Abstract
The rising prevalence of liver diseases poses a significant global health challenge. This study evaluates the hepatoprotective and antioxidant potential of the ethyl acetate fraction of Combretum paniculatum (EFCP) against carbon tetrachloride (CCl4)-induced hepatotoxicity in albino rats. Phytochemical analysis of EFCP was conducted using spectrophotometry and gas chromatography-mass spectrometry (GC-MS), while its in vitro antioxidant activity was assessed via 2,2-diphenyl-1-picrylhydrazyl (DPPH) and ferric reducing-antioxidant power (FRAP) assays. Rats were divided into five groups: Group 1 (normal control) received no treatment, while Groups 2-5 were administered CCl4 (100 mg/kg body weight). Group 2 remained untreated, whereas Groups 3-5 received silymarin (100 mg/kg b.w.) or EFCP (100 and 200 mg/kg b.w.) for 14 days. Biochemical and histological analyses were conducted to assess liver and kidney function, lipid profile, oxidative stress markers, and haematological parameters. EFCP was rich in bioactive phytochemicals and exhibited strong antioxidant activity (IC50 values: 3.47 μg/mL for DPPH, 207.70 μg/mL for FRAP). CCl4 intoxication significantly increased malondialdehyde (MDA), liver enzymes, bilirubin, and lipid profile markers while depleting antioxidant defenses and high-density lipoprotein cholesterol (HDL-C). Additionally, haematological and kidney function abnormalities were observed. EFCP treatment improved antioxidant status, HDL-C levels, and haematological parameters while reducing MDA, lipid profile, liver, and kidney dysfunction markers. Histopathological analysis revealed restored hepatic architecture with reduced hepatocyte necrosis, tubular degeneration, and inflammation in EFCP-treated groups. These findings suggest that EFCP possesses potent hepatoprotective properties and may serve as a promising therapeutic agent against hepatotoxicity and oxidative stress-related liver damage.
This is a visual representation of the abstract.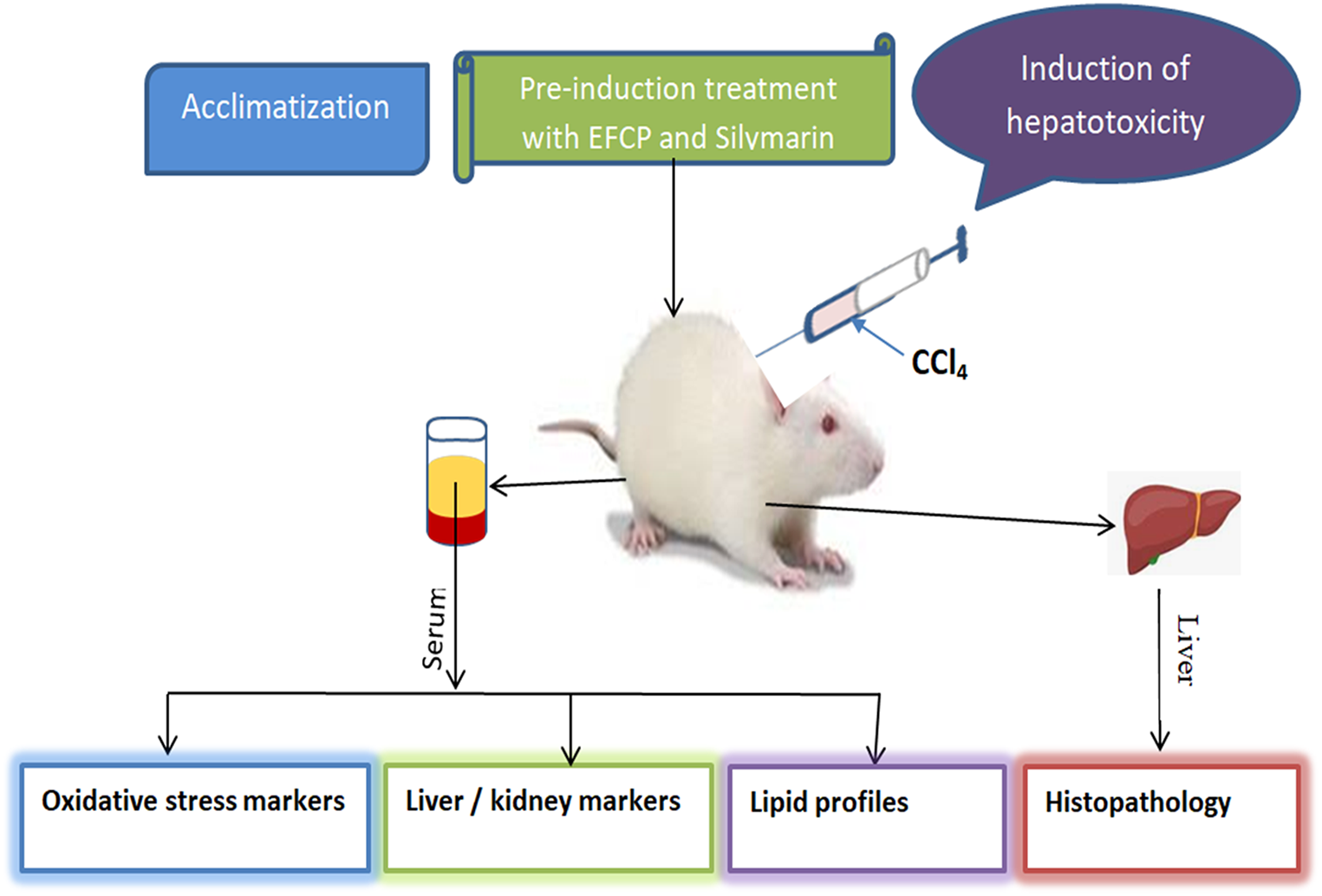
Keywords
Introduction
The liver plays a central role in metabolizing xenobiotics and endogenous substrates, thereby maintaining homeostasis. 1 Additionally, it is crucial for bile secretion and formation, immune defense, blood volume regulation, and growth signaling pathways. 2 However, liver diseases have become a critical global health challenge, accounting for approximately 2 million deaths annually, representing 4% of all global fatalities. The mortality rate has surged by 64% over two decades, particularly in high-income nations. 3 This increasing burden is further exacerbated by metabolic syndrome, with the global prevalence of non-alcoholic fatty liver disease (NAFLD) reaching 30.05% and projected to double by 2030. 4 Additionally, alcohol-related liver diseases (ALD) with global mortality rate estimated at around 25% may develop into severe diseases not limited to hepatic cirrhosis, steatohepatitis, and hepatocellular carcinoma 5
Frequent exposure to drugs, chemicals and environmental toxins remains a significant etiological factor contributing to acute hepatic injury6,7 Toxicant-mediated liver damage is characterized by extensive hepatocyte necrosis, neutrophil infiltration into parenchymal cells, hepatocellular membrane disruption, oxidative stress, and activation of hepatic stellate cells, culminating in progressive liver dysfunction. 8 Notably, human exposure to hepatotoxic agents such as acetaminophen (a common analgesic and antipyretic), ethanol (in alcoholic beverages), and carbon tetrachloride (CCl4) (present in contaminated water, air, soil, and food) plays a major role in the onset and progression of liver diseases. 9
Carbon tetrachloride (CCl4), also known as tetrachloromethane, is a halogenated organic toxicant extensively used in experimental hepatotoxicity models due to its potent liver-damaging effects. It is metabolized in the liver via the cytochrome P450 2E1 (CYP2E1) enzyme into the highly reactive trichloromethyl radical (CCl3). This radical subsequently interacts with molecular oxygen, forming trichloromethyl peroxyl radical (CCl3O2•), a key initiator of lipid peroxidation. 10 Apart from suppressing hepatocyte protein synthesis, CCl4-derived radicals cause extensive oxidative damage, disrupt biochemical pathways, compromise membrane integrity, and deplete endogenous antioxidant defenses, leading to widespread hepatic injury.
Emerging evidence has demonstrated that CCl4-induced hepatotoxicity is mediated through multiple molecular pathways, including toll-like receptor 4 (TLR4), autophagy, p53, transforming growth factor-β1 (TGF-β1), mitogen-activated protein kinase (MAPK), and nuclear factor-κB (NF-κB) signaling. 11 These pathways collectively regulate antioxidant responses and inflammation. Oxidative stress, a key feature of CCl4 toxicity, arises when prooxidant levels exceed the antioxidant defense capacity, leading to redox imbalance. 12 Given the liver's crucial role in metabolism and detoxification, it is particularly susceptible to oxidative damage. Conventionally, mitigating liver dysfunction involves enhancing endogenous antioxidant systems such as catalase (CAT), superoxide dismutase (SOD), and glutathione (GSH) to counteract excessive reactive metabolite generation. 9
Recently, there has been growing interest in medicinal plants as alternative therapies for hepatotoxicity, primarily due to their potent antioxidant properties, high therapeutic efficacy, safety, biocompatibility, cost-effectiveness, and multi-targeted mechanisms of action.5,13–15 Among these, Combretum paniculatum, a flowering plant of the Combretaceae family native to West Africa, has been traditionally used by the Igbo people for managing hepatic enlargement and jaundice. It has also been reported to possess medicinal properties for treating diarrhea, anesthetic conditions, ringworm infections, joint pain, chronic dysentery, and liver disorders. 16 Additionally, pharmacological studies indicate that C. paniculatum showed no toxic effect up to a dose of 5000 mg/kg and exerts anti-inflammatory, antimicrobial, anticancer, and antioxidative effects 16 It has demonstrated gastroprotective activity against indomethacin-induced ulcers and antimicrobial efficacy against hepatopathy-associated pathogens like Escherichia coli. 17
The selection of the ethyl acetate fraction of C. paniculatum in this study aligns with phytochemical evidence showing that Combretaceae extracts exhibit 42% higher phenolic content compared to aqueous preparations, optimizing the isolation of non-polar antioxidants capable of scavenging lipid-soluble CCl3 radicals. 18 This study, therefore, investigates protective effects of Combretum paniculatum fraction on CCl4-induced acute liver injury. A comprehensive biochemical analysis was conducted to evaluate antioxidant status, lipid profile, liver and kidney function markers, electrolytes, haematological indices, and histopathological alterations. These markers aim to elucidate the molecular mechanisms underlying EFCP's protective effects and assess its potential as a plant-derived therapeutic candidate for oxidative stress-induced liver damage.
Materials and Methods
Plant Collection and Identification of Plant Material
Fresh leaves of Combretum paniculatum were collected from Nsukka Local Government Area, Enugu State, Nigeria, between November and December 2021. Taxonomic identification was performed by Mr Alfred Ozioko at the International Centre for Ethnomedicine and Drug Development (InterCEDD). A voucher specimen was deposited at the research institute under the reference number INTERCEDD/19099.
Processing and Preparation of the Crude Extract
The collected leaves were air-dried and ground into a fine powder. A total of 300 g of the powdered leaves was macerated in 2 L of 70% ethanol and 30% distilled water (v/v) for 72 h with intermittent shaking and stirring. The extract was first filtered through muslin cloth and subsequently through Whatman No. 1 filter paper. The filtrate was concentrated to dryness under reduced pressure using a rotary vacuum evaporator at 45 °C to obtain 32 g of the extract (10.67% yield).
Solvent-solvent Partitioning of the Crude Extract
The crude extract was fractionated using solvents of varying polarity. First, 30 g of the extract was dissolved in 200 mL of aqueous ethanol (20% v/v) and transferred into a separating funnel. An equal volume (200 mL) of n-hexane was added, and the mixture was vigorously shaken and allowed to stand for 20 min, enabling phase separation. The n-hexane fraction was collected, and the remaining aqueous-ethanol fraction was further partitioned with 200 mL of ethyl acetate following the same procedure. All fractions were evaporated to dryness and subjected to preliminary analysis. The ethyl acetate fraction of C. paniculatum (EFCP) was selected for this study due to its superior antioxidant activity.
Spectrophotometric and Gas Chromatography-Mass Spectroscopy Phytochemical Analysis of EFCP
Phytochemical quantification of EFCP was conducted using a UV-visible spectrophotometer to determine the concentrations of alkaloids, phenols, tannins, terpenoids, flavonoids, glycosides, and reducing sugars, following standard methods described by Harborne 19 and Trease & Evans. 20 Gas chromatography-mass spectrometry (GC-MS) analysis was performed using an electron ionization device operating at 70 eV. Helium gas (99.995% purity) was used as the carrier at a constant flow rate of 1 mL/min. The initial column temperature was set at 50 °C and ramped up to 150 °C at a rate of 3 °C per minute, with a holding time of 10 min. The temperature was then further increased to 300 °C at a rate of 10 °C per minute. A 1-µL aliquot of the EFCP solution (1%) was injected in splitless mode. Compound identification was achieved by comparing the retention times (RT) and mass spectra with reference standards from the Replib and Mainlab databases. The relative concentrations of each compound were expressed as percentages.
The Acute Toxicity and Tolerated Dose of EFCP
The safety profile and maximum tolerated dose of EFCP were assessed following Lorke's 21 experimental protocol. Eighteen Wistar rats were randomized into six groups (n = 3) and orally administered EFCP at doses of 10, 100, 1000, 1600, 2800, and 5000 mg/kg body weight. The animals were observed for 48 h for any signs of toxicity, behavioral changes, or mortality.
Experimental Animals
Twenty-five albino Wistar rats (90 ± 5.14 g) were obtained from the Animal House of the Faculty of Veterinary Medicine, University of Nigeria, Nsukka, Nigeria. The rats were housed in a controlled laboratory environment in stainless steel cages under a 12-h light/dark cycle at a temperature of 23-26 °C, with unrestricted access to water and a standard rodent diet. They were acclimatized for two weeks prior to the experiment. All procedures adhered to the National Institutes of Health Guide for the Care and Use of Laboratory Animals 22 and were approved by the Faculty of Biological Sciences Ethical Committee (Approval No.: UNN/FBS/EC/1082). The reporting of this study conforms to the ARRIVE 2.0 guidelines for animal research, 23 which provide a standardized framework for reporting animal experiments to ensure transparency, reproducibility, and ethical integrity.
Study Design, Induction of Toxicity, and Treatment
The rats were randomly assigned into five groups (n = 5 per group). Group 1 served as the normal control and received neither CCl4 nor treatment. Groups 2-5 were administered CCl4 (100 mg/kg body weight) orally on days 1 and 2, 24 h apart, to induce hepatotoxicity. Group 2 remained untreated (CCl4 control), while Group 3 received the standard hepatoprotective agent, silymarin (100 mg/kg b.w.). Groups 4 and 5 were treated with EFCP at doses of 100 mg/kg b.w. and 200 mg/kg b.w., respectively. All treatments were administered orally for 14 consecutive days. On day 15, the rats were sacrificed, and blood and liver samples were collected for biochemical and histological analyses.
Anesthesia and Euthanasia
To ensure the humane treatment of animals, anesthesia and euthanasia procedures were conducted in accordance with ethical guidelines. At the conclusion of the experiment, the rats were euthanized humanely using an intraperitoneal injection of sodium pentobarbital (200 mg/kg), a barbiturate agent, following the guidelines of the American Veterinary Medical Association (AVMA). Prior to disposal of the carcasses, the animals’ breathing patterns, pupillary responses to light, and heartbeats were carefully examined to confirm death, ensuring compliance with ethical standards for animal research.
Determination of Serum Oxidative Stress Parameters
Lipid peroxidation was assessed by quantifying malondialdehyde (MDA) levels using the method described by Wallin et al 24 method. MDA, which forms a complex with thiobarbituric acid (TBA) under acidic conditions, was measured spectrophotometrically at 532 nm. Superoxide dismutase (SOD) activity was evaluated based on its ability to catalyze the dismutation of superoxide radicals to hydrogen peroxide in an alkaline medium, following Fridovich's 25 protocol. Catalase (CAT) activity was determined using Aebi's 26 method by measuring the decomposition of hydrogen peroxide (H2O2) into water (H2O) and oxygen (O2), with absorbance recorded at 240 nm. Glutathione (GSH) levels were quantified according to Beutler's 27 method, while vitamin E and vitamin C concentrations were determined following protocols outlined by ref. 28 and , 29 respectively.
Determination of Serum Liver Markers
The activities of aspartate transaminase (AST), alanine transaminase (ALT), and alkaline phosphatase (ALP) were determined using the method of Reitman and Frankel. 30 AST activity was measured based on the formation of 2,4-dinitrophenylhydrazine from oxaloacetate hydrazine, while ALT activity was determined by quantifying pyruvate hydrazone formation at 546 nm. ALP activity was assessed by measuring the formation of a pink-colored complex in an alkaline medium upon hydrolysis of phenolphthalein monophosphate. Total protein (TP) and albumin (AB) concentrations were determined using the methods of Tietz 31 and Doumas 32 methods, respectively. Total bilirubin (TB) levels were measured at 405 nm by assessing the release of albumin-bound bilirubin upon reaction with diazotized sulfanilic acid.
Determination of Serum Lipid Profile
Serum lipid parameters were analyzed using standard methods. High-density lipoprotein cholesterol (HDL-C) was measured using Quimica Clinical Aplicada (QCA) kits, following the method of Albers et al 33 methods. This method involves precipitating low-density lipoprotein (LDL) and very low-density lipoprotein (VLDL) using a polysaccharide in the presence of divalent cations, followed by absorbance measurement at 546 nm. LDL-C was calculated as the difference between total cholesterol and cholesterol in the supernatant after LDL precipitation with polyvinyl sulfate (PVS). Triacylglycerol (TAG) levels were determined using Randox commercial kits following the method of ref. 33 while total cholesterol was measured using the procedure described in ref. 34 with Quimica Clinical Aplicada (QCA) commercial kits.
Determination of Serum Kidney Status
Serum urea and creatinine levels were measured using Randox commercial kits following the method described in ref. 35 Serum electrolyte levels, including sodium (Na+), potassium (K+), bicarbonate (HCO3−), and chloride (Cl−), were analyzed using Micropoint Diagnostic kits according to the method of Tietz. 31
Determination of Serum Haematological Parameters
Haematological parameters, including hemoglobin (Hb), red blood cell count (RBC), white blood cell count (WBC), platelet count (PLT), and packed cell volume (PCV), were measured using a fully automated haematology analyzer. The analyzer detects electrical pulses generated when blood cells pass through a channel surrounded by electrodes. Each pulse represents an individual cell, and pulse height correlates with cell volume. The system utilizes reagents such as diluents, lysing agents, and cleaning solutions for optimal performance.
Determination of Inflammation and Cardiac Markers
Lactate dehydrogenase (LDH) activity was measured using Randox diagnostic assay kits following the manufacturer's instructions. The assay quantifies NADH formation from lactate oxidation to pyruvate, catalyzed by lactate dehydrogenase in the presence of NAD+. The rate of NADH formation, which is directly proportional to serum LDH activity, was monitored at 340 nm using an enzyme-linked immunosorbent assay (ELISA) kit. C-reactive protein (CRP) levels were also assessed using an ELISA kit.
Histological Examination
Liver tissue samples were fixed in 10% buffered formalin and processed through a series of ascending ethanol concentrations (50%, 70%, 90%, and absolute ethanol ≥99.5%) for dehydration. Samples were subsequently cleared with xylene (two changes, one hour each) and embedded in paraffin wax. Tissue sections (5 μm thick) were cut, mounted on glass slides, and deparaffinized before staining with hematoxylin and eosin (H&E). Histopathological examinations were conducted using a BIO CEL500× light microscope at ×100 and ×400 magnification. Photomicrographs were captured and analyzed for histopathological alterations.
Statistical Analysis
Data were analyzed using one-way analysis of variance (ANOVA) followed by post hoc multiple comparisons to determine significant differences among experimental groups. Statistical significance thresholds were set at P < .05, P < .01, P < .001, or P < .0001. Results were expressed as mean ± standard deviation (SD). Nonlinear regression curves were generated using GraphPad Prism (Version 9.5) to determine the half-maximal inhibitory concentration (IC50) and regression coefficient (R²) values.

Chromatogram of Phytochemicals Identified from EFCP Using GC-MS.
Results and Discussion
Phytochemicals Composition of EFCP
Phytochemicals play a crucial role in the pharmaceutical and food industries due to their diverse biological and therapeutic properties. Increasing interest in medicinal plants has driven the screening of their phytochemical constituents as potential agents for developing novel treatments for liver diseases. 11 In this study, EFCP was found to be rich in phenols, flavonoids, reducing sugars, and alkaloids, with moderate amounts of tannins and terpenoids, while glycosides and steroids were present in lower concentrations (Table 1).
Quantitative Phytochemical Compositions of EFCP.

Phenolic compounds and flavonoids are well-documented for their antioxidant properties, including metal ion chelation, free radical scavenging, and modulation of signaling pathways and cellular mediators.36,37 Alkaloids, which were abundantly present in EFCP, have garnered significant research interest due to their wide-ranging pharmacological activities, including antimicrobial, anticancer, immunomodulatory, anti-inflammatory, and antioxidant effects. 38
Gas chromatography-mass spectrometry (GC-MS) analysis further revealed that EFCP contains bioactive compounds with diverse pharmacological properties, including anti-inflammatory, anticancer, antimicrobial, antioxidant, and hepatoprotective activities (Figure 1, Table 2). Among the key bioactive constituents, pregnenolone, identified as a major component of EFCP, has been reported to exhibit anti-inflammatory properties, enhance immune function, and protect neuronal health. Additionally, 9-octadecanoic acid and hexadecanoic acid, both present in EFCP, have demonstrated anticancer, anti-androgenic, anti-inflammatory, 5-alpha reductase-inhibitory, hypocholesterolemic, and antioxidant activities. 39
Compounds Identified in Ethyl Acetate Fraction of C. paniculatum Using GC-MS.

Results are displayed as means ± SD (n = 3).
Squalene, another compound detected in EFCP, is recognized as a potent antioxidant known for its ability to quench singlet oxygen, thereby protecting skin from peroxidation. Beyond its antioxidative function, squalene has been shown to exert anticancer, antimicrobial, cardioprotective, immunomodulatory, hypocholesterolemic, and anti-inflammatory effects.39,40 Furthermore, methyl dihydroisosteviol, which was also identified in EFCP, has been widely studied for its antiviral, antibacterial, anticancer, neuroprotective, antihypertensive, antidiabetic, cardioprotective, anti-inflammatory, and antioxidant properties. 41
Notably, steviol and its derivatives have been reported to modulate nuclear factor-κB (NF-κB) expression in CCl4-induced liver cirrhosis and fibrosis. These compounds exert their hepatoprotective effects by suppressing excessive pro-inflammatory cytokine production and inhibiting profibrogenic signaling pathways. 41 The presence of these bioactive compounds in EFCP underscores its potential as a promising natural therapeutic agent for oxidative stress-related liver disorders.
In Vitro Antioxidant Activities of EFCP
The DPPH scavenging activity is a well-established model for investigating the antioxidant potentials of both synthetic and natural agents.42,43 It quantifies a test compound's hydrogen/electron-donating capacity to stabilize the DPPH radical (violet) to the hydrazine form (yellow). In this study, EFCP demonstrated strong DPPH radical scavenging activity, surpassing the standard antioxidant ascorbic acid, with IC50 values of 3.47 and 4.45 µg/mL, respectively. The varying concentrations of EFCP and ascorbic acid positively correlated with their inhibitory capacities, as evidenced by their regression coefficients of 0.9791 and 0.8833, respectively (Figure 2A).

DPPH Radical (A) and FRAP (B) Scavenging Activities of EFCP. The Results are Displayed as Mean ± SD (n = 3).
Additionally, FRAP assay further confirmed the antioxidant capability of EFCP, as it reduced the ferricyanide/Fe complex into the Fe/ferrous form. The FRAP result, expressed in gallic acid equivalent (GAE), showed an IC50 value of 207.70 µg/mL with a regression coefficient of 0.9577 (Figure 2B). These findings suggest that EFCP can effectively neutralize free radicals generated by CCl4, thereby preventing lipid peroxidation and associated health complications. The potent antioxidant activity of EFCP is likely due to its high phenolic content, as phenols exert their antioxidative effects through hydrogen donation and the subsequent delocalization of unpaired electrons along multiple phenolic rings.44,45
Median Lethal Dose and Acute Toxicity Study of EFCP
Medicinal plants are increasingly used as alternatives to conventional treatments to mitigate the adverse effects of synthetic drugs on human organs, including the liver. 11 However, toxicological assessments of medicinal plants are essential to ensure their safety for human use. Acute toxicity studies help determine safe dosage ranges for animal studies and clinical trials and identify potential behavioral or physiological adverse effects. 38 In this study, the acute toxicity assessment revealed that EFCP was safe up to a dosage of 5000 mg/kg body weight, as no observable behavioral or morphological abnormalities, nor mortality, were recorded at this concentration (Table 3). This finding aligns with previous research on the acute toxicity of C. paniculatum ethanol extract. 16 Based on these results, we proceeded to evaluate the effects of EFCP on the biochemical and histological profiles of CCl4-intoxicated rats.
Median Lethal Dose and Acute Toxicity Study (LD50) of EFCP.

n = 3
Effects of EFCP on Oxidative Stress Biomarkers
Oxidative stress biomarkers provide a direct measure of the redox balance within cells following exposure to toxicants such as CCl4. 46 Malondialdehyde (MDA) is one of the most widely used biomarkers for assessing lipid peroxidation and oxidative stress.1,40 Cellular defense against oxidative damage primarily relies on enzymatic antioxidants, including catalase (CAT) and superoxide dismutase (SOD), which play critical roles in detoxifying reactive oxygen species. SOD catalyzes the conversion of superoxide anions into hydrogen peroxide (H2O2), which is subsequently broken down by CAT into water and oxygen, thus preventing oxidative damage. 40 Cellular defense against oxidative damage primarily relies on enzymatic antioxidants, including catalase (CAT) and superoxide dismutase (SOD), which play critical roles in detoxifying reactive oxygen species. SOD catalyzes the conversion of superoxide anions into hydrogen peroxide (H2O2), which is subsequently broken down by CAT into water and oxygen, thus preventing oxidative damage. 47
The reduction in antioxidant enzyme activity observed in the untreated rats may be due to excessive oxidative stress, which alters enzyme expression and function or leads to their overutilization in detoxifying CCl4 metabolites. Interestingly, EFCP treatment significantly restored these biochemical parameters (Figure 3). The marked depletion of MDA levels in EFCP-treated rats suggests that EFCP effectively inhibits the propagation of lipid peroxides, a critical step in oxidative chain reactions. Concurrently, the improvement in SOD and CAT activities in the treated groups indicates a reinforcement of the endogenous antioxidant defense system. Furthermore, the significant increases in non-enzymatic antioxidants such as GSH, vitamins C, and E in the EFCP-treated groups highlight the fraction's ability to replenish antioxidant reserves, thereby amplifying its protective effects.47,48

Effect of EFCP on serum Oxidative Stress Parameters of CCl4 Intoxicated Rats. The Results are Displayed in the Mean ± SD (n = 5). * (P < .05), ** (P < .01), *** (P < .001), **** (P < .0001) Denote Significant Differences Compared with the Untreated Group. MDA; Malondialdehyde; SOD; superoxide dismutase; CAT; catalase; GSH; glutathione.
Effects of EFCP on Liver Markers
It has been established in previous research that CCl4 induces hepatotoxicity via activation of liver resident macrophages (Kupffer cells). 47 Subsequently, Kupfer cells enhance the release of neutrophils, which are involved in generating excessive ROS and inflammatory cascades in the hepatocytes, leading to hepatotoxicity. 47 Compared with the normal rats, the untreated CCl4 group 2 had significantly higher ALT, AST, and ALP activities, and TB levels. In contrast, the TP and AB levels were lower in the untreated rats than in the normal rats in group 1 (Table 4). Damages to hepatocyte membranes release intracellular enzymes, ALT and AST, into the bloodstream. Hence, clinically, they are important signs of damage to hepatocellular units. 7 Similarly, increased ALP and TB levels signify cholestasis and impaired bile flow, further substantiating liver dysfunction. Treatment with EFCP markedly reduced these aberrations, indicating its hepatoprotective potentials. Apparently, the recorded decrease in MDA in the treated rats might have reduced lipid peroxidation, which prevented hepatic damage and subsequent leakage of the liver enzymes into circulation. The decrease in protein level under liver damage is primarily due to sequestration of circulating albumin in ascitic fluid and extracellular space. Importantly, albumin is the most significant prognostic factor of liver damage. In fact, patients with liver damages undergoes irreversible and reversible posttranslational changes in properties of not only quality of albumin but also decrease in serum albumin. 49 The improvement in TP and AB levels in treated groups further supports the ability of the fraction to restore liver synthetic function. These effects are probably caused by phytochemicals like flavonoids, tannins, and phenolic acids, which are known to remove reactive oxygen species (ROS) and bind metal ions. 40
Effect of EFCP on Serum Liver Markers of CCl4 Intoxicated Rats.

The results are displayed in the mean ± SD (n = 5). * (P < .05), ** (P < .01), *** (P < .001), **** (P < .0001) denote significant differences compared with the untreated group. AST; aspartate transaminase ALT; alanine transaminase, ALP: alkaline phosphatase, TP; total protein, TB; total bilirubin, AB; albumin.
Effects of EFCP on Lipid Panels
Changes in lipid panels such as TC, TAG, LDL-C, and HDL-C provide vital clues on animals’ predispositions to cardiovascular diseases (CVDs) and their associated complications.50,51 Ideally, the liver coordinates the metabolism of lipids in metabolic organs, especially the adipose tissue, under steady-state physiological conditions through tightly regulated signalling pathways to form free fatty acids stored as non-toxic TAG. 52 However, liver damage enhances fatty acid uptake by hepatocytes to synthesise TAG. 52 In this study, CCl4 intoxication provoked a significant increase in serum levels of TC, TAG, and LDL-C and a decrease in HDL-C in the untreated rats compared to the normal rats (Figure 4). These results are in tandem with the findings of Chu, et al 1 who also recorded decreased HDL-c and the increased TG, TC, and LDL-c in rats exposure to Microcystin-LR. This marked increase in LDL-C, TAG, and TC levels after CCl4 intoxication could be attributed to increased synthesis, lipid peroxidation, and hydrolysis of polyunsaturated fatty acids. In addition, changes in cholesterol efflux proteins, reverse cholesterol transport, apolipoproteins, and acute phase proteins are significant factors contributing to the reduced HDL-C in animal cells. 53 Thus, it could be inferred that CCl4 metabolites altered the lipid metabolism regulatory pathways, leading to dyslipidemia. Our in vivo lipid peroxidation assay results, which recorded an elevated level of MDA in the untreated group, support this finding. We realised that treatment with EFCP effectively elevated the HDL-C level while concurrently reducing LDL-C, TAG, and TC levels, compared to the untreated group ((Figure 4). Remarkably, prior studies have registered that HDL-C helps to export lipids to the liver for excretion via the bile and as such circumvent the onset of CVDs. 53 This result is in line with those of Njoku et al, 50 and Altinok-Yipel et al 54 who found the potency of Costus afer extract and some algae species in alleviating hyperlipidemia, respectively. The fraction's antioxidant components could be in charge of the lipid lowering effects. Specifically, flavonoids, tannins, and phenols have demonstrated antioxidant actions in several clinical and animal studies. Moreover, squalene identified in EFCP is a powerful antioxidant known to reduce cholesterol levels by increasing faecal excretion and inhibiting lipid peroxidation. 39 Also, 9-octadecanoic acid and hexadecanoic acid, abundant in the EFCP, help prevent the onset of atherosclerosis by lowering LDL. 55

Effects of EFCP on Serum Lipid Profile of CCl4 Intoxicated Rats. The Results are Displayed in the Mean±SD (n=5). * (P < .05), ** (P < .01), *** (P <.001), **** (P < .0001) Denote Significant Differences Compared With the Untreated Group.
Effects of EFCP on Kidney Parameters
Emerging research outcomes have revealed that CCl4 autoxidation triggers renal oxidative stress and consequently tubular, tubuloe-interstitial, and glomerular dysfunction. 44 Besides, the disrupted redox equilibrium in favor of oxidants makes renal tissues susceptible to lipid peroxidation, protein modification, and DNA damage, which culminates in cellular necrosis, cell death, and renal failure.44,56 Here, we realized that CCl4 exerted nephrotoxic effects on the kidney tissues, leading to a marked increase in the levels of urea, creatinine, Na+, K+, HCO3, and Cl− in group 2 rats relative to group 1. However, treatment with the fraction normalised these parameters, with group 5 demonstrating the most marked improvements (Table 5). The elevation in urea and creatinine, which are key indicators of renal damage after CCl4 intoxication, suggests renal damage possibly due to lipid peroxidation, changes in creatinine phosphate or muscle mass, protein oxidation, decline in creatinine clearance and renal utilization of newly synthesized creatinine. 56 According to Baigh and Khan,, 56 the increase in creatinine level mediated by CCl4 could provokes multiple renal dysfunctions such as overload of the nephron due to alteration in tubular absorption, reduced renal utilisation of available creatinine, and enhanced energy expenditure leading to renal damage. The EFCP-treated groups showed a reduction in these biomarkers, suggesting a potential nephroprotective role through the attenuation of oxidative stress in renal tissues. It's possible that EFCP lessened this effect by its antioxidant properties, which sucked up or trapped the extra ROS that CCl4 produced. This probably stopped the oxidative peroxidation of renal membranes, which would have caused damage to the kidneys’ structure and function. Also, higher levels of Na+, K+, HCO3, and Cl− in the CCl4 group that wasn't treated show metabolic acidosis and problems with ion transport, which are both signs of kidney damage. 47 Interestingly, restoring these parameters back to normal in the treated groups shows that the fraction can keep acid-base homeostasis and renal tubular function, which in turn keeps ionic balance.
Effect of EFCP on Serum Kidney Parameters of CCl4 Intoxicated Rats.

The results are displayed in the mean ± SD (n = 5). * (P < .05), ** (P < .01), *** (P < .001), **** (P < .0001) denote significant differences compared with the untreated group while ns stands for non-significance.
Effects of EFCP on Haematological Parameters
Exposure to toxicants like CCl4 triggers modification of the body's cellular components, including the haematological parameters. Hence, the measurement of haematological indices after CCl4 exposure helps to quantify the extent of cellular and tissue damage. 57 Our haematological parameter results (Figure 5) revealed a significant reduction in Hb, RBC, and PCV and an increase in WBC and platelet counts in group 2 compared to group 1. The observed decline in Hb, RBC, and PCV counts in the untreated rats reflects the presence of red blood cell haemolysis and anaemia, possibly due to oxidative damage to haematopoietic tissues and impaired erythropoiesis. Elevated WBC and platelet counts, on the other hand, reflect an inflammatory response to tissue injury and thrombocytopenia. 57 It is worth mentioning that groups 4 and 5 treated with 100 and 200 mg/kg b.w. of EFCP, respectively exhibited marked improvements in these parameters, comparable to silymarin. This finding further strengthens the protective effects of EFCP on cellular membranes. This action could be correlated to its antioxidant potentials. Invariably, the fraction could have enhanced haematopoiesis or protected against oxidative damage to haematopoietic cells. Flavonoids and phenolic acids, through their anti-inflammatory and antioxidant activities, may contribute to these effects by reducing inflammation and promoting cellular repair. 40

Effect of EFCP on serum Heamatological Parameters of CCl4 Intoxicated Rats. The results are displayed in the mean ± SD (n = 5). * (P < .05), ** (P < .01), *** (P < .001), **** (P < .0001) denote significant differences compared with the untreated group.
Effects of EFCP on Cardiac and Inflammation Markers
Both occupational and environmental toxicant exposures can cause cardiac toxicity. Generally, under ideal physiological conditions, the body maintains homeostasis by sequestering the LDH in the cytoplasm of cardiomyocytes. Sadly, cardiac insult or damage triggers the release of LDH into the blood circulation, making it an effective biomarker of myocardial damage. Additionally, CPR is known as a clinically significant biomarker of acute and chronic inflammation in several diseases, such as CVDs. 58 Thus, we assessed the effect of CCl`4 intoxication on the cardiac and inflammatory markers to ascertain the extent of cardiac damage. This study recorded a significant increase in LDH and CRP in the untreated rats compared with the normal rats. However, treatment with the fraction and silymarin counteracted this by provoking a significant decrease in LDH activity and CRP levels relative to the untreated rats (Figure 6). The high affinity of the cardiac tissue to CYP450-mediated oxidation of CCl4 usually disrupts the heart membrane through peroxidation of membrane phospholipids and proteins, leading to leakage of cardiac enzymes into circulation as observed in the untreated group. 59 This corroborates with the findings of Njoku et al 50 who also realized elevated LDL activity following CCl4 intoxication. Moreover, the increase in CRP suggests myocardial vascular inflammation. Phytochemicals may contribute to the registered reduction in these important biomarkers after treatment. Research evidence has demonstrated that attenuation of oxidative damage, inflammation, and epigenetic modifications, together with regulation of contractile function and signal transduction pathways, are the key mechanisms of the cardioprotective effects of phytochemicals. 60

Effect of EFCP on Haematological Parameters of CCl4 Intoxicated Rats. The Results are Displayed in the Mean±SD (n=5). * (P <.05), ** (P < .01), *** (P < .001), **** (P <.0001) Denote Significant Differences Compared With the Untreated Group.
Effects of EFCP on Liver Histopathology
It is worth mentioning that the histological examination provides further insights into the cellular and structural changes associated with CCl4-induced hepatotoxicity, just have been reported in similar study. 61 In this study, the presence of central vein atrophy and mild sinusoidal dilation observed in the untreated rats further reflects the extent of vascular and parenchymal damage. The liver sections from groups 4 and 5 that were treated with EFCP, on the other hand, show a lot of normal liver architecture restoration, with well-preserved hepatocytes, intact sinusoidal spaces, and less vascular damage (Figure 7). This further confirms hepatoprotective effects of EFCP. Interestingly, the histological findings of the treated rats corroborate the biochemical data, revealing enhanced antioxidant enzyme activities (eg, SOD, CAT, GSH) and decreased lipid peroxidation markers. Furthermore, the presence of central vein atrophy and mild sinusoidal dilation also aligns with elevated serum biomarkers such as ALT, AST, and ALP recorded in the untreated group. The lack of immune cell infiltration and the decrease in inflammation are also signs that the treatment is working to reduce inflammation. This might be achieved by changing the levels of pro-inflammatory cytokines and oxidative stress pathways. Thus, suggesting a robust hepatoprotective potential, possibly mediated by the antioxidant bioactive compounds in C. paniculatum. The near-complete normalisation of liver structure in the high-dose treatment group highlights the dose-dependent efficacy of the EFCP, which is favourable for the standard hepatoprotective drug (silymarin).

Effect of EFCP on Liver Histopathology of CCl4 Intoxicated Rats. Key: A Definition of the Liver is Displayed on the Slides, Along With its Histological Constituents—the Parenchyma, Stroma, and Sinusoids. Hepatocytes (Black Arrow) Constitute the Parenchyma, Arteries and Connective Tissue Make Up the Stroma, and Capillaries That Run Between Hepatocytes are Called Sinusoids (Red Arrow).
Study Limitation and Implications
While the findings from this study are promising, several limitations should be acknowledged. First, the study was conducted exclusively on an animal model, which may not fully replicate human liver pathophysiology. Second, although the ethyl acetate fraction of Combretum paniculatum showed significant hepatoprotective and antioxidant effects, the exact molecular mechanisms and the individual contributions of each bioactive compound remain to be elucidated. These limitations suggest the need for further targeted investigations.
Nonetheless, the results of this study highlight EFCP's potential as a natural therapeutic agent for managing oxidative stress-related liver damage. This provides a foundation for future translational research, including compound isolation, molecular pathway studies, and eventual clinical trials aimed at validating its use in human liver disorders.
Conclusion
This study provides compelling evidence that the ethyl acetate fraction of Combretum paniculatum (EFCP) mitigates CCl4-induced hepatic and renal injuries through antioxidant and anti-inflammatory mechanisms. EFCP restored biochemical, haematological, and histological parameters, reinforcing its therapeutic potential. Further exploration into the isolation of its active constituents and assessment in clinical settings is recommended to harness its full medicinal value.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251390705 - Supplemental material for Protective Effects of Combretum paniculatum Fraction on CCl4-Induced Acute Liver Injury
Supplemental material, sj-docx-1-npx-10.1177_1934578X251390705 for Protective Effects of Combretum paniculatum Fraction on CCl4-Induced Acute Liver Injury by Ifeoma Felicia Chukwuma, Solomon Adole Ochepo, Victor Onukwube Apeh, Florence Nkechi Nworah, Okechukwu Ignatius Eze and Timothy Prince Chidike Ezeorba in Natural Product Communications
Footnotes
Ethical Approval
This study was approved by the Faculty of Biological Sciences, University of Nigeria, Ethical Committee (Approval No.: UNN/FBS/EC/1082) on 5th April 2022.
Statement of Human and Animal Rights
All experimental procedures involving animals were conducted following the Institutional Animal Care guidelines at the University of Nigeria, in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. To ensure humane treatment, anesthesia and euthanasia were performed using an intraperitoneal injection of sodium pentobarbital (200 mg/kg), a barbiturate agent, following the American Veterinary Medical Association (AVMA) guidelines. Animal death was confirmed through an assessment of breathing patterns, pupillary responses to light, and heartbeats before carcass disposal.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All data obtained in this study are included in the manuscript.
Statement of Informed Consent
This study does not involve human subjects, and informed consent is not applicable
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
