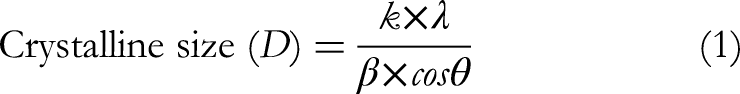Abstract
Objective
The study aimed to synthesize zinc oxide nanoparticles (ZnO-NPs) using zinc acetate as the raw material and leaf extract of Plantago lanceolata both as reducing and stabilizing agent, and to evaluate their antibacterial, photocatalytic, as well as antioxidant activities.
Methods
ZnO-NPs were biosynthesized with zinc acetate and P. lanceolata leaf extract. This is the first report on the green synthesis of ZnO-NPs using P. lanceolata leaf extract as a bio-reductant and stabilizer. Characterization involved UV-Vis spectroscopy, zeta potential measurements, TEM, XRD, and FTIR. Photocatalytic activity was assessed via methylene blue degradation under UV light. Antioxidant activity was evaluated using the DPPH assay. Antibacterial testing was conducted against S. aureus, K.pneumoniae, S. epidermidis, and E. coli.
Results
UV-Vis absorption peak were observed at 370 for biosynthesis of ZnO-NPs nm, indicating nanoparticles formation, with an optical band gap energy ranging from 3.57 to 3.61 eV. Zeta potential was −30.5 mV, confirming good colloidal stability. TEM showed irregularly shaped nanoparticles averaging 23 nm, and XRD confirmed a hexagonal wurtzite crystal structure, with an average crystallite size of 23 nm. FTIR verified the presence of flavonoids and polyphenols as capping agents. Photocatalytic degradation of MB reached 76.65% efficiency after 75 min at 500 µg/mL concentration, with degradation increasing from 14.09% at 100 µg/mL to 60.27% at 75 min. Antioxidant activity showed concentration-dependent radical scavenging, with an IC50 of 76.24 µg/mL and inhibition percentages ranging from 50.23 ± 1.74% at 100 µg/mL to 90.31 ± 0.51% at 500 µg/mL. Antibacterial assays revealed notable zones of inhibition, with K. pneumoniae showing the highest (30.1 ± 1.11 mm), surpassing the control (28.00 mm), and values for other strains including E. coli (21.21 ± 1.15 mm), S. aureus (28.11 ± 1.08 mm), and S. epidermidis (25.34 ± 1.15 mm) at 500 µg/mL.
Conclusion
These findings shows that P. lanceolata-mediated ZnO-NPs possess excellent photocatalytic, antibacterial as well as antioxidant activities, highlighting their potential for eco-friendly applications in wastewater treatment and biomedical fields.
Introduction
In the twenty-first century, nanotechnology and nanopharmacology have emerged as pivotal fields of research and development, focusing on the manipulation and study of matter at the atomic scale, typically in the range of 1 to 100 nm.1–4 When materials reduced to the nanoscale, surface-to-volume ratios increase considerably, enhancing surface effects and making analysis easier.5,6 Nanomaterials with larger surface areas exhibit superior physicochemical and biological properties compared to those with smaller surface areas, which opens up a world of possibilities in many scientific field. 7 The increase in surface area of a nanoparticle give a characteristics properties to have high reactivities compared to material with low surface area. 8 Because of these characteristics, nanoparticles are particularly well-suited for pharmacological study. 9
Among the many different kinds of nanoparticles, metal oxide (MO) nanoparticles stand out as being particularly promising due to their especial biological, physical, and chemical properties like solubility, chemical stability, and adhesiveness. 10 Techniques for preparing MO nanoparticles may include the use of hazardous chemicals that are harmful to the environment, animals, and plants if ecologically friendly ingredients are not used. On the other hand, synthesize MO nanoparticles with green methods such as plant extracts provides a simple, affordable, eco-friendly, and non-toxic substitute. 10 For nanoparticle synthesis, plant extracts contain a variety of active biomolecules like fiber, flavonoids, alkaloids, polyphenols, carbohydrates and lipids. They work both as reducing and capping agent that stabilize nanoparticles and reduce metal ions.11–13 For the synthesis of nanoparticle using physical, chemical, and biological methods involves larger amount energy, higher temperature, and non-green organic solvents like sodium borohydride and hydrazine as reducing agents. 10 Because of this, researchers have been paying more attention to the topic of green chemistry.
Metal oxide nanoparticles such as ZnO, MgO, Fe2O3, CuO, Ag2O, CaO, NiO, and ZrO2 have recently attracted considerable attention for different activities for a biomedical and environmental potential.14–18 They are a promising candidates in biomedicine, with demonstrated anti-cancer, antibacterial, anti-inflammatory, antioxidant as well as anti-diabetic peroperties. 19 In particular, the versatile properties of ZnONPs have been explored in various targeted applications, including electrical conductivity, antibacterial and antifungal activities, catalytic functions, and use in the cosmetic industry 20 Green manufacturing of ZnO nanoparticles using plant extracts can increase these properties, 21 for example, Shnawa et al (2023) 22 evaluated the antioxidant and antibacterial qualities of ZnONPs made from aqueous Ziziphus spina-christi leaf extract and found that the ZnONPs exhibited concentration-dependent antibacterial activity against tested microorganisms. Alghamdi et al (2023), 23 showed higher antioxidant and antibacterial activites of ZnONPs synthesized using C. argentea extracts compared to ZnONPs. Additionally, ZnO nanoparticles biosynthesized using P. indica leaf extract showed highest antibacterial activity properties. 24
P. lanceolata, often called Buckhorn, English Plantain, Narrowleaf Plantain, or Ribwort Plantain, is one of Plantaginaceae family used as traditional medicinal plants. 25 This herb is widely used in both formal and conventional medical systems because of its well-known medicinal properties. 26 Numerous bioactive chemical elements of P. lanceolata, such as iridoid glycosides, polyphenols, polysaccharides, terpenoids, alkaloids, tannins, flavonoids, as well as fatty acids have been found by ethnobotanical investigations and contribute to its therapeutic properties. 27 P. lanceolata possesses a range of biological characteristics, including as antibacterial, anti-inflammatory, wound-healing, antiulcerogenic, cytotoxic effects, anti-cance. 28 However, ZnO nanoparticles mediated by P. lanceolata and their potential antioxidant, antibacterial, and photocatalytic activity have not been studied, despite the fact that P. lanceolate has been employed for a variety of therapeutic purposes. Therefore, the current study uses zinc acetate as starting material and P. lanceolata leaf extracts as a stabilizing and reducing agent to synthesize ZnONPs was carried out for the purpose of antioxidant, antibacterial and Photocatalytic activities which is used for Ecofriendly application in wastewater treatment such as photocatalytic degradation of pollutants like methylene blue and biomedical field such as combating bacterial infections and oxidative related condition.
Material and Methods
Plant Collection
The leaves of P. lanceolata were collected in the South Gondar zone of the Amhara area, which is roughly 588 kilometers from Addis Ababa, Ethiopia's capital. Dr Melkamu Wondaferah of the Botany Department at Addis Ababa University in Addis Ababa, Ethiopia, taxonomically recognized the plant components that were gathered. The National Herbarium was given voucher number 4076 after receiving voucher specimens of the collected plant materials. The healthy leaves were gathered and thoroughly cleansed with tap water to get rid of any dust particles from the leaf of P. lanceolata. The leaves were cleaned with tap water and then let to air dry for 15 to 20 days. Using a mortar and pestle, the dried P. lanceolata leaves were ground into a fine powder and kept in an airtight container for later usage at room temperature based on the procedures used by Shankar, et al (2004) with some modification. 29
Preparation of Plant Extract
The chemical composition of the P. lanceolata leaf extract including major phytochemicals like flavonoids, phenolics, alkaloids, and terpenoids, has been detailed in our previous studies.28,30 The plant extraction from the leaf P. lanceolata was carried out based on the methods used by Shankar, et al (2004). 29 100 ml of distilled water were added to a conical flask containing 8 grams of finely ground P. lanceolata leaves. The flask was then heated continuously at 60 °C on a hotplate for 10 min to facilitate the extraction of bioactive compounds. To prevent contamination and evaporation during heating, the mouth of the flask was tightly sealed with aluminum foil. The mixture was heated and then left to cool naturally to room temperature. To get rid of particles and create a clean solution, the cooled extract was filtered using Whatman filter paper number 1. The filtrate was stored at room temperature in a sterile, airtight container for further laboratory activities. These settings were used to retain the stability and bioactivity of this aqueous extract for use in pharmacological testing, components. Studies showed that the aqueous extraction of bioactive compounds from plant can enhance biological functions. 31
Biosynthesis of Zinc Oxide Nanoparticles
Biosynthesis of ZnONPs was carried out standard methods based by Khajuria et al (2025) 32 and Hong et al (2006). 33 A conical flask containing 60 ml of P. lanceolata extract was placed on a hot plate equipped with a magnetic stirrer and heated to 75 oC with continuous stirring for 20 min to ensure uniform mixing (Figure 1). A dropwise addition of a 20 mM zinc acetate solution was made to the extract while the temperature was maintained at 70 °C. ZnONPs production was aided by the exact pH adjustment of the reaction mixture using an aqueous solution of sodium hydroxide (NaOH). The gradual appearance of turbidity in the solution showed that ZnONPs were produced by the extract. After the reaction was finished, the nanoparticles were separated from the supernatant by centrifuging the precipitate for ten minutes at 4000 rpm. To eliminate any remaining moisture, the collected pellet was subsequently dried in an oven set to 70 °C. The dried P. lanceolata leaf extract-mediated ZnONPs (P-ZnONPs) were then kept in airtight, sealed containers to avoid contamination and guarantee stability for characterization, antioxidant, antibacterial and photocatalytic activities (Figure 1).

Synthesis of ZnO nanoparticles with leaf extract from P. lanceolata and subsequent assessment of their biological activity.
Characterization of Nanoparticles
UV–Vis Absorption
The UV-Vis absorbance spectrum of the P-ZnONPs was recorded using a UV-Vis spectrophotometer (JASCO V-670). The measurements revealed a prominent absorption peak in the range of 300–400 nm, which served as a key indicator for the successful synthesis of P-ZnONPs and provided essential insights into the formation of nanoparticle process. 34 The formation of nanoparticles was verified by visual indicators, such as a noticeable color change in the reaction mixture.
Transmission Electron Microscopic (TEM) study
TEM analysis was performed on the powder particles at an accelerating voltage of 200 kV, achieving a resolution of approximately 1 Å. For this purpose, the p-ZnONPs sample was dispersed in triple distilled water (TDW) using a probe sonicator. Subsequently, a droplet of the resulting suspension was deposited onto a carbon-coated copper grid and left to air-dry at room temperature before conducting the TEM characterization.
Fourier Transform Infrared Spectroscopy
Fourier Transform Infrared Spectroscopy (FTIR) spectroscopy was performed to characterize the important functional group used for reducing and capping agent of p-ZnONPs. The p-ZnONPs FTIR spectra were collected using a 400–4000 cm−1 wavenumber range using an FTIR spectrometer. FTIR analysis was carried out using an IS50 FTIR spectrometer. 35
X-ray Diffraction (XRD)
The crystallite size and morphology of the P-ZnONPs synthesized from zinc acetate were evaluated using XRD diffractometer equipped with monochromatic Cu Kα radiation (λ = 1.54056 Å) over a 2θ range of 5°–80°. The crystallite size was determined by applying the Scherrer equation (Equation 1).
36
Where D is a crystallite's size in Å, θ is the scattering angle in radians, k is Scherer's constant with a value of 0.9, λ (1.54178 Ao) is the wavelength of X-ray radiation, and β is the integral breadth of the maximum full width (FWHM).
Antioxidant Activity
The antioxidant activities of p-ZnONPs were performed using DPPH (2,2-diphenyl-1-picrylhydrazyl) free radical assay using a methods used Blois
37
and Desmarchelier et al
38
Upon addition of a hydrogen-donating substance (antioxidant material from P. lanceolate leaf extracts) to the alcoholic DPPH solution, the DPPH radical is reduced, resulting in a color change from purple to pale yellow. Accordingly, 2 mL of a freshly prepared 0.1 mM methanolic DPPH solution was mixed with 2 mL of P-ZnONPs solution at different concentrations of 25.0, 75.0, 100.0, and 125.0 μg/mL. After that, the test tubes were placed in the dark for 30 min. After incubation, the absorbance was measured at 517 nm. A concentration of ascorbic acid (AA) ranging from 25 to 125 μg was utilized as a reference or control. Through measuring the absorbance of the reaction mixture during measurement, a higher degree of free radical scavenging activities can be identifies.
39
Equation 2 was used to determine the DPPH radical scavenging properties of the samples using AA as a reference in order to evaluate their antioxidant capabilities.
Antibacterial Activity
Using the agar well diffusion method, the antibacterial susceptibility of p-ZnONPs was evaluated against four different bacteria species obtained from Gondar university which are isolated clinically such as S. abony, B. subtilis, S. aureus, and E. coli. The bacterial culture was evenly distributed across the hardened nutrient agar plates using a sterilized glass spreader dipped in the inoculum suspension. After that, a 6 mm well was made on the agar plates’ surface. After adding of the produced p-ZnONPs solution to the wells, the Petri plates were incubated for 24 h at 37 °C in order to assess the zone of inhibition. 40 In this experiment, streptomycin (10 µg/ml) served as the reference. Millimeters were used to measure the inhibitory zone's diameter and compare it to the control.
Photocatalytic Activity
UV irradiation was used to analyze the materials’ photocatalytic activity using previously reported procedures done by Fallah et al and Zhai et al41,42 depending on the photodegradation response of methylene blue (MB) dye. To investigate the photocatalytic activities of p-ZnO NPs, MB was subjected to high-intensity (365 nm, 115V-60 Hz, 2.5A, 100 W) Lamp UV irradiation. After forming a 10 ppm stock solution of MB in 500 ml of deionized water, 2 mg of different samples were added to 60 ml of a 10 ppm dye solution. Before photoirradiation, the solution was agitated with a magnetic stirrer for 15 min in the dark to form adsorption-desorption equilibrium in the system. After that, the combination was exposed to UV light for 15, 30, 45, and 75 min. After removing the testing samples, including p-ZnONp, from the colloidal mixture, the cleaned supernatant was produced by centrifuging at 5000 rpm for 15 min. The absorbance spectra of the supernatant from 350 to 850 nm wavelength were then measured using the Perkin Elmer UV-Vis spectroscope (USA). Using equation 3, the dye concentration during the degradation process was determined at 660 nm absorbance value.
Statistical Analysis
Origin 8 software was utilized to evaluate variance using a one-way ANOVA. The standard deviation (SD) and average of the three runs of each measurement, which were performed three times (n = 3), are displayed. Significance was determined using P ≤ 0.05.
Result
The aqueous extract of P. lanceolata leaves contains glucosides, flavonoids, tannins, phenolic compounds, alkaloids, saponins, as well as steroids from our previous research work.28,30 These phytochemicals possess P. lanceolata to have different pharmaceutical activities such as lipid peroxidation, metal ion reduction abilities, antibacterial, free radical scavenging or antioxidant, and stabilization abilities.28,30 In this study, phytochemicals obtained from aqueous leaf extract of P. lanceolata was utilized to cap Zn²+ ions, thereby promoting the nucleation of ZnONPs. The synthesized ZnONPs were then characterized employing various analytical techniques and evaluated for their potential photocatalytic, antioxidant, and antibacterial activities.
UV-Vis spectrum
A visible color change in the reaction mixture confirmed the biosynthesis of ZnONPs, which transitioned from white at the beginning to yellow at the end, indicating the formation of ZnONPs. The UV-Vis absorption spectrum (Figure 2) shows the synthesis of ZnONPs with zinc acetate as the zinc source, revealing a discernible absorption peak at a maximum wavelength of 370 nm, characteristic of ZnO nanoparticles. 35 The presence of ZnONPs was confirmed by a large absorption peak between 343 and 347 nm in the UV-Vis spectra. The optical band gap energy of the produced ZnO nanoparticles was estimated using a Tauc plot generated from the absorbance data, ranging from 3.57 to 3.61 eV. 43
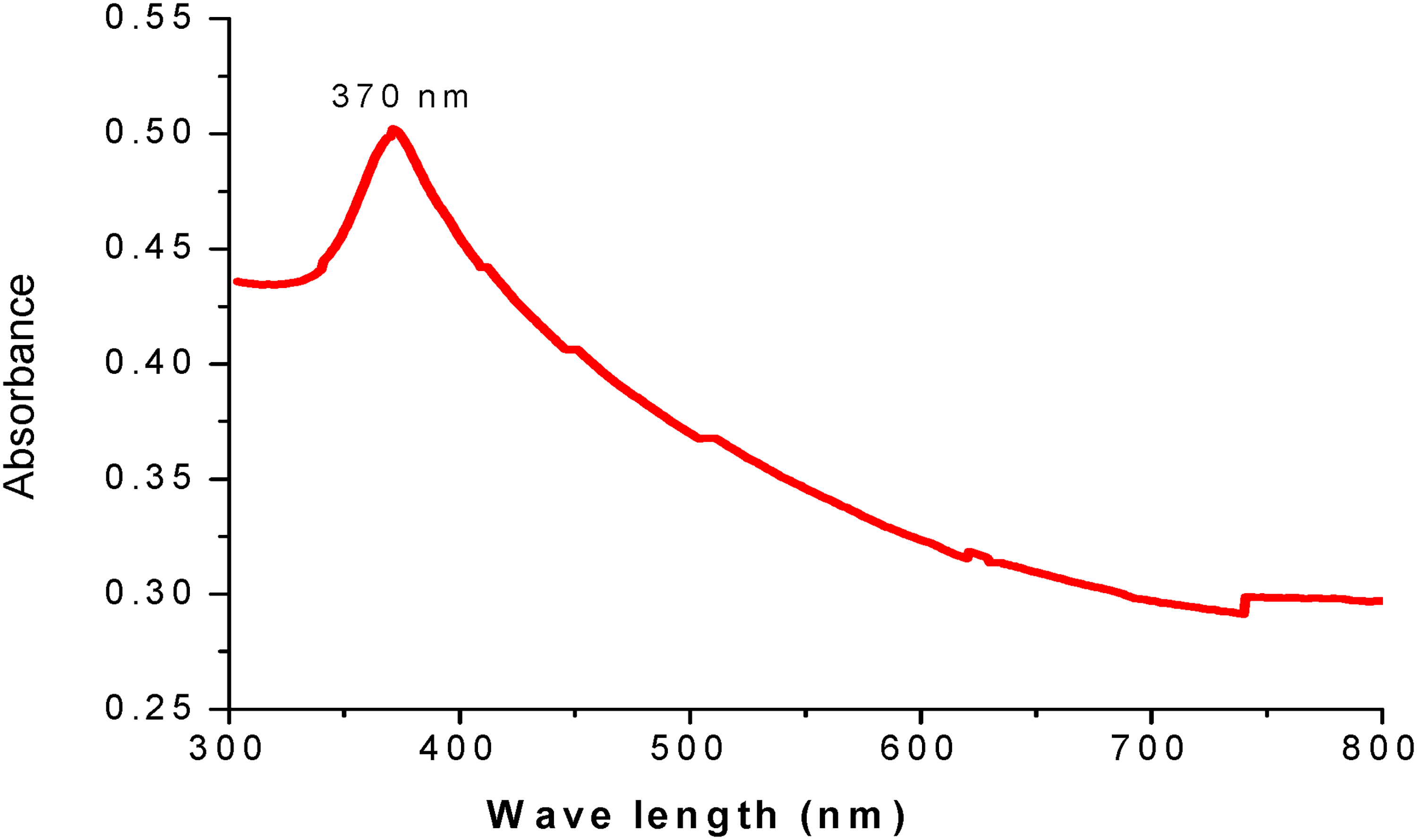
UV-VIS spectra for P. lanceolata mediated ZnONPs.
X-Ray Diffraction (XRD) Analysis
XRD is a powerful technique used to determine the crystal structure of materials by measuring interatomic distances, bond angles, and atomic arrangements within crystalline solids. 44 The XRD pattern of the synthesized p-ZnONPs (Figure 3) clearly demonstrates the crystalline nature of the leaf-mediated ZnONPs prepared using zinc acetate as the precursor. Six distinct diffraction peaks are observed at 2θ values of 32.519°, 34.380°, 36.487°, 47.810°, 56.737°, and 62.727° (Figure 3). Additionally, notable peaks are revealed at 32.837°, 32.962°, 36.514°, 47.712°, 56.921°, and 63.230°. These diffraction peaks were indexed to the hexagonal wurtzite crystal structure of ZnO, corresponding to the lattice planes (hkl) of (100), (002), (101), (102), (110), (103), and (112). This indexing is consistent with the standard reference pattern from the Joint Committee on Powder Diffraction Standards (JCPDS) card number 36-1451, confirming the successful synthesis of crystalline ZnONPs with the expected crystal structure. 45 The most intense signal at (101) was used to determine the average size of the ZnO nanoparticles using the Debye-Scherrer Equation.46,47 The average crystallite size of the ZnONPs produced using zinc acetate as the precursor was 23 nm. The ZnONPs synthesized via zinc acetate display a hexagonal wurtzite structure.
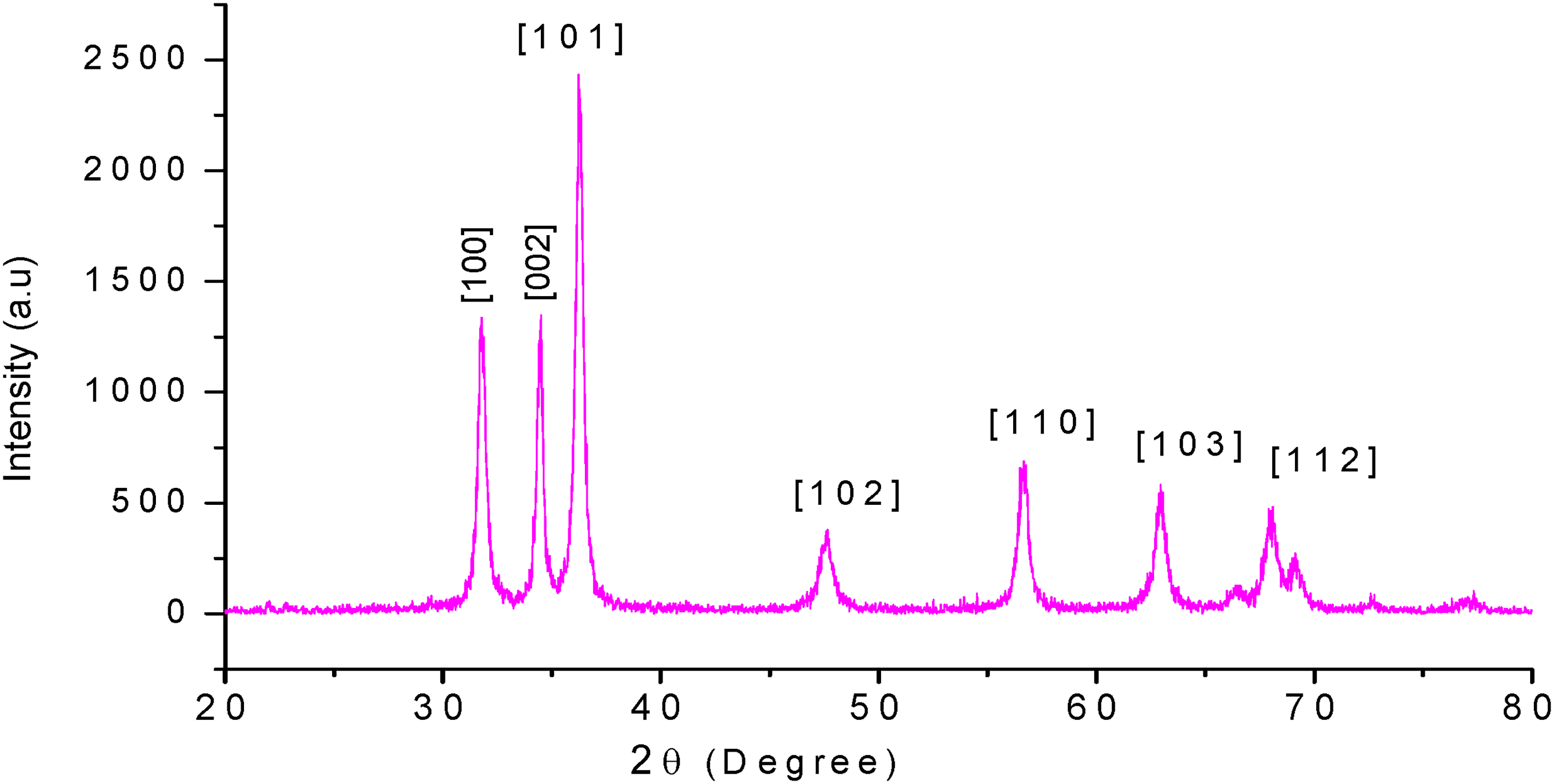
Different XRD peaks of P. lanceolata mediated ZnONPs.
DLS Analysis
For the synthesized p-ZnONPs, a zeta potential of −30.5 mV (Figure 4) was recorded, indicating good colloidal stability. A comparable result was obtained by Rajeshkumar et al (2018), 48 who reported a zeta potential of approximately −32 mV for ZnONPs synthesized using S. album leaf extract, demonstrating similar stability and surface charge characteristics. These results are in line with earlier research on biosynthesized ZnONPs using C. verrucosa leaf extract with a zeta potential value of −21 mV, and also agree with green synthesis of ZnONPs from S. album leaf extract and using C. halicacabum leaf extract. 49

Zeta potential (mV) for P. lanceolata mediated ZnONPs.
Transmission Electron Microscopy (TEM)
Transmission Electron Microscopy (TEM) is used to analysis both morphology and size distribution of the synthesized p-ZnONPs (Figure 5). The TEM image revealed predominantly irregular shapes, with an average particle size of approximately 23 nm. By comparison, ZnONPs synthesized using P. indica leaf extract exhibited spherical shapes and smaller dimensions, averaging 12.00 nm, as reported by Al-Askar et al (2023). 50 On the other hand, bigger ZnONPs with an average size of 46.6 nm and irregular forms were noted by Mishra et al (2023). 51

TEM image of p-ZnONPs using zinc acetate mediated nanoparticles (a) and their size distribution (b).
Fourier-Transform Infrared (FT-IR) Spectroscopy
FT-IR spectroscopy was employed to analyze the synthesized P-ZnONPs, revealing multiple distinct peaks corresponding to various functional groups (Figure 6). A strong absorption band observed at 3400 cm−¹ is attributed to O–H stretching vibrations, indicating the presence of structural polymeric relationships. 52 The stretching vibrations of aldehydic C-H and alkane C-H are responsible for the peaks at 2975 cm−¹. 53 The absorption at 1625 cm−¹ signifies C = O stretching vibrations, 54 while bending vibrations of methyl and methylene groups appear at 1405 cm−¹ and 1430 cm−¹, 54 respectively. Additionally, peaks at 1237 cm−¹ and 1050 cm−¹ are assigned to the C–OH and C–C stretching vibrations of carboxylic acids. 55 The peak at 660 cm−¹ is caused by the aromatic C = C vibrations. Absorptions that fall between 600 and 400 cm−¹ are common Zn–O vibrations. 56 The absence of peaks in PLLE and presence of peaks at 557 and 453 indicated that the synthesis of ZnONPs occurred on the surface of PLLE.

FT-IR peaks of P. lanceolata mediated ZnO NPs.
Application of P-ZnONPs
Photocatalytic Activities of Biosynthesis ZnONPs
The photocatalytic activity of p-ZnONPs1, p-ZnONPs2, p-ZnONPs3, p-ZnONPs4, and CCNC/ZnO5 (Figure 7) was evaluated through the solid-phase photodegradation of MB dye by monitoring its degradation under UV radiation over time. A blank test without any photocatalyst was conducted to establish a baseline and determine the extent of MB degradation caused solely by UV light. Figure 7a illustrates the photocatalytic activities of the p-ZnONPs samples over a 75-min irradiation period, showing that MB degradation increases with higher concentrations of p-ZnONPs in the order of p-ZnONPs1 to p-ZnONPs5. For p-ZnONPs1, p-ZnONPs2, p-ZnONPs3, p-ZnONPs4, and p-ZnONPs5, the corresponding absorption peak of MB at 2.27 gradually drops to 1.95, 1.58, 1.16, 0.79, and 0.53, respectively. The length of UV light exposure has a major impact on MB deterioration, as seen in Figure 7b. The UV absorbance of the p-ZnONPs sample was monitored at 15-min intervals for a total of 75 min. The absorbance of P-ZnONPs5 after 75 min was significantly lower than that after 15 min (Figure 7). The result indicated that the absorbance decreased steadily with increasing irradiation time from 15 min to 75 min. The decrease in absorbance of a sample indicated the gradual degradation of the MB dye.

UV-Vis spectrum alterations in the MB using the p-ZnONPs at varied concentration p-ZnONPs1, p-ZnONPs2, p-ZnONPs3, p-ZnONPs4 and p-ZnONPs5 (a) and UV-Vis light exposure time (0, 15, 30, 45, 60, and 75 min) for p-ZnONP5(b).
The rates of MB dye degradation by p-ZnONPs at different concentrations, ranging from 100 µg/ml to 500 µg/ml, are shown in Figure 8. With increasing ZnONPs concentration, the degradation efficiency rises dramatically, reaching 76.65% at 500 µg/ml (p-ZnONPs5) from 14.09% at 100 µg/ml (p-ZnONPs1). The degradation rate of methylene blue (MB) increases significantly with reaction time when treated with p-ZnONPs, starting from a minimal 3.42% at 15 min and reaching 60.27% at 75 min (Figure 8), indicating substantial dye breakdown over time. Using ZnONPs made from an aqueous leaf extract of S. acuta, Ramesh et al 57 obtained a photocatalyst for dye degradation of 70.25% in 40 min of exposure to sunshine; this is less than the current work.

Percentage degradation rate of MB due to concentration and reaction time variation.
Antioxidant Activities
The antioxidant properties of the green-synthesized p-ZnONPs was evaluated by measuring the decrease in DPPH absorbance at 517 nm, which reflects their capacity to scavenge free radicals generated by DPPH. 58 The observed shift from blue at the beginning to yellow at the end showed the nanoparticles’ antioxidant activities. In this study, the DPPH technique was used to measure the antioxidant activity of phytochemical-assisted biosynthesized p-ZnONPs. 59 Methanol was used as a solvent, and DPPH was used as a free radical, which gave a violet color to methanol. When biosynthesized p-ZnONPs and leaf extract are combined in an alcoholic solution, the violet color of DPPH is reduced; if the antioxidant property is high, then it changes to yellow after 30 min. Alcoholic solutions were prepared by combining ascorbic acid (AA), P. lanceolata leaf extract (PLLE), and biosynthesized p-ZnONPs, with concentrations of p-ZnONPs adjusted to 100, 200, 300, 400, and 500 µg/ml, respectively. The resulting solutions were then labeled as p-ZnONPs1, p-ZnONPs2, p-ZnONPs3, p-ZnONPs4, and p-ZnONPs5, respectively (Figure 9b). An equal quantity of DPPH was then added for the same duration. The percentage inhibition of DPPH for ascorbic acid (AA), PLLE, and biosynthesized p-ZnONPs samples at different concentrations is shown in Figure 9a. The percentage inhibition for biosynthesized p-ZnONPs were 50.23 ± 1.74 for p-ZnONPs1 and 90.31 ± 0.51 for p-ZnONPs5; however, for PLLE the percentage inhibition was 20.23 ± 0.21 at 100 µg/mL and 50.11 ± 0.21 at 500 µg/mL, which are less than those of biosynthesized p-ZnONPs samples. According to Figure 9, the inhibition percentages for AA at comparable concentrations were 58.23 ± 0.23 and 96.23 ± 0.43, respectively. The results presented in Figure 9 clearly indicate that the antioxidant activity of biosynthesized p-ZnONPs is concentration-dependent, with higher concentrations exhibiting greater activity.

Antioxidant power of p-ZnONPs samples where A = DPPH, B = AA, D = p-ZnO NPs5, E = p-ZnONPs4, F = p-ZnONPs3, G = p-ZnONPs2, H = p-ZnONPs1 and I = PLLE at 500 μg/mL.
Figure 10 shows the IC50 value for biosynthesized p-ZnONPs samples. A lower IC50 value indicates better antioxidant activity against DPPH free radicals. Our results showed that PLLE had an IC50 value of 495.5 μg/mL, which is significantly higher than the biosynthesized p-ZnONPs, with an IC50 value of 76.24 μg/mL, as shown in Figure 10. This means that biosynthesized p-ZnONPs have better antioxidant properties than PLLE. In comparison, other studies found varying IC50 values for ZnONPs made with different leaf extracts. The ZnONPs made with T. heyneana Wall. leaf extract had a much higher IC50 value (89.47-185.8 µg/ml) than biosynthesized p-ZnONPs. On the other hand, the ZnONPs made with P. lentum and A. altissima leaf extract had similar IC50 values (83.79 and 78.23 µg/mL) compared to biosynthesized ZnONPs. However, the ZnONPs made with C. dicoccum (L.) extract had a much lower IC50 value (32.85) than biosynthesized p-ZnONPs, indicating even better antioxidant properties.60–63 Alghamdi et al (2023) 23 showed a low IC50 value of C. argentea mediated ZnONPs with an IC50 value of 91.24 mg/ml, comparable to p-ZnONPs. Compared to well-known standard chemicals like ascorbic acid (AA) (IC50 = 26.11 mg/ml), p-ZnONPs has a higher IC50 value. By employing aqueous extracts of A. sativum, R. officinalis, and O. basilicum, Stan et al (2015) 64 produced ZnONPs by a both chemicals and biological methods (coprecipitation). The outcome demonstrated that ZnONPs made with extracts from the chosen plant species had higher antioxidant activity than ZnONPs made chemically. In the work of Dousti et al (2021), 65 when the green-produced ZnONPs mediated by F. parviflora had an IC50 value of 30.86 μg/ml, their ability to scavenge DPPH radicals enhanced. As the ZnONPs concentration climbed, the scavenging activity displayed a dose-dependent pattern.

IC50 value for biosynthesized p-ZnONPs.
Antibacterial Activity
The antibacterial activity of p-ZnONPs against four bacterial species—E. coli, K. pneumoniae, S. aureus, and S. epidermidis—was evaluated using the agar well diffusion method, with Tetracycline (TTC) as a positive control. The TTC control exhibited a consistent maximum zone of inhibition Zone of inhibition (ZOI) of approximately 28.00 ± 0.00 mm across all species. The ZnONPs samples (p-ZnONPs1 to p-ZnONPs4) demonstrated varying antibacterial effects depending on the bacterial species and nanoparticle formulation, as detailed in Table 1 and Figure 11. Notably, p-ZnONPs showed markedly different ZOI values compared to pure PLLE.

The zone of inhibition that ZnONPs against S. epidermidis (A), S. aureus (B), E. coli (C) and K. pneumonia (D). Where p-ZnONPs1(a), p-ZnONPs2(b), p-ZnONPs3 (c) and p-ZnONPs4(d).
The Zone of Inhibition That p-ZnONPs Against S. aureus, K. pneumonia, S. epidermidis, and E. coli.

The mean value ± SD was utilized after more than three examinations of each data set. Subscriptions a and b have values in the same column that differ significantly (P ≤ 0.05).
For E. coli, ZOI increased progressively from ZnONPs1 (17.21 ± 1.11 mm) to ZnONPs4 (21.21 ± 1.15 mm), though all remained below the TTC control. S. aureus displayed the strongest inhibition with p-ZnONPs1 (13.44 ± 1.10 mm), while ZnONPs4 achieved levels comparable to TTC. In S. epidermidis, p-ZnONPs1 and p-ZnONPs2 yielded lower ZOI values (∼17.55 to 18.11 mm), whereas p-ZnONPs3 and p-ZnONPs4 showed higher inhibition (23.18 to 25.34 mm), still below TTC. For K. pneumoniae, p-ZnONPs2, p-ZnONPs3, and p-ZnONPs4 produced ZOI values ranging from 26.55 to 30.1 mm, with some exceeding the TTC control.
Discussion
Based on their size and functionalization, nanoparticles have the rare capacity to change the properties of larger materials. 66 The bioactive compounds from P. lanceolata such as flavonoids and phenolic compounds exhibit versatile chelating and reducing properties, enabling them to cap Zn²+ ions and facilitate ZnO nucleation. Phenolic compounds form multiple chelating bonds that stabilize the ZnO nanoparticles after nucleation, whereas flavonoid groups initiate the capping process on Zn²+ ions, leading to the formation of nanoparticles with diverse size distributions. This approach enhances the physical and chemical properties of semiconducting ZnONPs. These findings align with prior studies, including Sharmila et al (2019), 12 who examined the capacity of T. castanifolia to synthesize ZnONPs, and Abdelbaky et al (2022), 67 who reported a green synthesis of ZnONPs using P. odoratissimum as a mediator, characterized via UV-Vis spectrophotometry, with an absorbance peak at approximately 370 nm. Additionally, Nozipho et al (2024) 7 confirmed that M. pruriens mediates the formation of ZnONPs by observing a high absorption peak at 350 nm. Electronic transitions from the valence band, which is predominantly made up of oxygen 2p orbitals (O2p), to the conduction band, which is primarily made up of zinc 3d orbitals (Zn 3d), are represented by this band gap. This absorption characteristic is consistent with previous research result on synthesis of ZnONPs.68,69
XRD is a powerful technique used to determine the crystal structure of materials by measuring interatomic distances, bond angles, and atomic arrangements within crystalline solids. The final particle size roughly matched results from previous research result on synthesis of ZnONPs.68,69 This confirms the purity and structural integrity of the biosynthesized nanoparticles, highlighting the effectiveness of the green synthesis method using P. lanceolata extract in producing well-defined ZnO nanostructures suitable for various applications.
The smaller size of P-ZnONPs is due to stabilized effect of phytochemicals present in P. lanceolata extract leaf extract. 50 Research work in Al-Askar et al (2023) 50 confirmed that, the smaller size and more uniform spherical shape of ZnONPs might have been caused by differences in the phytochemical composition of the plant extract, which influences the stability and reduction kinetics of nanoparticles. Furthermore, synthesis parameters like temperature, pH, and raw material concentration can have a significant impact on particle size and shape, which could assist to explain the observed discrepancies.70,71 The observed variation in nanoparticle characteristics could arise from differences in synthesis conditions or the specific types of biological agents employed. Additionally, less efficient capping or stabilization provided by the components of the plant extract may contribute to larger particle sizes and irregular morphologies, potentially leading to aggregation or uncontrolled growth.70,71 The variety of phytochemicals found in various plant extracts, which can function as both capping and reducing agents and impact nucleation rates, growth mechanisms, and particle stabilization, is a major contributor to this variability. This emphasizes how crucial it is to optimize synthesis conditions in order to obtain the necessary nanoparticle qualities for use in biomedicine, environmental remediation, and catalysis.
The stability and surface charge of p-ZnONPs are effectively indicated by their zeta potential values (−30.5 mV) (Figure 4). The hydroxyl groups found in bioactive compounds in the P. lanceolata leaf extract are mainly responsible for the negative charge, 30 which inhibits aggregation and improves dispersion stability. The reducing agents included in the leaf extract, such as the flavonoid and phenolic components are probably responsible for the negative charge potential of the produced ZnONPs. 30 The key information found in Zeta potential provides the stability and charge distribution of the produced nanoparticles. As crucial factors for possible uses in environmental cleanup, biological areas, and catalysis, the current study emphasizes the importance of surface charge distribution and nanoparticle durability. the result showed that, the green synthesis method employing P. lanceolata extract provides a sustainable means to synthesize ZnONPs with desired physicochemical characteristics by guaranteeing stable and evenly distributed nanoparticles.
The FTIR peaks showed that phytochemicals such as polyphenols, flavonoids, and carboxylic acids are responsible as stabilizing and capping agents and are essential for the production of P- ZnONPs. The stability and functionality of P-ZnONPs depend on the chemical interactions and bonding during their synthesis. 72 This study offers valuable insights into these processes, highlighting how plant-derived compounds facilitate green synthesis and enhance nanoparticle properties for potential applications in environmental and biomedical fields.
Photocatalytic activities of bio-Fabricated ZnONPs as indicated in Figure 7 measured based on UV absorption. A lower absorption peak denotes higher degradation. The trend reflects the ZnO concentration in each sample, as increased ZnO concentration increases the generation of electron-hole pairs (e− and h+) under UV irradiation. These charge carriers interact with oxygen (O2) and water (H2O) molecules adsorbed on the catalyst surface to produce reactive oxygen species such as hydroxyl radicals (•OH) and superoxide anions (O2−), which are highly effective in the oxidative breakdown of MB dye molecules. Consequently, higher ZnO concentrations lead to increased production of these reactive species, resulting in more efficient photocatalytic degradation of MB.73–75 This is due to the enhanced photocatalytic activity of the ZnONPs, which promotes the generation of reactive oxygen species (O2− and •OH) under prolonged UV exposure, leading to more efficient breakdown of the MB dye. The gradual generation of reactive species accounts for the observed increase in the degradation rate as the reaction progressed. These findings underscore the potential of bio-fabricated ZnONPs for efficient dye degradation in wastewater treatment applications.
Higher ZnONPs concentrations offer more active sites for photocatalytic processes, which increases the production of reactive oxygen species (ROS) such as superoxide anions and hydroxyl radicals, which more efficiently break down the dye molecules. Because there are fewer nanoparticles available at lower concentrations, there is less interaction with MB molecules and less photocatalytic activity. Higher concentrations of nanoparticles also enhance light absorption and raise the likelihood of electron-hole pair formation, which increases degradation efficiency. The plant extract used in the synthesis acts as a reducing and stabilizing agent, enhancing the surface properties of p-ZnONPs through biofunctionalization, which improves the adsorption of MB molecules and facilitates efficient generation of reactive oxygen species (ROS) such as hydroxyl radicals (•OH) and superoxide anions (O2•−) under light exposure. These ROS are responsible for attacking and degrading the dye molecules into less harmful substances. Longer reaction times allow for increased ROS production and more extensive interaction between the dye and nanoparticles, resulting in enhanced photocatalytic activity. Based on the preceding observations, a potential mechanism for the photocatalytic performance of p-ZnONPs involves ZnO absorbing ultraviolet light to generate photogenerated electron-hole pairs. The resulting photogenerated hole (h+) and electron (e−) could go to the p-ZnONPs surface, interacting with both H2O and O2 to produce O2− and •OH, which could take part in the direct oxidation that leads to dye degradation.42,76–78 This mechanism highlights the superior performance of the biosynthesized ZnONPs compared to other studies, suggesting their potential for advanced environmental remediation applications.
Antioxidants help prevent oxidative damage by donating electrons to free radicals, thus neutralizing their detrimental effects on biological processes. As observed in Figure 9, antioxidant chemicals transform the violet-colored DPPH radical into its yellow form by donating hydrogen atoms or electrons, resulting in DPPH-H. This color change is monitored spectrophotometrically to assess the scavenging activity of the synthesized p-ZnONPs. 7 s shown in Figure 9, the p-ZnONPs exhibit enhanced antioxidant potential compared to the P. lanceolata leaf extract, underscoring the effectiveness of nanoparticle synthesis in boosting antioxidant activities. The antioxidant activities performed in using ZnONPs mediated by Mallotus philippinens done by Khajuria et al (2025). 32 They indicated that They indicated that the ZnONPs synthesized using M. philippinensis extract demonstrated superior antioxidant capacity, as evidenced by higher DPPH radical scavenging activity (up to 84.05% at 125 µg concentration) and enhanced reducing power compared to the crude extract alone, attributed to the nanoparticles’ increased surface area and bioactive compound stabilization. However, when compared to the standard ascorbic acid, the biosynthesized p-ZnONPs exhibit relatively lower antioxidant activity. This suggests that while the green synthesis enhances the extract's properties, further optimization may be needed to match or exceed standard antioxidants, potentially through refining synthesis parameters or exploring synergistic effects for broader biomedical applications.
The significantly lower IC50 value of biosynthesized p-ZnONPs (76.24 μg/mL) compared to PLLE (495.5 μg/mL) underscores the enhancement in antioxidant efficacy achieved through nanoparticle formulation, likely due to increased surface area and biofunctionalization from plant phytochemicals. This improvement aligns with trends in green synthesis, where biological methods often yield nanoparticles with superior radical scavenging compared to chemical approaches, as evidenced by Stan et al (2015). 64 However, the higher IC50 of p-ZnONPs relative to ascorbic acid (26.11 mg/ml) and some plant-mediated ZnONPs such as C. dicoccum at 32.85 µg/mL suggests room for optimization, possibly through adjusting synthesis parameters or combining extracts.60–63 The dose-dependent scavenging activity observed in Dousti et al's 65 study reinforces the concentration-dependent behavior, highlighting the potential of these nanoparticles for therapeutic applications in oxidative stress-related conditions, while comparative analyses emphasize the variability influenced by plant source and synthesis method. 79
Regarding the antibacterial activities of P-ZnONPs, the findings indicate that green-synthesized p-ZnONPs exhibit substantial antibacterial effects. As observed in Table 1, Gram-negative bacteria such as E. coli and K. pneumoniae generally show larger ZOI compared to Gram-positive strains like S. aureus and S. epidermidis. The progressive increase in activity from p-ZnONPs1 to p-ZnONPs4 suggests that optimizing nanoparticle properties enhances their effectiveness. These results align with studies by Khajuria et al (2025) 32 and Mushtaq et al (2023), 80 who reported enhanced antibacterial activity of green-synthesized ZnONPs against E. coli, S. aureus, and P. aeruginosa (increases of 22%, 66%, and 44%, respectively) compared to herbal extracts. However, the current research shows higher inhibition values than those reported by Ihsan et al (2023) 81 for C. zedoaria (18-19 mm) and by Dousti et al (2021) 65 for ZnONPs synthesized using F. parviflora extract (24.6 ± 0.72 mm for S. aureus and 13.2 ± 0.81 mm for E. coli).
The stronger activity against gram-negative bacteria is likely due to the nanoparticles’ large surface area, facilitating better interaction and bactericidal effects. 82 Differences in susceptibility may stem from cell wall structures. Gram-positive bacteria have thicker peptidoglycan layers, while gram-negative have a single layer. Nanoparticles bind to negatively charged bacterial cell walls via electrostatic attraction, disrupting permeability and integrity. 83 ZOI exceeding TTC for K. pneumoniae is one example of the observed variability that could point to bacterial resistance or formulation adaptation. These findings demonstrate the potential of green-synthesized p-ZnONPs as antibacterial agents, indicating the need for additional research into their mechanisms and patterns of resistance.
Researchers have suggested several potential antibacterial mechanisms for the interaction of ZnO NPs with bacteria. There are hypotheses that smaller NPs’ enhanced surface reactivity and faster cell penetration cause them to release Zn2+. The release of Zn2+ from ZnONPs, which is known to impede a number of bacterial cell functions, including active transport, metabolism, and enzyme activity, is a basic idea behind antibacterial actions. 84 ZnONPs emit Zn2+ ions, which adhere to and harm the bacterial cell wall. Additionally, it disrupts the bacterial cell membrane's layer of lipids, proteins, and carbohydrates. Additionally, DNA damage brought on by Zn2+ ion attacks may cause bacteria to die. Their attack breaks down DNA, which stops bacteria from growing and kills them. The nanoscale dimensions of the material enhance its surface area, thereby amplifying its antibacterial activity, while the small size facilitates easy entry into cells. 85 The generation of reactive oxygen species (ROS), which causes oxidative stress and cell damage or death, is the source of the other suggested antibacterial activity. ROS production is frequently used by ZnONPs as an antibacterial activities. 86 Upon accumulating on the surfaces of bacterial cell walls, the nanoparticles penetrate the bacterial interior via various protein channels, where they become activated and initiate the generation of reactive oxygen species (ROS) and metal ions. High ROS production finally destroys the integrity of the cell wall and disrupts the nucleoids of bacterial cells.87,88 The deposition of Zn2+ ions in the cytoplasm and cell membranes, as well as the interaction of ROS or hydrogen peroxides (H2O2) with bacterial cell membranes, may be the reason why ZnONPs exhibit their antibacterial activity. They generate H2O2, which is mixed with bacterial proteins and lipid bilayers. The oxidative damage in the cellular component may be caused by H2O2 in the cytoplasm. 89 As Fan et al (2021) 90 showed in their finding the size and composition of nanoparticles can affect their toxicity. Because of their small size, nanoparticles may be transported quickly and easily, which increases ROS generation and nanotoxicity. 91 The lethal activity of ZnONPs due to their attachment to bacterial cell membranes and accumulation inside the cytoplasm, which damages the integrity of the cell membrane and causes the leakage of cell contents, ultimately leading to cell death, is another suggested explanation. 88
Limitation of the Study
While this study effectively demonstrates the green synthesis of ZnONPs using P. lanceolata leaf extract and their photocatalytic, antioxidant and antibacterial activities, the following limitations exist. The in vitro focus restricts insights into in vivo performance, bioavailability, and toxicity. Photocatalytic reusability, radical trapping experiment and broader bacterial strain testing were omitted. Limiting mechanistic and practical understanding. Narrow concentration or time ranges may overlook optimal conditions and the absence of cytotoxicity or environmental impact studies raises safety concerns. These gaps highlight opportunities for future research to enhance applicability.
Conclusion and Recommendations
The aqueous leaf extract of P. lanceolata was used as a natural reducing and stabilizing agent in this study to successfully demonstrate the green production of ZnONPs, with zinc acetate acting as the zinc precursor. XRD and TEM investigations verified that the biosynthesized ZnONPs had a hexagonal wurtzite crystalline structure with an average particle size of roughly 23 nm. The presence of bioactive compound such as polyphenols and flavonoids on the nanoparticle surface was verified by FTIR, indicating their crucial role in nanoparticle formation and stabilization. Strong antioxidant activity and significant antibacterial efficacy against both Gram positive (S. aureus, S. epidermidis) and Gram negative (E. coli, K. pneumoniae) bacteria were demonstrated by the synthesized ZnONPs, with DPPH radical scavenging comparable to that of standard ascorbic acid. However, Gram-negative strains were generally more susceptible. Additionally, the ZnONPs demonstrated outstanding photocatalytic efficiency, degrading methylene blue dye by up to 91.52% in 75 min when exposed to UV light, indicating their potential for use in environmental remediation applications. The P. lanceolata extract's tiny particle size, large surface area, and efficient biofunctionalization are responsible for the increased biological and photocatalytic activities. All things considered, this economical and environmentally friendly green synthesis method presents a viable path toward the production of biocompatible ZnONPs with multifunctional qualities appropriate for environmental and medicinal uses. The practical application of these biogenic ZnONPs will be further advanced by future research concentrating on in vivo assessments and comprehensive mechanistic insights.
Footnotes
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Authors’ Contributions
Limenew Abate Worku contributed to the investigation, methodology, and software aspects of the study. Additionally, Limenew Abate Worku provided supervision throughout the research process and was responsible for writing the manuscript.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The datasets generated and/or analysed during the current study are available within the manuscript.