Abstract
Objectives
This research focuses on the green synthesis of ZnO nanostructures using
Methods
ZnO nanostructures were synthesized via a green, eco-friendly, and time-effective method. Characterizations included P-XRD for phase and crystallite size determination, FTIR spectroscopy for functional group analysis, FIB-SEM and HR-TEM for surface morphology, UV-Vis spectroscopy for optical properties, and electrochemical tests for evaluating super capacitor performance.
Results
XRD analysis confirmed the tetragonal Wurtzite hexagonal phase of ZnO nanostructures, with an average crystallite size of 35 nm calculated using the Debye-Scherrer equation. FTIR confirmed the formation of ZnO, while FIB-SEM and HR-TEM revealed a spongy, agglomerated surface morphology. UV-Vis analysis showed a reduction in the energy band gap with increasing calcination temperatures. Electrochemical analysis demonstrated that ZnO nanostructures calcinated at 580 °C exhibited superior charge/discharge capacities, cycling stability, and nearly 100% coulombic efficiency for up to 1000 cycles at a current density of 5 A/g, outperforming those prepared at 280 °C and 480 °C.
Conclusion
The biosynthesized ZnO nanostructures, especially those calcinated at 580 °C, show excellent potential as electrode materials for high-performance super capacitors due to their enhanced electrochemical performance and long-term cycling stability.
Introduction
Nanomaterials are one of the furthermost as well as fastly emerging areas in the fields of material sciences. Nanomaterial, controlled to nano-crystalline sizes (less < 100 nm), could show atomic-like properties that resulted from high surface energies because of their larger surface areas as well as broader band gaps in-between valence band(VB) as well as conduction band (CB) while they are separated to near atom-sizes. 1
Metallic oxides have tough photo-catalytic and photo-oxidizing characteristics because of the existence of several active sites as well as fabrications of hydroxyl-radicals on their superficial. 2 In the previous decades, many physical as well as chemical methods utilized in the production of metallic oxide nanoparticles have been established, comprising for the purposes of evaluating thermal decompositions, chemical vapor depositions, electro-chemical depositions, electro-phoretic depositions, co-precipitations, ultrasounds, pulsed-laser depositions, and anodization. 3 The conservative physical methods, as well as chemical approaches, are cheaper, poisonous, and lead to contaminating the environment and creating numerous drawbacks for the applications of nanomaterials. To overcome these problems, it is essential to find an eco-friendly approach for the preparation of metallic and metallic oxide nanomaterials. The biosynthesis method has begun to be a cost-effective alternative to conservative preparation techniques. Nowadays, the usages of bio-organic compounds extracted from leaf stems, flowers, fruits, or seeds as reductants have been stated by numerous scholars.4–6 Separately from the plant extract, unadulterated organic compounds attained from natural harvests like as andro-grapholide, Carboxylic-curdlan, Ellagic-acid, and geranial have also been utilized for creating reducing agents that help in the immediate reductions of metallic ions in nano-structured metallic oxides. 7 Contrary to biological entities, plant extracts are more reliable, simple, and eco-friendly compared with other biological entities in the green synthesis of nanoparticles.8,9 The plant-mediated green synthesis also offers a number of advantages compared to other biological materials for the synthesis of nanoparticles. These advantages include one-spot synthesis of nanoparticles, robustness, eco-friendliness, natural capping and reducing agents, ease of availability, safety, cost-effectiveness, suitability for large-scale synthesis, and no requirement for cell cultures. 10 The largescale production of green-synthesized nanoparticles from the lab to pilot-plant level was still in the infant stages compared with conventional methods applied.11,12
The plant extract used for the synthesis of the nanoparticles in this study is Albizia gummifera, commonly known as
Higher-capacitor nanomaterials were utilized to improve the energy storage as well as rates of capabilities of metallic oxide pseudo-capacitors. These types of active nanomaterials with novel ways of preparations are crucial for discovering surface reactions in the finding of enhanced energy storage site. 13 Oxides of transition metals, such as Ru,14,15 Mn, 16 Fe 16 as well as Sn, in several oxidation states, have been extensively investigated having to their higher pseudo-capacitances. In addition, ZnO-NPs are very auspicious among oxides having to their eco-friendliness, wide availability, as well as ideal capacitive characteristics. 17 Furthermore, these kinds of oxides have verified the ability to self-assembles in several nano-structured surface morphologies. Likewise, electrodes with of nano-structured surface morphology may display ominously improved electrochemical performances due to the smaller diffusions pathway, fast charge carriers transfer, as well as superior numbers of electro-chemically active site. 18 One thing that is new in this study is that most of the electrodes reported were prepared from transition metals which are challenging to stabilize and expensive chemicals usage. Instead of using such metal oxides, the researchers investigated easy, eco-friendly, cost and time-effective methods of synthesis of nanomaterials for this particular application.
However, it was perceived in earlier reports that the specific capacitances of metallic oxide grounded super-capacitors fall greatly within an improvement in the scan rates because of their poor electronic conductivities. Hence, it is worth adding metallic oxide to carbon-based material to manufacture the composite that can enhance the advantages of both parts as well as reduce the limitations of respective components. In this research, biosynthesized ZnO has been studied to serve as working electrode materials for high-performance super-capacitor applications. C-ZnO nanostructure was equipped by utilizing ZnO slurry as well as NMP as a solvent. In specific, ZnO nanostructures were used to create the ZnO slurries. The super-capacitor performances of C-ZnO nanostructure were tested utilizing cyclic voltammetry (CV) as well as a charging-discharging procedure at constant currents. The study aimed to prepare and characterizations of ZnO nanoparticles at various calcination temperatures using novel plant
Materials and Methods
Materials
Zinc acetate-dihydrate with 98.9% purity was purchased from Sigma (Table 1). Aldrich and were utilized as the sources. 0.3 M of zinc salt was mixed in 50 mL of DI water as well as encouraged for a few min with a magnetic stirrer. The aqueous leaf of
Chemicals Used in Present Research and Their Information.

Slurry-making Procedure
During the preparation of samples for the electrode, 20% of carbon, 10% of PVDF-(poly-vinylidene fluorides), and 70% of biosynthesized ZnO were added together and grinded into the mixtures samples by adding droplets of NMP-(N-Methyl-2-Pyrrolidones) as solvents. The biosynthesized materials were layered in 1.5 mg carbon paper and dried in an air oven at 60 °C overnight, and the prepared electrode was tested as a working electrode in the cyclic-voltammetry (CV) characterizations. The observed data were recorded in 1 M KOH as electrolytes.
Electro-chemical Measurements
The cyclic-voltammetry (CV) and galvano-static charge-discharge (GCD) measurements of C-ZnO nanostructure were accomplished in a three-electrode symmetric cell configuration accumulated into cells. The electrochemical record of the devices was conducted by CV as well as GCD measurement using a PARSTAT-MC Sequence. The CV measurement was measured at several scan-rates of 10 to 50 mV/s in the potential ranges of −0.5 to +0.5 Vs. Galvano-static charge/discharge curvatures were gained at several current densities of 5 to 25 Ag−1 in the potential ranges of −0.5 to +0.5 Vs.
Statistical Analysis
In data analysis the Origin Pro software Version 8 and Excel were used for plotting and analyzing the electrochemical data, such as current-voltage curves, capacitance values, and other relevant parameters. Include details on how data were processed and represented graphically.
Characterization Techniques
The phytochemical analysis of the sample was analyzed using Fourier transform infrared (FTIR) spectroscopy. Spectrum was verified to distinguish functional groups elaborated in biosynthesized nanoparticle preparation and were attained through a Perkin Elmer FTIR spectro-photometer (Norwalk, CT, USA) at a resolution of 5/cm in a diffuse transmittance manner using potassium bromide(KBr) pellet. Focused ion beam-scanning electron microscopy (FIB-SEM), High-resolution transmission electron microscope (HR-TEM), and Energy dispersive spectroscopy (EDS) were used to examine the surface morphology, size, and crystalline structure of the Zinc oxide nanostructures and optical properties were analyzed using UV-Vis spectroscopy. X-ray diffraction of the nanoparticles was obtained using a Rigaku X-ray diffraction meter (XRD) to confirm the crystalline nature of the obtained silver oxide nanoparticles.14,19
Result and Discussions
Morphological Analysis
The surface morphological behavior as well as the particle size distributions of the particles ZnO nanostructure were measured utilizing FIB-SEM as shown in Figure 1(a) and (c) at different magnification.

Typical FIB-SEM images of ZnO NPs.
The nanostructure ZnO exhibited some agglomerations. The high magnifications FIB-SEM images displayed and the ZnO nanostructure in the ranges of 20-70 nm as shown in Figure 1(b) and (d). The even dispersions of nanoparticles in the electrode improved the sponginess ZnO nanoparticles and shaped a showing grid for charge transfers. These leaky structures might provide supplementary inner surfaces to the electrolyte ion with improved specific-capacitance values. 20 In a further study, HR-TEM was achieved to define the morphological and nanostructural of biosynthesized ZnO nanoparticles. The HR-TEM micrographs of the ZnO nanoparticles are shown in Figure 2(a)–(d). The average crystallite size was determined utilizing HR-TEM to be in the ranges of 20-80 nm which is nearly related to the result gained from FIB-SEM images. The diffraction patterns of the ZnO Nanostructures Figure 2(a) and (b) showed the crystallite nature and the EDs show the presence of Zn and O and their percentage compositions.

(a) HR-TEM image, (b) SAED patterns, (c) mapping of elements and (d) EDS spectrums of Zn and O in biosynthesized ZnO NPs.
Structural Study
The crystallite phases and structural properties of biosynthesized ZnO nanoparticles were studied and recognized through X-ray diffractions. The material shows sharper as well as thinner peaks at the lower calcination temperature of 380 °C for 4 h which confirms the formation of ZnO nanoparticles. But, as the calcination temperatures were increased to 480, 580, and 680 °C separately, the nature crystals of biosynthesized ZnO NPs were more improved as displayed in Figure 3 denoted as S1, S2, S3, and S4. These incidences were ascribed to particle size increases as outcomes of sustained applications of heats.21,22 The XRD patterns confirmed that the synthesized ZnO nanoparticles exhibited the characteristic wurtzite structure, which is widely known for its excellent stability and favorable properties for various applications, including supercapacitors. 23 The wurtzite phase significantly impacts the material's structural and electrochemical properties. This structure contributes to a high surface area, enhanced electrical conductivity, and improved charge storage capacity, making it ideal for energy storage applications. Additionally, the wurtzite phase facilitates efficient electron transport and contributes to the overall stability of the ZnO nanoparticles under cycling conditions, essential for supercapacitor performance. 24 Higher calcination temperatures were found to promote better crystallinity and larger crystallite sizes, which, in turn, enhance the electrochemical performance of the ZnO nanoparticles. These changes are critical for optimizing the material's properties, as larger crystallite sizes and improved crystallinity lead to better charge transport, higher capacitance, and greater stability. Therefore, controlling the calcination temperature is essential for tuning the structural properties of ZnO nanostructures, ensuring their suitability for supercapacitor applications. These structural modifications directly affect the charge storage behavior and overall efficiency of the material, making them crucial for achieving optimal performance in energy storage devices. 25
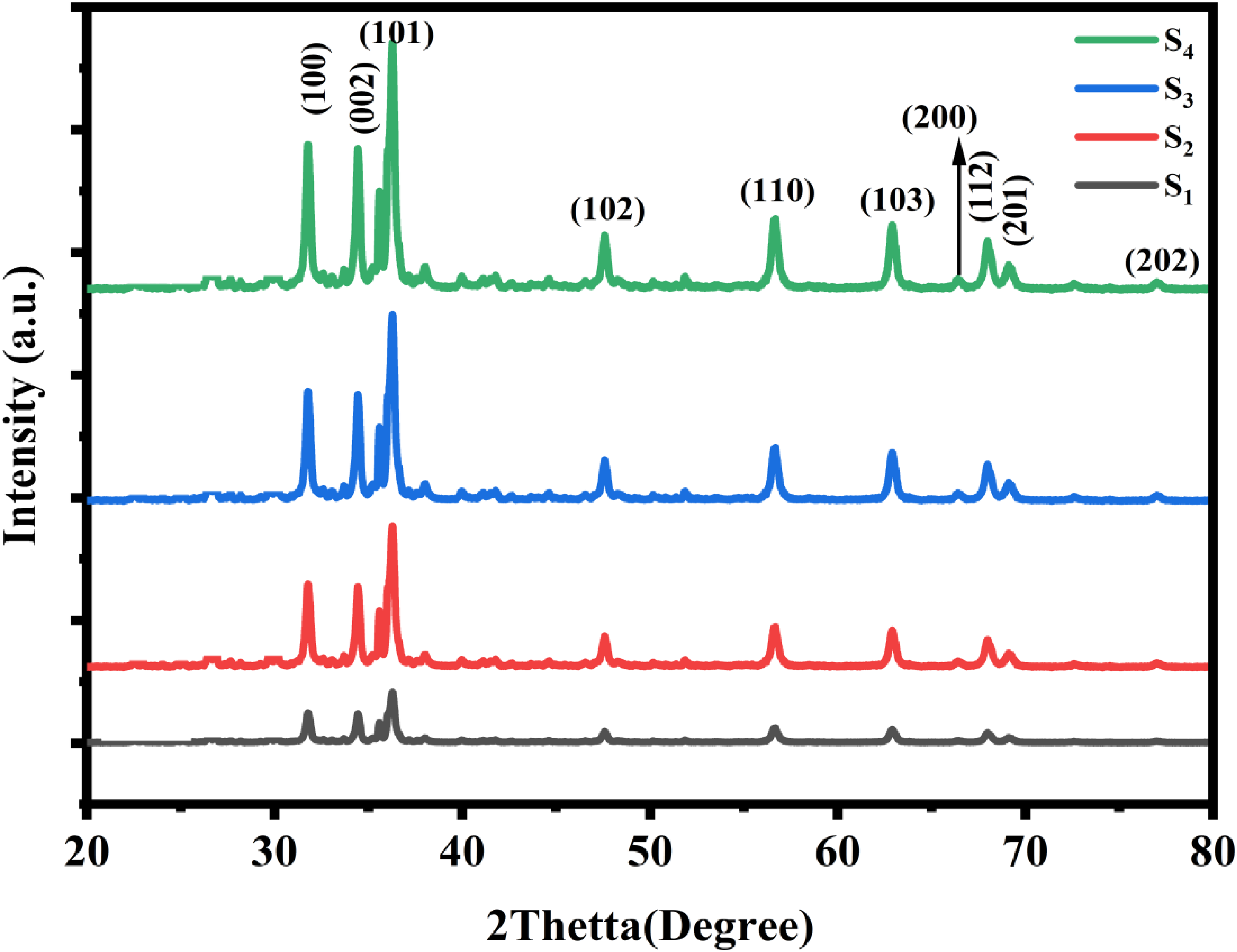
XRD patterns of biosynthesized Zno Nanoparticles at different calcination temperatures.
The PXRD results were in virtuous agreement with that of HR-TEM micrographs are revealed in Figure 2 and the Bragg peaks positions of ZnO nanoparticles were utilized to get the interplanar spacing (d) that gives the diffractions peak. The obtainability of several diffraction peaks of (100), (002), (101), (102), (110), (103), (200), (112), (201), and (202) in the patterns suggests which ZnO nanoparticles reveal hexagonal Wurtzite structures as described in the J-C-P-D-S card no. −00-036-1451. 26
The average crystallite size of ZnO NPs was determined by the Debye-Scherer equation and obtained at 35 nm and lattice parameters were calculated as 

Optical Analysis
UV-Vis spectroscopy is used to analyze the optical properties of materials. Figure 4(a) shows the absorbances of all samples observed at around 380 nm which indicates the absorbance of the samples in visible light. It is observed that the energy band gap of ZnO decreases effectively with increased calcination temperatures. These phenomena can be explained on the basis of several factors. Higher calcination temperatures promote better crystallization and growth of ZnO particles, thus reducing the defects and dislocations in the crystal lattice. Often, these defects and dislocations introduce localized states within the band gap, resulting in an increase in its width. Hence, their reduction causes shrinkage in the band gap. At higher temperatures, the ZnO grains will be larger in size with fewer grain boundaries. Considering that the electronic structure of the grain boundaries can form potential barriers that could change the characteristic features of the band gaps, it follows that at higher temperatures, with enlarging grain sizes and fewer grain boundaries, many grain boundaries will deliver a more homogeneous electronic structure and probably a reduced band gap. Calcination at higher temperatures can result in a reduction in the number of surface states or oxygen vacancies, which are known to be the cause of band gap changes. The presence of oxygen vacancies introduces additional energy levels within the band gap, increasing the band gap. A reduced count of such states can lower the value of the band gap. These effects can be experimentally validated through techniques such as UV-Vis spectroscopy, which enables the direct measurement of band gaps displayed in Figure 4(b). That is, with an increasing calcination temperature, UV-Vis spectroscopy would most likely demonstrate a shift to a longer wavelength redshift in the absorption edges, generally speaking of reduced band gaps.

Absorbance and Tauc relation of ZnO nanoparticles.
As the calcination temperature increases, the crystallinity of the ZnO nanoparticles improves, leading to a reduction in defects and a narrowing of the band gap. 28 This reduction is beneficial for super capacitor performance, as it facilitates easier electron movement during charge and discharge cycles, thereby improving charge storage and overall electrochemical performance. The lower bandgap allows for more efficient charge transport, which enhances the capacitance and cycling stability of the material, making it more suitable for energy storage applications. 29
Phytochemical Analysis
The FT-IR spectrums (Figure 5) were observed from FTIR analysis of S1, S2, S3 and S4. Based on the frequency vibration observed from FTIR the samples were 3467 broad peak, 2916, 1628, 1370, 1211, 1049, 672 cm−1 associated to the hydroxyl (O-H) broad peak, alkanes group(C-H), Alkenes group(C-C = C), Nitro-compounds(NO2 stretch), Aryl and Aryl Halides (C-F stretch) and C-Br stretch respectively. The functional groups observed for S1-S4 were 3444, 1622, 1370, and 623 506 cm−1 associated with O-H stretching, C-H vibrations, Nitro-compounds(NO2), C-Br stretches, and Metallic bonds respectively. The FTIR analysis of Ag2O NPs showed peaks at 3461, 2332, 1616, 1376, and 641 cm−1 associated with O-H stretching, C

FTIR analysis of ZnO nanostructures at different calcination temperatures.
High-performance Super Conductor Tests
The electrochemical measurements of S1, S3, and S4 electrodes are studied in three electrode systems, containing of the as-synthesized S1 and S4 working electrodes, platinum was used as a counter electrode and Ag/AgCl was used as reference electrodes determined in 1 M KOH basic solution. Figure 6 displays CV curves of S1, S3, and S4 in the existence of salt solution of KOH electrolyte solutions at a scan rate of 5 mV/s. The oxidation peak was observed at 0.4 V having to the transformations of Samples into Zn(OH)2, and the reduction peak was obtained at perceived at 0.25 V due to reverse reactions. Here, compared to that of S1, S3, and S4 with bare carbon paper, response currents are very few nearly equivalent to slight in bare carbon paper because capacitances are directly proportional to the areas under CV curves. Hence, this occurrence agreed that the influences of bare carbon paper in total capacitances are nearly slight and is reliable with the stated literature. 30 The Equations (3), (4) and (5) display the charge storages mechanisms of synthesized electrodes, rises in accordance of Faradaic redox reactions. 31

CV carves of the electrodes at different scan rates.
The faradaic redox reaction of a ZnO electrode in a KOH electrolyte involves reduction and oxidation processes at the electrode surface during charge/discharge cycles. Upon discharge, ZnO will be reduced and react with water in the KOH electrolyte to form zincate ions by releasing hydroxide ions:

Figure 6 displays the electro-chemical performances through CV schemes of as-synthesized S1, S3, and S4 electrodes in 1M-KOH aqueous electrolytes. It confirmed that the electrochemical performances of the S4 electrode became greater in alkaline-KOH electrolytes. This is attributed to enhanced crystallinity and better structural properties. Higher calcination temperatures ensure better crystallization and hence a reduced presence of defects and grain boundaries that may act against electron and ion transport, which reduces internal resistance and increases electrical conductivity, thus better charge transfer is promoted. 34 Besides, the elevated temperatures may also increase the surface area and pore volume of ZnO nanostructures and, accordingly, increase the active sites available for electrochemical reactions. This enhances capacitance and the energy storage capability of the super-capacitor. The greatly enhanced crystallinity combined with improved electrical conductivity and increased surface area played a key role in leading to the best performance for ZnO NPs synthesized at higher calcination temperatures. 35
Galvano-static charge-discharge (GCD) curves in the 1M-KOH salt solution are presented in Figure 7 for S1, S3 and S4 electrodes. From the GCD curves the Cp of the electrodes is calculated by using Equation (6).
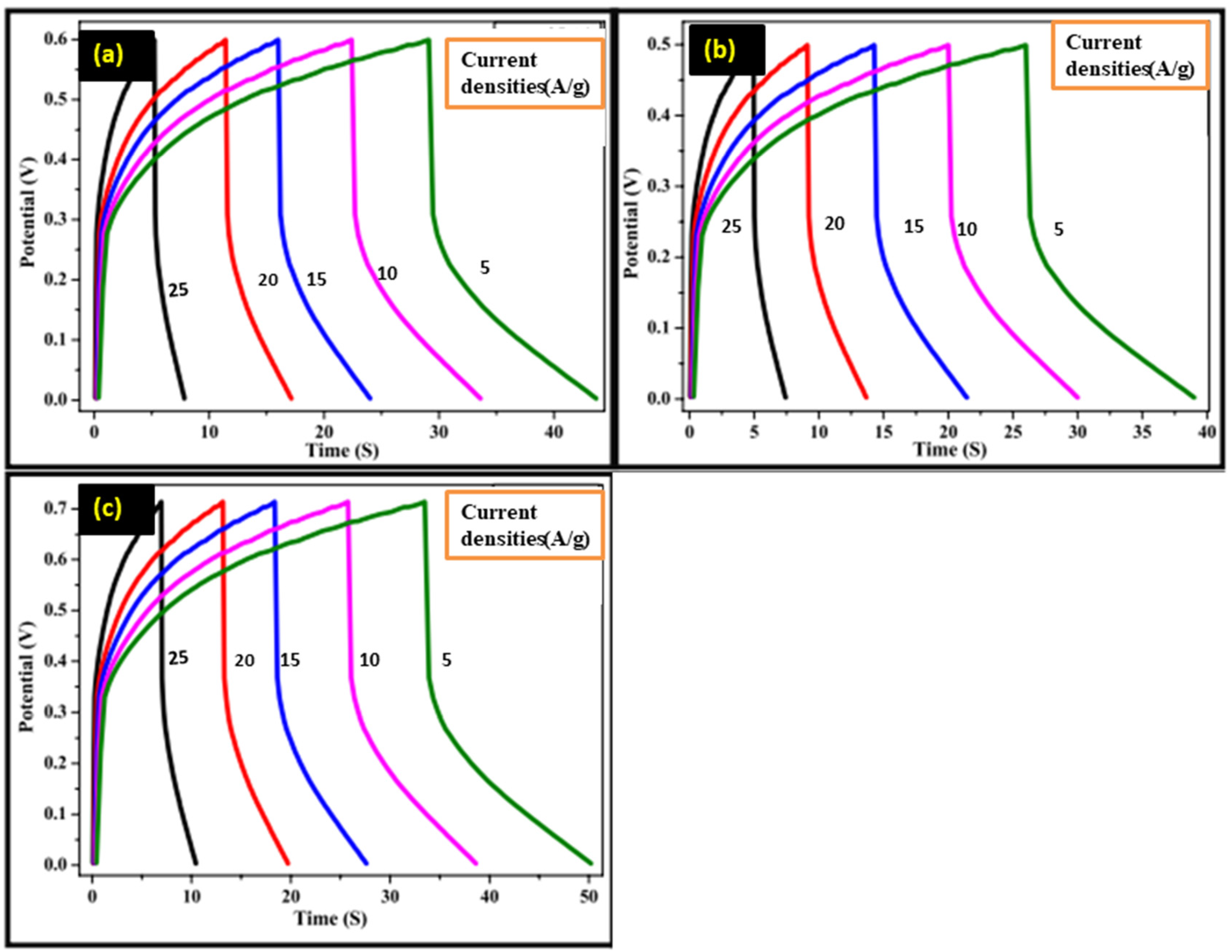
Galvano-static charge and discharge profiles.
Specific capacitances of the found to be 385 F/g, 601 F/g, and 879 F/g, respectively at the scan rate of 10 mV/s.
Here, m is the current, scan rate, and mass of electrodes, respectively. These are relative changes in peak heights for each sample, which may be due to differences in effective surface areas and conditions of active materials. Results show that the electrode made with ZnO nanoparticles calcined at the higher temperature exhibited the highest value of specific capacitance. 36 The increased calcination temperature enhanced crystallinity and surface property, leading to improved performance. 37
Electrochemical impedance spectroscopy (EIS) calibrations for S1, S3 as well as S4 electrodes were conducted with three electrode assemblies in one molarity of KOH with the frequencies s between 1 MHz as well as 100. The EIS spectrums could be perceived in Figure 8(a). The real constituent Z-real exposes the ohmic behavior when the imaginary parts (Z-img) relay to the capacitive characteristics. 38 Characteristically, semi-circles with greater radii refers to greater charge transfer resistances of the electrode. 39 Consequently, EIS outcome conclude that the charge transfer-resistance of S4 electrode is greatly reduced than that of S1 and S3, showing further effective nanostructure of this material in the charge-transfer procedure. Therefore EIS outcomes set lower-charge transfer resistances for S4 electrode as associated to S1 and S3 electrode, and therefore retain more capacitive behavior. The impedance is reduced in the case of ZnO synthesized at higher temperatures, represented by the lower radii impedance semicircle. It can be explained through the enhanced crystallinity and hence charge carrier mobility. The increased energy supplied by calcining ZnO at higher temperatures offers better crystallization and growth of the ZnO grains, ensuring fewer grain boundaries and defects. These usually provide a barrier to charge carrier movement, acting to enhance impedance. Consequently, on increasing the calcination temperature, a decrease in grain boundaries and defects provides efficient charge transport, which brings about a reduced radii impedance semicircle. Literally, this aspect can be validated through an analysis of the microstructure and electrical properties of ZnO synthesized at different temperatures. Improved crystallinity of ZnO calcined at higher temperatures, reflected by sharper and more intense peaks, can be further confirmed using an X-ray diffraction technique. Hence, this relationship between microstructure and impedance suggests that an increase in calcination temperature should raise the electrical performance of ZnO by reducing charge carrier movement barriers. The enhanced cycling stabilities found nearly 100% columbic efficiency can be maintained for up to 1000 cycles under a high current density of 5 A/g as shown Figure 8(b). This gives the composites great potential for rechargeable super-capacitors with long-time durability after cycling.
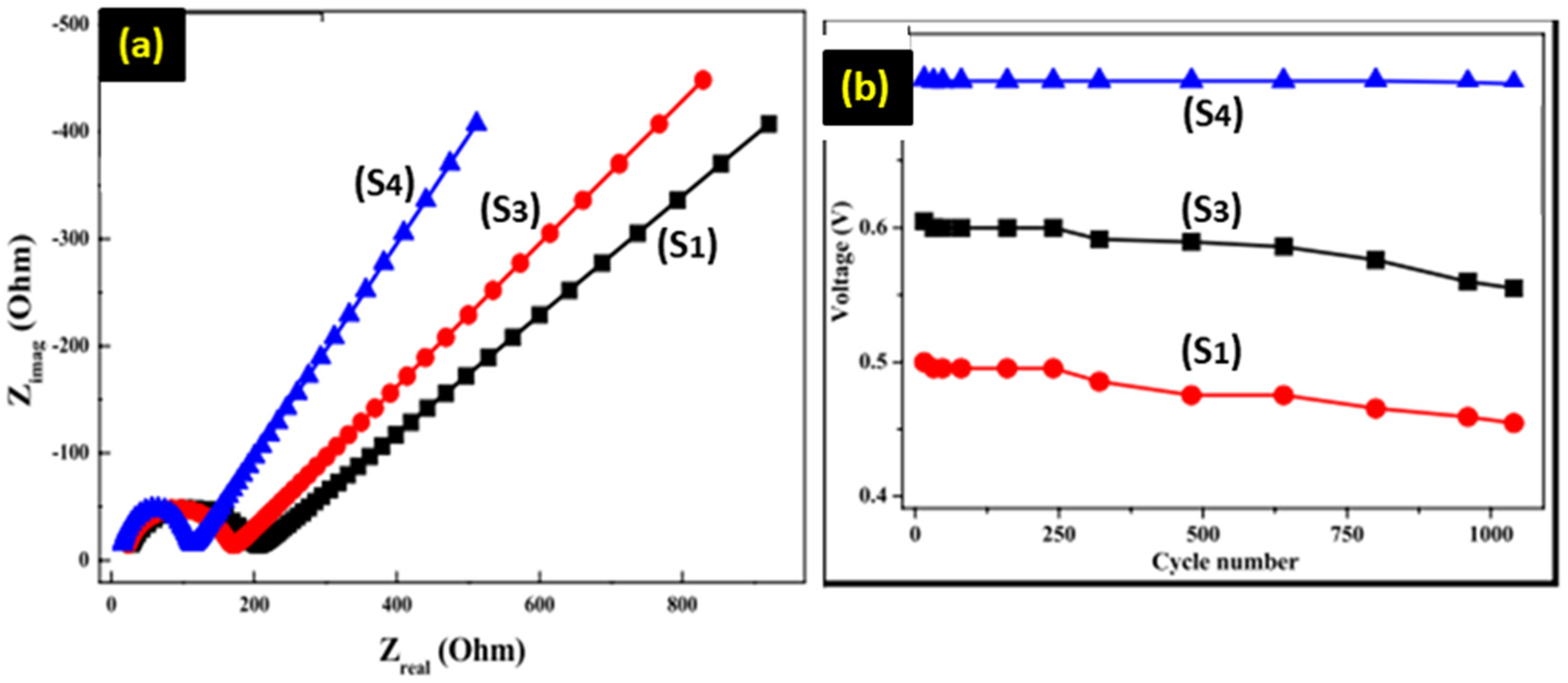
The EIS spectrums of electrodes.
Compared to other works in green synthesis and the conventional methods of synthesizing ZnO-based supercapacitors, the competitive charge/discharge capacities and cycling stability were demonstrated by ZnO nanoparticles synthesized using
Limitation of the Study
One important limitation is that the composition of bioactive compounds in the leaf extract used as the reducing agent may vary. Such variations can arise due to differences in environmental factors, such as geographic location, seasonality, and harvesting methods, which may affect the reproducibility and consistency of the synthesis process. These variabilities inherent in the processes need to be cautiously controlled or accounted for to ensure uniformity in the nanoparticles produced. Besides, although the green synthesis route exhibits ecological and economic advantages, scaling up to industrial levels still poses a challenge. This is because it requires very precise control of reaction conditions, and larger-scale setups may have inefficiencies.
Moreover, the present work focused on testing the supercapacitor performance of the synthesized nanoparticles under laboratory conditions. However, the optimization of the electrode preparation method to obtain the best electrochemical performance is one of the aspects that still requires further investigation. Factors like electrode composition, thickness, and homogeneity can strongly influence charge transport and storage behavior. Addressing these aspects would enhance the practical applicability of the material for real-world energy storage devices. While describing these, the limitations will also delineate a clear pathway for forthcoming studies on how such green-synthesized nanomaterials should be improved in terms of their synthesis, characterization, and application. These discussions further set a context for interested researchers to make further improvements in this area of research.
Conclusions
In this study, ZnO nanostructures at different calcination temperatures were successfully synthesized by green synthesis root using P. macrosolen L. leaf extract. The XRD diffraction peaks of all ZnO nanostructure coordinated very well with the tetragonal Wurtzite hexagonal phases, separately, as exposed by P-XRD graphs. FTIR spectroscopy sets the formations of ZnO nanostructure. FIB-SEM and HR-TEM showed spongy surface and agglomerated surface morphology for all samples. The biosynthesized ZnO nanostructures calcinated at 580 °C performed very good electro-chemical as an electrode material for super-capacitors associated with of samples prepared at 280, and 480 °C, which are analyzed by improved charge/discharge capacities and enhanced cycling stabilities. Nearly 100% columbic efficiency can be maintained for up to 1000 cycles under a high current density of 5 A/g. This gives the composites great potential for rechargeable super-capacitors with long-time durability after cycling.
Footnotes
Acknowledgements
Our sincere gratitude goes to the Indian Institute of Technology, Department of Material Science and Metallurgical Engineering (Combinatorial Laboratory), and Dambi Dollo University for their support in the laboratory work conducted for this article. Additionally, we would like to express our appreciation to the Joint PhD Program of The Ministry of Education Ethiopia and the Ministry of Innovation and Technology for providing Dambi Dollo University with the opportunity to engage in a joint PhD supervision program with IIT, Hyderabad.
Author Contributions
Conceptualization, AS. G, LT. J, N. N and K. R.; Data curation, AS. G, LT. J, N. N and K. R.; Analysis and Validation, AS. G, LT. J, N. N and K. R.; Formal analysis, AS. G, LT. J, N. N and K. R; Investigation, AS. G, LT. J, N. N and K. R.; Methodology, AS. G, LT. J, N. N and K. R.; Project administration, K.R. Resources AS. G, LT. J, N. N and K. R.; Software, AS. G, LT. J, N. N and K. R., Supervision, K. R.; Validation, AS. G, LT. J, N. N and K. R.; Visualization, AS. G, LT. J, N. N and K. R.; Writing—original draft, AS. G, LT. J, N. N and K. R., Data Visualization, Editing and Rewriting, AS. G, LT. J, N. N and K. R.
Data Availability
The datasets used and analyzed during the current study are available from the corresponding author on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal participants.
Statement of Informed Consent
Not Applicable.
