Abstract
Objectives
The aim of the project was the isolation, structure elucidation, and cytotoxic evaluation of compounds from Annona squamosa L. seeds.
Methods
Chromatographic techniques were used to isolate cytotoxic compounds from the methanol extract of A. squamosa seeds. The structures of compounds were elucidated by analyses of mass spectra and nuclear magnetic resonance data. Compounds were evaluated using cytotoxic assays.
Results
One new lignanamide, squamosamide, and seven known compounds, cannabisin M (
Conclusions
The seeds of A. squamosa yielded one new lignanamide and seven known compounds, several of which demonstrate potent cytotoxicity associated with apoptosis. Compounds
Introduction
The genus Annona (Annonaceae) comprises shrubs and small to large trees distributed in tropical and subtropical regions.
1
The genus comprises approximately 120 species distributed in Central and South America, Africa, Asia, and Australia.
2
Four species have been recorded and described in Vietnam, namely A. muricata, A. reticulata, A. glabra, and A. squamosa.
3
Annona squamosa L., commonly known as sugar-apple or sweetsop, is a small tropical-subtropical tree cultivated widely in Central and South America, India, Mexico, Taiwan, Vietnam, and other regions. It is typically 3–8 m in height, has a branched habit, and begins fruiting approximately in its third year.
4
In folk practices, the seeds are ground into powder and applied externally to eradicate head lice and other ectoparasites; seed paste is used for skin ulcers and abscesses; and seed extracts are administered in small doses as vermifuges.
5
In some regions, crushed seeds are also employed as insecticides. Phytochemical investigations of A. squamosa have revealed the presence of acetogenins, diterpenes, alkaloids, and cyclic peptides. These compounds exhibit a broad spectrum of biological activities, such as anticancer,6,7 antimicrobial,
8
anti-inflammatory,
9
antioxidant, antimalarial,
10
and insecticidal effects.
11
As part of our ongoing research to discover cytotoxic compounds from this species, we herein report the isolation, structural elucidation, and evaluation of the cytotoxic activity of one new lignanamide and seven known compounds, cannabisin M (

Chemical structures of compounds
Material and Methods
General
See Supplemental Material.
Plant Material
The seeds of Annona squamosa L. (Annonaceae) were collected in Lang Son province, Vietnam, in August 2024 and identified by one of the authors, Prof. Ninh Khac Ban, Institute of Chemistry, VAST. A voucher specimen (CDRD23) was deposited at the Institute of Chemistry, VAST.
Extraction and Purification of Compounds
The extraction and prefractionation procedures were described in the previous work. 19 Powdered seeds of A. squamosa (3.0 kg) were extracted with methanol (8 L) by ultrasonication (2 h × 2 at 50 °C) to afford a methanol extract (AS, 400 g), which was suspended in water (2.0 L) and successively partitioned with dichloromethane and then ethyl acetate (1:1, v/v; twice) to give the dichloromethane fraction (AS1, 216 g), the ethyl acetate fraction (AS2, 87 g), and the aqueous layer (AS3). AS2 was subjected to a silica gel column chromatography (CC) eluting with a gradient solvent of n-hexane/acetone (40:1, 10:1, 5:1, v/v), followed by dichloromethane/methanol (10:1, 5:1, v/v), to furnish five fractions: AS2A (16.0 g), AS2B (11.4 g), AS2C (25.2 g), AS2D (27.0 g), and AS2E (2.0 g).
AS2B was further separated by a silica gel CC using n-hexane/acetone (7:1, v/v) to give AS2B1 (0.5 g), AS2B2 (2.4 g), AS2B3 (1.4 g), and AS2B4 (1.7 g). AS2B2 was chromatographed on an RP-18 CC eluting with acetone/water (4:1, v/v) to yield compound
AS2D2 was chromatographed on an RP-18 CC eluting with acetone/water (1:2, v/v) to give AS2D2A (130 mg) and AS2D2B (166 mg). Further purification of AS2D2A on an HPLC system (J'sphere H-80 column (250 × 20 mm), 50% methanol in water, and flow rate of 3.0 mL/min) yielded compounds
The aqueous layer AS3 was fractionated on a Diaion HP-20 CC by stepwise elution with methanol in water (25, 50, 75, and 100%; 1 L each) to give four fractions: AS3A (2.4 g), AS3B (3.0 g), AS3C (8.0 g), and AS3D (4.2 g). AS3C was chromatographed on a silica gel CC using gradient solvent of dichloromethane/methanol (20:1, 10:1, 5:1, 2.5:1, v/v) to give AS3C1 (0.9 g), AS3C2 (1.0 g), AS3C3 (2.2 g), and AS3C4 (1.4 g). AS3C2 was chromatographed on a silica gel CC using solvent of dichloromethane/acetone (2.5:1, v/v) to give AS3C2A (360 mg), AS3C2B (1.5 g), and AS3C2C (860 mg). AS3C2B was chromatographed on an RP-18 CC eluting with methanol/water (1:1, v/v) to give AS3C2B1 (310 mg) and AS3C2B2 (736 mg). AS3C2B1 was chromatographed on an HPLC system (J'sphere H-80 column (250 × 20 mm), 16% acetonitrile in water, and flow rate of 3.0 mL/min) to afford compound
Squamosamide (1)
White amorphous powder;
The 1H- and 13C-NMR Spectral Data for Compounds
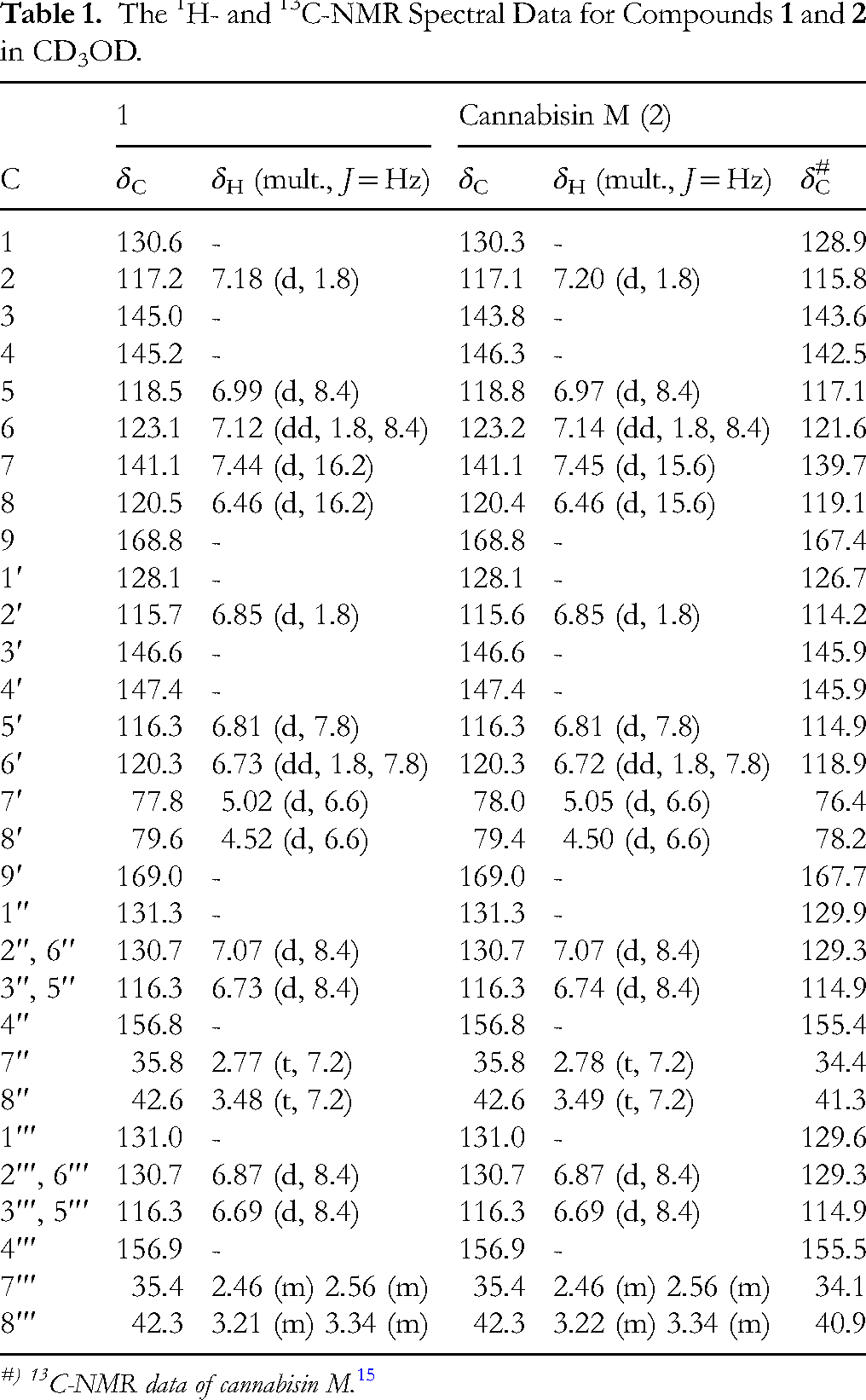
#) 13C-NMR data of cannabisin M. 15
Cell Culture and Viability Assay
Human oral cancer (KB) cells: ATCC® (CCL-17TM); human lung cancer(PC9) cells purchased from Merck Millipore (Cat. No. 90071810). PC9 and KB cells were maintained in RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS) and 1% antibiotics (penicillin/streptomycin) at 37 °C in a humidified atmosphere containing 5% CO2. Cells were seeded in 96-well plates at a density of 5 × 10³ cells/well and incubated overnight.20,21 Compounds at 30 µM were administered twice over a period of 48 h. Cell viability was measured using the MTS assay (CellTiter 96® AQueous One Solution, Promega). Absorbance was measured at 490 nm using a microplate reader. Mitoxantrone was used as a reference compound. Compounds that reduced cell viability by more than 50% were selected for further assays.
Caspase-3 Activity Assay
PC9 cells were seeded into 96-well plates at a density of 1 × 104 cells/well and incubated overnight. Cells were treated with the selected compounds at 10 µM for 48 h. Caspase-3 activity was measured using a caspase-3 colorimetric assay kit (Sigma-Aldrich) following the manufacturer's instructions. 22 Briefly, cells were lysed, centrifuged, and the supernatant was mixed with assay buffer and caspase-3 substrate (DEVD-pNA). Absorbance was measured at 400 nm using a microplate reader. Caspase-3 activity was calculated based on standard curves generated using known concentrations of p-nitroaniline (pNA).
Cleaved PARP1 Activity Assay
PC9 cells were seeded in 96-well plates at 1 × 104 cells/well and allowed to adhere overnight. Cells were then treated with compounds at 10 µM for 48 h. Cleaved PARP1 activity was determined using a human cleaved PARP1 ELISA kit (Abcam) according to the manufacturer's instructions. 22 Briefly, cells were lysed, centrifuged, and supernatants were collected. The cleaved PARP1 concentration was measured at 450 nm absorbance using a microplate reader.
Statistical Analysis
All experiments were conducted in triplicate and data are expressed as the mean ± standard deviation (SD). Statistical analyses were performed using GraphPad Prism 8 software. Differences between control and treated groups were assessed using one-way analysis of variance (ANOVA) followed by Tukey's multiple comparison tests. A p-value of less than 0.05 was considered statistically significant.
Results
Compound

The key HMBC and COSY correlations of compound
Compounds
The Effects of Compounds

Mitoxantrone was used a positive control.
Caspase 3 and Cleaved PARP1 Activities of Selected Compounds in PC9 Cells.

Discussion
Previous studies have reported acetogenins, diterpenes, alkaloids, phenolics, and cyclic peptides from A. squamosa.
4
The present study yielded one new lignanamide, squamosamide and seven known compounds (
All compounds were evaluated for cytotoxic activity on PC9 and KB cancer cell lines. Mitoxantrone was used as a positive control and showed cytotoxic activity with IC50 values of 6.7 ± 0.4 µM (PC9) and 6.8 ± 0.9 µM (KB). Compounds
Conclusions
In summary, phytochemical investigation on the methanol extract of A. squamosa seeds led to the isolation of one new lignanamide, squamosamide and seven known compounds,
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251403961 - Supplemental material for Lignanamides and other constituents from the seeds of Annona squamosa L. and their cytotoxic activity
Supplemental material, sj-docx-1-npx-10.1177_1934578X251403961 for Lignanamides and other constituents from the seeds of Annona squamosa L. and their cytotoxic activity by Nguyen Thi Thu Ha, Nguyen Thi Tu Oanh, Vu Mai Thao, Tran Huu Giap, Vu Kim Thu, SeonJu Park, Ninh Khac Ban, Nguyen Thi Minh Hang and Nguyen Xuan Nhiem in Natural Product Communications
Footnotes
Acknowledgements
The authors would like to thank Mr Dang Vu Luong, Institute of Chemistry, VAST for recording NMR spectra. HR-ESI-MS spectra were obtained using a Waters ACQUITY UPLC system connected to a Xevo G2-XS QTOF at the Korea Basic Science Institute (KBSI, Metropolitan Seoul Center).
Ethical Approval
Ethical approval is not applicable to this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Vietnam Academy of Science and Technology (VAST) under grant number NCVCC38.03/24–25.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
