Abstract
Background
Peptic ulcer affects up to 10% of the world's population and remains a significant health concern. While conventional treatments are available, their side effects and costs have led to increased interest in herbal alternatives.
Objectives
Vi Trang BH, a traditional Vietnamese decoction containing nine medicinal herbs, was evaluated for its protective effects against peptic ulcers using two experimental models in rats.
Methods
The study utilized indomethacin (40 mg/kg) and a dual-dose cysteamine protocol (400 mg/kg each dose) to induce peptic ulcers. Rats were pretreated with Vi Trang BH decoction at doses of 40 ml/kg and 80 ml/kg body weight for seven days.
Results
The results demonstrated that Vi Trang BH significantly reduced ulcer indices in the indomethacin model (protective indices: 71.79% and 69.23% for respective doses) and showed moderate improvement in the cysteamine model (protective indices: 27.52% and 30.28%). The decoction exhibited hepatoprotective effects by regulating liver enzymes in both models. Additionally, histopathological analyses confirmed the protective effects on gastric and duodenal tissues.
Conclusion
Vi Trang BH demonstrated significant protective effects against peptic ulcers, particularly in the indomethacin-induced model, suggesting its potential as a promising therapeutic agent for peptic ulcer treatment.
Introduction
Peptic ulcer is an acid-induced lesion of the digestive tract, typically located in the stomach or proximal duodenum, characterized by denuded mucosa with defects extending into the submucosa or muscularis propria. 1 With a global prevalence of approximately 10%, peptic ulcer disease has become a significant health concern. 2 The condition may affect various parts of the digestive system, including the lower esophagus, distal duodenum, or jejunum.
Several risk factors contribute to peptic ulcer development, with Helicobacter pylori infection and non-steroidal anti-inflammatory drugs (NSAIDs) use accounting for the majority of cases. Other risk factors include alcohol and tobacco consumption, and Zollinger-Ellison syndrome. 3 While conventional treatments such as proton pump inhibitors (PPIs), histamine-2 receptor antagonists (H2 blockers), and antacids are commonly prescribed, these medications often come with high costs and potential adverse effects. 4
In recent years, there has been growing interest in medicinal plants as alternative treatments for peptic ulcers. Approximately 50% of the Western population uses herbal medicines as alternative therapy, with 10% specifically using them for digestive disorders. 5 This preference can be attributed to herbal treatments costing only about one-sixth of conventional Western medicine and generally having fewer side effects. 6 Multiple studies have demonstrated the efficacy of herbal medicines in treating digestive diseases, including peptic ulcers. 6
In traditional medicine theory, peptic ulcers and gastroesophageal reflux are categorized under Stomach Pain and Stomach Rebellion conditions, which encompass various manifestations such as Qi Stagnation, Liver Qi Invading the Stomach, and Spleen-Stomach Deficiency with Cold. The primary pathological mechanisms involve damage to the Qi, manifesting as pain from Qi obstruction, rebellious Qi causing symptoms like belching and acid regurgitation, and in severe cases, Qi stagnation transforming into fire and heat. 7
The Vi Trang BH decoction is a polyherbal formulation composed of nine medicinal plants, each selected for its specific therapeutic properties, in accordance with traditional medicine principles. According to traditional medicine principles, the formula is structured to restore digestive harmony. Five components are included to regulate Qi, relieve pain, and address reflux syndrome: Fructus Aurantii and Pericarpium Citri reticulatae alleviate bloating and epigastric pain by resolving Qi stagnation; Cortex Magnoliae officinalis works with them to reduce abdominal fullness; Radix Linderae warms the stomach to disperse cold and alleviate pain; and Radix Saussureae lappae provides direct analgesic effects. To support digestive strength, Fructus Amomi is included to warm and invigorate the spleen. The formula also incorporates herbs with broader actions: Folium et Ramulus Crotonis tonkinensis, used in Vietnamese tradition, provides anti-inflammatory and detoxifying effects. Radix Bupleuri chinensis is crucial for addressing stress-related digestive complaints by dispersing Liver Qi stagnation. Finally, Radix et Rhizoma glycyrrhizae (licorice) strengthens the spleen and tonifies Qi, while also acting as a harmonizing agent and providing well-documented anti-inflammatory and mucosal healing properties. 8 While individual components of Vi Trang BH have shown effectiveness in treating peptic ulcers, no studies have evaluated the therapeutic effect of these ingredients in combination. This formulation is designed to work synergistically, addressing not only the symptoms of peptic ulcers but also the underlying imbalances in digestive function, such as Qi stagnation, cold accumulation, and liver-spleen disharmony. Animal models, particularly rodents, are valuable tools for evaluating anti-ulcer activity due to their genetic similarities to humans. Common experimental models include pylorus ligation, stress ulcer induction, and substance-induced peptic ulcers using agents such as alcohol, indomethacin, acetic acid, or cysteamine. 9
Indomethacin, an NSAID, induces ulcers by inhibiting cyclooxygenase (COX) enzymes, reducing prostaglandin production, and compromising the protective mucosal barrier. 9 Cysteamine, first described by Selye and Szabo for inducing peptic ulcers in rats, increases gastric acid secretion by depleting tissue somatostatin and enhancing gastrin release while inhibiting Brunner gland secretion, which is crucial for duodenal mucosal defense. 10 Given that the pathogenesis of peptic ulcers involves a complex imbalance between mucosal defensive factors and aggressive luminal factors, a comprehensive evaluation of a potential therapeutic agent requires assessing its effects against both types of insults. Therefore, to thoroughly characterize the anti-ulcer potential of Vi Trang BH, this study employed two mechanistically distinct and clinically relevant experimental models. The indomethacin-induced ulcer model was selected to investigate the decoction's cytoprotective and anti-inflammatory properties by inducing gastric ulcers caused by the depletion of endogenous protective prostaglandins, a common pathway for NSAID-related gastropathy. In addition, the cysteamine-induced ulcer model was utilized to assess the efficacy of the decoction against ulcers driven by gastric acid hypersecretion and impaired duodenal neutralization. Using these two complementary models enables a more comprehensive mechanistic profile, facilitating the determination of whether Vi Trang BH acts by strengthening mucosal defense, reducing acid secretion, or both.
Materials and Methods
The Investigational Product
Vi Trang BH remedy consists of nine medicinal plants including Fructus Aurantii 12 grams, Cortex Magnoliae officinalis 12 grams, Radix Saussureae lappae 12 grams, Radix Linderae 12 grams, Pericarpium Citri reticulatae 12 grams, Fructus Amomi 8 grams, Folium et Ramulus Crotonis tonkinensis 12 grams, Radix Bupleuri chinensis 12 grams and Radix et Rhizoma Glycyrrhizae 4 grams. The herbal plants met the standards in the Chinese Pharmacopoeia 2015 and the Vietnam Pharmacopoeia V.11,12 The fresh plant materials were first washed and boiled twice at 100 °C for 90 min each with deionized water to collect the extracted liquid. Then the total amount of extracted liquid was condensed to a concentration of 96 grams/50 mL and was preserved in 100-mL glass bottles (storage conditions: temperature (2-8 °C), humidity (under 75%).
To characterize its phytochemical profile, the Vi Trang BH decoction was analyzed for total phenolic and flavonoid content. The total phenolic content, determined using the Folin-Ciocalteu method, 13 was 3.41 ± 0.04 milligrams of gallic acid equivalents (GAE) per milliliter. Furthermore, the total flavonoid content was quantified via the Dowd method, 14 yielding a concentration of 194.4 ± 14.3 micrograms of quercetin equivalents (QE) per milliliter. Both values represent the mean ± SD of five independent determinations.
Chemicals and Reagents
Indomethacin and cysteamine were obtained from Sigma (Aldrich) Chemicals Pvt. Ltd USA and used as inducers for peptic ulcers in rats. Misoprostol 200 mcg tablets (STELLA, Vietnam) and famotidine 40 mg tablets (Vidiphar Central Pharmaceutical Jsc., Vietnam), were used as the positive control compound. Pentobarbital was obtained from Sigma (Aldrich) Chemicals Pvt. Ltd USA and used as the anesthetic agent.
Experimental Animals
Adult Wistar rats (weighed 200 ± 20 grams) of both sexes were raised for 7 days before researching to adapt to the environment and breeding conditions and during the experiment at the laboratory of the Department of Pharmacology, Hanoi Medical University. The temperature in the experimental animal room was 22 °C ± 3 °C. The relative humidity was at a minimum of 30% and preferably would not exceed a maximum of 70%, other than during room cleaning. The daily artificial lighting sequence would be 12 h of light/dark. Food and water were provided ad libitum. All protocols used in this study involving animals were approved by the Scientific Board Committee of Hanoi Medical University (ref number: IRB00003121), under ethics code No. 2727/QĐ-ĐHYHN, dated June 18, 2024.
Experimental Design
All methods were carried out in accordance with ARRIVE guidelines and the National Institutes of Health guide for the care and use of laboratory animals.
Indomethacin-Induced Peptic Ulcers
The experimental study was conducted using Wistar rats randomly divided into five groups (n = 10 per group). The animals received daily oral treatment for seven consecutive days according to their group allocation. Group I (normal control) and Group II (model) received distilled water (10 ml/kg/day). Group III, serving as the positive control group, was administered misoprostol (50 μg/kg/day). 15 Groups IV and V received Vi Trang BH decoction at doses of 40 ml/kg/day and 80 ml/kg/day, respectively. All treatments were administered via oral gavage. The selected doses were based on Vi Trang BH's safety profile from previous toxicity studies, dosage guidelines for its herbal constituents from the Vietnamese Pharmacopoeia fifth edition, the Chinese Pharmacopoeia 2015, and traditional medicinal usage.11,12
Following the seven-day pretreatment period, animals were fasted for 18 h while maintaining free access to water. One hour after the final treatment dose, peptic ulcers were induced in Groups II through V by oral administration of indomethacin (40 mg/kg).16,17 Six hours post-indomethacin administration, the animals were euthanized by pentobarbital (20 mg/kg, i.p.) for subsequent evaluations.
Blood samples were collected via cardiac puncture for biochemical analysis, including the measurement of liver function markers (AST, ALT) and kidney function parameters (creatinine and urea). The stomach and duodenum were carefully excised and opened along the greater curvature. The specimens were gently rinsed with 0.9% NaCl solution and fixed for macroscopic examination. The ulcer index (UI) of peptic ulcers was evaluated according to the scoring system described by Raish M et al,
18
where: 0 indicates no lesions (normal stomach); 0.5 represents hyperemia (red coloration); 1 signifies hemorrhagic spots; 2 corresponds to 1–5 small ulcers; 3 indicates many small ulcers; 4 represents many small and large ulcers; and 5 denotes stomach full of ulcers with perforations. The protective index (PI) was calculated using the following formula:
PI = (UImodel-UItreated) × 100)/(UImodel)
18
(where UImodel represents the ulcer index of the model group and UItreated represents the ulcer index of the treatment groups.)
For histopathological examination, six out of ten gastric tissue samples were fixed in 10% neutral buffered formalin and processed using standard histological techniques. The fixed tissues were embedded in paraffin, sectioned at 5-μm thickness, and stained with hematoxylin and eosin (H&E). The stained sections were examined under a light microscope for detailed histological assessment. Microscopic scoring was performed according to the scale established by Simões S et al, 19 with parameters detailed in Table 1. The total microscopic score was calculated by summing individual parameter scores, with a maximum possible score of 15. The histopathological examination was scored by a single pathologist, who remained blinded to all treatment groups throughout the histological preparation and evaluation process.
Histological Scoring System for the Microscopic Evaluation of Gastric Lesions According to the Classification of Simões et al. 19

Cysteamine-Induced Peptic Ulcers
The experimental protocol utilized Wistar rats randomly allocated into five groups (n = 10 per group). Animals received daily oral treatment for seven consecutive days according to their group assignment. The normal control group (Group I) and model group (Group II) received distilled water. Group III, serving as the positive control, was administered famotidine (50 mg/kg body weight/day). 20 Groups IV and V received Vi Trang BH decoction at doses of 40 ml/kg and 80 ml/kg body weight/day, respectively. All treatments were administered via oral gavage. The selected doses were based on Vi Trang BH's safety profile from previous toxicity studies, dosage guidelines for its herbal constituents from the Vietnamese Pharmacopoeia fifth edition, the Chinese Pharmacopoeia 2015, and traditional medicinal usage.11,12
One hour following the final treatment dose, peptic ulcers were induced in Groups II through V using a dual-dose cysteamine protocol. Two doses of cysteamine (400 mg/kg each) were administered orally at a four-hour interval.21,22 Following the first cysteamine dose, animals were fasted for 18 h while maintaining free access to water. Twenty-four hours after the initial cysteamine administration, the animals were euthanized by pentobarbital (20 mg/kg, i.p.) for subsequent evaluations.
Blood samples were collected via cardiac puncture for biochemical analysis, including liver function markers (AST, ALT) and kidney function parameters (creatinine and urea). For macroscopic assessment, the stomach and duodenum were carefully excised, opened along the greater curvature, and gently rinsed with 0.9% NaCl solution. The specimens were fixed and examined for ulcerative lesions according to Thiemer's classification23,24,25: Grade I - presence of edema, hyperemia, and single submucosal punctiform hemorrhage; Grade II - submucosal hemorrhagic lesions with small erosions; and Grade III - deep ulcers with erosions and invasive lesions.
The ulcer index (UI) was calculated using the formula:
UI = (number of Grade I lesions × 1) + (number of Grade II lesions × 2) + (number of Grade III lesions × 3).
The protective index (PI) was determined using the equation:
PI = (UImodel-UItreated) × 100)/(UImodel)
(where UImodel represents the ulcer index of the model group and UItreated represents the ulcer index of the treatment groups)
For histopathological examination, six out of ten gastric tissue samples were fixed in 10% formalin, embedded in paraffin, and sectioned at 5-μm thickness. The sections were stained with hematoxylin and eosin (H&E) and examined under a light microscope. Microscopic scoring was performed according to the scale established by Simões S et al, with detailed parameters presented in Table 1. 19 The histopathological examination was scored by a single pathologist, who remained blinded to all treatment groups throughout the histological preparation and evaluation process.
Statistical Analysis
Statistical analyses were carried out using SPSS version. 26.0. The data were expressed as the mean ± standard deviation (SD). Multiple groups were compared by one-way analysis of variance (ANOVA) followed by Bonferroni's post-hoc comparisons tests. In the analysis of histological grading, nonparametric tests (Mann-Whitney U test) were used. The p-values of < 0.05 were considered to be statistically significant.
Results
General Observations Throughout the Studies
Throughout both experimental periods, all rats remained alive and healthy, with normal weight gain. No adverse effects were observed in any treatment group, with no notable changes in vital signs, physical appearance (skin and fur condition), or daily behavioral patterns. All animals maintained normal activity levels and feeding behavior until the time of sacrifice at the end of the experiments.
Effects of Vi Trang BH Decoction on Indomethacin-Induced Peptic Ulcers in Rats
Macroscopic evaluation revealed marked reductions in ulcer incidence, ulcer index, and the number of gastric and duodenal lesions in Vi Trang BH-treated groups compared to the model group. The protective indices were 71.79% and 69.23% for the 40 mL/kg and 80 mL/kg doses, respectively (Table 2 and Figure 1). Both doses showed greater protective effects than misoprostol (35.90%), the positive control.

Macroscopic Appearance of Gastric Mucosa in the Indomethacin-Induced Ulcer Model. Representative Images Show the Stomachs From Each Experimental Group. The Model Group Exhibits Severe Hemorrhagic Lesions and Swelling Due to Blood Accumulation. Treatment with Misoprostol or Vi Trang BH (40 and 80 mL/kg) Markedly Reduced the Severity and Number of Ulcers. Black Arrow Indicates a Large, Hemorrhagic Ulcer; Blue Dashed Arrows Point to Smaller Erosions.
Effects of Vi Trang BH Decoction on the Macroscopic Indices of Indomethacin-Induced Peptic Ulcers.

***p < 0.001 compared with the model group.
Histological analysis further confirmed these findings. Both doses of Vi Trang BH decoction significantly improved gastric and duodenal lesions. Notably, rats treated with 40 mL/kg of Vi Trang BH showed complete protection against mucosal erosion, with all samples (6/6) classified as Grade 0, compared to only 2/6 samples in the model group. Inflammatory changes were also substantially reduced in the Vi Trang BH-treated groups, with the 40 mL/kg dose group showing predominantly Grade 1 inflammation (4/6) and Grade 2 inflammation (2/6), while the model group exhibited more severe inflammation with Grade 2 (4/6) and Grade 3 (2/6). No hemorrhagic injury was observed in any of the samples (Table 3 and Figure 2).

Histological Analysis of Gastric Tissue in the Indomethacin-Induced Ulcer Model. Representative Micrographs (H&E Staining; Magnifications 40× (A1-E1), 100× (A2-E2), 400 × (A3-E3)). The Model Group (B1-3) Shows Significant Pathological Changes, Including Deep Mucosal Erosion, Submucosal Edema, and Severe Inflammatory Cell Infiltration. These Changes Were Substantially Ameliorated in the Misoprostol (C1-3) and Vi Trang BH-Treated Groups (D1-3 & E1-3), Which Showed Preserved Mucosal Architecture and Reduced Inflammation. Black Ovals Denote Areas of Mucosal Erosion; Black Arrows Indicate Dense Infiltration of Inflammatory Granulocytes, and Red Dashed Oval Denotes an Apoptosis Injury.
Effects of Vi Trang BH Decoction on Microscopic Lesions in Indomethacin-Induced Peptic Ulcers.

*, ** compared to group I (p < 0.05, p < 0.01).
Δ Groups III, IV, and V were compared to group II (p < 0.05).
Effects of Vi Trang BH Decoction on Cysteamine-Induced Peptic Ulcers in Rats
Vi Trang BH decoction administered at doses of 40 mL/kg b.w/day and 80 mL/kg b.w/day for seven consecutive days showed improvement in cysteamine-induced peptic ulcers, though the effects were less pronounced than in the indomethacin model.
Macroscopic evaluation indicated that both doses of Vi Trang BH reduced the ulcer index compared to the model group, with protective indices of 27.52% and 30.28% for the 40 mL/kg and 80 mL/kg doses, respectively (Table 4 and Figure 3). These values were comparable to famotidine (28.44%), the positive control.

Macroscopic Appearance of Gastric Mucosa in the Cysteamine-Induced Ulcer Model. Representative Images Showing Gastric Surfaces. The Model Group Displays Deep Ulcerative Lesions and Surrounding Hyperemia. Treatment with Famotidine and Vi Trang BH (40 and 80 mL/kg) Resulted in a Visible Reduction in Ulcer Severity, Leading to a Smoother and Healthier-Appearing Mucosal Surface. Black Arrow Indicates a Deep Ulcer; Blue Dashed Arrows Highlight Smaller, Superficial Erosions.
Effects of Vi Trang BH Decoction on the Macroscopic Indices of Cysteamine-Induced Peptic Ulcers.

Histological analysis corroborated these findings, demonstrating improvements in gastric and duodenal lesions in the Vi Trang BH-treated groups. The treatment groups showed significantly better erosion depth scores, with most samples (5/6) exhibiting only Grade 1 erosions compared to the model group. For ulcer depth, the 40 mL/kg dose reduced severity with fewer Grade 3 lesions (0/6) compared to the model group (2/6), while the 80 mL/kg dose showed even greater improvement with 4/6 samples classified as Grade 0. No hemorrhagic injury was observed in any of the samples (Table 5 and Figure 4).

Histological Analysis of Gastric Tissue in the Cysteamine-Induced Ulcer Model. Representative Micrographs (H&E Staining; Magnifications 40× (A1-E1), 100× (A2-E2), 400× (A3-E3)). The Model Group (B1-3) Exhibits Severe Disruption of the Mucosal Architecture, Including Deep Erosions that Penetrate the Submucosa and Significant Inflammatory Infiltration. Famotidine (C1-3) and Vi Trang BH Treatments (D1-3 & E1-3) Helped Preserve Mucosal Integrity and Reduced the Inflammatory Response. Black Ovals Highlight Areas of Mucosal Erosion and Ulceration; Black Arrows Point to the Infiltration of Inflammatory Cells in the Submucosa; Red Dashed Oval Denotes an Apoptosis Injury, and Blue Dotted Oval Highlights a Peptic Ulcer.
Effects of Vi Trang BH Decoction on Microscopic Lesions in Cysteamine-Induced Peptic Ulcers.
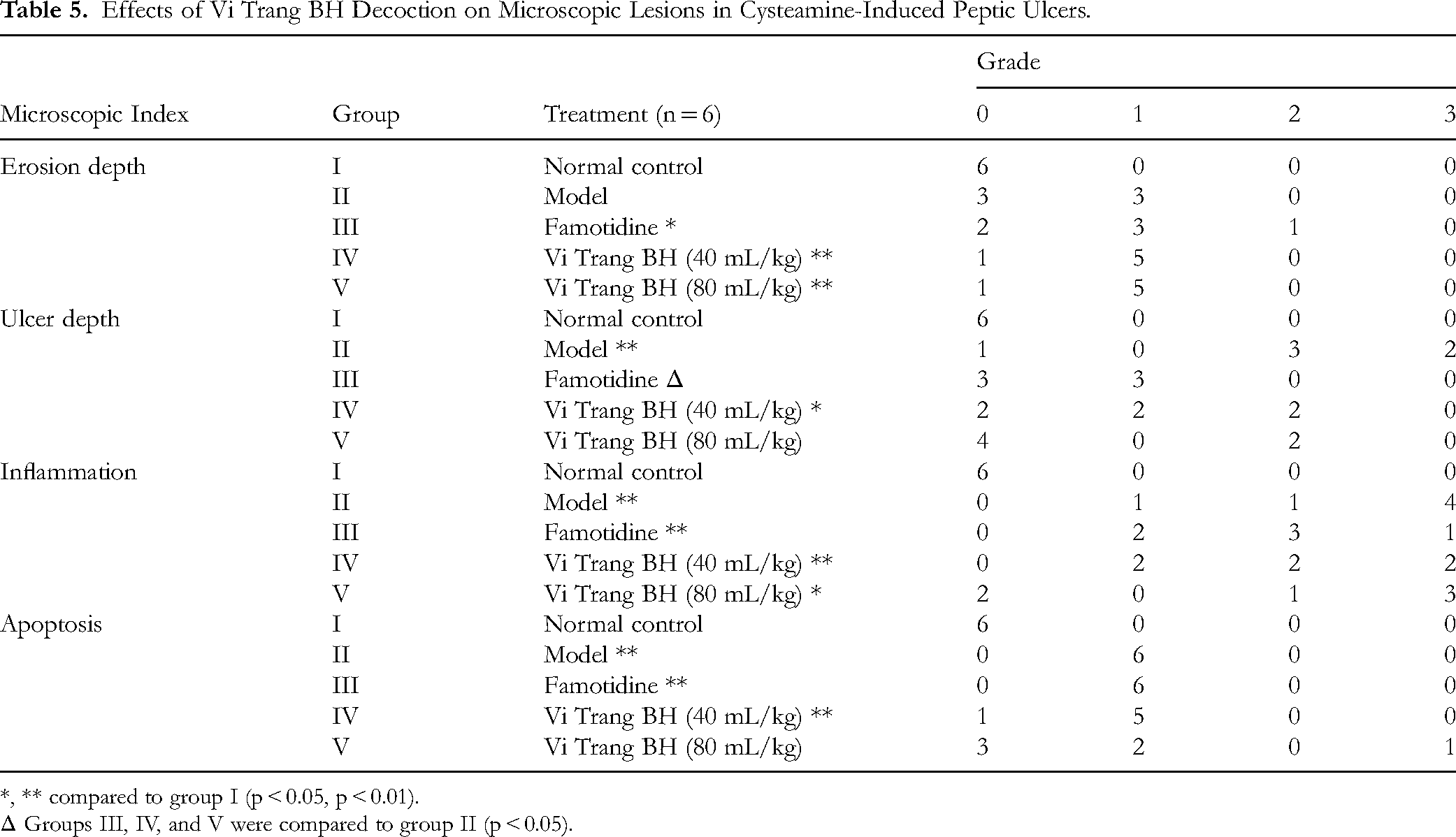
*, ** compared to group I (p < 0.05, p < 0.01).
Δ Groups III, IV, and V were compared to group II (p < 0.05).
Effects of Vi Trang BH Decoction on Biochemical Parameters
As shown in Table 6, Vi Trang BH decoction ameliorated the elevated blood levels of hepatic enzymes in both experimental models. In the indomethacin model, AST and ALT levels were significantly reduced in both Vi Trang BH-treated groups compared to the model group (p < 0.05 for AST, p < 0.01 for ALT). Similarly, in the cysteamine model, the 40 mL/kg dose tended to decrease AST levels; however, this trend is not significant (p > 0.05). Regarding renal function parameters, treatment regimens showed minimal effects on creatinine and urea levels.
Effects of Vi Trang BH on Biochemical Markers of Liver and Kidney Function.

All values are expressed as mean ± standard deviation (SD).
#, ##, ### compared to the normal control group (p < 0.05, p < 0.01, p < 0.001).
*, ** compared to the model group (p < 0.05, p < 0.01).
Discussion
Peptic ulcers represent a significant global health challenge in the twenty-first century, with changing lifestyle patterns and dietary habits contributing to their increasing prevalence. Recent epidemiological studies suggest that peptic ulcer disease affects approximately 5–10% of the global population. 26 The limitations of conventional treatments, including adverse effects and high costs associated with synthetic drugs, have sparked renewed interest in natural therapeutic alternatives. The use of herbal medicines in gastrointestinal disorders has increased significantly over the past decade, with studies reporting efficacy rates comparable to conventional treatments in some cases. 27
This study investigated the protective effects of Vi Trang BH remedy, a novel combination of nine traditional medicinal plants, in both indomethacin- and cysteamine-induced peptic ulcer models. A key strength of this study is its use of two mechanistically distinct experimental models—indomethacin- and cysteamine-induced ulcers—to provide a comprehensive assessment of the protective effects of Vi Trang BH. The choice of these models was deliberate, as they mimic different clinical etiologies of peptic ulcer disease.
The indomethacin model represents a well-established approach to studying NSAID-induced gastric damage. Indomethacin's ulcerogenic effects operate through prostaglandin synthesis inhibition via COX-1 and COX-2 blockade, 28 leading to reduced mucosal protection and increased acid vulnerability. 29 NSAID-induced ulcers are a major public health concern, with chronic NSAID use being a key risk factor for peptic ulcer development. 30 Our results showed that Vi Trang BH at both tested doses (40 mL/kg and 80 mL/kg) significantly reduced ulcer indices and improved microscopic parameters, comparable to misoprostol (50 µg/kg), the reference drug used in the indomethacin model. Misoprostol, a synthetic prostaglandin E1 analog, is recognized for its protective effects on gastrointestinal mucosal cells, which include inhibiting gastric acid secretion, enhancing mucus and bicarbonate secretion, increasing blood flow, and promoting mucosal cell regeneration. 31 The dose of 50 µg/kg used in this study aligns with preclinical studies in rats, where misoprostol has been shown to reduce NSAID-induced gastric lesions effectively.15,32 This suggests that the chosen dose is clinically relevant and suitable for evaluating gastroprotective effects in this model. This level of protection against NSAID-induced damage suggests potential clinical applications in prophylactic treatment for patients requiring long-term NSAID therapy. Vi Trang BH's superior protective indices (71.79% and 69.23% for 40 mL/kg and 80 mL/kg, respectively, compared to 35.90% for misoprostol) indicate that its multi-component herbal formulation may offer enhanced cytoprotection and reinforcement of the gastric mucosal barrier, potentially through synergistic effects of its constituents. Additionally, its anti-inflammatory effects are demonstrated by a decrease in the microscopic inflammatory index, which aligns with the current understanding of ulcer pathogenesis. 29
In the cysteamine-induced peptic ulcer model, cysteamine acts by decreasing somatostatin levels in the duodenal mucosa, which leads to an increase in the production of reactive oxygen species, reduces the elimination of free radicals, and elevates the expression of endothelin-1, a vasoconstrictor. These changes impair blood perfusion, resulting in reduced duodenal mucosal blood flow, increased tissue ischemia, and lowered blood oxygen levels.21,33 Additionally, cysteamine inhibits mucus secretion from Brunner's glands in the duodenum, stimulates gastric acid secretion, slows gastric emptying, and increases serum gastrin levels.34,35 This model is therefore particularly suited for assessing the anti-secretory and acid-neutralizing properties of a treatment. This study showed that cysteamine, at a dose of 400 mg/kg twice, resulted in significant peptic ulceration, with 100% of the rats developing ulcers. Famotidine, a histamine-2 receptor antagonist used as the reference drug control in the cysteamine model at 50 mg/kg, reduces gastric acid secretion by competitively inhibiting histamine binding to H2 receptors on parietal cells. The dose of 50 mg/kg is consistent with preclinical studies demonstrating significant anti-ulcer effects in rats, 20 indicating compelling control when used to evaluate the antacid activity of the investigational product. Both doses of the Vi Trang BH remedy showed a tendency to reduce the ulcer index compared with the model group and improved microscopic peptic lesions, with protective indices of 27.52% and 30.28% for 40 mL/kg and 80 mL/kg, respectively.
By utilizing both models, this study demonstrates the multifaceted nature of Vi Trang BH. The pronounced success in the indomethacin model points to a potent cytoprotective effect, while the moderate activity in the cysteamine model indicates a secondary, supportive role in modulating gastric acid. This dual-model approach thus provides a more complete and clinically relevant profile of the decoction's therapeutic potential than either model could alone. The comparative pharmacodynamic analysis indicates that Vi Trang BH's efficacy may stem from its multi-targeted approach. The herbal formulation's broader spectrum of action, supported by its diverse phytochemical constituents, may explain its promising performance in the current studies. These findings align well with the effects of some ingredients included in the Vi Trang BH formula. For instance, research by Wu Y et al has demonstrated that licorice flavonoids from the Glycyrrhiza rhizome alleviate gastric ulcers by modulating the gut microbiota and stimulating mucus cell regeneration. 36 Another study on licorice suggests that it enhances gastrointestinal prostaglandin levels, increases mucus secretion, and contains carbenoxolone, which reduces gastrin secretion. 37 Additionally, Saussurea lappa root possesses significant antiulcer properties, possibly due to its cytoprotective action or its ability to strengthen the gastric and duodenal mucosa, enhancing mucosal defense. 38 Saikosaponins in Bupleuri chinensis have also demonstrated anti-ulcer effects by decreasing NF-κB and TNF-α levels. 39 The ethanol extract from Magnolia officinalis bark, at a dose of 30 mg/kg, exhibits gastroprotective properties against acute ethanol-induced gastric ulceration by inhibiting oxidation and targeting the NF-κB inflammatory signaling pathway. 40 Besides, Linderae Radix has a protective effect related to inhibiting the JAK-STAT signaling pathway, improving intestinal inflammation, and reducing intestinal epithelial cell apoptosis. 41 The protective effects of Citri reticulatae pericarpium (tangerine peel) on the digestive system, such as modulating gut microbiota, promoting gastrointestinal motility, and inhibiting gastric ulcers, have been demonstrated. 42 Furthermore, it has been shown that the anti-ulcerogenic activity and the mechanism underlying it are associated with its antioxidant properties. 43 According to Yana Lv et al, Amomi fructus may influence inflammatory response and apoptosis by regulating the PI3 K/AKT signaling pathway. 44
In addition to its effects on peptic ulcers, our results also suggest that Vi Trang BH decoction has a positive impact on liver function. Indomethacin may cause an elevation in liver enzymes by inhibiting COX, altering liver metabolism, increasing oxidative stress, and directly damaging liver cells.45,46 In this present study, both indomethacin and cysteamine increased liver enzymes, as demonstrated in the model group. These findings show that Vi Trang BH remedy has a regulatory effect on liver enzymes, addressing the drawbacks of synthetic drugs. These results are consistent with the effects of some ingredients in the Vi Trang BH formula. Flavonoids from Fructus Aurantii significantly decreased serum levels of AST and ALT in rats with 5-fluorouracil-induced hepatotoxicity. 47 The oral administration of the ethanolic extract of Saussureae lappae significantly protected the hepatic cells from damage in rabbits. 48 According to Wang et al, Radix Bupleuri could protect against acetaminophen-induced liver injury based on CYP450 inhibition. 49 Additionally, Linderae Radix extracts improved the histopathological status and decreased the serum levels of ALT and AST in a rat model of alcoholic liver injury. 50 Therefore, the ability of Vi Trang BH to significantly attenuate the increase in AST and ALT in both models demonstrates a clear protective action against this chemically induced liver damage. This effect is not merely an incidental finding but a therapeutically relevant property. This “hepatoprotective” action suggests that the herbal formulation may possess antioxidant or anti-inflammatory properties that mitigate the systemic toxicity of the ulcer-inducing agents, a critical advantage in managing patients, particularly those on chronic NSAID therapy. Since the observed hepatoprotective effects suggest a favorable safety profile, this is particularly relevant from a clinical standpoint, as an ideal anti-ulcer therapy should not only heal the primary lesion but also be protective against off-target organ damage. For the reasons above, it is suggested that the effect of regulating liver enzymes could also be further studied in the future.
The Vi Trang BH remedy combines various herbs, each with unique mechanisms of action, to enhance its therapeutic efficacy. Recent advances in ethnopharmacology indicate that multi-component herbal formulations may yield superior therapeutic outcomes compared to single-herb treatments. 51 Beyond the mechanistic insights, it is crucial to consider the translational significance of these findings for treating human peptic ulcers. The pronounced protective effect of Vi Trang BH in the indomethacin model is particularly relevant, as NSAID-induced gastropathy is a major clinical problem worldwide. 52 This suggests a potential role for the decoction as a prophylactic or adjunctive therapy for patients requiring long-term NSAID treatment, offering a potentially safer alternative to conventional agents. Furthermore, the decoction's efficacy against cysteamine-induced ulcers suggests its potential utility in managing ulcers associated with hypersecretory conditions. Additionally, the use of readily available herbal components presents a cost-effective alternative to conventional therapies. This therapeutic potential is strongly corroborated by the remedy's traditional use in Vietnamese medicine. Vi Trang BH was historically formulated to treat conditions classified as ‘Stomach Pain’ and ‘Stomach Rebellion,’ arising from ‘Qi Stagnation’ and disharmony between the liver and stomach. Our scientific findings, which demonstrate reduced mucosal lesions, suppressed inflammation, and hepatoprotective effects, provide modern biomedical validation for these traditional applications. While no formal clinical trials on this specific formulation have been conducted, the strong alignment between its traditional use and our robust preclinical data provides a compelling rationale for advancing Vi Trang BH into clinical studies to assess its efficacy and safety in human patients.
While this study successfully demonstrates the significant protective effects of Vi Trang BH against peptic ulcers, we acknowledge certain limitations. A primary limitation is the absence of direct measurement of specific molecular markers that would fully elucidate the decoction's mechanism of action. Our findings are based on macroscopic and histological assessments, which, although robust, do not provide a complete picture of the underlying anti-inflammatory and antioxidant pathways.
Therefore, future research should be directed toward these mechanistic studies and figuring out the active ingredients. Investigating the effects of the decoction on prostaglandin synthesis, its ability to scavenge free radicals through the activity of antioxidant enzymes like superoxide dismutase (SOD), and its influence on the expression of pro- and anti-inflammatory cytokines (eg, TNF-α, IL-6) would provide a more comprehensive understanding of its mode of action. Such studies would not only validate the hypotheses drawn from our current findings but also strengthen the scientific rationale for advancing Vi Trang BH toward clinical application.
Conclusion
Based on our experimental findings, Vi Trang BH remedy demonstrated significant protective effects against peptic ulcers, particularly in the indomethacin-induced model, with protective indices reaching up to 71.79%. The remedy's efficacy was confirmed through both macroscopic and histological analyses, showing notable improvements in gastric and duodenal lesions, while also exhibiting beneficial effects on liver enzyme regulation and minimal effects on creatinine and urea levels. These findings, along with the observed efficacy at both tested doses, provide compelling evidence supporting Vi Trang BH's development as a promising therapeutic option for peptic ulcer treatment.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251380048 - Supplemental material for Protective Effects of Vi Trang BH Decoction on Indomethacin- and Cysteamine-Induced Peptic Ulcers in Wistar Rats
Supplemental material, sj-docx-1-npx-10.1177_1934578X251380048 for Protective Effects of Vi Trang BH Decoction on Indomethacin- and Cysteamine-Induced Peptic Ulcers in Wistar Rats by Hung Tien Bui, Huynh Thi Bui, Van Anh Thi Pham, Phong Xuan Pham, Toan Van Nguyen, Tung Thanh Tran, Thuy Thi Nguyen, Linh Dieu Do, Quang Vinh Trinh and Duong Thuy Dau in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X251380048 - Supplemental material for Protective Effects of Vi Trang BH Decoction on Indomethacin- and Cysteamine-Induced Peptic Ulcers in Wistar Rats
Supplemental material, sj-docx-2-npx-10.1177_1934578X251380048 for Protective Effects of Vi Trang BH Decoction on Indomethacin- and Cysteamine-Induced Peptic Ulcers in Wistar Rats by Hung Tien Bui, Huynh Thi Bui, Van Anh Thi Pham, Phong Xuan Pham, Toan Van Nguyen, Tung Thanh Tran, Thuy Thi Nguyen, Linh Dieu Do, Quang Vinh Trinh and Duong Thuy Dau in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X251380048 - Supplemental material for Protective Effects of Vi Trang BH Decoction on Indomethacin- and Cysteamine-Induced Peptic Ulcers in Wistar Rats
Supplemental material, sj-docx-3-npx-10.1177_1934578X251380048 for Protective Effects of Vi Trang BH Decoction on Indomethacin- and Cysteamine-Induced Peptic Ulcers in Wistar Rats by Hung Tien Bui, Huynh Thi Bui, Van Anh Thi Pham, Phong Xuan Pham, Toan Van Nguyen, Tung Thanh Tran, Thuy Thi Nguyen, Linh Dieu Do, Quang Vinh Trinh and Duong Thuy Dau in Natural Product Communications
Footnotes
Acknowledgments
This study was supported by other departments and faculties at Hanoi Medical University (HMU), Hanoi, Vietnam. The authors, therefore, acknowledge with thanks the HMU staff for useful advice and technical support.
ORCID iDs
Ethical Considerations
All experimental procedures were approved by the Scientific Board Committee of Hanoi Medical University (reference number: IRB00003121) with approval number 2727/QĐ-ĐHYHN.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
CRediT Authorship Contribution Statement
Hung Tien Bui: Conceptualization, Formal analysis, Methodology, Project administration, Supervision, Writing – original draft; Huynh Thi Bui: Data curation, Investigation, Methodology, Writing – original draft; Van Anh Thi Pham: Formal analysis, Methodology, Project administration, Supervision, Writing – review & editing; Phong Xuan Pham: Methodology, Supervision, Visualization; Toan Van Nguyen: Investigation, Resources, Visualization; Tung Thanh Tran: Conceptualization, Resources, Software, Supervision; Thuy Thi Nguyen: Formal analysis, Investigation, Resources; Linh Dieu Do: Resources, Software, Writing – review & editing; Quang Vinh Trinh: Data curation, Software, Formal analysis, Writing – review & editing; Duong Thuy Dau: Conceptualization, Investigation, Project administration, Supervision, Validation, Visualization, Writing – review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data that support the findings of this study are available from the corresponding author, Dr Duong Thuy Dau, upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
