Abstract
Zicao, a traditional Chinese herbal medicine documented since the Shennong Ben Cao Jing (Classic of the Divine Husbandman's Materia Medica) in the Eastern Han Dynasty, exhibits significant anti-inflammatory, antitumor, and antibacterial activities. Modern pharmacological research confirms its efficacy in these areas. Commercially available Zicao is derived from multiple species—primarily Arnebia euchroma, Lithospermum erythrorhizon, and Onosma paniculatum—resulting in substantial quality variations. Among these, A. euchroma is recognized as the superior medicinal source. However, its supply is severely constrained by scarce wild resources and challenges in artificial cultivation, leading to frequent substitution with inferior varieties.
This review elucidates critical distinctions between A. euchroma and other Zicao species in native habitats, chemical composition, emphasizing A. euchroma possesses unique chemical advantages, characterized by high concentrations of bioactive naphthoquinones (notably β, β-dimethylacrylalkannin, β-hydroxyisovalerylalkannin, and β-acetoxyisovalerylalkannin), polysaccharides, and phenolic acids. These constituents underpin its enhanced anti-inflammatory, antitumor, antioxidant, antimicrobial, neuroprotective, and dermatological activities compared to alternative Zicao sources.
By integrating traditional knowledge with modern research, this review provides a scientific foundation for prioritizing A. euchroma in clinical and pharmaceutical applications of Zicao.
This is a visual representation of the abstract.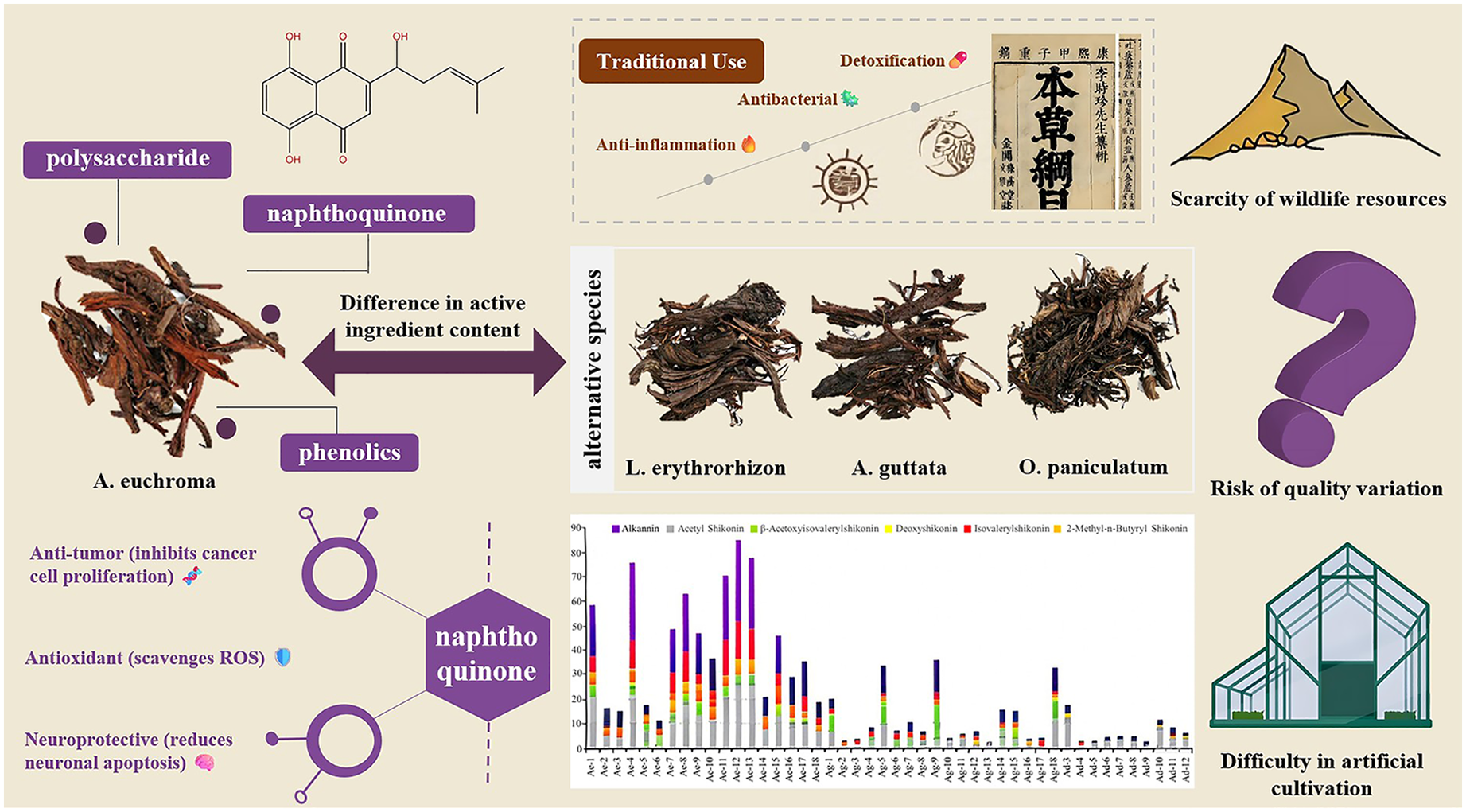
Introduction
Zicao, a traditional Chinese herb used widely for centuries, is first documented in the classic Shennong's Classic of Materia Medica. It is described as “cooling blood, activating circulation, clearing heat, detoxifying, resolving rashes, and dispelling spots”. The Compendium of Materia Medica further specifies its use for “macular rashes, pox toxins, sores, and carbuncles”. Clinically, it is often paired with herbs like Paeonia lactiflora and Moutan cortex; Zicao Decoction from Standards of Diagnosis and Treatment, for example, addresses inadequate measles eruption. Externally, it is boiled with sesame oil to make Zicao oil, used for burns and similar conditions.1,2
In Xinjiang's traditional Uyghur medicine, Zicao is a core herb for skin diseases, using its roots medicinally. Representative formulas include Suziafu Tablets for psoriasis and vitiligo, and Kaliziran Tincture—a topical tincture to relieve skin itching and erythema. 3 Use of the herb extends beyond China. In the Indian Himalayas, root powder is blended with glycerin to make ointments for burns and ulcers, or mixed with mustard oil as a hair conditioner. In Central Asia, including Iran and Turkey, Zicao extracts mixed with olive oil treat eczema and other skin ailments. It also serves as a natural dye in these regions, used in religious rituals and cloth dyeing. 4
These long-standing traditional uses offer key historical context for modern research into its anti-inflammatory, wound-healing, and skin-healing properties. Modern pharmacology confirms its diverse activities: hemostasis, 5 anti-tumor effects, 6 antibacterial action, and anti-inflammatory activity. 7 It has attracted increasing research attention, holding significant status in both traditional Chinese medicine and modern pharmaceutical studies.
However, Zicao can refer to the roots of various medicinal plant species, including Arnebia euchroma, Lithospermum erythrorhizon, and Onosma paniculatum. According to the 2020 edition of the Pharmacopoeia of the People's Republic of China, A. euchroma and Arnebia guttata have replaced L. erythrorhizon as authentic sources of medicinal Zicao. A. euchroma, a national second-class protected plant species in China, is considered a superior medicinal resource to other Zicao species such as L. erythrorhizon and O. paniculatum because of its abundance of bioactive compounds, including β, β-dimethylacrylalkannin, β-hydroxyisovalerylalkannin, and β-acetoxyisovalerylalkannin. 8 However, A. euchroma, which is mainly distributed in the high-altitude mountainous regions of the Xinjiang Uygur Autonomous Region, is in short supply owing to limited wild resources and challenges in artificial cultivation; this has resulted in the frequent adulteration of raw materials and inconsistent quality of Zicao products.
The pharmacological activities of Zicao are predominantly attributed to its various chemical constituents, particularly naphthoquinones (such as shikonin (SK), isovalerylshikonin, and acetylalkannin), polysaccharides, and phenolic acids. Naphthoquinones have received extensive research attention owing to their anti-inflammatory, antibacterial, and antitumor properties. Moreover, recent studies have demonstrated the potential of Zicao polysaccharides and phenolic acids to modulate immune responses and exert antioxidant effects, further expanding their therapeutic applications.
Despite increasing research on Zicao, the complex chemical composition and geographical variation of Zicao species lead to inconsistencies in the quality and efficacy of medicinal preparations. Furthermore, the mechanisms underlying the pharmacological activities of the key constituents have not been fully elucidated. Therefore, this review first elucidates differences in the native habitats and chemical composition of different Zicao species and plant parts, then summarizes research progress on the bioactive substances and pharmacological effects of A. euchroma.
Native Habitats, Medicinal Parts, and Naphthoquinone Composition of Different Zicao Species
Differences in Native Habitats and Growth Conditions
The main Zicao species on the market, namely Arnebia euchroma, A. decumbens, A. guttata, Lithospermum erythrorhizon, and Onosma Paniculatum, exhibit significant differences in their geographic distribution, origin and character (Table 1), which directly influence the content of their active ingredients and medicinal efficacy. The diverse distribution and origin of Zicao species play crucial roles in determining their active ingredient content and medicinal properties.
Natural Habitats of Different Zicao Species.

A. euchroma is a perennial herb. Its stems, 15–40 cm tall, are densely hispid (covered with stiff hairs). The plant bears linear to linear-lanceolate leaves and produces deep purple, funnelform-campanulate flowers. The nutlets are broadly ovoid, dark brown, with coarse reticulation, which are predominantly distributed on the northern and southern slopes of the Tianshan Mountains, in Bazhou, Kezhou, Aksu, and Kashgar in southern Xinjiang, and in Ili Kazakh Autonomous Prefecture, Bozhou, Changji Hui Autonomous Prefecture, Tacheng, and Urumqi in northern Xinjiang. 9
A. guttata is a perennial herb whose roots contain purple pigments. Its stems (10-25 cm tall) are much-branched and densely hispid with stiff hairs. The leaves are spatulate-linear and densely covered with white stiff hairs on both surfaces. It produces yellow, funnelform-campanulate corollas with purple spots. The nutlets are triangular-ovoid, pale yellowish-brown, and tuberculate. typically grows in alpine meadows at altitudes of 2500–3400 m, with average annual temperatures of 3 °C and large significant diurnal temperature differences owing to strong sunshine. A. guttata is also found on the slopes and deserts of northern and southern Xinjiang at altitudes of 500–3000 m. The main A. guttata production areas include Ulat Houqi and Karakin Banner (specific regions of Inner Mongolia) and Altay and Heshuo in Xinjiang. 10
A. decumbens is an annual herb with roots containing trace amounts of purple pigments. Stems (15-30 cm tall) are branched from the base and hispid with stiff hairs. Leaves are linear-oblong, sparsely strigose on both surfaces. It produces yellow, funnelform-campanulate flowers with calyces becoming accrescent and hardened in fruit. Nutlets are triangular-ovoid, brown, and densely tuberculate. Distributed in low-altitude sandy areas of northern Xinjiang, particularly in the sandy wastelands on the northern slopes north slopes of the Tianshan Mountains and low-altitude regions of the Altai Mountains. 11
L. erythrorhizon is a perennial herb with roots rich in purple pigments. The stems (40-90 cm tall) develop ascending branches in the upper part and are strigose throughout. Leaves are ovate-lanceolate, strigose on both surfaces. It produces white flowers with faucal appendages (corolla scales). Nutlets are ovoid, smooth, and ventrally grooved, which occurs in the middle and lower reaches of the Yangtze River and Liaoning. 12
O. paniculatum is a biennial herb, turning black upon drying. Stems 40–80 cm tall, simple (unbranched), densely hispid with rigid hairs. Basal leaves rosulate, linear-lanceolate; cauline leaves narrowly elliptic with hastate-amplexicaul bases. Flowers blue-purple, funnelform-campanulate. Nutlets dark brown, densely tuberculate. Distributed in Sichuan, Yunnan, and Guizhou and thrives in the gravelly areas of grassy slopes at altitudes of 2000–3600 m. 13
Distinguishing these Zicao species requires examination of multiple characteristics. A. euchroma is most conspicuously characterized by its exceptionally stout roots and deep purple corollas. Its stems are few, often with basal sheaths formed by remnant leaf bases, and nutlets exhibit coarse reticulation. In contrast, A. decumbens is an annual species with roots containing minimal purple pigments, pure yellow corollas, and densely tuberculate nutlets. A. guttata, while also yellow-flowered, displays distinct differences: its stems are profusely branched and densely covered with white, bristly hairs featuring discoid bases; leaves are typically spatulate-linear; and corolla limbs bear purple maculations. O. paniculatum possesses a unique trait—the entire plant turns black upon drying. L. erythrorhizon, presents white corollas and tall stature, ovate-lanceolate leaves, and nutlets with smooth, lustrous surfaces and a pronounced ventral groove—strikingly contrasting with the tuberculate or reticulate nutlets typical of other congeners.
Differences in Naphthoquinone Active Components among Zicao Species
Although A. euchroma, A. decumbens, A. guttata, O. paniculatum, and L. erythrorhizo are all Zicao species, they occupy markedly divergent habitats. A. euchroma and A. guttata thrive in high-altitude environments characterized by intense UV radiation, cold temperatures, and aridity, whereas L. erythrorhizon inhabits lower-altitude, more temperate regions. This pronounced ecological contrast likely imposes strong selective pressures on secondary metabolite biosynthesis. Plants in harsh, high-UV conditions frequently evolve enhanced production of protective compounds such as naphthoquinones, which possess intrinsic chromophoric and antioxidant properties. This ecological perspective explains the significantly higher naphthoquinone yields observed in the Xinjiang species (A. euchroma, A. guttata) compared to L. erythrorhizon, aligning with reported differences in their specific naphthoquinone profiles.14,15 The common naphthoquinone compounds in Zicao are as follows (Figure 1): (1) SK, (2) acetylshikonin (ASK), (3) 1-methoxyacetylshikonin, (4) propionylshikonin, (5) isobutyrylshikonin, (6) isovalerylshikonin, (7) β,β-dimethylacrylshikonin (DMAKN), (8) teracrylshikonin, (9) angelylshikonin, (10) β-hydroxyisovalerylshikonin, (11) β-acetoxyisovalerylshikonin, (12) butyrylshikonin, (13) valerylshikonin, (14) 5-acetoxy-valerylshikonin, (15) α,α-dimethylpropionylshikonin, (16) α-methylene-butenoylshikonin, (17) β-acetoxy-α,β-dimethybutyrylshikonin, (18) (Z)2-methylene-3-butenoylshikonin, (19) (E)2-methylene-3-butenoylshikonin, (20) (E)4-hydroxy-2,4-dimethylpent-penteneacylshikonin, (21) alkannin, (22) acetylalkannin, (23) β,β-dimethylacrylalkannin, (24) β-hydroxyisovalerylalkannin, (25) β-acetoxyisovalerylalkannin, and (26) deoxyalkannin. Table 2 shows the bioactive components present in different Zicao species (Table 2).

Common Naphthoquinone Compounds in Zicao.
Naphthoquinone Chemistry of Different Zicao Species.

The naphthoquinone content of A. euchroma (2.12-6.73%) is considerably higher than that of A. guttata (0.13-0.81%) and L. erythrorhizon (0.18-0.39%).14,15 For example, the content of compound (1) (shikonin) in A. euchroma (2.01%) is much higher than that in A. guttata (0.21%), L. erythrorhizon (0.38%), and O. paniculatum (0.10%). 16 Moreover, A. euchroma contains some unique naphthoquinone compounds, such as (11) β-acetoxyisovalerylshikonin and (23) β,β-dimethylacrylalkannin. 17
In addition to distinguishing different species of Zicao by their morphological characteristics, we can also differentiate various types of Zicao by measuring the differences in the content and types of these compounds via HPLC.
A. euchroma contains diverse naphthoquinones at high concentrations, whereas L. erythrorhizon exhibits moderate diversity but low abundance, and O. paniculatum exhibits limited diversity and low abundance. These differences in naphthoquinone profiles are related to the pharmacological activities. Naphthoquinone compounds are closely associated with hemostatic, anti-inflammatory, antibacterial, and antioxidant properties. Owing to its higher and more diverse naphthoquinone content, A. euchroma likely exhibits greater pharmacological activity than other species.18–21 This chemical superiority provides a scientific basis for the preferential clinical and pharmaceutical application of A. euchroma.
Chemical Differences in the Medicinal Parts of the Zicao Plant Species
Arnebia euchroma
The chemical composition of Zicao also varies among the different plant parts of A. euchroma, namely the roots, root bark, stems, and leaves. Although the roots and root bark contain very similar naphthoquinone compounds,15,22,23 the roots have lower contents of shikonin, isovalerylshikonin, and β, β-dimethylacrylalkannin. Specifically, these components are significantly more abundant in the root bark of A. euchroma from Bazhou and Ili, Xinjiang. In contrast, the stems and leaves contain fewer naphthoquinone compounds (Table 3).15,24 Moreover, naphthoquinone concentrations are higher in the roots than in the stems and leaves. 25 Specifically, the average content of naphthoquinone compounds is 3.631% in the roots but only 1.516% in the stems and leaves. The root bark of A. euchroma contains the greatest most tremendous variety and concentration of naphthoquinone compounds,18,20 followed by the roots, which still possess significant medicinal value (Table 3).
Naphthoquinone Chemistry of Different Parts of Arnebia euchroma.

Other Zicao Species
Substantial flavonoid variations have also been observed in different parts of A. guttata. 26 The flavonoid content is highest in the flowers, with an average value of 10.054%, which is approximately double that in the leaves (average 5.192%) and 5.6 times that in the stems (average 1.790%). The flowers of A. guttata are therefore the primary source of flavonoids, exhibiting substantial therapeutic potential. The roots of L. erythrorhizon contain various active ingredients, 27 primarily naphthoquinones, phenols, and phenolic acids, whereas the active constituents of the stems and leaves are predominantly diterpenoids and flavonoids. Moreover, L. erythrorhizon seeds contain pyrrolizidine alkaloids, which are toxic and may cause liver damage. The pharmacological activities of these active ingredients vary widely, providing a rich basis for further research and development of L. erythrorhizon.
Bioactive Substances and Pharmacological Effects of Arnebia euchroma
The major active components in A. euchroma are naphthoquinone compounds, polysaccharides, and phenolic acid compounds, which boast unique pharmacological characteristics and advantages. Naphthoquinone compounds have particularly notable pharmacological activities, including anti-inflammation, antibacterial, antioxidant, antitumor, and wound-healing effects. Polysaccharides predominantly exhibit antioxidant and anti-inflammatory properties, whereas phenolic acid compounds typically exert anti-inflammatory and antiviral effects.
Pharmacological Activities of Naphthoquinones
The primary naphthoquinone compounds in A. euchroma are SK, ASK, DMAKN, deoxyshikonin (DSK), and isovalerylshikonin. In addition to their anti-inflammatory and antitumor pharmacological activities, these naphthoquinone compounds exhibit unique therapeutic properties; for example, SK demonstrates antioxidant effects and promotes wound healing, ASK shows contraceptive and antibacterial pharmacological activities, and isovalerylshikonin exerts preventive effects against Alzheimer's disease and insecticidal properties.
Anti-Inflammatory Effects
SK exhibits potent anti-inflammatory activity. Lian treated a collagen-induced arthritis mouse model with SK and found that SK alleviates inflammation in arthritis models by selectively inhibiting the polarization of pro-inflammatory M1 macrophages while preserving the differentiation of anti-inflammatory M2 macrophages. 28 Additionally, Han treated mice with DSS-induced ulcerative colitis using SK, and Lu treated mice with xylene-induced inflammation using SK. Their findings revealed that the efficacy of SK involves suppressing TNF-α release by inhibiting LPS-induced NF-κB nuclear translocation and proteasome activity.29,30
ASK, similarly, exhibits broad anti-inflammatory effects but via distinct and multifaceted mechanisms. Jayasooriya treated LPS-stimulated BV2 microglial cell models with ASK and found that ASK suppresses NF-κB activation by targeting the ROS/PI3 K/Akt pathway, while concurrently inducing HO-1 expression to reduce NO and PGE2 levels in microglia. 31 Zorman treated LPS-stimulated macrophages with ASK and found that ASK also specifically impairs NLRP3 inflammasome activation, blocking IL-1β maturation. 32 Furthermore, ASK effectively reduces key pro-inflammatory cytokines (TNF-α, IL-1β, IL-6, MCP-1) and mediators (IgE, IgG1, Th2 cytokines) in diverse models including acute inflammation, steatohepatitis, pulmonary inflammation, and allergic rhinitis, primarily through NF-κB inhibition and Nrf2/Nur77 axis activation.33,34,35
Collectively, the mechanistic profiles of SK and ASK suggest distinct therapeutic niches. SK's selective modulation of macrophage polarization, coupled with its neuroprotective effects, implicates potential utility in macrophage-centric diseases or neuroinflammatory disorders. Conversely, ASK exhibits a multifaceted mechanism—targeting leukotriene biosynthesis, specifically inhibiting NLRP3 inflammasome activation, inducing HO-1 expression, and activating the Nrf2/Nur77 pathways—resulting in broader anti-inflammatory and immunomodulatory coverage, suggests ASK may be better suited for complex inflammatory conditions involving dysregulation of multiple pathways. Subsequent direct comparative studies in relevant disease models, particularly models of metabolic inflammation, are necessary to more definitively delineate their therapeutic divergence.
Antitumor Effects
Zicao naphthoquinones—SK, DMAKN, ASK, DSK, and isobutyrylshikonin—inhibit tumor growth via a multi-pathway, multi-target strategy. Their key mechanisms encompass the induction of programmed cell death, suppression of pro-survival signaling, reversal of drug resistance, and precise modulation of the cell cycle.
Gong treated hepatocellular carcinoma (HCC) with SK and found that SK induces apoptosis in HCC through both intrinsic (mitochondrial permeabilization, cytochrome C release) and extrinsic (death receptor activation) pathways, dependent on Bcl-2 family balance and ROS amplification.36,37 This dual targeting may help overcome apoptosis resistance, though ROS dependence could limit efficacy in tumors with altered redox states. Notably, SK concurrently inhibits PI3 K/AKT/mTOR and MAPK/ERK survival pathways while activating pro-apoptotic JNK, 38 suggesting multi-targeted mechanisms distinct from single-pathway agents.
DMAKN shows dose-dependent bimodal effects in HCC. Shen used THP-1 cells as a model: after inducing their differentiation into M0 macrophages with PMA, he further induced them into M1 macrophages with LPS/IFN-γ and into M2 macrophages with IL-4/IL-13, respectively, while administering DMAKN at different concentrations. He then co-cultured the treated macrophages with HepG2 and Huh7 hepatocellular carcinoma cells, and found that low doses of DMAKN induce JNK/ROS-mediated cell cycle arrest, while high doses trigger necroptosis via ROS hyperaccumulation.39,40 This suggests therapeutic outcomes may be tailored through dose adjustment based on tumor context.
Lin treated non-small cell lung cancer cells (H1299, A549) and normal lung fibroblasts (MRC-5) with different concentrations of ASK and found that ASK shifts from inducing apoptosis to preferentially inducing necroptosis via RIPK1/RIPK3/MLKL activation. 41 Meanwhile, Hao treated CML cell line (K562) and normal mammalian Vero cells with ASK and found that ASK simultaneously enhances apoptotic sensitivity through Bax/Bak upregulation and Bcl-2 suppression. 42 Such dual activation of death pathways could bypass treatment resistance. ASK also attenuates tumor-promoting inflammation and disrupts cell cycle regulation.
Hsieh treated human osteosarcoma cells (U2OS and HOS) with different concentrations of DSK and found that DSK triggers apoptosis through p38 MAPK activation, which is amplified by HO-1 upregulation in osteosarcoma and tongue carcinomas.43,44 It also inhibits PI3K-Akt-mTOR-mediated energy metabolism in colorectal cancer and AML45,46 – directly targeting cancer metabolism. Critically, DSK reverses multidrug resistance in NSCLC via ABCB1 transporter inhibition, 47 addressing a key clinical challenge.
Zhong treated human colon cancer HT29 cells with isobutyrylshikonin and found that isobutyrylshikonin specifically induces G2/M arrest in colon cancer in a concentration- and time-dependent manner, 48 showing derivative-specific cell cycle targeting distinct from SK's G1/S effects.
Naphthoquinones exert anticancer effects by simultaneously triggering the apoptosis–necrosis dual pathway and suppressing multiple survival signals. This dual mechanism not only circumvents drug resistance but also imposes precise cell-cycle control, thereby providing a robust foundation for the development of novel, multi-target natural anticancer agents.
Antioxidant Effects
SK combats oxidative stress through three distinct pathways: direct scavenging of radicals (stabilizing DPPH and ABTS+), interruption of lipid peroxidation cascades via complexation with unsaturated fatty acids, 49 and suppression of inducible NO synthase, thereby reducing NO production and downstream radical amplification. SK's multi-targeted approach addresses oxidative damage at different stages, potentially offering broader protection than single-mechanism antioxidants.
ASK also demonstrates potent antioxidant activity, but its primary mechanism differs. Li treated the hippocampus of a subacute aging mouse model of Alzheimer's disease induced by D-galactose with ASK and found that ASK significantly enhances endogenous enzymatic defenses: it reduces lipid peroxidation (measured by malondialdehyde levels) and elevates superoxide dismutase activity. 50 ASK's focus on boosting cellular enzymatic systems represents a complementary strategy to direct radical quenching.
Both SK and ASK are effective antioxidants. SK primarily acts through direct radical neutralization and modulation of NO pathways. ASK, conversely, emphasizes fortification of endogenous enzymatic defenses. This divergence in primary mechanisms—SK's direct interception versus ASK's cellular defense enhancement—points to potential mechanistic complementarity. Such complementary actions warrant investigation for synergistic applications, particularly in contexts like neurodegenerative or age-related pathologies where multi-faceted oxidative stress is implicated.
Antimicrobial Effects
ASK exhibits broad-spectrum antimicrobial activity against diverse pathogens, including fungi (Candida albicans, Cryptococcus neoformans) and bacteria (Staphylococcus aureus, Pseudomonas aeruginosa).51–53 This activity profile suggests potential utility against polymicrobial infections. Inhibition of Gram-positive bacteria occurs at low concentrations. However, significant suppression of Gram-negative bacteria and fungi requires substantially higher concentrations. 54 Such a high threshold may limit systemic application against these pathogens.
Combining ASK with agents like polymyxin enhances antimicrobial efficacy. 55 Yet, elevated acetylcholinesterase activity in these combinations indicates increased neurotoxicity risk, creating a critical therapeutic trade-off.
Both SK and DMAKN also demonstrate bactericidal effects against S. aureus. Wan used the broth dilution method and found that SK acts through multiple mechanisms: depleting intracellular ATP, disrupting membrane integrity, and inducing morphological changes. 56 This multi-targeted approach potentially reduces the risk of resistance development compared to agents with a single mechanism.
Neuroprotective Effects
Lu treated mt-Keima neurons with DSK and found that DSK activates protective mitophagy in primary cortical neurons without causing mitochondrial damage. 57 This selective clearance of dysfunctional mitochondria represents a targeted neuroprotective strategy distinct from generic antioxidant approaches. Chen treated primary rat cortical neurons with SK and found that SK counters oxygen-glucose deprivation-induced apoptosis in cortical neurons, primarily via PI3 K/Akt pathway activation. 58 By targeting this central survival signaling axis, SK may protect neurons against ischemic injury – a key pathological process in stroke.
Wang treated SH-SY5Y and PC12 cell models with ASK, then induced oxidative stress with hydrogen peroxide, and found that ASK exerts neuroprotection through dual complementary mechanisms: HO-1 upregulation reduces oxidative neuronal damage, 59 while SIRT1/p53/p21 axis modulation promotes neuronal survival. 60 Such coordinated action on oxidative stress and cell death pathways could enhance therapeutic efficacy in neurodegenerative contexts. ASK additionally stabilizes neurotransmitter levels through monoamine oxidase inhibition.
Ding treated rat pheochromocytoma cells (PC12 cells) induced by β-amyloid protein (Aβ25-35) with isovalerylshikonin and found that isovalerylshikonin reduces acetylcholinesterase activity in Aβ-injured neuronal cells. 61 This specific modulation of cholinergic function directly addresses a core pathological feature of Alzheimer's disease, supporting its potential as a disease-modifying agent.
Other Effects
ASK exhibits reversible contraceptive effects; oral administration significantly reduced pregnancy rates in rats, with fertility fully restored after treatment cessation. 62 SK plays a critical role in accelerating wound healing in infected models, achieved by activating granulation tissue formation, promoting follicular/sebaceous gland regeneration, and upregulating epidermal growth factor expression. 63 Isobutyrylshikonin and isovalerylshikonin display insecticidal activity against Spodoptera littoralis. Isovalerylshikonin shows superior potency, correlating with stronger acetylcholinesterase inhibitory activity, likely due to the isovaleryl side chain enhancing target binding. 64
Pharmacological Activities of Polysaccharides
Radix Arnebiae polysaccharide (RAP) is a structurally complex natural compound. Zhu isolated two RAP fractions from Zicao. These fractions had average molecular weights of 28,746 Da and 4877 Da. Their number-average molecular weights were 27,336 Da and 1152 Da. 65 Bo purified an RAP component using hot water extraction, ethanol precipitation, and TCA deproteinization. This component had a molecular weight of approximately 1.23 × 104 Da. Structural analysis identified RAP as a heteropolysaccharide. Its monosaccharide composition is galactose (53.8%), arabinose (21.3%), glucose (11.7%), mannose (6.8%), rhamnose (4.3%), and fucose (2.2%). 66 This unique, complex structure underpins RAP's diverse pharmacological effects. These include immunomodulation, antitumor, antioxidant, and anti-inflammatory activities.
Within the active substance system of A. euchroma, RAP is a significant component. Its mechanism of action differs markedly from naphthoquinones. This difference is particularly evident in immunomodulation. Research by Li demonstrates this. Oral administration of RAP (50 mg/kg/day for 7 days) effectively restored peritoneal macrophage phagocytosis in immunosuppressed mice. It also promoted T-lymphocyte proliferation. 67 These findings reveal RAP activates both innate and adaptive immunity. This immunomodulatory property broadens the application scope of A. euchroma. RAP holds relevance for diseases requiring immune response modulation. Its effects may synergize with the anti-inflammatory and anticancer actions of naphthoquinones.
RAP's antioxidant activity is another fundamental aspect of its efficacy. Studies used RAP to treat carbon tetrachloride-induced liver injury models. RAP treatment reduced levels of oxidative stress markers (NO, malondialdehyde). It simultaneously enhanced the activity of endogenous antioxidant enzymes (superoxide dismutase, glutathione). 68 This dual action—scavenging reactive oxygen species and boosting cellular defenses—demonstrates its capacity to maintain redox balance. Liu elucidated RAP's anti-inflammatory mechanism. RAP dose-dependently inhibits the production of pro-inflammatory cytokines (TNF-α, IL-6, IL-1β). At the transcriptional level, it suppresses LPS-induced expression of TNF-α and IL-18. 69 This ability to intervene in the inflammatory cascade at transcriptional and post-translational levels further underscores its multi-target nature.
RAP's structural complexity supports its multifaceted bioactivities. It plays a vital role among the active constituents of A. euchroma. Its distinct immunomodulatory pathways complement the direct anti-inflammatory/anticancer effects of naphthoquinones. This complementarity expands the therapeutic potential of A. euchroma. Nevertheless, current investigations have exposed a gap. RAP's structural and functional differences remain poorly characterized. This limitation hinders deeper investigation into the overall synergistic mechanisms governing A. euchroma's efficacy. Future research should prioritize establishing precise structure-activity relationships for these polysaccharides. It must also explore the molecular basis of their synergy with naphthoquinones. This focus is essential to fully unlock RAP's medicinal value.
Pharmacological Activities of Phenolic Acid Compounds
Recent phytochemical investigations have identified four principal phenolic acids (Figure 2) in Radix Arnebiae: rosmarinic acid (27), gallic acid (28), lithospermic acid (LA; 29), and caffeic acid tetramer (CAT; 30). Notably, CAT accounts for 18.55–25.49% of the total phenolic content, whereas gallic acid and LA are relatively understudied, with no prior literature available. LA and CAT exhibit broad-spectrum pharmacological effects, including anti-inflammatory, antitumor, antiviral, and cardioprotective properties. Mechanistic studies have revealed that LA attenuates endothelial inflammation by inhibiting NF-κB phosphorylation and effectively suppressing high glucose-induced pro-inflammatory cytokine expression. 70 Complementarily, CAT demonstrates dual anti-inflammatory action in LPS-activated macrophages, reducing NO production and downregulating pro-inflammatory cytokine mRNA levels via MAPK/NF-κB pathway inhibition. 71 Structurally, LA acts as a non-covalent inhibitor of HIV-1 nucleocapsid proteins, independent of redox mechanisms, as confirmed by molecular modeling, 72 whereas CAT exhibits direct antiviral activity against HIV replication in cellular models. 73

Phenolic Acid Compounds in Zicao.
In oncology, LA derivatives exhibit dose-dependent cytotoxicity against glioblastoma cells (U87/T98) and impair tumor cell viability. 74 Cardiovascular studies have highlighted the role of LA in mitigating myocardial ischemia reperfusion injury by activating the Nrf2/HO-1 antioxidant pathway and AMPKα phosphorylation, as evidenced by reduced infarct size and improved cardiac function in animal models. 75
These findings, which have been validated through cellular assays, animal models, and advanced molecular techniques, systematically elucidate the multi-target mechanisms of the phenolic acid compounds in Radix Arnebiae. By integrating TCM applications with modern pharmacological insights, these studies establish a foundation for developing novel anti-inflammatory, antiviral, and anticancer therapeutics derived from natural products.
Conclusion and Future Directions
Zicao, a cornerstone of TCM with historical records dating back to the Eastern Han Dynasty, has attracted substantial scientific interest because of its multi-functional bioactivity. The 2020 Pharmacopoeia of the People's Republic of China designates A. euchroma and A. guttata as official sources of medicinal Zicao, highlighting their rich content of bioactive naphthoquinones (eg, SK, isovalerylshikonin, β,β-dimethylacrylalkannin), polysaccharides, and phenolic acids. These phytochemicals endow Zicao with potent anti-inflammatory, antitumor, antioxidant, antimicrobial, and neuroprotective properties.
From a TCM quality control perspective, accurate identification of the genuine medicinal Zicao (A. euchroma and A. guttata) and distinguishing them from substitute species are crucial for ensuring raw material quality in procurement and quality inspection. Comparative analysis reveals distinct morphological (eg, root characteristics) and phytochemical markers (eg, specific naphthoquinone ratios) between the official species and common substitutes. Furthermore, existing literature suggests significant variations in the profiles and concentrations of bioactive constituents, particularly naphthoquinones like shikonin derivatives, among Zicao materials sourced from different geographical regions. These observed differences lead to the reasonable hypothesis that environmental factors and genetic variations contribute to this chemotypic diversity. The genuine species, especially A. euchroma and A. guttata, are characterized by the presence of unique or significantly enriched bioactive compounds (eg, specific acetylshikonin esters found predominantly in A. euchroma), which are strongly associated with their characteristic pharmacological activities, including potent anti-inflammatory effects, and significant antiviral activity.
Among the official species, A. euchroma is particularly notable because of its unique geographical distribution and environmental adaptability, yielding significantly higher concentrations of active constituents than other Zicao taxa. This chemotypic superiority highlights A. euchroma as a promising resource for clinical and pharmaceutical applications. However, unsustainable harvesting practices, challenges in large-scale cultivation, and market adulteration have resulted in erratic quality and supply chain instability, threatening the long-term utilization of A. euchroma.
Although contemporary research has advanced our understanding of the multi-target mechanisms of Zicao, which include modulation of the NF-κB, MAPK, and PI3 K/Akt signaling pathways, critical knowledge gaps persist. Key unresolved issues include 1) incomplete characterization of naphthoquinone biosynthetic pathways, 2) limited understanding of in vivo metabolic profiles and constituent synergy, and 3) reliance on single-component quality metrics (eg, naphthoquinone content) that fail to reflect holistic medicinal properties. Therefore, addressing these challenges requires the development of integrative quality control systems that incorporate chemical fingerprinting, genomic markers, and bioactive profiles.
Emerging biotechnological approaches offer a transformative potential for Zicao research. Molecular biology tools can unravel the biosynthesis of bioactive compounds, thereby enabling the development of synthetic biology-based production platforms. Metabolic engineering can enhance compound yield or generate novel derivatives with improved therapeutic indices. Concurrently, artificial intelligence-driven high-throughput screening and systems biology analyses can accelerate the discovery of Zicao-derived drugs with optimized efficacy and safety profiles. To ensure sustainable resource utilization, the development of standardized cultivation protocols and good agricultural practice-compliant planting bases should be prioritized. Diversifying applications into the functional foods, cosmeceuticals, and precision medicine sectors will maximize economic value while preserving traditional medicinal uses. Furthermore, strengthening international collaboration and harmonizing regulatory frameworks will facilitate the global acceptance of Zicao-based therapies and foster TCM modernization. Through interdisciplinary innovation and industry-academia partnerships, Zicao shows substantial potential as a strategic resource for addressing unmet global health needs, particularly chronic disease management and antimicrobial resistance.
Footnotes
Author Contributions
LT wrote the manuscript, WXY conducted the preliminary research and revised the manuscript, LN and WSS revised the manuscript, HJ conducted part of the research, DY and LL reviewed and edited the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
