Abstract
Objective/Background
Lilium species possess notable medicinal properties, but their use is often limited by a bitter taste that reduces consumer acceptance. The molecular mechanisms underlying this bitterness remain unclear. The aim of this study was to investigate the role of cyanide accumulation in the bitterness of Lilium bulbs, with a focus on cyanide detoxification by the β-cyanoalanine synthase (β-CAS) enzyme.
Methods
Transcriptome sequencing was performed on three Lilium cultivars, a notably bitter Lilium brownii cultivar (YAO) and two nearly nonbitter Lilium davidii cultivars (LZ and KZ), to identify key genes involved in bitterness. This analysis led to the identification of β-cyanoalanine synthase as a candidate gene. The cyanide content was subsequently quantified in the bulbs of each cultivar, and in vitro enzyme activity assays were conducted to determine the catalytic function of β-cyanoalanine synthase.
Results
Compared with the other cultivars, the bitter cultivar presented significantly lower expression of the β-cyanoalanine synthase gene. Cyanide content analysis revealed that the bitter cultivar accumulated the greatest amount of cyanide, which was positively correlated with bitterness. Enzyme assays confirmed that β-cyanoalanine synthase catalyses the conversion of cysteine and cyanide into β-cyanoalanine, indicating its role in cyanide detoxification.
Conclusion
Reduced β-cyanoalanine synthase expression in the bitter cultivar leads to cyanide accumulation in bulbs, contributing to bitterness. These findings offer a molecular basis for improving Lilium taste through genetic modulation of β-cyanoalanine synthase expression.
Introduction
Lilium species are perennial herbaceous plants renowned for their medicinal value and edibility. 1 Their bulbs are rich in starch and bioactive compounds such as polysaccharides, steroidal saponins, and alkaloids, which confer benefits that include lung moisturization, cough relief, calming the mind, and detoxification. 1 The cultivation area of lilies in China exceeds 200 km2. However, the bitter compounds in lily bulbs negatively affect their taste, reducing consumer acceptance and limiting their market potential and widespread adoption. Investigating the mechanisms underlying bitterness in lilies is therefore crucial for developing high-quality varieties with improved taste.
Bitter compounds are widely present in plants and function as secondary metabolites, imparting unique bitterness to plants while playing key roles in growth, development, and environmental interactions.2–4 Common bitter compounds in plants include alkaloids, phenolic compounds, terpenes, flavonoids, and cyanogenic compounds, all of which exhibit diverse structures and functions. 5 Previous studies have suggested that bitterness in lily bulbs primarily results from bitter amino acids, steroidal saponins, and steroidal alkaloids.6–8 However, the results of these studies remain inconsistent, and the precise mechanism underlying bitterness in lilies has yet to be elucidated.
This study investigated the relationship between bitterness in lily bulbs and the presence of cyanide, a previously overlooked compound. Cyanide, a highly toxic bitter compound, is closely linked to plant defence mechanisms. 9 Hydrogen cyanide (HCN) effectively inhibits cytochrome oxidase activity in the respiratory chain, exerting strong toxic effects on herbivores and pathogens. 10 In plants, free cyanide is primarily stored in the form of cyanogenic glycosides, which, upon physical damage or biological stress, are rapidly hydrolysed by specific enzymes to release HCN. 11 Typical cyanogenic plants include Manihot esculenta, Sorghum bicolor, and Prunus dulcis, and cyanogenic glycoside contents vary across species and environmental conditions.12–14 To prevent self-inflicted damage, plants have evolved metabolic pathways for HCN detoxification. The classic cyanide metabolism pathway is the β-cyanoalanine synthase (β-CAS) pathway.15–17 β-CAS catalyses the first step in this pathway, in which cyanide replaces the thiol group of cysteine to produce β-cyanoalanine and release H2S as a byproduct.15–17 β-Cyanoalanine is ultimately metabolized into aspartic acid and ammonia. 15
Despite previous studies attributing Lilium bulb bitterness to amino acids, saponins, and alkaloids, the role of cyanide—a well-known bitter compound in other plants—remains unexplored.6–8 Given the inconsistent results in the literature and the potential link between cyanide metabolism and bitterness, the aim of this study was to (1) investigate whether cyanide accumulation contributes to bitterness in Lilium bulbs and (2) identify key genes (eg, β-CAS) involved in cyanide detoxification. By integrating transcriptomics, enzymatic assays, and cyanide quantification, we provide mechanistic insights into bitterness formation, offering a foundation for the breeding of sweeter Lilium varieties through metabolic engineering.
Materials and Methods
Transcriptome Analysis
Lanzhoubaihe (LZ), adopted from Lanzhou City, Gansu Province, belongs to Lilium davidii and is nonbitter. Kaizhoubaihe (KZ), adopted from Youyang County, Chongqing City, belongs to Lilium davidii and has a slightly bitter taste. Yaobaihe (YAO), adopted from Youyang County, Chongqing City, belongs to Lilium brownii and has a strong bitter taste. These three lily cultivars were cultivated uniformly in the experimental field of the Medicinal Botanical Garden at Southwest University. The experimental planting site was located at 29.82°N latitude and 106.42°E longitude and contained sandy loam soil. Bulb cuttings were planted in early March 2023, and harvesting was completed in July of the same year. During the cultivation period, the daily temperatures ranged between 20 and 32 °C. For each variety, ten plants were cultivated, and three individuals exhibiting consistent growth and optimal health were randomly selected for transcriptome sequencing and cyanide content analysis. Mature lily bulbs were harvested, flash-frozen at −80 °C, and stored immediately for subsequent transcriptome analysis and hydrogen cyanide content determination.
The BGI sequencing platform was used for the sequencing of total RNA, which was performed by the BENAGEN Company (Wuhan, China; https://www.benagen.com/). The experimental procedure was conducted in triplicate. The raw reads of the LZ, KZ, and YAO transcriptome data were uploaded to the National Genomics Data Center (https://ngdc.cncb.ac.cn/?lang=zh; BioProject: PRJCA033382). Raw reads were filtered with fastp. Differential expression analysis using DESeq2 was employed to identify genes that were differentially expressed with a selection criteria of log2FC > 2, padj <0.05, and FDR <0.01. 18 Subsequently, a Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis was conducted to determine which pathways were significantly enriched with respect to the differentially expressed genes compared with all annotated genes. 19
Determination of the Cyanide Content in Lilium Bulbs
A Spectroquant® Cyanide Test Kit (Merck, Germany) was used to determine the content of free cyanide ions in the Lilium bulbs. The procedure was based on the test kit's manual with some modifications (https://www.sigmaaldrich.cn/CN/zh/product/mm/109701). The detailed steps are as follows: First, 1 g of fresh Lilium bulb sample was accurately weighed, ground in a mortar, and diluted with water to a final volume of 10 mL. One dose of reagent CN-1 was added and mixed thoroughly. The sample was sealed, digested at 120 °C for 30 min, and then cooled to room temperature. Three drops of reagent CN-2 were added and mixed immediately. The pretreated sample was then ready for analysis of the cyanide ion content. To perform the analysis, 5 mL of the pretreated sample was added to 1 level green microspoon of reagent CN-3 and mixed rapidly. Afterwards, 1 level blue microspoon of reagent CN-4 was added, and the sample was shaken vigorously until the reagent dissolved completely. After a 10-min reaction time, 1 mL of the sample was transferred to a 10 mm cuvette, and the absorbance was measured at 578 nm using a spectrophotometer. The Cyanide Standard Solution Certipur® (Merck, Germany) served as the standard for free cyanide ions. The cyanide ion content in the lily bulbs was calculated using a standard curve.
CAS Protein Purification and Enzyme Activity Analysis
The activity of β-CAS in Lilium brownii plants was determined by heterologous expression and purification of β-CAS in Escherichia coli, followed by in vitro enzyme activity assays. The complete cDNA sequence of β-CAS was obtained from the transcriptome data of Lilium brownii. According to previous research, the β-CAS gene contains a mitochondrial localization signal, which was removed in a prokaryotic expression experiment. 20 Primers (β-CAS-F: aactttaagaaggagatataccatggcaatcccgacacagag, β-CAS-R: tggtggtggtgctcgagctcgacatccacaggctgca; the underlines represent the homologous recombination sequences) were designed to clone the β-CAS gene, and the β-CAS gene was inserted into the prokaryotic expression vector pET28a. The recombinant plasmid was subsequently transformed into E. coli BL21 (DE3) to obtain the engineered strain for protein expression. The 6×histidine-tagged protein was induced and purified following standard protocols. 21
The in vitro enzyme activity of CAS was measured as follows: A 500 μL system containing 50 mM Tris-HCl buffer (pH 8.0), 1 mM cysteine, and 1 mM potassium cyanide was used for each reaction. Fifty micrograms of purified protein was added to each reaction, with the negative control containing equal amounts of heat-inactivated protein. 17 The samples were incubated at 30 °C for 1 h, after which the reaction products were filtered through a 0.22 μm filter. The products were then analysed using a high-resolution Orbitrap Exploris™ 120 mass spectrometer (Thermo, MA, USA) coupled with a Vanquish Flex UPLC system. 22 The column temperature was set to 35 °C, and the samples were separated using a Thermo Hypersil GOLD column (2.1 × 100 mm, 1.9 μm). A binary isocratic elution method was used, with a flow rate of 0.3 mL/min. Elution was performed using a 0.1% formic acid aqueous solution and acetonitrile as the mobile phase, with a ratio of 98:2 (formic acid aqueous solution to acetonitrile), and the mass spectrometer parameters were based on those of a previous study. 23
Results
Insufficient Expression of the CAS Gene in YAO
To understand the bitterness of Lilium bulbs, the three cultivars were subjected to transcriptome sequencing (Figure 1). Differential expression analysis revealed 5276 upregulated genes and 6693 downregulated genes between LZ and YAO (Figure 1B) and 6081 upregulated genes and 6552 downregulated genes between KZ and YAO (Figure 1C). A Venn diagram revealed 4059 upregulated genes and 3660 downregulated genes in common (Figure 1D). KEGG enrichment analysis revealed a common pathway related to bitterness, named cyanoamino acid metabolism (Figure 1F). However, its role in the bitterness of Lilium has been largely overlooked in previous research.

Transcriptome Analysis Indicated That β-CAS is Insufficiently Expressed in YAO. A. PCA of Three Cultivars. B. LZ Versus YAO DEG Volcano Map. C. KZ Versus YAO DEG Volcano Map. D. Venn Diagram Showing the Number of Upregulated DEGs. E. Venn Diagram Showing the Number of Downregulated DEGs. F. KEGG Enrichment Analysis of the Shared DEGs Whose Expression was Upregulated. G. KEGG Enrichment Analysis of the Shared DEGs Whose Expression was Downregulated.
TRINITY_DN1357, a gene whose expression was significantly downregulated in YAO compared with that in LZ and KZ, attracted our attention (Figure 1B–C and Figure 2B). The gene function annotation results indicated that it encoded L-3-cyanoalanine synthase (β-CAS; β-cyanoalanine synthase; EC 4.4.1.9), which is the principal enzyme for cyanide detoxification in plants. β-CAS catalyses the first step in the cyanide assimilation pathway, which involves substitution of the sulfhydryl moiety of cysteine with cyanide to yield the nitrile β-cyanoalanine and concomitant release of H2S (Figure 2A). Therefore, we speculate that insufficient expression of the β-CAS gene in YAO leads to high accumulation of free cyanide, resulting in a bitter taste.

Insufficient Expression of the β-CAS Gene in YAO Led to High Accumulation of Free Cyanide. A. The Cyanide Assimilation Reaction Catalysed by β-CAS. B. FPKM Values of TRINITY_DN1357_c0_g1 in LZ, KZ and YAO. C. Cyanide Content in LZ, KZ and YAO. Data are Presented as the Mean±SD; n≥3. Different Letters on Columns Indicate Significant Differences at the Level of P <0.05 According to Duncan's Test.
High Accumulation of Cyanide in YAO
Comparative transcriptome analysis revealed significant differences in the expression levels of β-CAS genes involved in cyanide metabolism among the three Lilium materials. It is hypothesized that the content of free cyanide ions is among the factors contributing to the bitterness of YAO Lilium. A detection kit was used to measure the free cyanide ion content in the bulbs of the three Lilium materials. The free cyanide ion content was highest in YAO, reaching 10.29 μg/g FW (fresh weight), slightly lower in KZ at 3.61 μg/g FW, and lowest in the nonbitter LZ material at only 1.22 μg/g FW (Figure 2C). These findings demonstrate a positive correlation between the free cyanide ion content and the perception of bitterness among the three Lilium materials, suggesting that free cyanide ions are among the key substances influencing the bitter taste of Lilium.
Functional Characterization of LbCAS
The results of the free cyanate content indicate that the accumulation of free cyanate leads to a bitter taste in Lilium. It is speculated that the decreased expression level of the β-CAS gene promotes cyanate accumulation. Therefore, we further verified the function of β-CAS using in vitro enzyme activity assays. The coding region of the β-CAS gene was cloned from LZ, and after removal of the N-terminal signal peptide, it was inserted into the multicloning site of the pET28a prokaryotic expression vector to obtain the β-CAS prokaryotic expression construct. Induced expression was performed in E. coli, and the β-CAS protein was purified using a nickel-affinity chromatography column. SDS‒PAGE analysis of the purified β-CAS protein revealed a distinct band above the 35 kDa position. (Figure 3A), which is consistent with the theoretical molecular weight of β-CAS of 36.9 kDa. Using cysteine and potassium cyanide as substrates, UPLC‒MS/MS was employed to detect the catalytic reaction products of β-CAS. In the β-CAS catalytic reaction system, a compound with a retention time of 0.90 min and a mass-to-charge ratio (m/z) of 115.0502 was detected (Figure 3C). This result matches the retention time (0.89 min) and m/z value (115.0502) of the β-cyanoalanine standard (Figure 3B). In contrast, in the boiled inactivated β-CAS control sample, no compound with a m/z of 115.0502 was detected (Figure 3D). To further confirm the structure of the β-CAS catalytic product, the enzymatic reaction product was subjected to secondary mass spectrometry analysis. The secondary mass spectrometry fragmentation data of the β-CAS enzymatic product were consistent with those of the β-cyanoalanine standard (Figure 3E–F). The in vitro enzyme activity data indicate that β-CAS can catalyse the conversion of cysteine and cyanate into β-cyanoalanine.
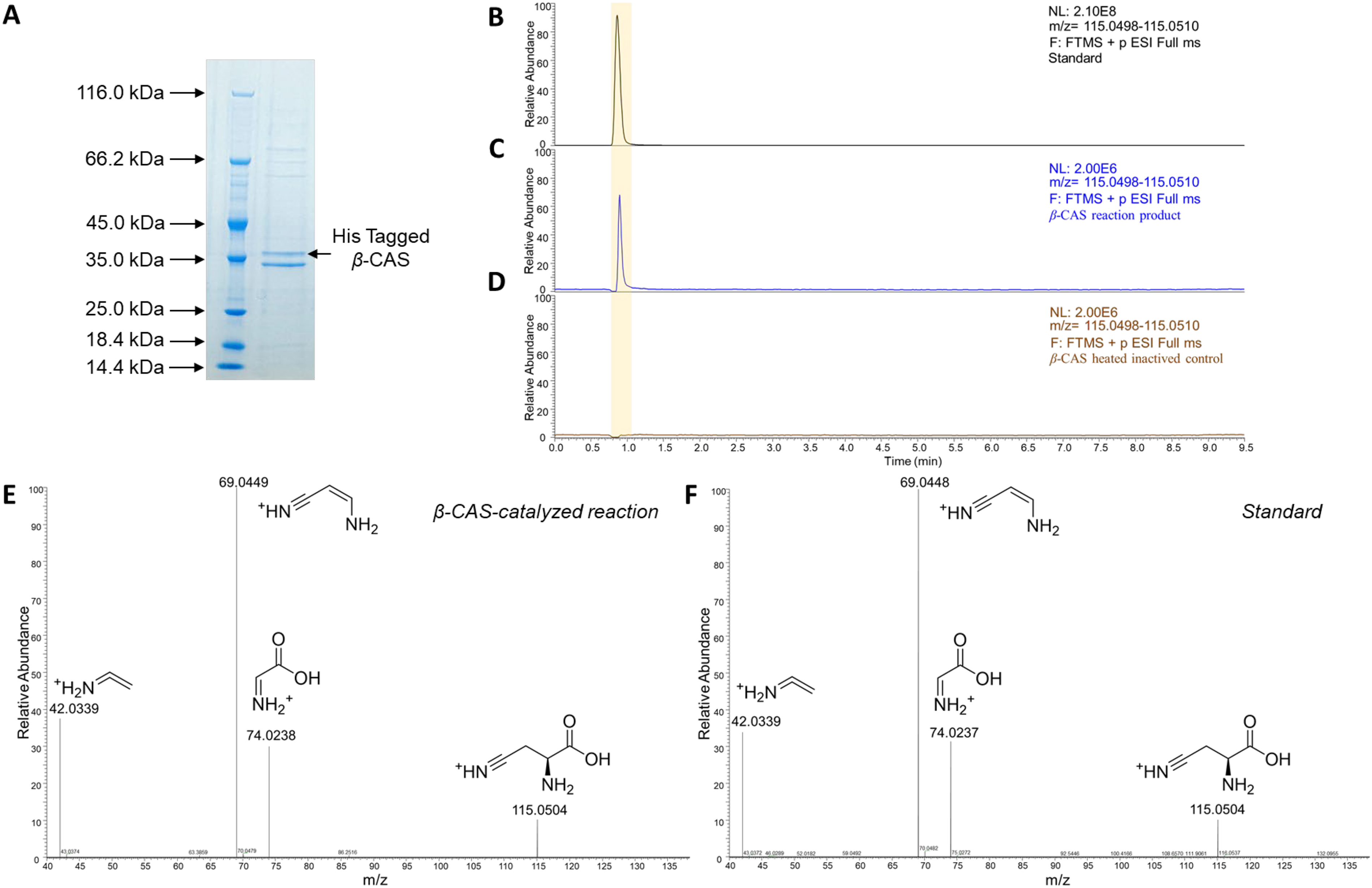
Purification and In Vitro Enzyme Activity Assay of the β-CAS Protein. A. SDS‒PAGE Analysis of β-CAS Protein Purified by Nickel Ion Affinity Chromatography. B. Extracted Ion Chromatogram of the β-Cyanoalanine Standard. C. Extracted Ion Chromatogram of the Product from the β-CAS-Catalysed Reaction. D. Extracted Ion Chromatogram of the Heat-Inactivated β-CAS Control. E. Secondary Mass Spectrometry (MS/MS) Analysis of the Product from the β-CAS-Catalysed Reaction. F. MS/MS Spectrum of the β-Cyanoalanine Standard.
Discussion
Role of Cyanide in Plants
Plants produce various bitter-tasting secondary metabolites, including structurally diverse compounds such as alkaloids, phenolic compounds, terpenes, flavonoids, and cyanogenic glycosides, each with distinct ecological and physiological functions. 5 With respect to Lilium species specifically, existing research has shown that the characteristic bitterness in bulb tissues is predominantly attributed to three classes of compounds: bitter amino acids, steroidal saponins, and steroidal alkaloids.6–8 Notably, previous studies have reported inconsistent conclusions regarding the specific class of compounds responsible for bitterness in Lilium bulbs. However, our findings demonstrate that cyanide accumulation due to impaired β-CAS activity is a key contributor to bitterness in Lilium brownii (YAO), which aligns with reports in other cyanogenic plants, such as bitter almonds (Prunus dulcis) and cassava (Manihot esculenta).24–26
Cyanide is widely recognized as a secondary metabolite in plants and is often associated with defence mechanisms against herbivores and pathogens. 27 Many plant species, including bitter almonds, bamboo shoots, and cassava, produce cyanogenic glycosides, which release HCN upon receiving tissue damage.24–26 This serves as an effective chemical deterrent, preventing excessive predation. Additionally, cyanide is involved in plant physiology beyond defence. Studies have shown that at low concentrations, cyanide functions as a signalling molecule that regulates processes such as seed germination, root development, and stress responses.28–32 However, excessive accumulation can be detrimental, disrupting cellular respiration by inhibiting cytochrome c oxidase (COX) in mitochondria. 33 To avoid self-toxicity, plants use β-cyanoalanine synthase (β-CAS) to detoxify HCN by catalysing the conversion of cyanide and cysteine into β-cyanoalanine (with H2S as a byproduct), which is subsequently metabolized to aspartate and ammonia.15–17 Understanding the dual role of cyanide in plants provides valuable insights into its regulation and potential manipulation to balance defence and plant growth.
Medicinal Value and Toxicity of Cyanide in Humans
Cyanide has a complex effect on human health and is both a potent toxin and a potential therapeutic agent. 34 Its toxicity arises from its ability to inhibit mitochondrial electron transport, leading to cellular asphyxiation. 33 Acute cyanide poisoning can be fatal, while chronic exposure—such as from cyanogenic food sources—has been linked to neurological disorders and thyroid dysfunction. Despite its toxicity, cyanide has medicinal applications. 34 Sodium nitroprusside, a cyanide-releasing compound, is used as a vasodilator to treat hypertension and heart failure. 35 Additionally, cyanogenic glycosides from plants have been explored for use in anticancer therapies, such as leveraging controlled cyanide release to selectively target tumour cells. 36 Therefore, the medicinal value of Lilium brownii (Yaobaihe) may also be related to its high cyanide accumulation. The dual nature of cyanide highlights the importance of precise regulation in both medical and dietary contexts to maximize benefits while minimizing risks.
Controlling Cyanide Content Through Metabolic Engineering and Breeding
Regulating cyanide levels in plants is crucial for ensuring food safety and improving crop quality. Traditional breeding approaches have been used to develop low-cyanide cultivars of crops such as cassava, mitigating the risks of cyanide poisoning during human consumption. 37 In recent years, metabolic engineering has emerged as a powerful tool for modulating cyanide biosynthesis and detoxification pathways. Overexpressing the key gene β-cyanoalanine synthase (β-CAS) has demonstrated potential in reducing cyanide accumulation while maintaining plant defence capabilities.31,38 Our findings suggest that insufficient HCN metabolism contributes to the bitterness of Lilium bulbs (Figure 1 and Figure 2). The function of the key enzyme β-CAS from Lilium was validated through in vitro enzymatic activity assays (Figure 2). By integrating advanced biotechnological approaches with selective breeding and targeting β-CAS as a key regulatory gene, it is possible to develop plant varieties with optimized cyanide levels, increasing both agricultural safety and economic value.
Bitterness Beyond Cyanide: Limitations and Future Directions
While this study provides evidence linking cyanide accumulation to the bitterness of Lilium bulbs and identifies β-CAS as a key gene involved in cyanide detoxification, several limitations should be noted. First, the analysis was conducted on a limited number of cultivars, which may not fully capture the genetic and metabolic diversity present in the Lilium genus. Further validation across a broader range of bitter and nonbitter cultivars is necessary to generalize the findings. Second, although the role of β-CAS was functionally verified through in vitro enzymatic assays, in planta functional validation—such as gene silencing or overexpression experiments in Lilium—was not performed. Such studies could provide more direct evidence of the causal relationship between β-CAS activity and bitterness. Third, the focus of this study was the free cyanide content, while the potential contributions of cyanogenic glycosides and other bitter compounds remain unexplored. Integrating metabolomic analyses targeting a wider array of bitter-related compounds could provide a more comprehensive understanding of bitterness in Lilium. The aim of future research should be to address these limitations by incorporating more cultivars, conducting genetic manipulation experiments, and expanding metabolite profiling to elucidate the mechanisms underlying taste regulation in Lilium species.
Conclusion
In summary, these findings suggest that insufficient β-CAS expression in YAO leads to cyanide buildup, contributing to a bitter taste. These insights into cyanide metabolism not only deepen our understanding of bitterness in Lilium bulbs but also provide a foundation for applied strategies in food security and medicinal research.
Supplemental Material
sj-jpg-1-npx-10.1177_1934578X251392006 - Supplemental material for Accumulation of Hydrogen Cyanide Contributes to Bitterness in Lilium Bulbs
Supplemental material, sj-jpg-1-npx-10.1177_1934578X251392006 for Accumulation of Hydrogen Cyanide Contributes to Bitterness in Lilium Bulbs by Xianhui Huang, Junlan Zeng, Ting Li, Yuan Pan, Feng Xie, Yicheng Wang, Lingjiang Zeng, Dongchao Zhao, Xiuhua Li, Zhihua Liao, Daxia Chen and Chunxian Yang in Natural Product Communications
Footnotes
Acknowledgments
Thanks to the Integrated Science Center of Germplasm Creation in Western China (CHONGQING) Science City for providing mass spectrometry for metabolome analysis. Thanks for the support of the Academy for Advanced Interdisciplinary Studies Device Sharing Service Platform, Southwest University for transcriptome analysis.
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
Investigation, X.H., J.Z., T.L., Y.P., F.X., and Y.W.; methodology, L.Z. and D.Z.; Writing—Original Draft, J.Z.; Resources, C.Y., D.C., and Z.L.; Writing—Review and Editing, X. L., and C.Y. Conceptualization and Funding acquisition, C.Y., J. Z. and D.C. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Special Key Project of Chongqing Technology Innovation and Application Development (cstc2021jscx-gksbX0015) and Key project at central government level: The ability establishment of sustainable use for valuable Chinese medicine resources (2060302).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
