Abstract
Background & Objective
Paraquat (PQ), a widely used heterocyclic herbicide, has caused many deaths due to acute lung fibrosis, which demands more effective therapies. Since multiple co-activated pathways are involved in the pathogenesis of fibrosis, targeting these pathways seems to be a reasonable strategy for a successful treatment. The present study aimed to evaluate the therapeutic effect of a combination of pirfenidone and silymarin in paraquat-induced lung fibrosis in rats.
Methods
Forty Wistar rats were randomly divided into five groups (control, PQ, SM, PQ + SM, and negative control group). Pulmonary fibrosis was induced by a single dose of 20 mg/kg PQ in rats and followed by oral treatment regimens. The comparison of effects was performed by evaluating histopathological changes, hydroxyproline content, tissue oxidative stress parameters, and the expression of pro-inflammatory and pro-fibrotic genes, including TGF-β1, TNF-α, TIMP-1, and MMP-2, using real-time RT-PCR.
Results
Pathological changes, including increased lung hydroxyproline levels, were observed in the lung tissues of the paraquat-treated group. Paraquat also caused oxidative stress and an increase in profibrotic gene expression. Pirfenidone and silymarin were able to solely recover the pathological changes caused by paraquat, decrease lipid peroxidation, and restore the antioxidant enzymes to near-normal values. The combination of pirfenidone and silymarin significantly reduced the hydroxyproline content. At the molecular level, the expressions of TGF-β1, TNF-α, TIMP-1, and MMP-2 genes were decreased in the treated rats. Concurrent therapy by pirfenidone plus silymarin showed more potent anti-fibrotic and anti-inflammatory effects.
Conclusion
In conclusion, pirfenidone plus silymarin holds promise as a combination therapy for treating PQ-induced pulmonary fibrosis.
Highlights
Paraquat is a widely used herbicide, can cause severe acute pulmonary fibrosis.
Pirfenidone and Silymarin reduce paraquat-induced pulmonary fibrosis.
Combination therapy by pirfenidone plus silymarin more potently, reduce paraquat-induced pulmonary fibrosis.
This combination therapy reduces oxidative stress and lipid peroxidation.
This combination therapy reduces the pro inflammatory and pro fibrotic gene expression, such as TGF-β1, TNF-α, TIMP-1 and MMP-2.
Introduction
Pesticide poisoning has become a serious public health concern in most developing countries. 1 Paraquat (PQ) is a highly poisonous, widely used herbicide that can cause severe lung damage in humans and animals. Exposure to PQ results in pulmonary accumulation of PQ, causing acute lung injury and ultimately lung fibrosis, which is characterized by overproduction and deposition of extracellular matrix and scarring. 2
Despite considerable advances in understanding the pathogenic mechanisms and molecular pathways responsible for PQ-related lung fibrosis, it represents a significant challenge to pulmonary patients and physicians. Since multiple co-activated pathways are involved in the pathogenesis of pulmonary fibrosis, targeting a single pathway with a single therapy is not effective. Combination therapy, which aims to exert its potentiating effects, is one of the rationale strategies proposed to increase response. 3 Here, we examined the combination of pirfenidone and silymarin as a novel oral treatment for curing PQ-induced lung injury. This treatment protocol was guided by the pathogenesis of pulmonary fibrosis, based on preclinical and experimental findings in rodents, as well as in vitro experiments. Since oxidative stress and inflammation are considered two main culprits in the progression of lung scarring and fibrosis,4–8 it seems that by targeting these pathways, the fibrosis could be limited and/or prevented more successfully.
Pirfenidone [5-methyl-1-phenyl-2-(1H)—pyridone] is an orally active small molecule that exhibits potent anti-fibrotic and anti-inflammatory activities, and recently, different clinical trials have evaluated the therapeutic effect of pirfenidone (PF) for idiopathic pulmonary fibrosis (IPF).9,10 Its primary mechanism involves downregulation of Transforming Growth Factor β signalling, alongside with the suppression of proinflammatory cytokines, TNF-α and reactive oxygen species (ROS) production.11,12 The most powerful trigger of this process is transforming growth factor-beta (TGF-β) which acts as a paracrine factor released by cells of the immune systems, in particular, macrophages, which appear early when a protective or reparative response is required. 13
Silymarin, a flavonoid obtained from the fruits and seeds of the milk thistle (Silybum marianum L. Gaertn.), is composed mainly of silibinin (≈90%) together with small amounts of other silibinin stereoisomers, such as isosilybin, dihydrosilybin, silydianin, and silychristin, which have potent antioxidant and anti-inflammatory properties.14,15 It can scavenge free radicals, such as hydroxyl, superoxide, and hydrogen peroxide, reduce lipid peroxidation, and enhance SOD activity by the different mechanism reviewed by Surai. 16 Varieties of pharmacological properties, including anti-inflammatory, anti-oxidant, and anti-fibrotic effects, have been reported for this compound. Silymarin also exhibits potent anti-fibrotic properties against various toxicants, including halothane, galactosamine, thioacetamide, paladin, carbon tetrachloride, paraquat, and bleomycin.14,17–20 The anti-inflammatory and anti-fibrotic effects of silymarin are mainly mediated by inhibition of NF-κB and related pathways, and decreased expression of proinflammatory interleukins (IL-2 and IL-4), TNF-α and IFN-γ as well as downregulation of procollagen α1(I) and TIMP-1.21,22
The present study was designed to evaluate the anti-fibrotic effects of pirfenidone and silymarin, both alone and in combination, in a paraquat (PQ)-induced lung fibrosis model. Histopathologic examinations, hydroxyproline content, lung tissue oxidative stress, and the expression of inflammatory and fibrotic genes, including TGF-β1, TNF-α, TIMP-1, and MMP-2, in the lung tissues assessed these effects.
Materials and Methods
Materials
Paraquat, Pirfenidone, Silymarin, hydroxyproline, 4-dimethylaminobenzaldehyde, chloramine T, and malondialdehyde (MDA) were purchased from Sigma–Aldrich Chemical Co. (USA). TRIzol® RNA isolation reagent and HyperScript™ first strand cDNA synthesis kit were purchased from Invitrogen (Germany). SYBR Green Master Mix was obtained from Takara (Japan). All other chemicals were bought from Merck (Germany).
Animals and Treatments
Forty Male Wistar rats of 8-12 weeks old were randomized and divided into 5 groups with eight rats in each group, and all groups were maintained in standard laboratory conditions at 21 ± 21 °C under a 12/12 light–dark cycle. Pulmonary fibrosis was induced by intraperitoneal (i.p.) administration of a single dose of 20 mg/kg PQ.23,24 Pulmonary fibrosis developed in 2 weeks (Day14), confirmed by histopathological assessment of lungs and diagnosis of interstitial collagen overproduction. 25 Then experiment was performed in rats were equally and randomly divided into five groups, namely PQ-induced fibrosis rats (PQ group which received single dose of 20 mg/kg PQ), Pirfenidone treatment (which received 200 mg/kg PF for 14 constitutive days), Silymarin treatment (which received 50 mg/kg SM for 14 constitutive days), Pirfenidone plus Silymarin treatment (which received 100 mg/kg PF plus 25 mg/kg SM for 14 constitutive days) and normal animals receiving vehicle (water) by gavage as negative control. The treatment regimens were administered to the respective groups after 14 days, following the establishment of pulmonary fibrosis. Pirfenidone and Silymarin were administered by gavage. The doses of pirfenidone and silymarin were selected based on clinical trials in patients and experimental studies,26–29 and in the combination group, the doses of pirfenidone and silymarin were halved compared to the single- drug groups to evaluate potential potentiating effects and to reduce the risk of adverse effects.
Rats were maintained and treated humanely according to the guidelines on ethical standards for investigation, which were approved by the Animal Experimentation Ethics Committee of Kerman Neuroscience Research Center (EC/KNRC/90).
Sample Collection and Analytical Procedures
After the treatment period (28 days), the rats were anesthetized by intraperitoneal injection of Ketamine/Xylazine (Ketamine, 100 mg/kg; xylazine, 10 mg/kg). The lung was quickly removed and immediately divided into two halves. The right lung was stored at −80 °C for analysis of gene expression, oxidative stress, and Hydroxyproline content. The left lung was dipped in 10% buffered formalin for histopathological observations.
Histopathological Analysis
After embedding the fixed lung tissues into liquid paraffin, 5 µm thick tissue sections were prepared. The sections were stained by Hematoxylin and Eosin (H & E) and Masson's trichrome staining methods. The presence and grading of inflammation 30 and fibrosis 25 were evaluated in the stained sections based on the scoring indicated in previous studies and also our previous studies. 24 Briefly, a qualitative evaluation based on a simpler grading system (absent, present, focal) or (absent, mild, moderate, severe) indicated in the previous studies was also used for the specific histopathologic characteristics. All histologic sections were blindly evaluated by an expert histopathologist.
Determination of Oxidative Stress Parameters
Preliminary preparation of lung tissue samples for biochemical evaluation was done with 0.1 M Tris–HCl buffer (pH 7.4) at 4 °C using a tissue homogenizer. The resulting tissue homogenates were used for biochemical measurements. The malondialdehyde in the lung tissue was measured on the basis of reaction with thiobarbituric acid in the acidic condition to produce a pink chromophore with a maximum absorbance at 532 nm. Different concentrations of tetra butyl ammonium prepared as a standard solution. 24 The activities of SOD was measured according to the method of Beyer and Fridovich with modification, on the basis of the ability of the enzyme to inhibit the autoxidation of pyrogallol with minor modifications 31 and CAT activity was determined using the method of Aebi. 32
Measurement of Collagen
The hydroxyproline content was quantified colorimetrically as a measure of collagen deposited in the lung tissue and fibrosis, using the method of Reddy & Enwemeka 33 with minor modification. 24
Determination of Fibrotic Gene Expression by Real-Time RT-PCR
Total RNA was extracted from pulmonary tissues using TRIzol® reagent according to the manufacturer's protocol. Samples (2μg RNA) were reverse-transcribed using a HyperScript™ first-strand cDNA synthesis kit. Synthesized cDNA was used in real-time RT-PCR (lightcycler® 96 Roche, Germany) experiments using SYBR GREEN Supermix and analyzed with lightcycler® 96 Software. The sequences of primes are as follows 34 : rat TGF-β1(5′-GCTCGCTTTGTACAACAGCA-3′ and 5′-GAGTTCTACGTGTTGCTCCA-3′), rat MMP-2 (5′-CTGGGCAACAAGTATGAGAG-3′and 5′GTGTAGGTGTAGATAGGGGC-3′), rat TIMP-1(5′-GCCTCTGGCATCCTCTTG-3′and 5′-TGCGGTTCTGGGACTTGT-3′).
Specificity was confirmed by electrophoretic analysis of the reaction products and by inclusion of template- or reverse transcriptase-free controls. To normalize the amount of total RNA present in each reaction, β-actin cDNA was used as an internal standard.
Statistical Analysis
The quantitative data were presented as mean ± SEM. Differences between the means were analyzed using one-way analysis of variance (ANOVA) followed by Tukey HSD post-hoc test. The differences are considered statistically significant when p < 0.05. The data were analyzed using SPSS 18.0.
Results
Histological Studies
The histopathological changes in lung tissues were evaluated by H&E or Masson trichrome staining. In the negative control group, the lung tissues exhibited a normal alveolar structure without inflammation. PQ exposure induced a marked inflammatory response characterized by the aggregation and infiltration of inflammatory cells (lymphocytes and histiocytes) in the alveolar space and septum, resulting in the substantial thickening and degradation of the standard alveolar structure. Masson trichrome staining revealed markedly increased collagen deposition, predominantly in the thickened alveolar regions and small bronchioles (Figure 1). PF and SM treatment attenuated the marked interstitial thickening, inflammatory responses and collagen accumulation induced by PQ. Combination therapy by PF plus SM, alleviated the inflammatory and fibrotic lesions more than each of them separately. All the lesions are depicted in Figure 1 and Table 1.
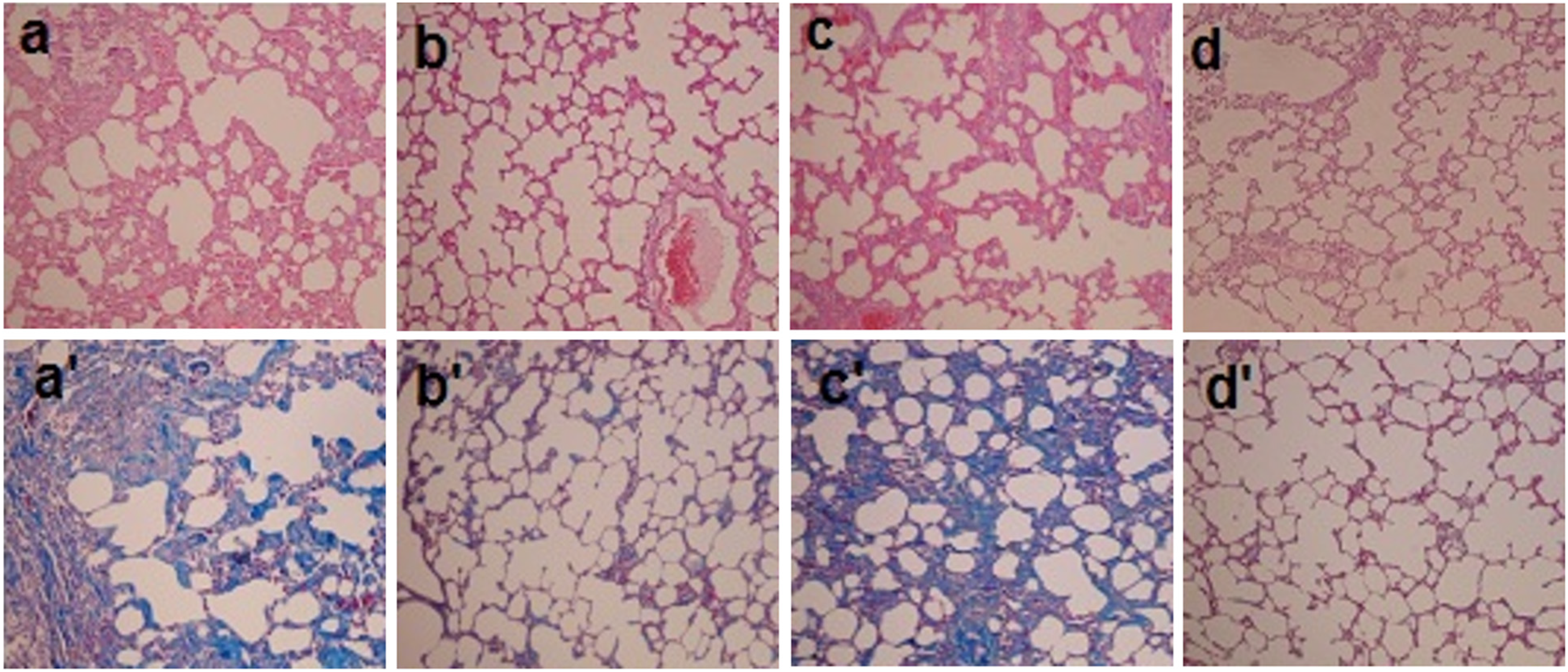
Photomicrographs of hematoxylin and eosin (a, b, c, d) and Masson's trichrome (a', b', c', d') stained lung tissues from each group. PQ group: received PQ without treatment shows highest level of fibrosis; PQ+PF group: received PQ and PF (200 mg/kg) for 14 days as a treatment; PQ+SM group: received PQ and SM (50 mg/kg) for 14 days as a treatment; PQ+PF+SM group: received PQ and PF (100 mg/kg) plus SM (25 mg/kg) for 14 days as a treatment (magnification ×100).
Histopathological Lesions of the Lung Tissue After Paraquat Toxicity and Therapeutic Effects of Pirfenidone, Silymarin, and Pirfenidone-Silymarin Combination.

Oxidative Stress Parameters
The levels of lipid peroxidation and the antioxidant activities of SOD and CAT were depicted in the lung tissue of the control and experimental rats (Figure 2). Lipid peroxidation in the lung tissue showed a significant rise in the PQ-treated animals. A decrease in the enzymatic activities of SOD and CAT accompanied this effect. Administration of both PF and SM significantly decreased lipid peroxidation in lung samples (p < 0.001) and restored SOD and CAT activity to near normal levels. The co-administration of PF and SM exacerbates these effects. Significance levels are separately depicted in Figure 2.
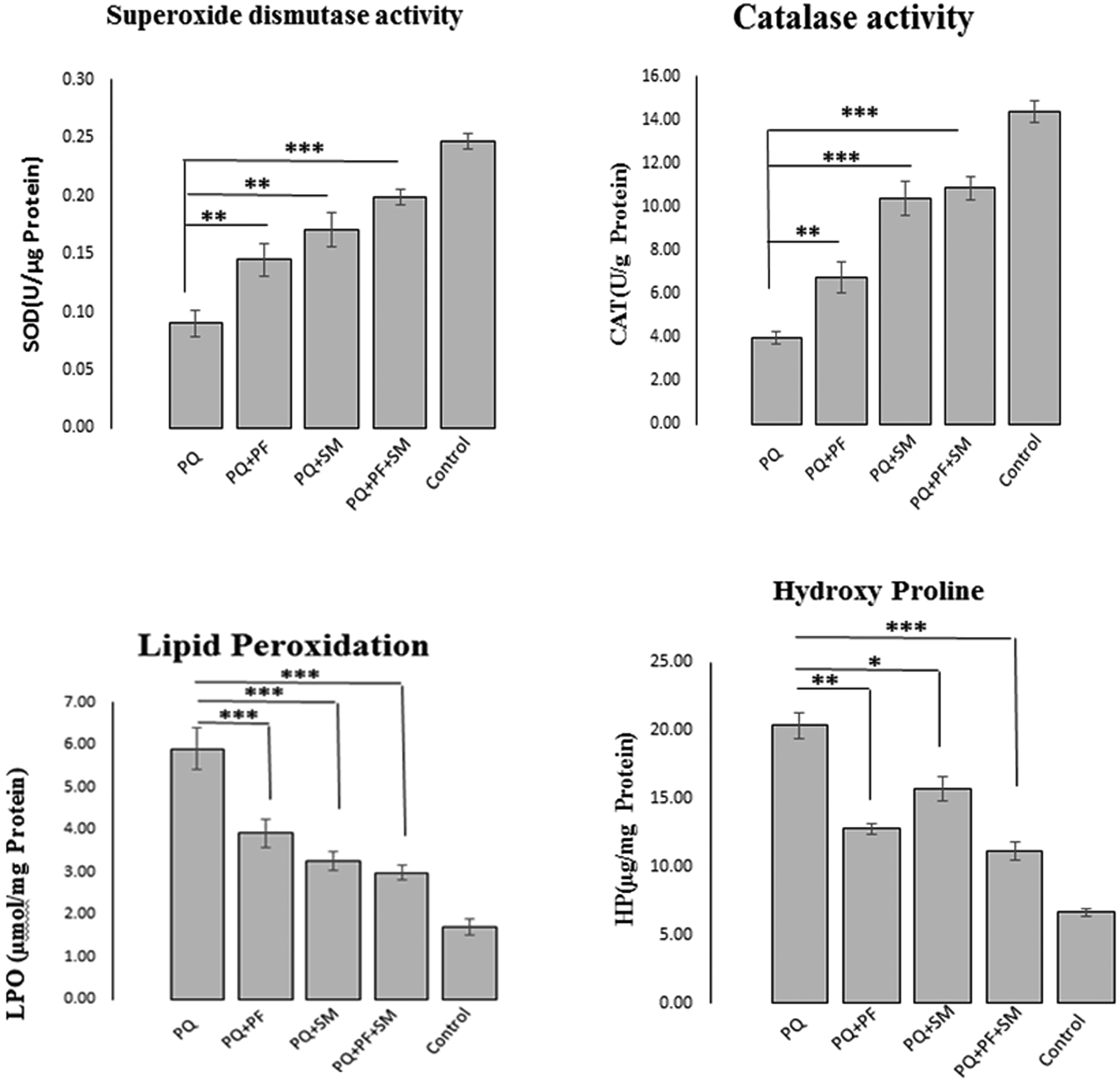
Influence of PQ toxicity and therapeutic effects of pirfenidone, silymarin, and pirfenidone-silymarin combination. On oxidative stress parameters and hydroxyl proline content. Data are expressed as the mean ± SEM (two replicates in each assay), n=8. *P < 0.05, **P < 0.01 and ***P < 0.001 in comparison with the PQ group.
Hydroxyproline Assay
Hydroxyproline was measured as a marker of fibrosis in the lung tissue. As revealed in Figure 2, the hydroxyproline content in the lungs of the PQ-treated rats significantly increased in contrast to the control group. Administration of PF (p < 0.01) and SM (p < 0.05) could significantly reduce the content of hydroxyproline in lung tissues. These findings were more distinctive in combined therapy by PF-SM (p < 0.001), which all confirmed the histopathological results.
Pulmonary TGF-1 and TNF-α Gene Expression
To further investigate the specific anti-fibrotic and anti-inflammatory effects of PF and SM, and to compare them with their combination therapy, the expression patterns of inflammatory and fibrosis-related cytokines in lung tissues were evaluated by RT-PCR. TGF-β1, as the main pro-fibrotic cytokine, has been demonstrated to be the most potent and direct stimulator of fibroblast proliferation and collagen production, and is requisite for pulmonary fibrosis of various etiologies. 35 As shown in Figure 3, PQ exposure progressively increased TGF-β1, while PF and SM treatment significantly decreased lung TGF-β1 mRNA levels (p < 0.001). TNF-α, which plays an essential role in PQ-induced pulmonary inflammation, was analyzed. As anticipated, PQ increased lung TNF-α expression, which was significantly inhibited by both PF (p < 0.001) and SM (p < 0.01) (Figure 3). Combination therapy with PF plus SM, compared to each one alone, reduced the expression of both TGF-β1 and TNF-α genes, indicating more potent pro-fibrotic and pro-inflammatory effects.
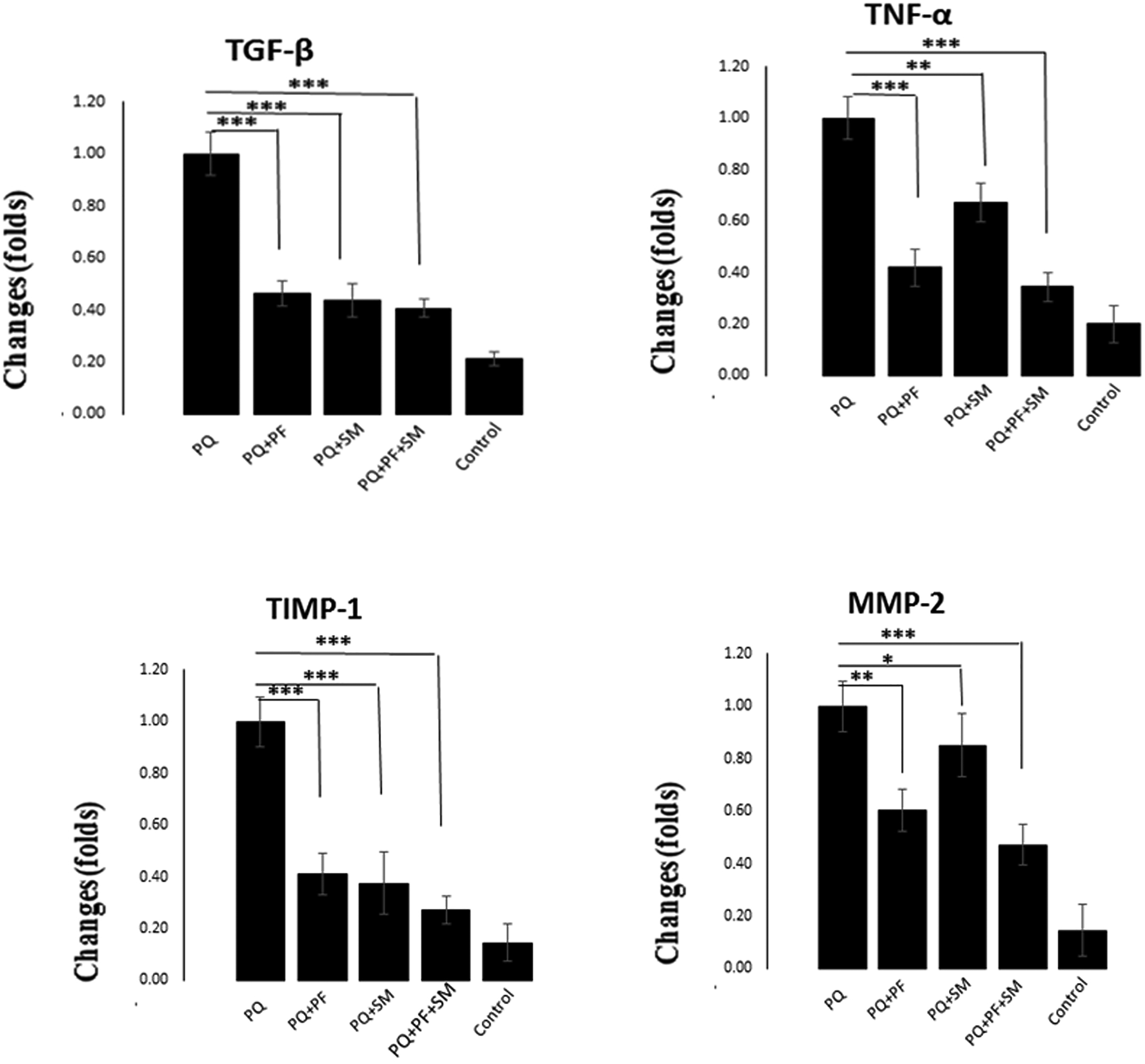
Influence of therapeutic effects of pirfenidone, silymarin, and pirfenidone-silymarin combination on the expression of fibrotic genes in PQ-induced lung fibrosis. The rats were treated for 14 days after the development of fibrosis. The expressions of TGF-β, TNF-α, TIMP-1, and MMP-2 mRNA were determined in the lungs of the rats by real-time RT-PCR. The data represent the means ± SE (two replicates per assay) for 8 rats. *P < 0.05, ***P < 0.001 and **P < 0.01 in comparison with the PQ group.
Pulmonary TIMP-1 and MMP-2 Gene Expression
Following a fibrogenic stimulus, lung cells lose their morphology and function, and produce large amounts of ECM proteins due to the overexpression of MMPs and their physiologic inhibitors TIMPs, which are proven in fibrotic lung disease. Therefore, we detected the mRNA levels of key fibrotic marker genes, TIMP-1 and MMP-2. The mRNA expression of TIMP-1 and MMP-2 genes was increased in the PQ group. PF and SM solely decreased the expression of the TIMP-1 and MMP-2 genes. Subsequent to combination therapy by PF-SM, the mRNA expression of the TIMP-1 and MMP-2 obviously decreased (p < 0.001), which all supports its higher ability to inhibit the activation of ECM deposition and the progression of fibrosis.
Discussion
The present study evaluated the potential effect of PF plus SM in PQ-induced pulmonary injury. The findings indicated that the lung injury following PQ administration was characterized by alterations in several parameters, including a significant reduction in SOD and CAT activity, an elevation in lung lipid peroxidation, increased hydroxyproline content, and overexpression of pro-fibrotic and pro-inflammatory genes, such as TGF-β, TNF-α, TIMP-1, and MMP-2.
Production of free radicals, mainly including ROS, and thereby induction of oxidative stress, is considered the foundational mechanism of PQ-induced lung fibrosis.5,36 Oxygen-free radicals cause the oxidation and damage of genetic materials, proteins, and membrane lipids, and the production of oxidation markers, such as MDA, which is a marker of lipid peroxidation. So, it has been demonstrated that antioxidant agents provide beneficial effects on lung injury.5,37,38 In the present study, we observed a significant elevation in lipid peroxidation levels in the lung tissue of the PQ-treated animals, which was accompanied by a decrease in the enzymatic activities of SOD and CAT. As a single agent, both PF and SM have shown decreased lipid peroxidation and restored SOD and CAT activity to near normal levels. As previously demonstrated, SM is a potent antioxidant, and the anti-fibrotic effects of SM are thought to be mainly related to its free radical scavenger properties and antioxidant activity, and decreasing oxidative stress caused by PQ.29,39,40 The potent antioxidant role of pirfenidone has also been reported in many experimental models of fibrosis and cirrhosis.41,42 Here, the administration of PF plus SM produced more effective effects than either treatment alone, indicating reduced ROS formation as a primary mechanism by which the anti-fibrotic effects of this combination therapy were mediated. Moreover, PF plus SM more efficiently inhibited the hydroxyproline content. PQ-induced hydroxyproline content, as an indicator of collagen deposition, was significantly reduced with both PF and SM treatment. This effect can be attributed to probable mechanisms, such as the inhibition of free oxygen radical generation and the subsequent inflammatory process initiated after PQ poisoning, resulting in a reduction of fibroblast proliferation and hydroxyproline production.
Histopathological evaluation supported the results obtained from lipid peroxidation, antioxidant assays, and hydroxyproline measurements. PQ administration induced inflammatory cell aggregation and infiltration in the alveolar space and septum, substantially thickening and degrading standard alveolar structure, and collagen deposition in the thickened alveolar regions and small bronchioles. Both PF and SM suppressed PQ-induced inflammatory cell infiltration and collagen deposition in the lungs. These effects can be attributed to the antioxidant and anti-inflammatory properties of SM, as well as the antioxidant, anti-inflammatory, and anti-fibrotic properties of PF. Growing evidence strongly suggests that lung fibrosis is characterized by the excessive proliferation of alveolar mesenchymal cells, including myofibroblasts, and the aggregation of inflammatory cells, such as neutrophils and macrophages. 43 All these cells under oxidative stress produce TGF-β, which encourages up-regulation of matrix metalloproteinases (MMPs) in lung tissue. 44 MMP-2 is typically upregulated in both the early and late stages of lung fibrosis, and its overexpression serves as a marker for basement membrane disruption. 45 Since the healthy alveolar epithelium depends on the integrity of the basement membrane, destruction of the subepithelial basement membrane leads to alveolar destruction and lung fibrosis. 45 On the other hand, increased MMP-2 expression creates an imbalance between MMP and TIMP, which leads to the progression of oxidative stress. 46 Oxidative stress and overproduction of ROS in many chronic diseases, including lung fibrosis, result in protein oxidation and subsequently release of inflammatory signal molecules and peroxiredoxin 2 (PRDX2) as an inflammatory signal. The relationship between oxidative stress and inflammation has been established, and numerous pieces of evidence suggest that oxidative stress plays a pathogenic role in chronic inflammatory diseases.47,48 In this regard, antioxidants such as silymarin 49 and resveratrol 50 are reported to correct this imbalance. MMP-2 and TIMP-1 were verified as the molecular signatures during the progression of lung fibrosis. They were widely expressed in fibrotic lungs, especially in areas of hyperplastic epithelial cells covering intra-alveolar fibrosis, as well as by mesenchymal cells in the fibroblast foci. 45 Here, a significant increase in MMP-2 and TIMP-1 was observed in rats exposed to PQ. In this regard, the administration of PF plus SM resulted in a substantial decrease in MMP-2 and TIMP-1 levels, indicating the correction of PQ-induced overexpression of MMP-2 and TIMP-1 genes.
TGF-β is the main cytokine which mainly derived from macrophages and fibroblasts and involved in the process of fibrosis via the conversion of fibroblasts to myofibroblasts and stimulate the collagen synthesis. 8 PQ-induced lung fibrosis and other inflammatory and fibrotic diseases have been associated with increased level of TGF-β1 in human and laboratory animals. 51 The relationship and overlap between the actions of TNF-α and TGF-β1 in lung fibrosis is completely proved and it is shown that TNF-α has a critical role in regulation of TGF-β1 expression in lungs. TNF-α activates the extracellular regulated kinase (ERK)-specific pathway in fibroblasts and subsequently stabilization of TGF-β1 mRNA and increased expression of TGF-β1.52,53 Besides, ample of evidence has shown the reciprocal regulation of TGF-β and ROS in the organ fibrosis. 54 TGF-β1 rises the ROS production and reduce antioxidant enzymes activity, leading to a redox imbalance. ROS overproduction activates TGF-β1 and mediate many of TGF-β's fibrogenic effects. It is demonstrated that ROS not only apply its destructive effects through attack the cell membrane and organelles, but also it has been shown to activate latent TGF-β. Exposure of epithelial cells and fibroblasts to TGF-β1 increased cell cytotoxicity mediated by oxidative stress 55 as well as collagen production. 56 It means that when TGF-β is up-regulated, excessive fibrosis results from increased collagen deposition. As expected, here TGF-β is downregulated by both PF and SM, and especially by their combined therapy, suggesting that both PF and SM exert their anti-fibrotic effects partly through the inhibition of TGF-β1 signaling. As over-fibroproliferation and ECM deposition are critical determinants of fibrogenesis, TGF-β inhibition is an appropriate marker for depiction of reduced fibrosis development in experimental models.
TNF-α, the pro-inflammatory factor, is produced by activated monocytes, fibroblasts, and endothelial cells, and is increased by paraquat administration. Increased expression of TNF-α has been seen in the lungs of patients and animal models of pulmonary fibrosis.57,58 The present results indicate that the anti-inflammatory effects of PF and SM may also depend on a decrease in the production of pro-inflammatory cytokines.
Conclusion
PF plus SM offers potential as a combination therapy for treating PQ-induced pulmonary fibrosis. Although both PF and SM modulate the expression pattern of inflammatory and fibrosis-related genes as well as hydroxyproline content and antioxidant status in the lung tissues, their combination exaggerates all these therapeutic effects.
The weaknesses of the current study should not be overlooked. Considering the wide range of pharmacologic effects of PF and SM, there may be far more complex mechanisms than we mentioned in this manuscript. More mechanistically pathways and larger preclinical examinations in animals as well as human trials are necessary to further define the beneficial effects of PF plus SM. In addition, the therapeutic mechanisms of PF plus SM in lung injury treatment should be assessed in many different doses and models.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251405972 - Supplemental material for Pirfenidone–Silymarin Combination Mitigates Paraquat-Induced Lung Fibrosis via Oxidative and Pro-Fibrotic Pathway Modulation
Supplemental material, sj-docx-1-npx-10.1177_1934578X251405972 for Pirfenidone–Silymarin Combination Mitigates Paraquat-Induced Lung Fibrosis via Oxidative and Pro-Fibrotic Pathway Modulation by Rokhsana Rasooli, Ali Mandegary and Saeed Aghebat-Bekheir in Natural Product Communications
Footnotes
Acknowledgments
The authors acknowledge Prof. Mahmoud Ghazi-Khansari, Department of Pharmacology, School of Medicine, Tehran University of Medical Sciences, for providing Paraquat poison and advice that aided the successful completion of this study.
Ethical Approval
All animals were cared for humanely following the ethics standards for investigating experimental pain in animals, approved by the Animal Experimentation Ethics Committee of Kerman Neuroscience Research Center (EC/KNRC/90).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was fully supported by the Deputy of Research, Kerman University of Medical Sciences.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data will be made available on request.
Supplemental Material
Supplemental material for this article is available online.
