Abstract
Introduction
Traditional herbal therapies and supplements can be used in addition to or instead of standard allopathic medications. Over hundreds of medicinal plants may cure various human ailments such as cancers, memory impairments, gastrointestinal/liver disorders, peptic ulcers, inflammations, hypertension/cardiovascular disease, diabetes mellitus, hyperlipidemia, and microbial infections.1–3
Carvacrol (CAR) (5-isopropyl-2-methyl phenol) is the abundant monoterpene, moderately lipophilic phenol in essential oils (EOs) of many aromatic plants from the Lamiaceae family, including pepper, wild bergamot, oregano, and thyme. 4 It can be collected mainly from leaves of oregano/thyme plants that comprise about 50% to 80% of their EO using steam distillation/hydro distillation techniques. CAR's spicy taste makes it compatible with some diets, and it is the most seriously researched ordinary food preservative. 5
CAR is approved to own antioxidant,
6
anti-inflammatory,
7
and antimicrobial activities
8
and has a total impact on the intestinal microflora. In an animal study, CAR enhanced Lactobacillus inhabitants in the ileum and decreased necrotic enteritis produced by
The progress of different drug delivery systems is of substantial significance to overwhelm many restrictions, including weak bioavailability, instability, water insolubility, intestinal malabsorption and unspecific targeting. Incorporating nanoscience as a drug delivery system in traditional medical systems enriches the potential of herbal drugs for treating chronic diseases. 12 Consequently, the plant/herbal EOs are approved to be loaded/encapsulated at a limited amount in various types of nanoemulsions for many biomedical applications. 13 Nanoemulsions can lower the toxicity of EO due to its small-sized globules (reducing oil droplets to nanoscale dimensions), nano-encapsulation of oil droplets, facilitates oil droplet dispersion in aqueous solution,14,15 increase solubility and sustained release that results in a reduction of median lethal dose value.16,17
Also, CAR is generally recognized as safe (GRAS reference 2245) for food flavoring according to the Food and Drug Administration (FDA, USA). Its use remains hampered by its high volatility, low water solubility (0.330 g/L at 20 °C), poor bioavailability in body fluids, and liability to the effect of light, heat, and oxygen. 18 To overcome these problems, drug delivery systems such as self-nano-emulsifying drug delivery system (SNEDDS) were developed. To date, there is no clear animal study report on the biomedical activities of CAR-SNEDDS from the literature. Thus, we aimed to synthesize and characterize the CAR-SNEDDS and to determine its sub-chronic toxicity profile towards healthy rats using various parameters and techniques.
Materials and Methods
CAR Loaded SNEDDS Synthesis
SNEDDS (Blank) Formulation
About 2 mL of long-chain lipid (olive oil), 5 mL of surfactant (Tween-80), and 2 mL of co-surfactant (dimethyl sulfoxide) were homogenized by a vortex mixer at 25 °C for 3 h, then the volume was completed to 100 mL with distilled water. 19 Next, the combination was ultra-sonicated for 30 min and gently stirred for 72 h at 25 °C. The SNEDDS nanocomposite was centrifuged and filtered, then kept at room temperature for further use.20,21
CAR-SNEDDS (Loaded) Formulation
About 0.5 mL of CAR had been well homogenized in freshly mixed components of SNEDDS nanocomposite to ensure uniformity. The resulting CAR-SNEDDS was ultra-sonicated for 30 min and gently stirred for 72 h at 25 °C. The SNEDDS nanocomposite was centrifuged and filtered, then kept at room temperature for further use (Figure 1).

Carvacrol-loaded self-nano-emulsifying drug delivery system (CAR-SNEDDS) formulation.
Characterization of CAR-SNEDDS
Physical Characterization
The globule size, polydispersity index (PDI), and zeta potential (ZP) were reported 19 using Zetasizer Nano ZS (Horiba Jobn Jyovin-Sz 100). The nanocomposites were diluted with Milli Q water (1:10) at 25 °C, and the measurements were done in triplicates by dynamic light scattering. On the other hand, the morphological appearance of CAR-SNEDDS was found by transmission electron microscopy (TEM) by depositing the formulated nanocomposites on the copper grid (mesh size of 300) and coating them with carbon. Then, phosphotungstic acid (1%, w/v) was used to stain the samples, and after air-drying, the grids were probed into a TEM device (Hitachi H-7100, Japan).
Chemical Characterization
The phase purity/crystallinity of SNEDDS/CAR-SNEDDS was investigated using the x-ray diffractometer (Pananalytical X pert Pro, Germany) at 40 kV with nickel-filtered copper (λ = 1.542 Å) between 10° and 50° and the chemical conformation was described by Fourier-transform infrared (FTIR) spectrometry (Cary 640 Agilent, USA) between 400 and 4000 cm−1. Also, both nanocomposites were quantitated by UV–Vis spectrophotometry (Shimadzu UV-160, USA) at a 150 to 450 nm wavelength.
In vivo study
Male Sprague Dawley rats, aged >2 months with >100 gm weight, had been provided by the College of Medicine, University of Tikrit, Iraq. The rats were adapted to regular tap water and a standard rat chow for 1 week and were preserved in a room with a 12-h dark/light cycle at 25 ± 3 °C.
Sub-Chronic Toxicity Assay
Male Sprague Dawley rats (No. = 28) were arbitrarily allocated into 7 groups (No. = 4): (i) normal control rats received distilled water (CN: control negative); oral treatment with (ii) SNEDDS (LD: low dose, 30 mg/kg body weight); (iii) SNEDDS (median dose, 60 mg/kg body weight); (iv) SNEDDS (high dose, 90 mg/kg body weight); (v) CAR-SNEDDS (low dose, 30 mg/kg body weight); (vi) CAR-SNEDDS (median dose, 60 mg/kg body weight); (vii) CAR-SNEDDS (high dose, 90 mg/kg body weight). Force-feeding, ball-tipped needle was used for oral treatments of animals for 4 consecutive weeks. The body weights were measured weekly, and the rats were checked for clinical, behavioral, and toxicological symptoms.
Biochemical Parameters
On day 29, blood samples (5 mL/rat) were obtained via cardiac puncture under deep anesthesia, and serum was collected by centrifugation (3500 rpm for 10 min). The concentrations of serum total protein (TP), albumin, calcium, inorganic phosphate, sodium, potassium, chloride, alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), urea, creatinine were assessed using Roche diagnostic kits using Cobas 311, Germany.
Hematological Study
The remaining blood samples (1.0 mL) were put into ethylene-diamine tetra-acetic acid tubes, shaken well, and analyzed immediately. The total and differential white blood cells (WBC), red blood cells (RBC), hemoglobin, hematocrit, and platelets for each sample were tested using Cell Dyn, 3700, Abbot, USA.
Histopathological Examination
After sacrificing the animals through deep anesthesia using intraperitoneal injection of xylazine and ketamine mixture (0.2 mL), small pieces of the liver/kidneys were collected from each rat, rinsed in normal saline, and processed for histopathological examination using previously well-known methods. 22 Briefly, tissues were fixed/preserved in neutral buffered formalin (10%), processed, trimmed, embedded in paraffin, sectioned (5.0 mm), and stained with hematoxylin and eosin (H&E). Then, slides were observed under a standard light microscope, and photos were captured.
Statistical Analysis
The data were analyzed using Statistical Package for the Social Sciences (SPSS, version 26.0). Shapiro–Wilk test and histogram figure were used to determine the normal distribution status of the data. In addition, samples T-test and ANOVA tests were implemented to test the relationship between the normally distributed quantitative variables. In comparison, Mann–Whitney U and Kruskal–Wallis tests were used to test the non-normally distributed quantitative variables.
Results
In the current study, the nanocomposite emulsions were rapidly formulated and prepared by ultra-sonication/continuous stirring technique using various compounds and characterized using different parameters. As a result, SNEDDS appeared a slightly milky, clear color that turned to slight opalescence when loaded with CAR due to the nature of the oil (deep yellow) (Figure 2).

The appearance of carvacrol (left), self-nano-emulsifying drug delivery system (SNEDDS) (middle), and carvacrol-loaded self-nano-emulsifying drug delivery system (CAR-SNEDDS) (right).
Concerning the characterization of formulated nanocomposites, zeta sizer was applied to find the mean PS (158.93 ± 22.18 nm), ZP (−22.56 ± 1.77 mV), and PDI (0.553 ± 0.31) of CAR-SNEDDS. All these readings were smaller than blank SNEDDS (Table 1). Additionally, using TEM, CAR-SNEDDS globules appeared irregular, round to spherical, with various diameters (Figure 3).

Transmission electron microscopic (TEM) image of carvacrol-loaded self-nano-emulsifying drug delivery system (CAR-SNEDDS).
The Summary of Characterization of Blank and Loaded SNEDDS.

Abbreviations: CAR, carvacrol; SNEDDS, self-nano-emulsifying drug delivery system.
Moreover, the XRAD of blank and loaded NPs had identical spectra of the broad peak between 10° and 30°, and the broad diffraction peaks were at 21°; however, less intensity with more diffused x-ray scattering was observed for CAR-SNEDDS (Figure 4). At the same time, the FTIR spectrum showed peaks at 3494 (accessible OH), 2924 (stretching of C-H group of aldehydes/alkanes), 1737 (carbonyl group [C=O]), 1459 ((NH)−C=O group), 1352 (C=C aromatic ring), 1250 (C−OH), 948 and 636 (phenyl (C−H)) (Figure 5). Then, UV–Vis absorption spectra were measured between 200 and 450 nm wavelengths, and CAR-SNEDDS presented an intense absorption peak at 200 nm, indicating the presence of phenols. In contrast, another discrete absorption peak was found at 325 nm, ascribed to its ion content. SNEDDS showed 3 absorption peaks at different wavelengths, which indicates its blankness (Figure 6).

Powder x-ray diffraction (XRAD) patterns of self-nano-emulsifying drug delivery system (SNEDDS), and carvacrol-loaded self-nano-emulsifying drug delivery system (CAR-SNEDDS). BL: Blank SNEDDS, LO: Loaded SNEDDS with CAR.

Fourier-transform infrared (FTIR) spectroscopy patterns of self-nano-emulsifying drug delivery system (SNEDDS), and carvacrol-loaded self-nano-emulsifying drug delivery system (CAR-SNEDDS). BL: Blank SNEDDS, LO: Loaded SNEDDS with CAR.

Ultraviolet-Visible (UV–Vis) spectroscopy patterns of self-nano-emulsifying drug delivery system (SNEDDS) and carvacrol-loaded self-nano-emulsifying drug delivery system (CAR-SNEDDS). BL: Blank SNEDDS, LO: Loaded SNEDDS with CAR.
Regarding the animal study, no signs of toxicity like behavioral change, segregation, laying down, mortality, ruffled hair, cannibalism, or loss of appetite/water intake were seen clinically. Furthermore, the body weight of the rats in all groups of CAR-SNEDDS was significantly raised (
Body Weight Estimation of Rats Treated With Various Doses of Blank and Carvacrol Loaded Nanoparticles for Various Times.

Data have been analyzed using repeated measurement ANOVA test.
Abbreviations: CAR, carvacrol; SNEDDS, self-nano-emulsifying drug delivery system.
* Significant difference using Independent Samples T-test; ** highly significant difference using Mann–Whitney U test. Letter “a” indicates a significant difference compared to Day zero.
Biochemical Parameters of Rats Treated With Various Doses of Blank and Carvacrol Loaded Nanoparticles.

Data have been analyzed using Repeated Measurement ANOVA test.
Abbreviations: ALT, alanine transaminase; ALP, alkaline phosphatase; AST, aspartate aminotransferase; B. Wt, body weight; CAR, carvacrol; SNEDDS, self-nano-emulsifying drug delivery system; TP, total protein.
* Significant difference using independent samples T-test. Letter “a” indicates a significant difference compared to control negative group.
Blood Electrolytes of Rats Treated With Various Doses of Blank and Carvacrol Loaded Nanoparticles.
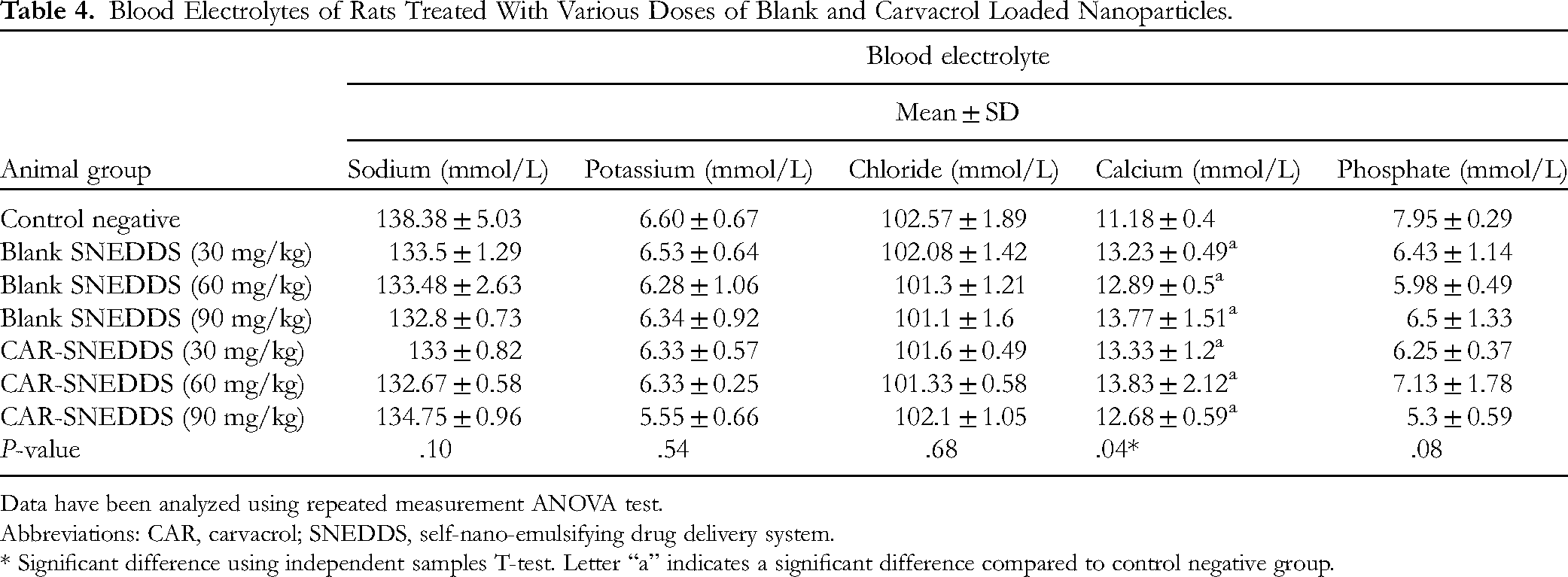
Data have been analyzed using repeated measurement ANOVA test.
Abbreviations: CAR, carvacrol; SNEDDS, self-nano-emulsifying drug delivery system.
* Significant difference using independent samples T-test. Letter “a” indicates a significant difference compared to control negative group.
Hematological Parameters of Rats Treated With Various Doses of Blank and Carvacrol Loaded Nanoparticles.
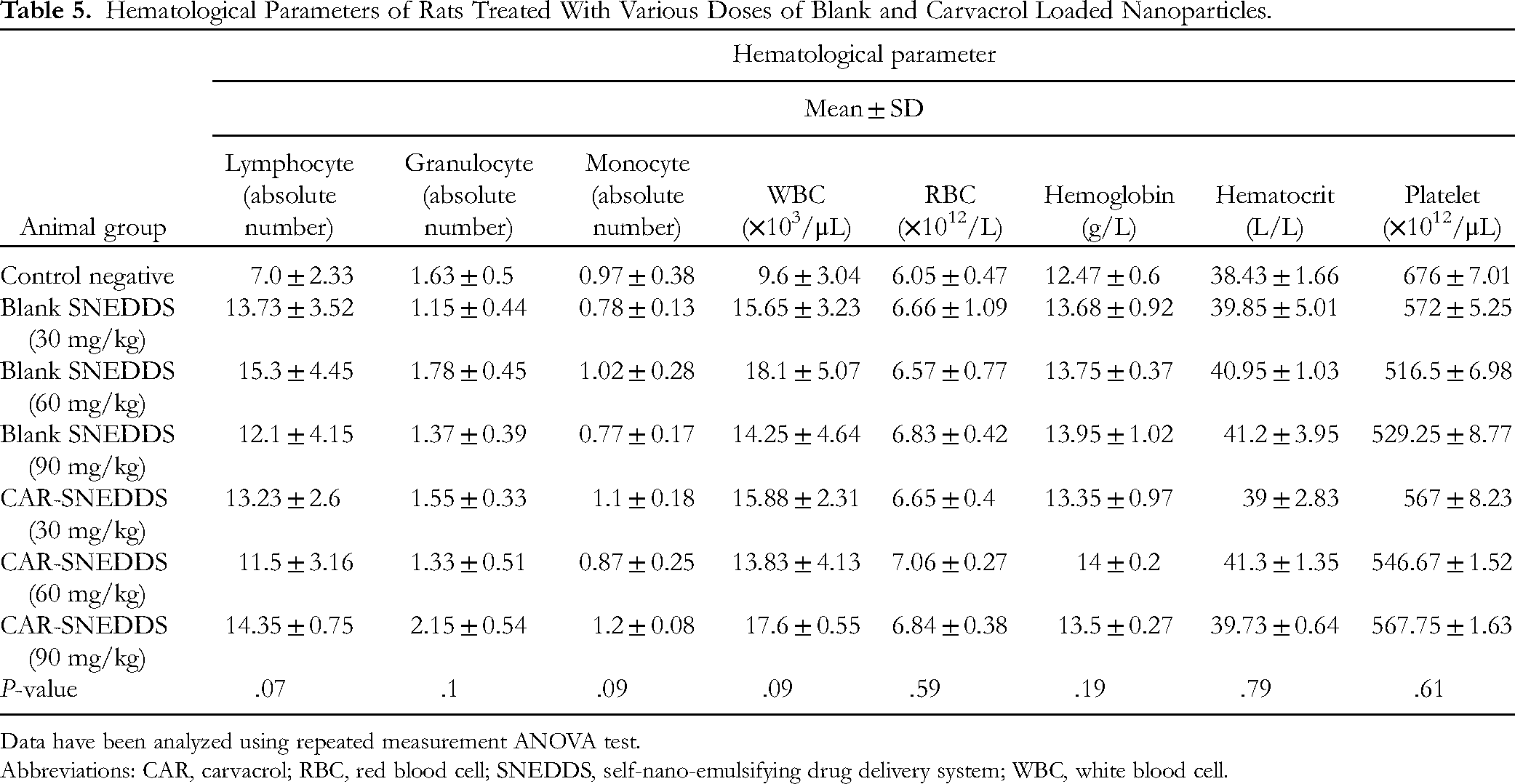
Data have been analyzed using repeated measurement ANOVA test.
Abbreviations: CAR, carvacrol; RBC, red blood cell; SNEDDS, self-nano-emulsifying drug delivery system; WBC, white blood cell.
Furthermore, histopathological observation showed no toxicological lesions with no noted differences in the renal and hepatic histological structures in the treated groups with various blank and loaded SNEDDS doses for 4 consecutive weeks. Liver sections of treated animals did not show destructed hepatic cell cords, polyploidization, the occurrence of inflammatory cells, focal lymphocytic infiltration, enlargement of sinusoidal space, eosinophilic cytoplasm, and pyknotic nuclei of hepatocytes. Also, there was no membrane leakage, necrosis, or apoptosis. Similarly, the overall structure of the liver, hepatocytes, sinusoids, portal triads, and central veins was typical (Figure 7). At the same time, the kidney sections of treated rats exhibited standard structure and appearance of glomeruli/tubules. In addition, the proximal convoluted tubules, distal convoluted tubules, and Bowman's capsule were intact with no interstitial inflammation (Figure 8).

Histopathological appearance of untreated animal's liver tissues (A) and treated animals with 30 mg/kg (B), 60 mg/kg (C), 90 mg/kg (D) self-nano-emulsifying drug delivery system (SNEDDS), and 30 mg/kg (E), 60 mg/kg (F), 90 mg/kg (G) carvacrol-loaded self-nano-emulsifying drug delivery system (CAR-SNEDDS) (H&E staining; 100×).

Histopathological appearance of untreated animal's kidney tissues (A), and treated animals with 30 mg/kg (B), 60 mg/kg (C), 90 mg/kg (D) self-nano-emulsifying drug delivery system (SNEDDS), and 30 mg/kg (E), 60 mg/kg (F), 90 mg/kg (G) carvacrol-loaded self-nano-emulsifying drug delivery system (CAR-SNEDDS) (H&E staining; 100×).
Discussion
The search for novel, potential, and harmless satisfying medicines ensued in integrating ethnopharmacological and traditional information and discovering the multiple plant/herbal components. 23 Thus, in this study, we aimed to formulate CAR nanoemulsions to encapsulate the oil droplets of CAR, facilitate its dispersion, enhance its solubility, and sustain its release.
Generally, PS and PDI are the main characteristics of NPs that directly correlate to the stability of NPs after being loaded with various components, materials, or drugs. PS influences the delivery of NPs, and a smaller PS results in a narrower PDI and vice versa. A suitable PS for CAR-SNEDDS has many advantages, including decreased liver uptake, prolonged blood circulation, and improved bioavailability. Tiny globules are also less phagocytosed by macrophages, leading to less destruction and clearance by the body. 24 The PS for SNEDDS loaded with CAR obtained in this study (158.93 ± 22.18 nm) is significantly smaller than that reported by Maryam et al who found the size of CAR-loaded HAS-NPs to be 230 ± 38 nm with monomodal distribution using emulsion/desolvation method, 25 while Shinde et al reported a PS of 250 nm for CAR loaded zein NPs using precipitation method. 26 These results indicate that SNEDDS is preferable to emulsion/desolvation and precipitation methods for loading CAR.
On the other hand, the PDI of CAR-NPs was also estimated as it is very critical for the stability of the delivery system, and a range of 0.1 to 1.0 is considered acceptable. Hence, the PDI value of the CAR-SNEDDS (0.553 ± 0.31) was lesser than blank SNEDDS (0.933 ± 0.27), designating that the nanoformulations were more homogeneous after loading of CAR. These results for CAR-SNEDDS are higher than that of Bidyarani et al (0.24 ± 0.07) 27 and Niza et al (0.17 ± 0.01), 18 who prepared different models of CAR-NPs. These results indicate a narrow size distribution and well-dispersed homogeneity of the formulated nanocomposites. 28
Reduced ZP reveals a reduced surface charge of NPS, which is very important for preserving the stability of the colloidal dispersion. Scientifically, ZP of +30 to −30 mV are accepted for NPs to have sufficient repulsive force, and less aggregation due to electrostatic repulsion leads to more stability. 29 However, the ZP of CAR-SNEDDS was between a limited range (−22.56 ± 1.77 mV) and less than the blank NPs (−10.66 ± 1.36), which means an increment of early CAR composite reduced the surface charge. These outcomes for CAR-SNEDDS are smaller than that found for CAR-loaded zein NPs (−15 mV), 26 CAR-loaded chitosan NPs (25-29 mV), 30 and PEI-coated PLA-loaded CAR (+54.7 ± 1.1), 18 while it is higher (−29.5 mV) than that found for CAR-HAS-NPs, 25 and rhamnolipid-stabilized CAR-zein NPs (−29.13 ± 1.60 mV). 27 These diversities might be related to the content of the nanoparticle and methods of preparation with environmental factors. Additionally, the TEM of CAR-NPs showed a nearly spherical shape similar to Keawchaoon and Yoksan's study, which prepared CAR-loaded chitosan NPS 30 and Bidyarani et al who formulated CAR-zein NPs. 27 Therefore, due to these features, no visible aggregations were found in the nanocomposites, as reported previously for CAR-NPs.27,30
Regarding the chemical characterization of the formulated nanocomposites, differential scanning calorimetry is the common profound technique to detect NP crystallization by assessing the thermal behavior and heat flow related to material transition. 24 Thus, the XRAD spectra were used to observe the crystalline content of the CAR-NPs, and we found 1 broad peak at 21° (most stable β-modification), which means that CAR no longer preserves its crystallinity and converts to amorphous, as well as dispersed in a lipid matrix. The nonexistence of crystalline structure in the CAR-SNEDDS would decline the loaded drug's tendency to discharge during storage, hence supporting high drug-loading capacity. In this regard, the XRAD pattern of Tin oxide-chitosan-PEG CAR by Tian et al showed many narrow peaks between 20 °C and 90 °C, 31 and the CAR-NLCs showed 2 endothermic peaks between 40 °C and 70 °C by Galvão et al. 32
Additionally, FTIR was done to determine the molecular contents of CAR-NPs, and the spectra of SNEDDS were almost similar to those of CAR-SNEDDS. O-H stretching between the region 3250 to 3600 cm−1 and aromatic C-H bond vibrations were found at around 2924, 948, and 636 cm−1, indicating the availability of CAR in the nanocomposite. Similar reasoning applies to the broadening of vibrational stretching of C-H bonds (2924 cm−1) in CAR-NPs. 26 Also, Vitali et al observed similar spectra for blank and CAR-loaded NPs, significantly increasing the intensity of the C-H stretching peak at 2870 to 2960 cm−1 in CAR-loaded chitosan NPs. 33 However, Tian et al found the wide O-H pulling at 3425 cm−1, symmetric C-H stretching vibration at 2924 cm−1, and distortion mode of O-H at 1633 cm−1 SCP-CAR-NPs. 31
Moreover, to determine the light absorption capacities of the nanocomposites, UV–Vis absorption spectra were recorded at wavelengths of 200 to 450 nm, and the maximum absorption peak of the CAR-SNEDDS was at 325 nm. In comparison, no absorption peak between 240 and 350 nm was reported. These outcomes indicated the presence of CAR in the loaded NPs, and the broad peak spectra of CAR-NPs were an indicator of both bound/unbound CAR. These results followed Vitali et al for CAR-loaded chitosan, 33 while Shinde et al showed no absorption peak between 250 and 400 nm for blank-NPs with maximum absorption at 274 nm for CAR-loaded zein lecithin-NPs. 26 However, Martínez-Hernández et al stated a maximum absorption peak at 275 nm for CAR-loaded chitosan, agreeing to CAR, but not observed for blank NPs. 34
In animal studies, body weight change is considered a vital toxicity index for tested laboratory animals, as significant body weight reduction is a dangerous sign of toxic products in treated animals. However, based on the literature, until now, there is no study on feeding either CAR or CAR-NPs to lab animals and their toxicological effects; thus, we compared our results to other disease models in laboratory animals or other animal species. In the current study, the rats’ body weight in all CAR-SNEDDS groups was significantly increased (
Furthermore, on the 29th day of this study, the ALT, AST, and ALP levels of all doses of CAR-SNEDDS compared with control negative were not significantly changed (
The level of the serum TP and albumin of all doses of CAR-SNEDDS compared to control negative were significantly elevated, which can be explained partly by the improvement in the synthetic liver function as albumin is exclusively synthesized by the liver
37
; also, the olive oil contained a minimal amount of protein (<0.25 mg/kg).
38
Similar outcomes were reported by Ahmadifar et al in fish-fed CAR-NPs.
35
In this study, blood urea and serum creatinine levels of all doses of CAR-NPs compared to control negative were not significantly changed (
The level of serum electrolytes in all doses of CAR-SNEDDS compared with control negative were not significantly changed (
The hematological parameters among all blank and CAR-SNEDDS groups compared with healthy control were insignificantly changed (
Regarding the histopathological study of the sacrificed animals’ livers and kidneys, no toxicity lesions were observed, which is complementary to the outcomes of hepatic/renal tests. These observations also confirm the safety of CAR-NPs caused mainly by hampering the toxicity effects of CAR by SNEDDS. These outcomes agreed with Ragab et al, who observed no noticeable lesions in the kidneys and livers of treated rats with CAR-NPs. 39
Moreover, sub-chronic toxicity study of SNEDDS loaded Bay leaf (
Conclusions
We realized that SNEDDS presented decent nanocomposite features and was a hopeful carrier system for the low soluble CAR. Loading CAR in the SNEDDS system did not significantly affect treated animals’ biochemical, hematological, and histopathological parameters at low and medium doses. At the same time, it showed little toxicity at high doses. More research must be directed to measure the pharmacokinetics, pharmacodynamics, and beneficial effects of CAR-SNEDDS
Footnotes
Acknowledgment
The authors are grateful to the Azna Laboratory, Sulaimaniyah, Iraq for providing facilities and services.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the scientific and ethical committees of the College of Medicine, University of Sulaimani, Sulaimaniyah, Iraq (No. 97 in 25/5/2021-CoM-UoS).
Statement of Human and Animal Rights
All experimental procedures involving animals were conducted according to Directive 2010/63/EU guidelines in Europe.
Statement of Informed Consent
No human subjects are in this article, and informed consent is not applicable.
