Abstract
Objective
The rising crisis of antimicrobial resistance necessitates the discovery of bioactive compounds from natural sources. Plants adapted to extreme environments are promising sources of unique secondary metabolites. This study investigates the chemical composition and antimicrobial potential of Atriplex leucoclada (Amaranthaceae), a halophyte native to the Egyptian desert.
Methods
The dried aerial parts of Atriplex leucoclada were exhaustively extracted with 80% aqueous methanol. The resulting crude extract was suspended in water and successively partitioned with n-hexane and ethyl acetate. The ethyl acetate-soluble fraction was subjected to multi-step chromatographic purification, including flash chromatography and preparative HPLC. The structures of purified metabolites were elucidated through comprehensive spectroscopic analysis, including nuclear magnetic resonance (NMR) and high-resolution electrospray ionization mass spectrometry (HRESIMS), and by comparison with literature data. The in vitro antibacterial activity of the extract and isolated compounds was evaluated against Bacillus subtilis and Escherichia coli using agar well diffusion and broth microdilution assays.
Results
Phytochemical investigation of A. leucoclada led to the isolation of six compounds
Conclusion
This study highlights the halophyte Atriplex leucoclada as a promising source of bioactive compounds. The significant antibacterial activity of compound
Introduction
Halophytes are plants adapted to survive in high-salinity environments. To endure such harsh conditions, they utilize a range of physiological and molecular adaptations, including the biosynthesis of specific enzymes, osmolytes, and secondary metabolites. 1 The Amaranthaceae family comprises the most abundant halophytes throughout the world. The genus Atriplex, belonging to the Amaranthaceae family, includes approximately 300 species of herbaceous halophytic plants. These species are widely distributed worldwide, particularly in the arid and semi-arid regions of Africa, Asia, and Europe. 2 Currently, there is growing interest in species of the genus Atriplex due to their rich profile of bioactive compounds. Studies have revealed that Atriplex species exhibit high nutritional value, owing to their high protein content, and thus can be used as alternative cereal grains. 3 Pharmacological investigations have demonstrated significant potential across several species; for example, A. semibaccata and A. vestita exhibit antifungal and anti-bronchitis properties, respectively, while A. hortensis is a source of vitamin A and has been used in folk medicine for respiratory, digestive, and urinary tract disorders, as well as for rheumatism due to its analgesic effects. Furthermore, A. halimus has shown efficacy in managing type 2 diabetes (T2DM) and is used in veterinary practices as an anthelmintic. Extracts of A. confertifolia display significant cytotoxicity against human breast cancer cell lines, while A. inflata and A. portulacoides have been reported to contain phytochemicals with fungicidal properties. 4 These biological activities are attributed to a diverse array of specific metabolites, that have been discovered in Atriplex species, including flavonoids,2,5 phenolics,6,7 saponins, 8 ecdysteroids, 9 and triterpenes. 10 Consequently, biological screening of Atriplex species has revealed anti-butyrylcholinesterase, 2 anti-inflammatory and antinociceptive,2,5 antidiabetic,8,11 antitumor, antimicrobial, 12 and hepatoprotective properties. 13
Atriplex leucoclada Bioss., commonly known as orache or raghal, is a perennial halophytic shrub renowned for its high salinity tolerance. Native to the Mediterranean region and the deserts of Egypt and Saudi Arabia, this species holds significant ecological value for soil stabilization and serves as a saline-tolerant livestock feed. 14 Despite the importance of this halophyte species, the detailed chemical composition and biological activities of A. leucoclada remain largely unexplored.
The emergence of antimicrobial resistance, driven by the inappropriate use of antibiotics and its harmful impact on the human microbiota, constitutes a major global health challenge. 15 The World Health Organization (WHO) has recognized antibiotic resistance as one of the most critical threats to humanity in the twenty-first century. 16 Notably, Enterobacteriaceae, such as Escherichia coli, Klebsiella spp., and Enterobacter spp., have emerged as superbugs of the twenty-first century, with associated mortality rates reaching 50%. It is estimated that annual deaths from drug-resistant infections could rise to 10 million by 2050. 17 Escherichia coli, an important member of the family Enterobacteriaceae, is a common gut bacterium and a significant human pathogen, capable of causing a wide range of serious infections such as urinary tract infections (UTIs), septicemia, pneumonia, and meningitis. Critically, over 50% of E. coli isolates in hospitals across five out of six WHO regions are now resistant to essential antibiotics including third-generation cephalosporins, making it a major antimicrobial resistance threat and a priority for global public health. 16 This urgent crisis has accelerated the search for natural products as promising therapeutic alternatives. Owing to their broad spectrum of bioactivity, lower cost, and greater accessibility compared to synthetic pharmaceuticals, plant-derived compounds have become a major focus in developing novel antibacterial strategies. 18 Therefore, as part of our ongoing research on bioactive metabolites from halophytes,19–24 this study aimed to characterize the chemical profile of the halophyte Atriplex leucoclada and evaluate the antimicrobial potential of its isolated compounds against two bacterial species including the Gram-negative Escherichia coli and the Gram-positive Bacillus subtilis. As a result, six compounds were isolated from the halophytic herb A. leucoclada and their antimicrobial activity were assessed.
Materials and Methods
Chemicals and Reagents
The extraction and purification processes utilized various solvents, including including n-hexane, dichloromethane (CH2Cl2), ethyl acetate (EtOAc), methanol, and acetonitrile, purchased from Wako Pure Chemical Industries (Osaka, Japan). Initial fractionation and purification were performed using a Biotage Selekt system (Uppsala, Sweden) equipped with normal-phase silica gel (Sfär HC, 100 g) and reversed-phase C18 (RP-C18) flash chromatography columns (40 g).
Final purification of the metabolites was achieved using a medium-pressure liquid chromatography system (MPLC Pure C-850 Flash Prep®, Büchi, Flawil, Switzerland) with ultraviolet (UV) and evaporative light scattering (ELSD) detectors. Preparative separation employed a reversed-phase HPLC column (Inertsil ODS-3, 5 µm, 20 × 250 mm, GL Sciences Inc., Japan). Thin-layer chromatography (TLC) was performed on silica gel 60 F₂₅₄ and RP-C18 plates from Merck (Darmstadt, Germany). Deuterated methanol (CD₃OD) for NMR analysis was purchased from Cambridge Isotope Laboratories (Andover, MA, USA). All other analytical-grade chemicals were purchased from Wako Pure Chemical Industries (Japan).
General Experimental Procedures
Nuclear magnetic resonance (NMR) spectra (1H: 600 MHz, 13C or APT: 150 MHz) were recorded on a Bruker DRX-600 spectrometer (Bruker Daltonics, USA) using deuterated methanol (CD₃OD) as solvent, with tetramethylsilane (TMS, δ 0.0 ppm) as an internal standard. High-resolution electrospray ionization mass spectrometry (HR-ESI-MS) data were obtained on an Agilent qTOF-LC-MS instrument (Agilent Technologies, USA). Thin-layer chromatography (TLC) analysis was performed on pre-coated silica gel plates, and compounds were visualized by spraying with 5% H₂SO₄ followed by heating at 110 °C.
Collection of Plant Material
Aerial parts of Atriplex leucoclada Boiss. were collected from desert areas in Maadi, Egypt (Wadi Degla desert land, 29°57′35′′ N, 31°19′44′′ E). The plant was identified by Prof. Ibrahim A. El-Garf, Professor of Botany, Faculty of Science, Cairo University, Egypt. A voucher specimen (ALA-2019-1) was deposited at the herbarium of the Pharmacognosy Department, Faculty of Pharmacy, Al-Azhar University, Cairo, Egypt.
Extraction and Isolation of Secondary Metabolites
The dried aerial parts (powder; 1 kg) was macerated three times (at 25 °C) in 80% aqueous methanol and stirred with mechanical stirrer. The combined extracts were concentrated in vacuo to get 135.6 g of a dark brown residue. The residue was then suspended in distilled H2O and subjected to extraction with n-hexane, ethyl acetate to yield n-hexane-soluble part (8.0 g), ethyl acetate-soluble part (16.0 g), and aqueous part (111.6 g). The ethyl acetate-soluble part (16.0 g) was partitioned with flash chromatography over a Biotage with Sfär column packed with normal-phase silica HC 100 g and eluted with a gradient mixture of n-hexane-ethyl acetate-methanol (1:0:0-0:0:1) to get 10 subfractions Eth-1 to Eth-10. Subfraction Eth-6 (1.6 g) was subjected to purification over a Flash RP-C18 (40 g) and eluted with H2O-methanol-0.1% HCO2H (1:0:0.1-0:1:0.1) with a flow rate 30 mL/min to afford compounds
Evaluation of Antimicrobial Activity Using the Disc Diffusion Assay
The antimicrobial activity of the total methanolic extract and compounds
Determination of Minimum Inhibitory Concentrations (MICs) Using Microdilution Method
The antibacterial activity of the tested samples against Staphylococcus aureus ATCC 25923 and Escherichia coli ATCC 25922 was evaluated using a quantitative microdilution method to determine the minimum inhibitory concentration (MIC).
26
Briefly, stock solutions of the extract and compounds
Statistical Analysis
All results in the present study were obtained from three independent biological experiments. The results were expressed as the means ± SD (n = 3). The statistical significance of differences between means was established using one-way analysis of variance (ANOVA) with Duncan's post hoc tests. p values < 0.05 indicate statistical significance.
Results
Identification of Isolated Compounds from A. leucoclada
The dried aerial parts of A. leucoclada were extracted with 80% aqueous methanol. The obtained crude extract was suspended in distilled H2O, and solvents of different polarities, including n-hexane and ethyl acetate, were used for partitioning the crude methanol extract. Metabolites were purified through several chromatographic procedures to get compounds
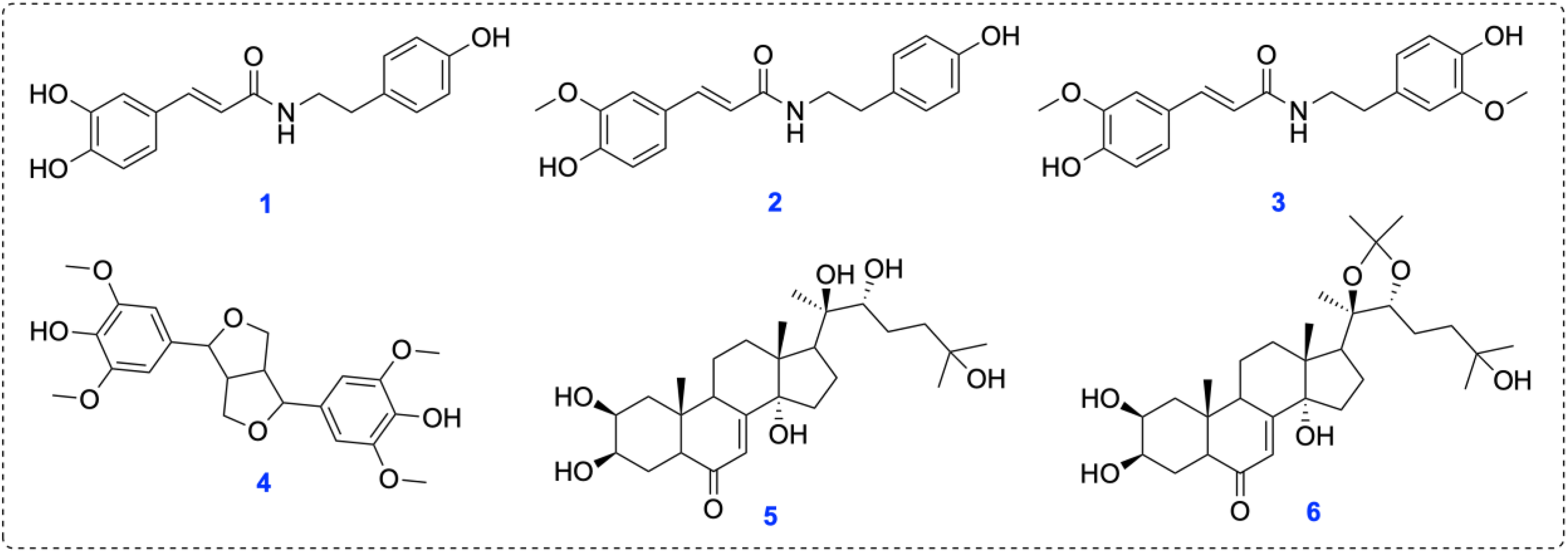
Structures of the isolated compounds from A. leucoclada.
N-trans-caffeoyltyramine (
1H NMR Spectral Data of Compounds
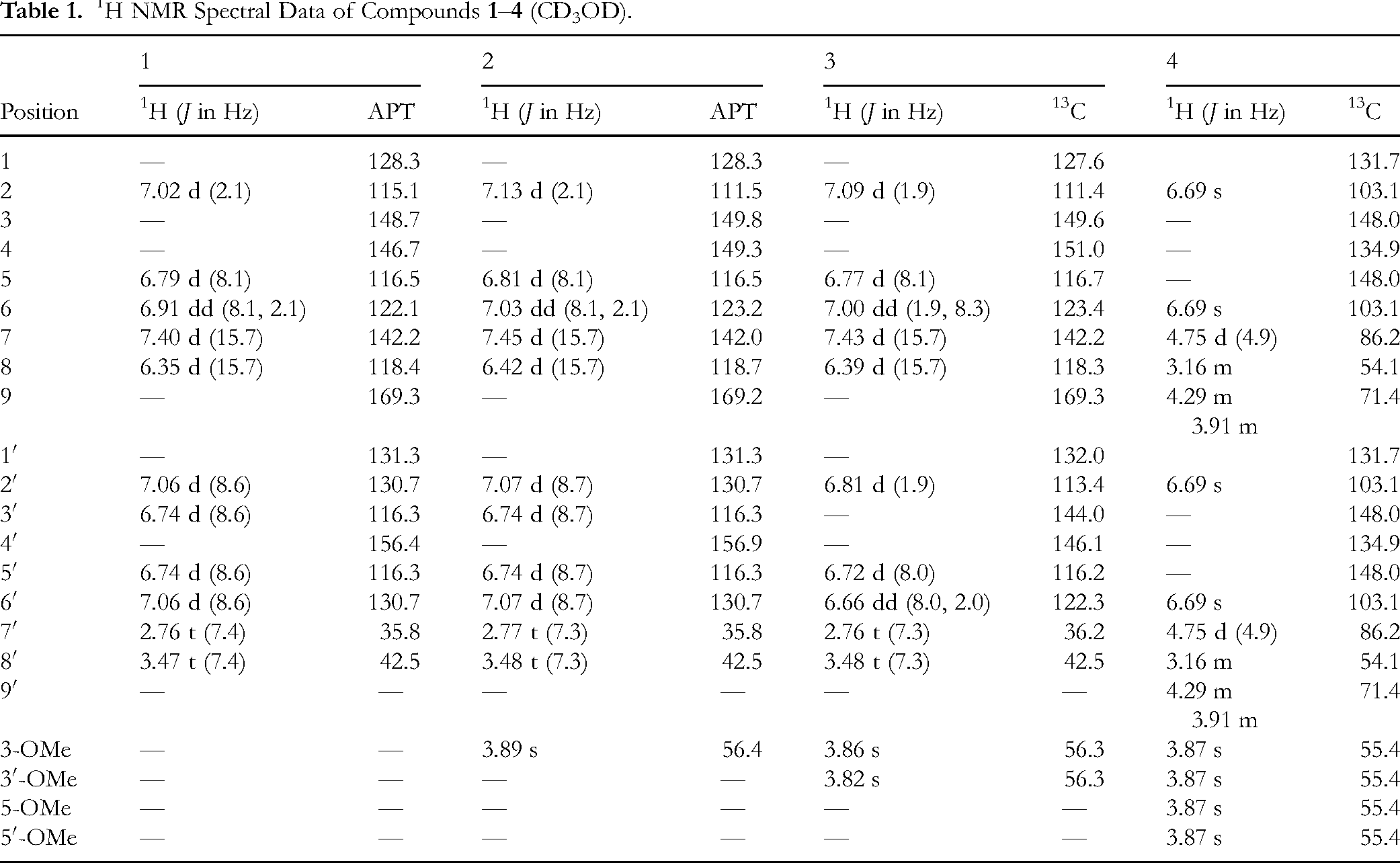
N-trans-feruloyl tyramine (
The molecular formula of N-trans-feruloyl-3-methoxytyramine (
Compound
Compound
1H NMR Spectral Data of Compounds

Compound
Importantly, the isolated compounds
Antimicrobial Activity of Metabolites from A. leucoclada
In the current study, the isolated compounds were evaluated for their antimicrobial activity (Figures 2 and 3). Importantly, the total methanolic extract of A. leucoclada at a concentration of 1 mg/mL showed antimicrobial activity against tested bacterial standard strains, B. subtilis and E. coli displaying clear IZDs of 16 and 14 mm, respectively. Among the tested isolates, compound

Evaluation of antibacterial effects of total methanolic extract and isolates

Antimicrobial activity of tested samples assessed by broth microdilution assay. (A) Activity against Bacillus subtilis ATCC 6633. Wells
The Inhibition Zone Diameters (IZDs) and Minimum Inhibitory Concentrations (MICs) of A. leucoclada Total Methanolic Extract and Isolates
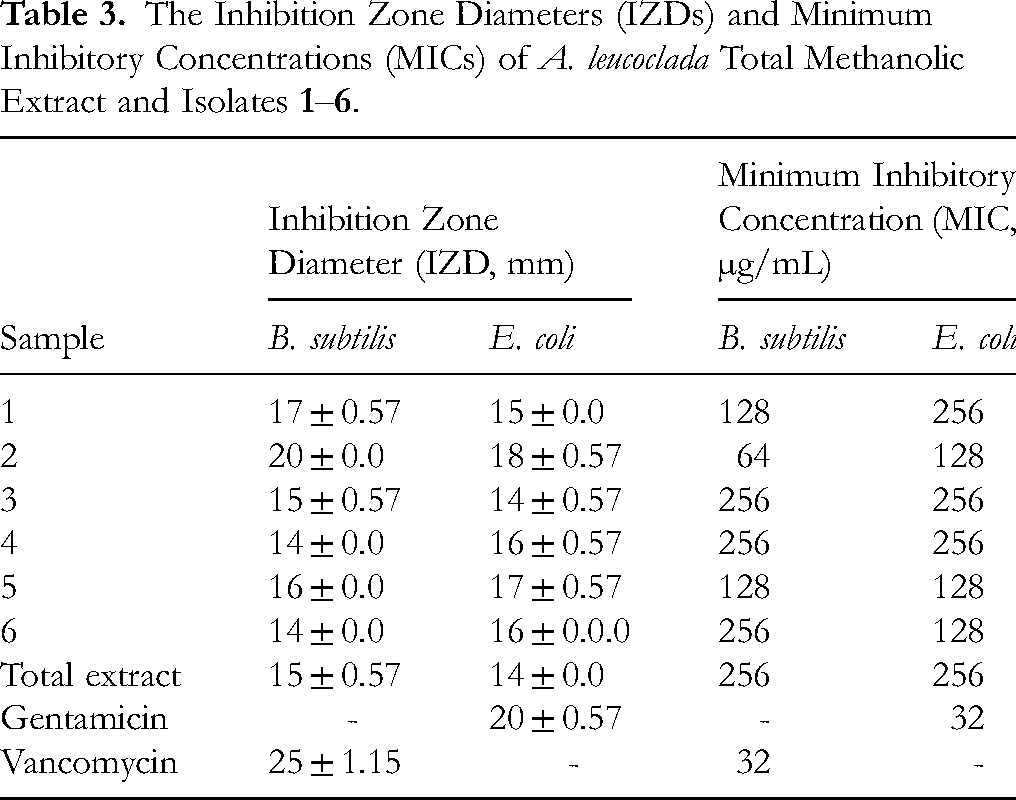
Moreover, using the broth microdilution method, the minimum inhibitory concentrations required to inhibit bacterial growth (MIC) against B. subtilis and E. coli were determined. Among the tested samples, the total methanolic extract obtained from A. leucoclada displayed the same MIC value 256 µg/mL against B. subtilis and E. coli. Furthermore, compound
Discussion
Natural products derived from plants are a crucial foundation of human medicine. Bioactive compounds from natural sources—including alkaloids, flavonoids, lignanamides, and ecdysteroids—possess diverse pharmacological activities. Within the Amaranthaceae family, numerous halophytic species are recognized as rich reservoirs of such compounds, holding considerable potential for various applications. Extracts from Amaranthaceae plants frequently demonstrate significant antimicrobial, neuroprotective, anticancer, and anti-inflammatory properties. 1
Previous studies on various Atriplex species (Amaranthaceae) report diverse metabolites, however, the chemical profile of A. leucoclada is poorly investigated. A previous study by Hayam S. Ahmed and colleagues analyzed the composition of defatted methanol and n-hexane extracts obtained from A. leucoclada. The results revealed the isolation of chemical compounds including 20-hydroxy ecdysone, phytol, β-sitosterol, stigmasterol, palmitic acid, luteolin, β-sitosterol-3-O-β-D-glucopyranoside, pallidol and isorhamnetin 3-O-β-galactopyranoside. 28 Moreover, the anti-inflammatory activity of the n-hexane-soluble part of A. leucoclada might be associated with the combined effect of 2-hexadecen-1-ol and hexadecanoic acid methyl ester, as well as ursodeoxycholic acid. 28 Our study aimed to expand the chemical profile of A. leucoclada and evaluate the antimicrobial activity of its isolated constituents. Hence, our investigation led to the identification of six compounds including lignanamides N-trans-caffeoyltyramine, N-trans-feruloyltyramine, and N-trans-feruloylmethoxytyramine; lignan syringaresinol; and ecdysteroids 20-hydroxyecdysone and 20-hydroxyecdysone-20,22-monoacetonide. Notably, lignanamide derivatives, syringaresinol, and 20-hydroxyecdysone-20,22-monoacetonide are reported from this species for the first time.
The biosynthesis of ecdysteroids is fundamentally controlled by sterol availability. Insects and arthropods cannot synthesize sterols de novo and must acquire C₂₇ or C₂₉ sterols from their diet, subsequently converting them into C₂₇ precursors required for ecdysteroid production. Conversely, plants have sterol biosynthesis pathway, mainly via cytosolic mevalonic acid (MVA) pathway, and can produce phytoecdysteroids (PEs) from C₂₇ sterol precursors such as cholesterol or lathosterol. Importantly, using acetyl-CoA, MVA pathway is considered the primary source of isoprenoid precursors for sterols’ production.
33
The biosynthesis of PEs

Proposed biosynthesis of compounds
Halophytes are a rich source of antimicrobial metabolites, demonstrating efficacy against both Gram-positive and Gram-negative pathogens. Bioactive constituents such as phenolics and flavonoids underlie their ability to inhibit the growth of bacteria including Staphylococcus aureus, Bacillus subtilis, Escherichia coli, and Pseudomonas aeruginosa, as evidenced by studies on some species.
34
Given the significance of bioactive compounds in plants that combat microbial infections, the evaluation of antimicrobial activities has become a key focus in phytochemical research. In the present study, the total methanolic extract of A. leucoclada (1 mg/mL) and compound
The novelty of this study lies in the detailed chemical investigation of Atriplex leucoclada, which led to the isolation of six metabolites and the evaluation of their in vitro antimicrobial activity. The chemical findings underscore the potential impact of environmental conditions on the plant's biosynthetic pathways. However, the exact mechanisms of action for the observed bioactivity remain unknown. Future work should therefore focus on elucidating these mechanisms, expanding the scope of pharmacological testing, and evaluating the clinical potential of this halophyte species and its metabolites. Specifically, biological evaluation against multidrug-resistant (MDR) strains and assays against diverse therapeutic targets are essential to fully reveal its potential as a source of novel therapeutic agents.
Conclusion
This study provides a phytochemical and biological investigation of the halophyte A. leucoclada, contributing to the understanding of its chemical diversity and therapeutic potential. Six compounds were successfully isolated and characterized from the ethyl acetate-soluble fraction of the methanolic extract. The identified metabolites, encompassing the alkaloids
In summary, this research validates A. leucoclada as a source of bioactive agents for further investigation. The significant antibacterial activity of compound
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261427064 - Supplemental material for Chemical Constituents and Antimicrobial Activity of the Halophyte Atriplex leucoclada
Supplemental material, sj-docx-1-npx-10.1177_1934578X261427064 for Chemical Constituents and Antimicrobial Activity of the Halophyte Atriplex leucoclada by Ahmed Othman, Fahd M. Abdelkarem, Yhiya Amen, Mohammed S. Abdulrahman and Kuniyoshi Shimizu in Natural Product Communications
Footnotes
Acknowledgements
The authors are indebted to Prof. Ibrahim A. El-Garf, Department of Botany, Faculty of Science, Cairo University, Egypt, for the identification of plant.
Ethical Approval
Ethical Approval is not applicable to this article.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All data generated or analyzed during this study are included in this published article.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
