oil. Of these compounds, flavonoids, sesquiterpenes, diterpenes, and tri-terpenoids and their glycosides are the major active components.
Flavonoids
Preliminary studies showed that A. theophrasti roots, stems, flowers, leaves, seeds, exocarps, and episperm are rich in flavonoids, which are the most widely distributed class of compounds in Chinese herbal medicine.15,16 A total of 29 flavonoids were isolated and identified from A. theophrasti.7,8,15–18 Figure 1 presents the leaves of A. theophrasti and its five main bioactive constituents. Table 1 lists the flavonoids in A. theophrasti. Their structures are shown in the Figures 2-4.
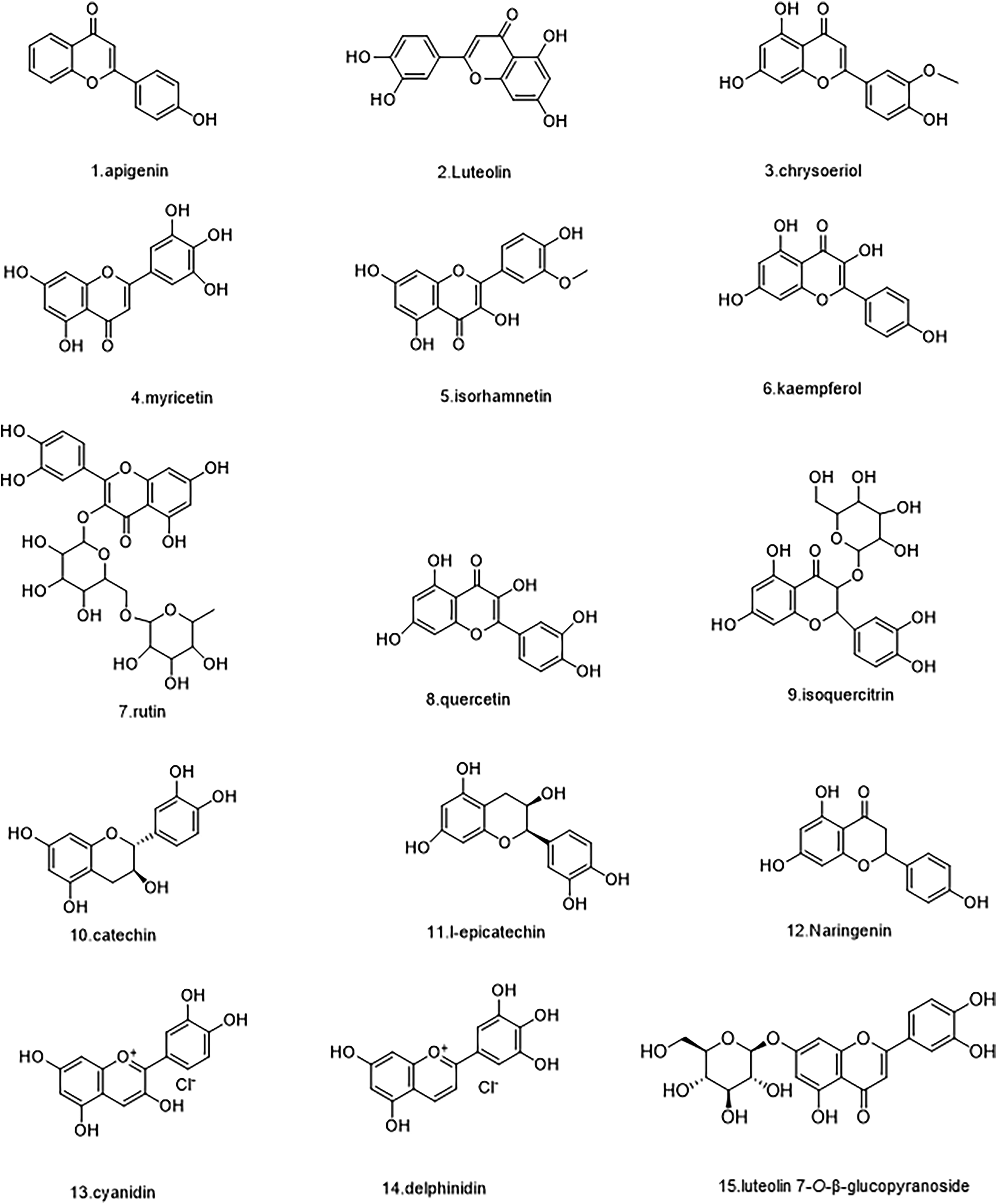
Structures of flavonoids compounds 1-15.

Structures of flavonoids compounds 16-23.

Structures of flavonoids compounds 24-29.
The Flavonoids in A. theophrasti.

Fatty Acid
Organic acids are class of chemical compounds containing carboxyl group which have antiviral and antibacterial effects. 7 Fatty acid are found in abundance in the seeds of A. theophrasti. According to reports, more than 27 fatty acids have been isolated from A. theophrasti, including linoleic acid, palmitic acid, and stearic acid.7,8,19,20 Table 2 lists the fatty acid in A. theophrasti. Their structures are shown in the Figures 5 and 6.

Structures of fatty acid compounds 30-43.
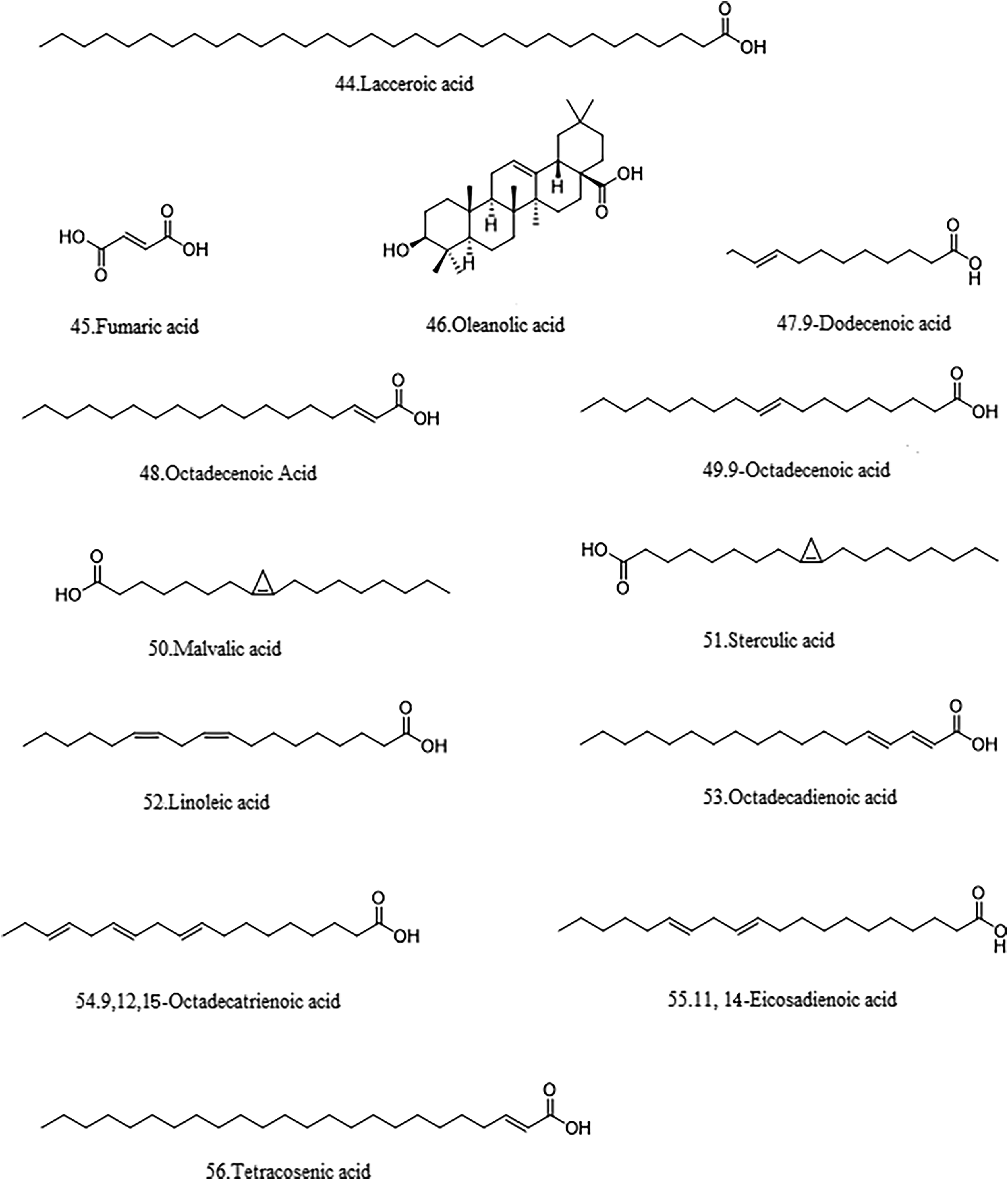
Structures of fatty acid compounds 44-56.
The Fatty Acid in A. theophrasti.

Essential Oil
Chen et al analyzed the chemical components of the essential oil from the A. theophrasti by gas chromatography-mass spectrometry (GC-MS) method. 21 A total of 45 compounds were identified from the volatile oils of A. theophrasti by GC-MS. Although the content and composition of volatile oil in A. theophrasti vary from production place to production place, the main representative chemical components of volatile oil in A. theophrasti are consistent, including phytol, palmitic acid, 2-pentadecanone-6, 10, 14-trimethyl, oleamide, lignocerane, and n-pentacosane. 22
Coumarins, Alkaloids, and Others
Apart from the above-described compounds (Tables 1 and 2), some chemical components also have been isolated from A. theophrasti. Kuo et al reported that A. theophrasti contains a variety of coumarins, such as methylcoumarate, trans-p-coumaric acid, and scopoletin. 15 Kuo et al also revealed that A. theophrasti possesses alkaloids, such as 1-lycoperodine and 1-methoxycarbonyl-β-carboline. 23
Pharmacological
Until now, scholars all over the world research and discovery pharmacological effects from A. theophrasti mainly focus on antibacterial activity, anti-inflammatory analgesia effect, antioxidant activity, and antitumor effect. The pharmacological activities of A. theophrasti are summarized in Figure 7.

The pharmacological activities of A. theophrasti.
Antibacterial Activity
At present, for bacterial infection, we generally choose relatively sensitive antibacterial drugs for treatment. The commonly used ones are roughly divided into β-β-lactams, aminoglycosides, macrolides, and fluoroquinolones, but at present, the resistance of bacteria to these antibacterial drugs has become the main driving force for the development of new antibacterial drugs. A wide variety of natural plants or drugs have attracted much attention because of their small toxic side effects and clear efficacy.24,25 Some researchers believe that most natural drugs have the effect of clearing heat, detoxification, anti-pathogenic microorganisms, and anti-endotoxin effects based on their main active ingredients. 26 A. theophrasti, considered to be a medicinal plant with antibacterial effect, has the effect of clearing heat and detoxification. Studies have shown that A. theophrasti root, stems, leaves, and flowers have a certain antibacterial effect (Table 3). Zhao W et al studied the bacteriostatic effect of ethanol extract ((1 g of raw medicinal material/mL) and water extract (1 g of raw medicinal material/mL) of root, stem, and leaf of A theophrastis by using in vitro bacteriostatic experiment (paper strip method), and the liquid medicine was diluted to five concentrations, respectively. The results of the in vitro experiment showed that each liquid had a certain antibacterial effect. In the antibacterial experiment in vivo, mice infected with different concentrations of Staphylococcus aureus and Escherichia coli and were intraperitoneally injected with various samples, in order to observe the minimum lethal dose of mice and the protective effect of A theophrastis on the dead-causing bacteria in mice infected. The results showed that there was a certain protective effect on the infecting lethal dose mice of Staphylococcus aureus and Escherichia coli (P < .05). They found that the water extract and ethanol extract of A. theophrasti stem and leaves could effectively inhibit Staphylococcus aureus and Escherichia coli. 27
Antibacterial Effect of Different Parts of A. theophrasti.

The ingredients of the antibacterial activity of A. theophrasti may be flavonoids. Kourbaniza and others diluted the flavonoids extract of A. theophrasti leaves, n-butanol extract (100 mg/mL), ethyl acetate extract (40 mg/mL), ether extract (18.7 mg/mL), and ethanol extract (0.5 mg/mL) by four and 10 times, respectively. Each sample solution was added to 0.04 mL of the medium containing each bacteria and cultured at 37 °C for 24 h to observe the bacterial growth. The results showed that each extract could effectively inhibit Escherichia coli, Bacillus thuringiensis, Staphylococcus aureus, and Bacillus subtilis. In addition, the sample solution was added to different fungal media at 28 °C for 48-72 h by the same method. As a result, it was found that each extraction solution can effectively inhibit Escherichia coli, Staphylococcus aureus, Bacillus subtilis, and Bacillus thuringiensis. In the same way, the samples were added to different fungal media and cultured at 28 °C for 48-72 h. And it was found that each extract also had a certain inhibitory effect on some fungi, including yeast, Aspergillus niger, and peninicillus. 28 Tian C et al found that Escherichia coli, Salmonella typhimurium, Staphylococcus aureus, and Streptococcus pneumoniae were inoculated onto MacConkey agar medium, SS agar medium, hypersalt mannitol medium, and improved Edward medium, respectively. Total flavonoid extract (TFE) was prepared with optimal extraction technology and was dissolved in sterile water to obtain an initial stock solution concentration of 2.0 g of raw medicinal material/mL. Ten serial twofold dilutions of the stock solution were prepared to produce a final concentration of 1.0 to 0.002 g of raw medicinal material/mL. The stock solution concentration of gentamicin was 1.28 mg/mL, and the final concentration ranged from 0.25 to 128 μg/mL. Antibacterial activity was screened by the microwell dilution method as described with modifications, and the MIC values, defined as the lowest concentration of samples showing clear wells or with complete inhibition of bacteria, were chosen as the primary evaluation indicators. The mediums were covered and incubated at 37 °C for 24 h. As shown in the antibacterial activities of TFE from A. theophrasti leaves, the order of the inhibitory effect of TFE on the four bacteria was as follows: first, Staphylococcus aureus, with an MIC value of 0.06 ± 0.01 g crude drug/mL; second, Streptococcus, with an MIC value of 0.26 ± 0.01 g crude drug/mL; third, Salmonella, with an MIC value of 0.51 ± 0.02 g crude drug/mL; and finally, Escherichia coli, with an MIC value of 1.02 ± 0.04 g crude drug/mL. These results suggested that the inhibitory effect of TFE on the four bacteria is greater than that of gentamicin. 30 Shi KM et al isolated and purified the nonvolatile components of A. theophrasti stems and leaves through silica gel column chromatography and preparation of liquid phase separation and identified three lactone compounds: methyl caffeate, sopratine and methyl ferulate through infrared, nuclear magnetic and high-resolution mass spectrometry. The volatile oil components and these three nonvolatile lactone components in the A. theophrasti were imported. In the bacteriostatic study of volatile oil components and these three nonvolatile lactone components in A. theophrasti is carried out, and set the solution concentration, respectively: 50, 25, 12.5, 6.25, 3.125, 1.563, 0.781, 0.391, 0.195, 0.098, and 0.049 mg/mL concentration of 11, AGAR diffusion method was used to determine the bacteriostatic effect on Escherichia coli, Bacillus subtilis, and Agrobacterium. The positive control uses 80µl amount of kanamycin with a concentration of 10μg/mL. After administration, the culture medium is cultured in a 37 °C incubator for 24-48 h. The diameter of the minimum bacteriostatic circle is determined by cross-sectional method, and the MTC value of the minimum antibacterial concentration is recorded at the same time. The results show that the three kinds of lactone compounds isolated in the A. theophrasti have a certain inhibitory effect on the test-supplying bacteria. Among them, they all have the strongest inhibitory effect on agrobacteria at a concentration as low as 0.195 mg/mL. Among them, methyl ferulic acid has shown obvious inhibition on all three test-suppressive bacteria. Secondly, it has a strong inhibitory effect on Escherichia coli and Bacillus subtella. The minimum bacteriostatic concentration MIC values are methyl caffeinate (1.563 mg/mL, 3.125 mg/mL, 0.391 mg/mL) and methyl ferulinate (0.781 mg/mL, 0.781 mg/mL, 0.195 mg/mL), sodamine (1.563 mg/mL, 0.781 mg/mL, 0.195 mg/mL). 29 Therefore, the flavonoids and lactones, as promising and potential antibacterial agents, have strong antibacterial activity in this plant.
Anti-Inflammatory Analgesia Effect
Inflammation is closely related to chronic diseases caused by most lifestyle factors. 31 In recent years, many active ingredients of natural drugs and their pharmacological effects have increasingly become a hot topic in research. Therefore, the search for anti-inflammatory drugs from many Chinese herbs with small toxic and side effects has attracted more and more attention.32–34 The folk use of A. theophrasti whole grass decoction is very effective in treating rheumatoid arthritis. A. theophrasti plants have a certain degree of anti-inflammatory and analgesic effect. Research further shows that A. theophrasti plants have good anti-inflammatory and analgesic effect, and prelimely clarifies the chemical components of effective anti-inflammatory and analgesic parts in stems and leaves of A. theophrasti including flavonoids, phenols, coumarins, and steroids.28,35 It was reported that flavonoids such as quincetin were effective for acute inflammation.36,37 Yang CX et al cultured mouse RAW 264 in vitro. Inflammation was induced by LPS. Inflammatory cells were treated with different concentrations of TFE from A. theophrasti leaves (50,100,200 μg/mL). The contents of interleukin 6 (IL-6), interleukin 10 (IL-10), interleukin 1β (IL-1β), and tumor necrosis factor-alpha (TNF-α) in the supernatant of inflammatory cells were measured by ELISA to evaluate the effect of TFE from A. theophrasti leaves on cytokines. Griess method and double antibody sandwich method were used to determine the content of nitric oxide (NO) and cyclooxygenase-2 (COX-2) in the supernatant of cells, and fluorescence quantitative PCR was used to detect the expression of COX-2 and inducible nitric oxide synthase (iNOS) genes to evaluate the effect of TFE from A. theophrasti leaves on COX-2 and iNOS. The results showed that 50, 100, and 200 μg/mL of TFE from A. theophrasti leaves could significantly reduce the content of RAW264.7. The contents of IL-6, IL-1β, and TNF-α in the supernatant of inflammatory cells, and the content of IL-10 was significantly increased by 200 μg/mL of the solution. The concentration of NO and COX-2 and the relative expression of COX-2 and iNOS mRNA were significantly decreased in a dose-dependent manner after treatment with 50, 100, and 200 μg/mL of TFE from A. theophrasti leaves. Based on previous studies, PCR and Western blot methods were used to investigate the regulation of TFE from A. theophrasti leaves on two important signaling pathways nuclear factor kappa-B (NF-κB) and mitogen-activated protein kinase (MAPK) at the gene and protein levels, respectively. TFE from A. theophrasti leaves could block the phosphorylation of IκB and p65NF-κB. The results showed that the total flavonoid content (TFC) could inhibit the activation of NF-κB signal transduction pathway induced by LPS by stabilizing IκB, thus achieving anti-inflammatory effect. The results showed that TFE from A. theophrasti leaves significantly inhibited the phosphorylation levels of ERK1/2, INK, and p38α, which indicated that TFE could inhibit the expression of inflammatory cytokines TNF-α, IL-6, and IL-1β by blocking the activation of MAPK signaling pathway. 38 In addition, Zhang Peng et al studied anti-inflammatory and analgesic factors of TFE from A. theophrasti leaves (250, 500, 1000 mg/kg·b·w) by establishing a model of acute lung injury (ALI) in mice. It was found that it had a certain regulatory effect on the influences of inflammatory factors (TNF-α, IL-1β, IL-6, and IL-10), inflammatory mediators (NO, pcolce2 (PCE2)) and oxidase content (malondialdehyde (MDA), superoxide dismutase (SOD)). The results showed that the contents of TNF-α, IL-1β, IL-6, NO, and PCE2 were significantly decreased in the low-, middle-, and high-dose groups (P < .01), and the contents of IL-10 were significantly increased in the middle- and high-dose groups (P < .01). It was also found that the expression of iNOS and COX-2 genes can be significantly reduced, which proved the anti-inflammatory effect of TFE from A. theophrasti leaves. 39 Tian C et al studied in vitro the activation and regulation of NF-κB pathway and MAPK pathway by TFE from A. theophrasti stems and leaves (50, 100, 200 μg/mL) after RAW264.7 cells were infected by LPS. Briefly, LPS, as an inducer, can activate macrophages by stimulating toll-like receptor 4 (TLR4) and triggering NF-κB and MAPK selectively. MyD88 and other small molecule proteins were enriched in TLRs after LPS treatment. In addition, TNF receptor-associated factor 6, a ubiquitin ligase, will produce a series of related enzymes through ubiquitination reactions. In the NF-κB signaling pathways, IκB kinase can regulate the expression of the downstream genes by degrading IκB, translocating the p65-p50 dimer into the nucleus and increasing DNA binding sites. TFE plays an important role in anti-inflammatory activities and not only increases IκB gene expression effectively and increases the p65-p50 dimer but also reduces the expression of the p65 gene. In the MAPK signaling pathways, TFE clearly displayed anti-inflammation activity via inhibition of phosphorylation of ERK1/2, JNK, and p38MAPK and prevention of their degradation into small molecular fragments and of combination with the DNA-targeted genes. 30 Tian C et al also found that its anti-inflammatory mechanism was activated by inhibiting the activation of NF-κB pathway and MAPK pathway through establishing a model of ALI in mice. The present study provides evidence that TFE from A. theophrasti leaves (0.25, 0.5 and 1.0 g/kg) displayed increased therapeutic effects against LPS-induced ALI by inhibiting the inflammatory response directly in ALI mice. The mechanisms of action were also revealed, including the following: reduction in MDA activity and increase in SOD activity; downregulation of the secretion of pro-inflammatory cytokines, such as IL-1β, IL-6, and TNF-α; upregulation of the secretion of anti-inflammatory cytokine IL10; reduction in the production of pro-inflammatory mediators NO and PGE2; diminution of the protein expression of pro-inflammatory mediators iNOS and COX-2; and suppression of NF-κB and MAPK signaling pathways by decreasing the protein phosphorylation of p65, IκB, ERK1/2, p38 MAPK, and JNK, 4 (Possible mechanisms of A. theophrasti are shown Figure 8).

The anti-inflammatory analgesia effect and possible mechanisms of A. theophrasti.
Antioxidant Activity
Oxidative stress, a state of oxidative imbalance in the body, is considered to be an important factor leading to aging and disease. At present, many components of plant medicines have certain antioxidant activity. According to current research, some chemical components in A. theophrasti have antioxidant activity. Tian CL et al found that the antioxidant activity of petroleum ether extract and ethyl acetate extract of A. theophrasti leaves is better 40 ; Zhao WY et al studied the extracts of roots, stems, and leaves of A. theophrasti, extracted total phenolic acids by Folin phenol colorimetric method, extracted total flavonoids by sodium nitrite-aluminum nitrate method, determined total antioxidant capacity by FRAP method, and detected 1,1-Diphenyl-2-picrylhydrazyl (DPPH) and 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) free radical scavenging capacity by microplate reader. The results showed that at the concentration of 1.5 mg/mL, the total antioxidant capacity (FRAP value) was in the order of leaf extract (487. 36 μmol/L), root extract (280. 23 μmol/L), and stem extract (172.61 μmol/L). The EC50 values of DPPH and ABTS radical scavenging activities of extracts from different parts of A. theophrasti were separately calculated as follows: extract from leaves of A. theophrasti (EC50 = 0.537 mg/mL) > extract from roots of A. theophrasti (EC50 = 0.907 mg/mL) > extract from stems of A. theophrasti (EC50 = 1.580 mg/mL), extract from leaves of A. theophrasti (EC50 = 0. 779 mg/mL) > extract from roots of A. theophrasti (EC50 = 1. 273 mg/mL) > extract from stems of A. theophrasti (EC50 = 1.927 mg/mL). The total phenolic content (TPC) was positively correlated with TFC, DPPH, and ABTS free radical scavenging activity and total antioxidant activity, and the correlation between TPC and several antioxidant indexes was higher than that between TFC and several antioxidant indexes, indicating that the antioxidant activity of A. theophrasti extracts was more related to total phenolic extract (TPE). 1
The main active ingredients in many Chinese herbs—flavonoids are excellent reactive oxygen scavengers and lipid antioxidants. Tian C et al studied the antioxidant effect of total flavonoids in A. theophrasti. In their research, the antioxidant activities of TFE were investigated using assays of ABTS radical scavenging activity, DPPH radical scavenging activity, and FRAP. To further understand the interrelationships between the antioxidant activity and TFC, the Pearson test was used to analyze the interaction between the two factors. The correlations were significant (P ≤ .05) between TFC and the antioxidant activity measured by the ABTS assay (r, − 1.000) and between TFC and the results of the DPPH assay (r, −0.999). A significant correlation (P ≤ .05) was found between the DPPH and ABTS assays (r, 1.000). Moreover, better correlations were found between FRAP and ABTS and FRAP and DPPH, with r values of −0.920 and −0.930, respectively. Furthermore, there was a better correlation (r, 0.912) between TFC and the values of the FRAP assay, although the correlation was not significant (P > 0 05). These results indicated that TFC contributes to the antioxidant activity and that the correlation is higher between the antioxidant activity and TFC. 30 In addition, Zhao CJ et al separated and enriched the main flavonoids (quercetin, kaempferol, isorhamnetin) from three kinds of A. theophrasti leaves by six different types of macroporous adsorption resins, HP20, AB-8, HPD400, X-5, D101, and NKA-9. The concentrations were set at 0.125, 0.25, 0.5, 1.0, 2.0, 4.0, and 8.0 mg/mL. The same method, including ABTS free radical scavenging assay, FRAP assay and DPPH free radical scavenging assay, was used to study the antioxidant activity in vitro. The TEAC value of ABTS experiment was 0.437 mmolTrolox/g; the FRAP value of FRAP experiment was 1.025 mmolFeSO4/g, the content of total flavonol glycosides was 26.2 mg/g. In the DPPH free radical scavenging experiment, the maximum scavenging rate of the sample was 83.6%, the IC50 value was 0.90 mg/mL, and the DPPH free radical scavenging ability was close to that of Vc. The results showed that the three flavonoids in the leaves of A. theophrasti had good antioxidant capacity. 8
Antitumor Effect
In recent years, the great development of TCM in the treatment of various cancers has attracted widespread attention from scholars at home and abroad, and there is an urgent need to find safe and effective antitumor drugs from TCM resources. 41 It is said in some folk that A. theophrasti has anti-tumor effect. At present, some scholars mentioned that syriacusin A in A. theophrasti root has cytotoxic activity on several human cancer cells such as melanoma, breast, lung, kidney, prostate, colon, and central nervous system, but their cytotoxicity is lower than that of doxorubicin, and there is no specific experimental report data in the document literature to support it. 42 At present, there is no experimental study on the antitumor effect of other chemical constituents of A. theophrasti, so more pharmacological experiments on the antitumor activity of A. theophrasti should be carried out in the future investigation.
Pharmacokinetics
Syriacusin A, as the main components of the A. theophrasti root, exhibits various pharmacological activities including inhibition of lipid peroxidation. 43 Syriacusin A also showed cytotoxicity against some human cancer cell lines with effective dose 50 (ED50) of 1.5-2.4 µg/mL. Chen X et al developed a simple and specific high-performance liquid chromatography method for the determination of Syriacusin A in rat plasma after intravenous administration of Syriacusin A at dose of 20 mg/kg.41,44 The pharmacokinetic parameters of Syriacusin A were t1/2 0. 884 h; ke 1. 412 h−1 and area under curve (AUC) (0−t) 1360.347 mg/L·h. 44
Conclusions and Future Perspectives
In summary, the review comprehensively summarizes the traditional uses, phytochemistry, pharmacological activity, and pharmacokinetics of Abutilon theophrasti Medic.(A. theophrasti) for the first of time. In terms of chemistry, many kinds of compounds including flavonoids, fatty acid, essential oil, coumarins, alkaloids, and others were isolated and identified. Among these constituents, rutine, hyperin, and quercetin are the most representative and dominant chemical constituents in A. theophrasti. 45 Therefore, future studies should also focus on quality standards and quality control of A. theophrasti, which is particularly important for stability of quality and clinical effects. Based on the phytochemistry and pharmacological activities, the active components in A. theophrasti, such as rutine, hyperin, and quercetin could be the potential quality control markers for the standardization of A. theophrasti, which should be further determined.
Moreover, a large number of studies indicated that A. theophrasti exhibit extensive bioactivities, including antibacterial, anti-inflammatory analgesia, antioxidant as well as the antitumor activities, etc. Researchers have investigated the various bioactivities of A. theophrasti. However, the in vivo metabolism of A. theophrasti remains restricted. And, current research on the isolated components of A. theophrasti remains restricted as well, and the correlations of these compounds with pharmacokinetic and in vivo metabolism are unknown. Extensive knowledge of the absorption and metabolic mechanisms as well as the bioavailability of A. theophrasti is essential if its pharmacological activities are to be understood in the near future. Further studies should focus on the pharmacodynamics activities in vivo using different animal models, as well as pharmacokinetics.
Although some ingredients have been confirmed to have multiple pharmacological activities, their mechanisms of action are still unclear. Further studies on the material basis and mechanism of action are clearly warranted. Hence, it is necessary to further assess the pharmacological activity of the extracts of A. theophrasti and to consider the synergistic effects among the compounds. In addition, the biological functions of characteristic components from A. theophrasti should be studied, which will better illustrate the pharmacological functions of the plant.
In conclusion, Abutilon theophrasti Medic.(A. theophrasti) is an ethnomedicine with a long history. Modern chemical and pharmacological chemical research suggest that A. theophrasti contains a variety of bioactive compounds and showed diverse biological functions, which is worthy of comprehensive and in-depth research in the near future. With the rapid development of information and technology, it is still important to preserve and develop the medicinal potential of folk medicine, and let them serve the human beings better in the near future.
Footnotes
Contributors' Statement
Design of the work: Lu Li, Linnuo Pang, Fei Chen, Luming Zhang, Fen Zhou, Rongrong Li, and Han Yan; literature review for traditional uses aspects: Lu Li, Linnuo Pang, and Fei Chen; literature review for chemical compositions aspects: Lu Li, Fen Zhou, and Luming Zhang; literature review for pharmacological aspects: Lu Li, Fen Zhou, and Rongrong Li; literature review for pharmacokinetics aspects: Han Yan, Fei Chen, and Fen Zhou; drafting the manuscript: Lu Li, Linnuo Pang, and Rongrong Li; critical revision of the manuscript: Lu Li, Rongrong Li, Fei Chen, and Han Yan.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
