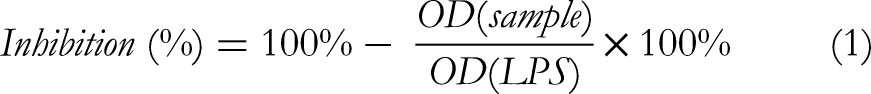Abstract
Introduction
Electrospinning is a method of using high electric voltage to generate nano- or micro-fibers from polymeric solutions or melts. 1 Electrospun fibers are widely used in many fields such as biomedicine, environment, energy, and packaging. One of the most important applications of electrospinning technique in the biomedical field is the encapsulation of bioactive ingredients.1–3 Nowadays, the application of bioactive compounds in functional foods and pharmaceuticals has attracted significant attention from scientists due to their wide variety of valuable bioactivities. 4 However, the unstable nature, sensitivity to light and temperature, poor water solubility, gastrointestinal degradation, and low bioavailability of some active components have led to limitations in their application and decreased therapeutic effectiveness. 5 Therefore, encapsulation of active ingredients in polymeric materials has been considered an effective method to protect them, enhance their stability and bioavailability as well as to achieve controlled release. 5 Many encapsulation techniques have been reported to generate nano- or microstructures including electrospinning, spray drying, freeze drying, gelation, emulsification, liposome, complexation, and so on.5,6 Among them, electrospun fibers are considered a flexible encapsulation method with several key advantages over other methods, including high encapsulation efficiency, stability, and targeted, sustained drug release profiles. 5
Numerous organic polymers, both natural and synthetic, have been successfully explored for encapsulation using the electrospinning approach. 7 Chitosan (CS), polylactic acid (PLA), and polyethylene glycol (PEG) are some of the most popular polymers used for the fabrication of electrospun nanofibers due to their biocompatibility, biodegradability, and safety. 7 Additionally, CS exhibits antibacterial and anti-inflammatory activity, whereas PLA has excellent mechanical strength and good fiber-forming ability.8–11 The addition of hydrophilic polymer PEG to the CS and PLA polymer blends was found to enhance compatibility and dispersibility between the two polymers. 12 PEG has been widely utilized in drug delivery to enhance the stability and solubility of drugs in vivo, decrease clearance rates from circulation, and improve drug efficacy. 13 To date, various natural bioactive compounds have been reported to be encapsulated in electrospun nanofibers such as carotenoids,14,15 phycocyanin,16,17 gallic acid, 18 catechins, 19 quercetin, 20 curcumin,21–23 vitamins,24,25 essential oils, 26 plant extracts.27,28
Curcumin (Cur), an active ingredient extracted from turmeric (Curcuma longa), is widely known for its anti-inflammatory, wound healing, gastroprotective, antiulcer, antioxidant, antibacterial, antifungal, antiviral, immunomodulatory, and anticancer properties.29,30 Curcumin has been widely used in various functional food products to support the treatment of gastritis.
Berberine (Ber) is one of the most interesting and promising natural agents extracted from Coscinium fenestratum, possessing valuable activities such as anti-inflammatory, antibacterial, neuroprotective, antidiarrheal, antidiabetic, and anticancer effects.31–33 Berberine exhibits antibacterial activity against a variety of bacteria, including H. pylori, Actinobacillus pneumoniae, Bacillus dysenteriae, and shows synergistic antibacterial effects with some antibiotics. 34 Interestingly, berberine exhibited a higher antibacterial effect against H. pylori than that of the two commercial antibiotics, metronidazole and tetracycline. 35
Rotundine (tetrahydropalmatine), a natural alkaloid from Stephania sp., has been reported to possess analgesic, anti-inflammatory, neuroprotective, anti-addiction, and antitumor effects. 36 Rotundine (Rot) has been demonstrated to have excellent analgesic activity in both experimental and clinical studies. 37
Gastritis is inflammation of the lining of the stomach. It can occur suddenly (acute gastritis) or develop gradually over time (chronic gastritis). 38 Chronic gastritis is a common and easily recurring digestive system disorder that causes pain, discomfort, and affects quality of life. 39 One of the causes of severe gastric ulcers is H. pylori. 38 However, antibiotic-resistant bacteria are currently becoming increasingly common, making the treatment of patients infected with H. pylori more challenging. 40 There are some types of synthetic medicines for the treatment of chronic gastritis, such as anti-gastric acid agents, antibiotics to kill H. pylori, and drugs to protect the gastric mucosa. 39 However, these synthetic drugs often trigger many serious adverse effects, and the disease may relapse after stopping the medication. Therefore, the application of natural bioactive ingredients in the prevention and treatment of gastritis has attracted much attention from scientists due to their high safety, low drug resistance, and stable efficacy. 39
In the current paper, we present the results of preparing the synergistic mixture of three natural bioactive compounds curcumin, berberine, and rotundine for the treatment of gastritis. Each active ingredient has a diverse spectrum of biological effects. The purpose of preparing this mixture of three active ingredients was to combine the strengths of each compound. The obtained mixture is expected to be more effective in treating gastritis than using only one parent compound. On the other hand, all three active agents were reported to be hydrophobic, with rapid clearance and low bioavailability in vivo.34,36,41 With the goal of enhancing bioavailability and enabling controlled release of the bioactive agents, we encapsulated the mixture of curcumin, berberine, and rotundine in electrospun polymer nanofibers CS/PEG/PLA. We also report the physicochemical properties, drug release characteristics, safety, and in vivo anti-gastric ulcer activity of the obtained electrospun nanofibers.
Experimental
Materials
Chitosan (CS, Mw 100 kD; degree of deacetylation = 99%) was purchased from Sigma-Aldrich (USA). Polylactic acid (PLA, Mw 200 kD), Polyethylene glycol (PEG, Mw 600 Da), acetic acid (≥ 99.5%), chloroform (≥ 99.5%), and ethanol (≥ 99.5%) were purchased from Merck (Germany). Curcumin, berberine, and rotundine (≥ 95%) were supplied by the Department of Pharmaceutical Chemistry and Organic Synthesis, Institute of Natural Products Chemistry, VAST, Vietnam. All the chemicals were used without further treatment.
RAW 264.7 cells were provided by Professor Domenico Delfino (Perugia University, Italy). Lipopolysaccharides (LPS) from Escherichia coli, sodium nitrite, sulfanilamide, N-1-napthyl ethylenediamine dihydrochloride, 3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyl tetrazolium bromide (MTT), Dexamethasone, phosphoric acid and dimethyl sulphoxide (DMSO) were purchased from Sigma-Aldrich (USA), Dulbecco's Modified Eagle's Medium (DMEM), fetal bovine serum (FBS) were obtained from Life Technologies (USA) for the nitrite assay.
1,1-diphenyl-2-picrylhydrazyl (DPPH), dimethyl sulfoxide (DMSO), ethanol, and ascorbic acid were purchased from Sigma-Aldrich (USA) for the antioxidant assay.
The reporting of this study conforms to ARRIVE 2.0 guidelines. 42 Healthy white mice, weighing about 22-25 grams, regardless of breed, were raised under standard conditions of temperature and light for an acute toxicity assay. Wistar white rats, weighing 200–250 grams, were kept in cages in a cool animal room, with unrestricted access to food and water at the Institute of Biotechnology, VAST, Vietnam for the anti-ulcer assay. Omeprazole, Braun 0.9% physiological saline solution, and formaldehyde were purchased from Sigma-Aldrich (USA) for pathology. This study was approved by the Scientific Council of the Institute of Biotechnology, VAST, Vietnam, on October fourth 2024. All animal procedures were performed in strict accordance with the guidelines of the Scientific Council of the Institute of Biotechnology, VAST, Vietnam, and Vietnamese ethical laws for the care and use of laboratory animals.
Preparation of a Mixture of Curcumin, Berberine and Rotundine (Cur/Ber/Rot Mixture)
Five different mixtures of the three compounds curcumin, berberine, and rotundine were prepared with varying weight ratios, including A1 (Cur/Ber/Rot 50:10:40), A2 (Cur/Ber/Rot 50:20:30), A3 (Cur/Ber/Rot 50:25:25), A4 (Cur/Ber/Rot 50:30:20), and A5 (Cur/Ber/Rot 50:40:10). Weigh three compounds curcumin, berberine, and rotundine in specific ratios to produce 1.0 gram of the mixture, then dissolve them in a minimum amount of ethanol. Evaporate the solvent to obtain a homogeneous mixture. Curcumin extracted from turmeric was chosen as the main component, with a content of 50% in the mixtures, due to two main reasons. Firstly, curcumin is a popular commercial active agent with good anti-inflammatory, wound healing, gastroprotective, antiulcer activities, as well as low cost, and is commonly used in many functional food products to support the treatment of gastritis.29,30 Secondly, Vietnam has a rich source of medicinal herbs, including turmeric, and possesses high-purity curcumin extraction technology. 43 Hence, Vietnam has been proactive in curcumin production. Therefore, using curcumin as the main component in the mixtures enhances the product's applicability in the future. The other two components, berberine and rotundine, accounted for 50% of the mixtures. The two components, berberine and rotundin accounted for 50% of the mixture, with varying ratios of 10:40, 20:30, 25:25, 30:20 and 40:10 in samples A1, A2, A3, A4, and A5, respectively. Five mixtures, A1, A2, A3, A4, and A5, were evaluated for anti-inflammatory and antioxidant activities to determine the optimal mixing ratio.
Nitrite Assay
Initially, RAW264.7 cells were cultured in DMEM medium supplemented with 10% FBS in 96-well plates at a density of 2 × 105 cells per well. The cells were then incubated in a humidified atmosphere with 5% CO2 at 37 °C for 24 h. Subsequently, the medium was replaced with DMEM (FBS-free) and incubated for an additional 3 h. The cells were treated with test samples for 2 h, followed by treatment with 10 µg/mL of LPS for 24 h. To assess the production of nitric oxide, the accumulated nitrite in the culture medium was measured using the Griess reaction. Briefly, 100 µL of the culture medium was mixed with 100 µL of Griess reagent, which consisted of 50 μL of 1% (w/v) sulfanilamide in 5% (v/v) phosphoric acid and 50 μL of 0.1% (w/v) N-1-naphthyl ethylenediamine dihydrochloride. This mixture was then incubated for 10 min at room temperature. The optical density of the mixture after the Griess reaction was measured at 540 nm using an ELISA reader.
44
The sample's ability to inhibit NO production was determined by the formula (1).
Where OD (sample) was the absorbance in wells treated with the test sample, OD (LPS) was the absorbance in wells of LPS-stimulated nitric oxide (NO) production.
The test was repeated three times to ensure accuracy. The IC50 value was determined using TableCurve 2D v4 software. The DMEM (FBS-free) medium was used as a blank, and dexamethasone was used as the positive control with a concentration range of 100, 20, 4, and 0.8 µM. Test samples (A1, A2, A3, A4, and A5) were evaluated with a concentration range of 500, 100, 20, 4, 0.8, and 0.16 µg/mL. The remaining cells in the cultured 96-well plates were subjected to an MTT assay to evaluate cell viability. 45
DPPH Assay
The antioxidant property of Cur/Ber/Rot mixtures at different ratios was evaluated using DPPH free radical scavenging method.
46
Test samples and the positive control, acid ascorbic, were dissolved in DMSO at the concentration ranges of 200, 100, 50, 25, 12.5 µg/mL and 100, 50, 25, 12.5, 6.25 µg/mL, respectively. DPPH was dissolved in ethanol. Mixtures of 10 µL sample solution and 190 µL DPPH solution were placed in 96-well plates and incubated in the dark for 30 min at room temperature. Changes in the absorbance of the samples were measured at 517 nm using a microplate reader (Infinite® 200 PRO, Switzerland). The experiment was repeated three times, and the absorbance value was the average of three replicate tests. The antioxidant capacity of samples was calculated using the following equation:
Where OD sample was the absorbance in wells treated with the test sample, OD negative was the absorbance of the DPPH and DMSO mixture. SC50 value was the sample concentration at which 50% of DPPH was scavenged. The SC50 values were determined using TableCurve AISN Software (Jandel Scientific).
Fabrication of Nanofiber Mats CS/PEG/PLA Loaded with a Cur/Ber/Rot Mixture
CS (0.1 g) was completely dissolved in 5 mL of 90% acetic acid solution with stirring at 50 °C to give CS solution with a concentration of 2% (w/v) (Solution 1). PLA (0.75 g) and PEG (0.05 g) were dissolved in chloroform at 40 °C with stirring to prepare the solution with a concentration of PLA and PEG of 15% and 1% (w/v), respectively (Solution 2). 0.089 g of Cur/Ber/Rot mixture (50:10:40, w/w/w), prepared by the method described above was then added to Solution 2 and stirred at 50 °C for 30 min to obtain Solution 3. Solution 1 was slowly added to Solution 3 with stirring at room temperature to produce a homogeneous solution for electrospinning (Solution 4).
The horizontal electrospinning process was carried out using an electrospinning machine (E02S, Foshan MBRT Nanofiberlabs, China). Solution 4 was loaded into a 5 mL syringe attached to a 22-gauge metal needle that was connected to a high voltage of 20 kV. The feeding rate of the solution was 1 mL/h. The distance from the needle tip to the drum collector was set at 15 cm, and the drum rotated at 500 rpm. The electrospinning process was carried out under controlled conditions of temperature (23-25
Characterization of the Nanofibers
Morphological Analysis
The morphological characteristics of the electrospun nanofibers were observed using a scanning electron microscope (Hitachi, S-4800) operated at a voltage of 5 kV and a current of 10 mA. The magnification was set from x2,000 to x100,000. The nanofiber diameters were measured using ImageJ software. The diameter distribution was plotted using Origin software. The average diameter was then calculated randomly from about 100 nanofibers using MS Excel software. 48
Chemical Analysis
Chemical characterization of the nanofibers was observed using Fourier Transform Infrared (FT-IR) spectrometer (PerkinElmer GX, USA) with a frequency range of 4000 to 600 cm−1. 49
Mechanical Analysis
The tensile strength of the nanofibers was measured using a Zwick Z2.5 material testing machine (Germany). Each sample was cut to dimensions of 15 mm in width and 50 mm in length. The thickness of the sample was determined by averaging three measurements taken at different positions on the sample using a digital micrometer. The test was conducted at a speed of 10 mm/min with a gauge length of 30 mm. Each measurement was repeated three times. 50
Wetting Study
The wetting property of the nanofibers was evaluated using the sessile drop method with an optical contact angle equipment (Dataphysics OCA 50, Germany). A small droplet (5 µL) of deionized water was placed on the surface of the nanofibers. 51 The contact angle was measured using a digital camera at 10 s after the droplet touched the nanofiber surface.52,53 Each measurement was repeated three times.
In Vitro Drug Release Study
The in vitro drug release from the nanofibers was evaluated using a UV-Vis spectrometer (EMC-61PCS-UV). The nanofibers (100 mg) were immersed in 300 mL of PBS buffer solution containing 30% ethanol (pH = 7.4) and kept at 37 °C in a shaking incubator at 100 rpm.54,55 At specific time intervals (1, 2, 3, 4, 8, 12, 16, 24, 48, and 72 h), 3 mL of the supernatant was withdrawn and replaced with an equal volume of fresh buffer, then analyzed for absorbance at wavelengths of 425, 350, and 232 nm, respectively. The calibration curves of curcumin, berberine, and rotundine were obtained by measuring different concentrations of each compound in PBS containing 30% ethanol under the same condition (pH 7.4, 37
Where Mt was calculated from the calibration curve and represents the amount of compound released up to time t, and Mo was the total amount of compound loaded into the nanofibers. Each experiment was performed in triplicate to obtain the average value.
Acute Toxicity Assay
Acute toxicity was conducted in accordance with the Organization for Economic Cooperation and Development (OECD) Guideline No. 420 and the Guidelines for preclinical and clinical testing of oriental medicines and medicinal herbs issued by the Vietnam Ministry of Health under Decision No. 141/QD-K2DT, dated October 27th, 2015.56,57 The nanofibers were ground into powder to be used as the sample for the acute toxicity test.
Preliminary test: ten white mice were completely starved for 16 h before receiving the sample at the highest dose of 10,000 mg/kg body weight. In the case of no mice died within 24 h of observation and monitoring, the official test would be immediately conducted according to the fixed dose procedure.
Official test: 20 white mice were divided into 2 groups (10 mice/group) and completely starved for 16 h before receiving the sample. Group 1 (control group) was treated with distilled water (0.5 mL per mouse). Group 2 was orally administered the sample at a dose of 5000 mg/kg body weight. The treated mice were observed continuously within the first 2 h. Their food and water consumption, body weight, mortality, and responses to light and sound were monitored and recorded for 14 days.
Ethanol-Induced Gastric Ulcer Model
Gastric ulcers in experimental rats were induced by administering ethanol according to the method described by Abdulla et al 58 Omeprazole, a proton pump inhibitor commonly used in the treatment of gastric ulcers, was used as the positive control. 32 Wistar white rats (200-250 g) were divided into 4 groups, each consisting of 8 rats. Group 1 (negative control) was orally administered physiological saline for 10 days (5 mL/kg). Group 2 (positive control) was orally administered omeprazole at a dose of 20 mg/kg for 10 days (5 mL/kg). Group 3 and 4 were orally administered the nanofibers loaded with Cur/Ber/Rot at doses of 250 and 500 mg/kg/day for 10 days (5 mL/kg), respectively. The rats were still fed and watered normally from day 1 to day 8. From day 9 onwards, they were fasted but continued to drink water normally. On day 10, after drinking saline or test sample for 1 h, the rats were given absolute ethanol at a dose of 5 mL/kg to induce gastric ulcers in all groups. One hour after ethanol administration, the rats were euthanized, and their stomachs were immediately removed for examination of gastric factors.
Macroscopic Evaluation
The stomachs were incised along the greater curve then washed with physiological saline to remove gastric contents and blood clots. Macroscopic evaluation of the stomachs was performed using a microscope with 25x magnification. Images of the stomachs were captured and saved to a computer hard drive. The anti-ulcer effect of the samples was evaluated based on three criteria: the number of ulcers or ulcer rate (UR), the area of ulcers, and ulcer scores.
The number of ulcers was counted and the ulcer rate (UR) in each group was calculated using the following formula:
UR = Number of ulcers / Number of rats in the group.
Stomach images were analyzed using ImageJ software (version 1.52a) from the National Institutes of Health (US) to calculate the total ulcer area for each rat.
Ulcer scoring was performed as follows: normal (0), red coloration (0.5), spot ulcer (1), hemorrhagic streak (1.5), deep ulcers (2), and perforation (3). 59
Histopathological Evaluation
After macroscopic evaluation, the stomach tissue was fixed in 10% neutral buffered formalin for 24 h and then immersed in paraffin using an embedding machine for routine paraffin blocking. The stomach sample was cut using a microtome (Sakura, Japan) at a thickness of 5 µm and double-stained with hematoxylin and eosin. Histopathological changes were evaluated using optical microscope according to Ortac et al. 60
Statistical Analysis
The data were processed using Excel and SPSS 20.0 software. Results are presented as mean ± SD (standard deviation). An independent t-test was used for statistical comparison between groups. Differences with P < 0.05 were considered statistically significant.
Results
Inhibition of Nitric Oxide Production
In the current study, mixtures of the three active ingredients curcumin, berberine, and rotundine at different mixing ratios (A1-A5) were screened for anti-inflammatory activity based on their ability to inhibit nitric oxide (NO) production. The results (Table 1) showed that the mixtures exhibited strong NO-inhibitory activity, with IC50 values ranging from 2.80 to 4.51 µg/mL, which were lower than that of the positive control, dexamethasone (IC50 = 14.10 µM). In addition, the NO-inhibitory activity of the three bioactive compounds was also evaluated individually. Curcumin was the most active, with an IC50 value of 3.20 ± 0.19 µg/mL. Rotundin showed moderate activity, with an IC50 value of 16.83 ± 1.06 µg/mL, while the IC50 value of berberine was not determined because the compound affected cell viability during the experiment. Interestingly, the A1 mixture (Cur/Ber/Rot 50:10:40) showed the strongest effect (IC50 = 2.80 ± 0.11 µg/mL), which was significantly better than curcumin alone (IC50 = 3.20 ± 0.19 µg/mL) (P < 0.05). Three mixtures A2 (Cur/Ber/Rot 50:20:30), A3 (Cur/Ber/Rot 50:25:25), and A4 (Cur/Ber/Rot 50:30:20) exhibited notable NO-inhibitory activity but were less potent than curcumin. The IC50 value of the remaining mixture, A5 (Cur/Ber/Rot 50:40:10), which had the highest berberine content (40%), was not determined due to its adverse effect on cell viability during the experiment.
Inhibition of NO Production of the Samples in LPS-Stimulated RAW264.7 Cells.

The results were expressed as mean ± SD of three independent experiments. Values with different superscripts letters (a-e) are significantly different (P < 0.05). NA: not available. Bold text and values represent the mixture with the best screening results.
Antioxidant Activity
Herein, we used the DPPH method to screen the antioxidant activity of mixtures A1, A2, A3, A4, and A5 in order to determine the optimal mixing ratio. Among the three active ingredients (curcumin, berberine, and rotundine), curcumin exhibited the strongest antioxidant activity, with an IC50 value of 81.73 ± 1.28 µg/mL, followed by berberine, while rotundine showed the weakest activity (Table 2). The positive control, ascorbic acid, had an IC50 value of 15.78 ± 0.84 µg/mL. All five samples A1, A2, A3, A4, and A5 showed good antioxidant activity, with IC50 values ranging from 78.55 to 89.39. Notably, mixture A1 exhibited the highest activity, with an IC50 value of 78.55 ± 1.49 µg/mL, which was significantly better than that of curcumin (P < 0.05).
Antioxidant Activity of the Samples Using DPPH Method.

The results were expressed as mean ± SD of three independent experiments. Values with different superscripts letters (a-f) are significantly different (P < 0.05). Bold text and values represent the mixture with the best screening results.
Characterization of the Nanofibers CS/PEG/PLA Loaded with 9% of Cur/Ber/Rot (50:10:40)
The mixture A1 (Cur/Ber/Rot at a ratio of 50:10:40) was selected as the optimal formulation based on its anti-inflammatory and antioxidant activities. The nanofibers CS/PEG/PLA loaded with 9% of this mixture (denoted as CS/PEG/PLA/Cur/Ber/Rot 9%) were fabricated via electrospinning from a homogeneous solution containing three polymers CS, PLA, and PEG and three active ingredients curcumin, berberine, and rotundine. A total loading content of 9% was identified as the highest concentration at which smooth nanofiber formation was still achieved using the electrospinning process.
Figure 1 shows that the surface of the nanofibers CS/PEG/PLA loaded with 9% of Cur/Ber/Rot (50:10:40) was smooth and uniform, without any bead formation. The average diameter of the nanofibers was 261 ± 88 nm.
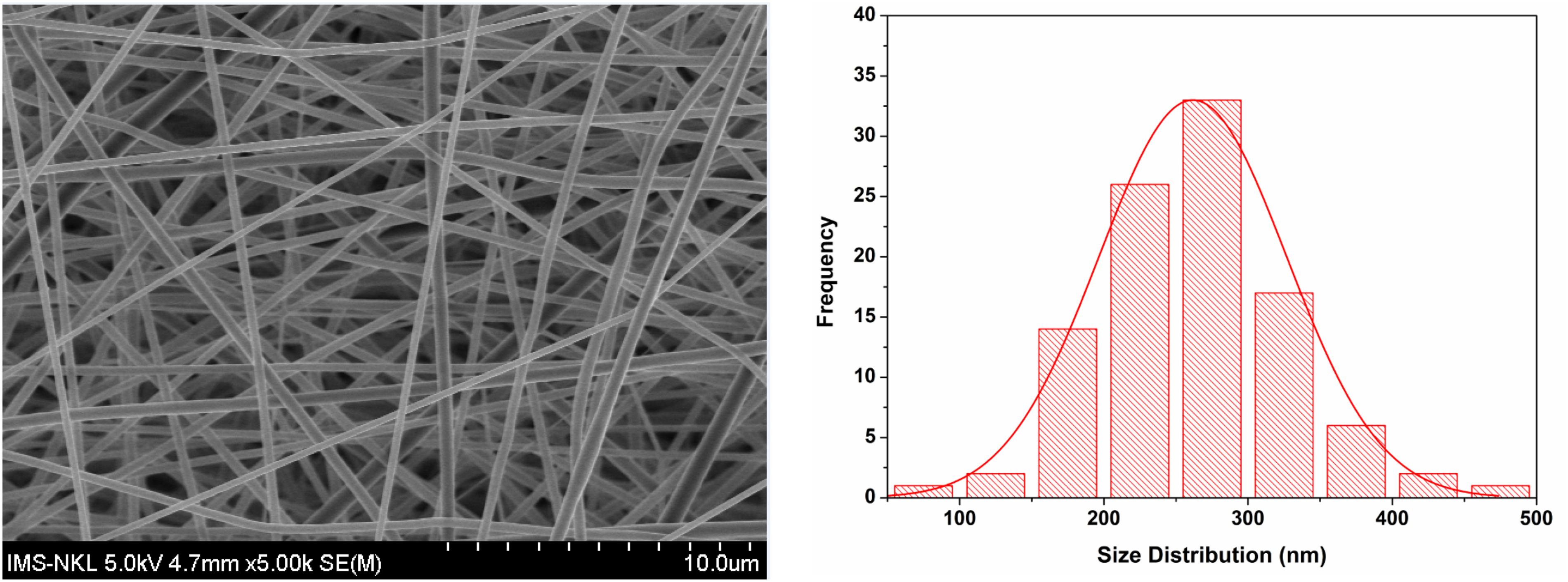
SEM Micrograph and Corresponding Diameter Distribution Curve of the Nanofibers CS/PEG/PLA/Cur/Ber/Rot 9%.
Figure 2 presents the FT-IR spectra of CS, PEG, PLA, berberine, curcumin, rotundine, and the nanofibers CS/PEG/PLA/Cur/Ber/Rot 9%. The FT-IR spectrum of CS revealed a strong broad absorption band around 3361 cm−1, corresponding to N-H and O-H stretching vibrations, a peak at 2869 cm−1 assigned to C-H stretching, and a strong doublet absorption at 1023 and 1066 cm−1 attributed to the pyranose structure. The spectrum of PEG exhibited two sharp peaks at 1089 cm−1 (C-O) and 2878 cm−1 (C-H). The FT-IR spectrum of PLA displayed characteristic stretching frequencies at 1747cm−1 (C = O), 2994 and 2944 cm−1 (C–H), and 1080 cm−1 (C-O). Berberine showed typical absorption peaks at 1503 cm−1 (aromatic C = C), 1270 and 1227 cm−1 (CH3), 1103 and 1033 cm−1 (C-O). The FT-IR spectrum of curcumin revealed major peaks at 1627 and 1599 cm−1 (aromatic C = C), 1503 cm−1 (C = O, aromatic C = C), 1426 cm−1 (CH olefin), 1274 and 1025 cm−1 (C-O). Rotundine exhibited a broad absorption range from 2747 to 3000 cm−1 (C-H), along with three peaks at 1454, 1493 and 1511 cm−1 corresponding to aromatic C = C, and distinct C-O peaks between 1227–1276 and at 1080 cm−1. The FT-IR spectrum of the nanofibers revealed typical peaks at 1752cm−1 (C = O), 1455 cm−1 (aromatic C = C) and 1044, 1085, 1182 cm−1 (C-O). The observed shifts in the characteristic adsorption peaks of functional groups in the nanofibers compared to those of the individual components (CS, PLA, PEG, berberine, curcumin, and rotundine) suggest interactions among the constituents. These shifts may be attributed to formation of hydrogen bonds involving -OH or -NH groups and oxygen or nitrogen atoms from the polymers and active compounds.

FTIR Spectra of CS (A) PEG (B) PLA (C), Berberine (D), Curcumin (E), Rotundine (F), the Nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% (G).
Mechanical analysis of the nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% (thickness: 82.6 ± 4.8 µm) exhibited a tensile strength of 2.31 ± 0.17 MPa, an elongation at break of 1.55 ± 0.61%, and a Young's modulus of 198.49 ± 6.52 MPa, calculated from the stress-strain curve (Figure 3).

The Stress-Strain Curve of the Nanofibers.
The water contact angle was measured to assess the hydrophilicity of the nanofibers. The results showed that the nanofibers were hydrophilic, with a contact angle of 73.9 ± 1.1°.
In Vitro Drug Release Property
The release profiles of curcumin, berberine, and rotundine from the nanofibers in PBS containing 30% ethanol (pH 7.4) at 37 °C are presented in Figure 4. Overall, the release of all three active compounds followed a biphasic pattern: an initial burst release followed by a sustained release. Among the three, rotundine exhibited the highest release rate, followed by curcumin, while berberine showed the lowest. During the burst release phase (within the first 4 h), 61.67% of rotundine, 53.61% of curcumin, and only 44.10% of berberine were released from the nanofibers. This initial rapid release is likely attributed to the presence of the drugs on or near the fiber surface.
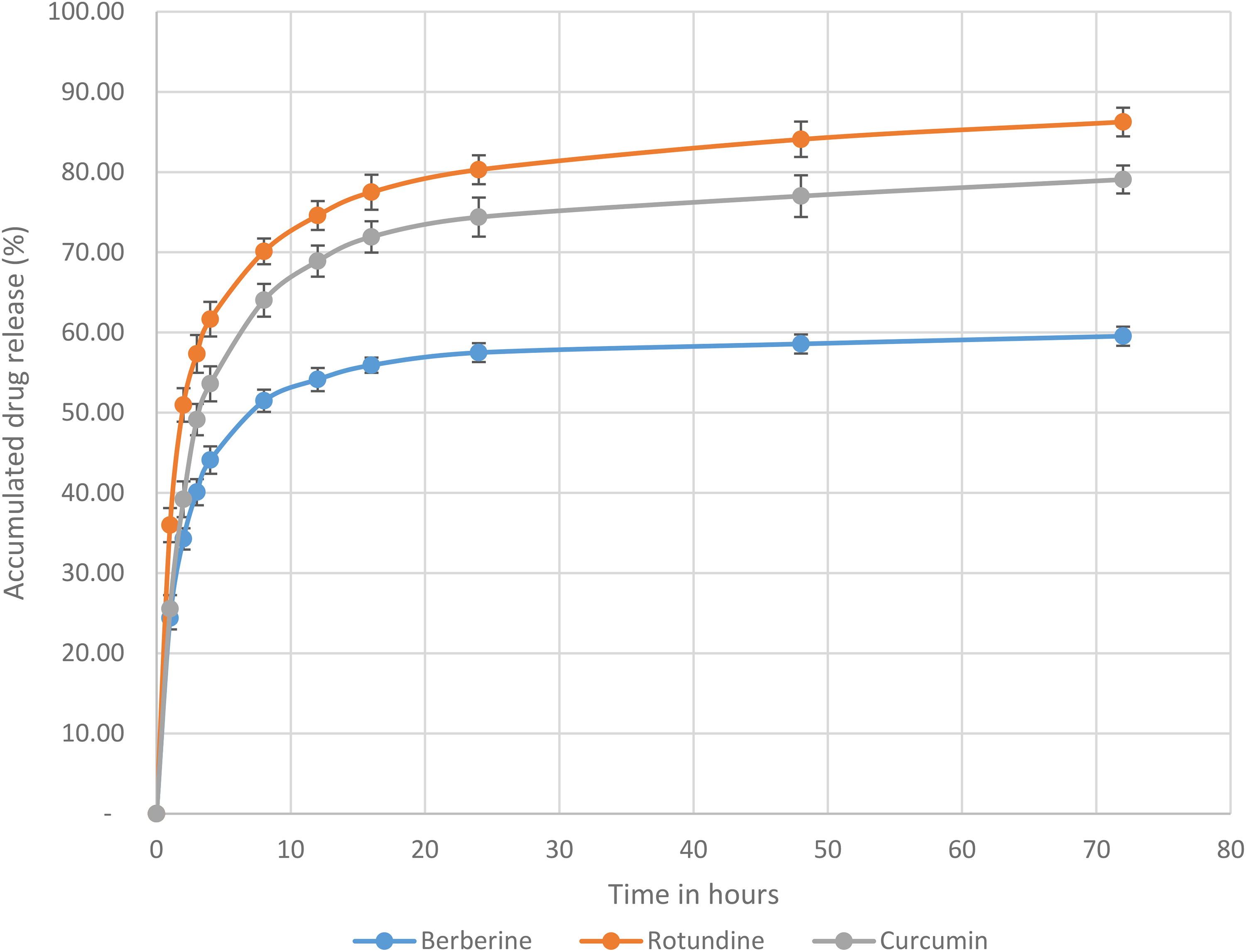
Release Profile of Curcumin, Berberine, and Rotundine from the Nanofibers.
Approximately 20% of rotundin and curcumin and 13% of berberine, were further released over the subsequent 20 h (from 4 to 24 h). By 24 h, around 80% of rotundine, 74% of curcumin, and 57% of berberine had been released from the nanofibers. At the end of the study (72 h), a total of 86% of rotundine, 79% of curcumin, and 59% of berberine had been released. This sustained drug release phase is likely governed by diffusion of the active compounds through the solid nanofibers and partial degradation of the polymer matrix. At all points during the study, the percentages of curcumin, berberine and rotundin released were statistically different (P < 0.05).
Acute Toxicity
In the acute toxicity study, oral administration of the nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% at a dose of 5000 mg/kg body weight caused no mortality or observable signs of toxicity in mice over a 14-day observation period. Treated animals maintained normal behavior, including locomotion, food intake, and appropriate responses to light and sound stimuli. No significant differences in body weight were observed between the control and treated groups (P > 0.05). According to the Globally Harmonized System of Classification and Labeling of Chemicals (GHS) and OECD guidelines for oral toxicity, the nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% at a dose of 5000 mg/kg body weight are classified as non-toxic and considered safe, as they did not induce any signs of acute oral toxicity in mice.
Anti-Gastric Ulcer Effect
The nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% were evaluated for their gastro-protective effects in an ethanol-induced gastric ulcer model. The results showed that at the tested doses of 250 and 500 mg/kg/day, the nanofibers did not adversely affect the general health status of the experimental rats. No mortality was observed, and all animals remained healthy, exhibited normal feeding and locomotor activity, and responded appropriately to external stimuli.
The anti-ulcer effects of the samples were evaluated based on both macroscopic and histopathological analyses of rat stomach tissues. In the macroscopic evaluation, three criteria including number of ulcers or ulcer rate (UR), ulcer area, and ulcer score were used to determine the severity of gastric lesions in rats. The results (Table 3) showed that in the negative control group (n = 8), 100% of rats developed gastric ulcers, with an average number of ulcers of 4.33 ± 1.15. The ulcer rate in this group was 0.54 ± 0.14. In the group treated with the nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% at a dose of 250 mg/kg/day, the total number of ulcers significantly decreased to 1.67 ± 0.58 compared to the negative control group. The ulcer rate in the group was 0.21 ± 0.07 (P < 0.05). Interestingly, in the group receiving the nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% at a dose of 500 mg/kg/day, no gastric ulcers were detected. These results indicate that the nanofibers at a dose of 500 mg/kg/day were as effective in preventing gastric ulcers in rats as the standard drug, omeprazole, at a dose of 20 mg/kg/day.
The Anti-Gastric Ulcer Effects of the Nanofibers in Rats.

The results are expressed as mean ± SD (n = 8 per group). *P < 0.05, ** P < 0.01 indicate statistically significant differences compared to the negative control group.
The results of gastric ulcer area measurement in experimental rats showed that the negative control group had the largest ulcer area, at 0.307 ± 0.055 cm2. In contrast, the group treated with the nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% at a dose of 250 mg/kg/day showed a significantly smaller ulcer area of 0.072 ± 0.013 cm2 compared to the negative control group (P < 0.05). Notably, no ulcers were observed in either the omeprazole-treated group (20 mg/kg/day) or the group treated with nanofibers at 500 mg/kg/day. These findings indicate that the nanofibers CS/PEG/PLA/Cur/Ber/Rot 9%, administered at both 250 and 500 mg/kg/day for 10 days was effective in reducing the ulcer areas in experimental rats.
The severity of gastric damage in experimental rats was evaluated based on ulcer scores. In the negative control group, most ulcers were classified as hemorrhagic streaks and deep ulcers, with an average ulcer score of 1.68 ± 0.55. In the group treated with the nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% at a dose of 250 mg/kg/day, the gastric lesions were primarily spot ulcers, with an ulcer score of 1.00 ± 0.41. In contrast, the majority of rats treated with the standard drug, omeprazole (20 mg/kg/day) and those treated with the nanofibers at a dose of 500 mg/kg/day exhibited only mild gastric lesions, classified as red coloration, with ulcer scores of 0.50 ± 0.26, 0.61 ± 0.35 respectively. The macroscopic images of stomachs from different groups are shown in Figure 5 (A-D). After evaluating the anti-ulcer effects of the nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% based on three criteria including number of ulcers, ulcer area, and ulcer score, the results showed that the nanofibers effectively prevented ulcer formation, reduced the number and area of gastric ulcers, and alleviated the severity of gastric lesions in rats with ethanol-induced gastric ulcers. Notably, at a dose of 500 mg/kg/day, the nanofibers exhibited anti-ulcer efficacy comparable to that of omeprazole administered at 20 mg/kg/day.
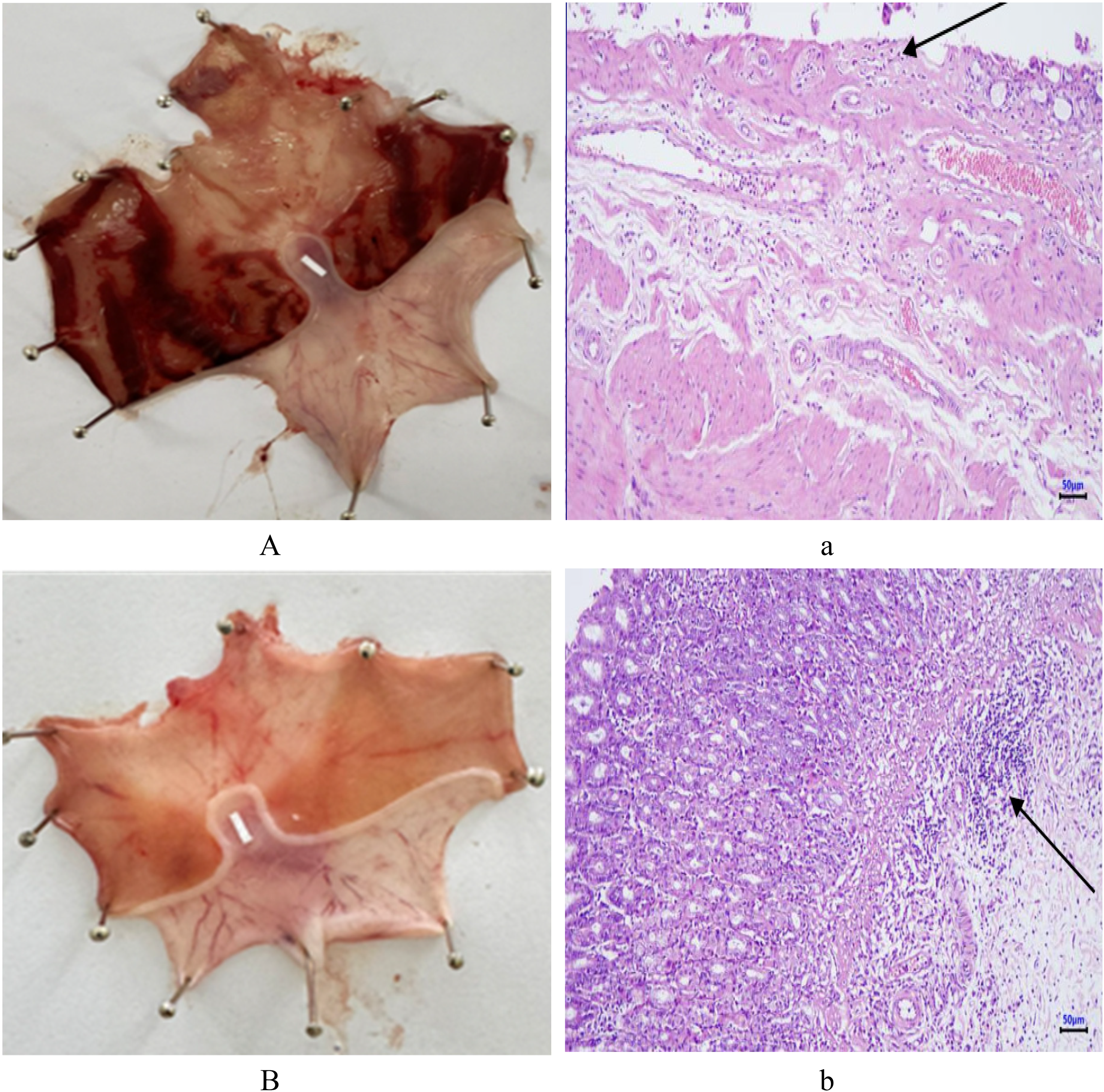
Macroscopic Images of Stomachs from: (A) Negative Control Group, (B) Group Treated with Omeprazole (20 mg/kg/day), (C) Group Treated with the Nanofibers (250 mg/kg/day), (D) Group Treated with the Nanofibers (500 mg/kg/day). Histological Appearance of Gastric mucosa from: (a) Negative Control Group, (b) Group Treated with Omeprazole (20 mg/kg/day), (c) Group Treated with the Nanofibers (250 mg/kg/day), (d) Group Treated with the Nanofibers (500 mg/kg/day).
In addition, histopathological analysis (Figure 5, a-d) also showed significant differences between the negative control group and the treatment groups. Histological sections of the stomach from the negative control group showed severe ulceration, characterized by peeling mucosa, ulcer bases extending deep into the submucosal layer, stromal infiltration with mononuclear inflammatory cells, and marked congestion of blood capillaries. In the experimental groups treated with the nanofibers at doses of 250 and 500 mg/kg/day, the histopathological examination revealed mild erosive gastritis. Ulceration and vascular congestion were markedly improved in both groups. There was no significant difference in the histopathological profiles between the two treatment groups. In the positive control group, mild inflammatory lesions and slight degeneration of the mucosal layer were observed. Thus, pretreatment with nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% at doses of 250 and 500 mg/kg/day for 10 days was effective in reducing the severity of gastric ulcers in rats with ethanol-induced gastritis.
In summary, evaluation of the anti-ulcer effects of the nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% based on three criteria including number of ulcers, ulcer area, and ulcer score, demonstrated that the nanofibers effectively prevented ulcer formation, reduced the number and area of gastric ulcers, and alleviated ulcer severity in rats with ethanol-induced gastric ulcers. The nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% at a dose of 500 mg/kg/day exhibited anti-gastric ulcer effects comparable to those of omeprazole at a dose of 20 mg/kg/day. It is noteworthy that the dose of nanofibers required to achieve similar therapeutic efficacy was substantially higher than that of omeprazole. The discrepancy may be attributed to differences in the mechanisms of gastroprotective effect between the curcumin, berberine and rotundin mixture and omeprazole. Another possible explanation is the relatively low drug loading in the nanofibers (9%). Additionally, since the drug-loaded nanofibers were designed for sustained or controlled release, a higher initial dose may be necessary to maintain effective therapeutic concentrations over an extended period.
Discussion
Nitric oxide (NO) is a key signaling molecule in the immune system. It is produced by various cell types, particularly endothelial cells, macrophages, and neutrophils, in response to inflammatory stimuli. Under normal physiological conditions, NO exerts anti-inflammatory effects. However, during inflammation, excessive NO production can contribute to tissue damage and exacerbate the inflammatory response. Therefore, inhibition of NO production is considered an important therapeutic strategy for managing inflammatory diseases, including gastritis.61,62 Synergy occurs when two or more drugs or compounds are combined to produce a total effect greater than the sum of their individual effects. 63 Several previous studies have reported that curcumin, when combined with other agents such as piperine, 64 tetramethylpyrazine, resveratrol, 65 docosahexaenoic acid, and eicosapentaenoic acid, 66 exert synergistic anti-inflammatory effects. It is likely that the synergistic interaction among curcumin, berberine, and rotundine in mixture A1 contributed to its greater inhibitory effect on NO production compared to each component alone.
Oxidative stress refers to an imbalance between the production of reactive oxygen species (ROS) and the body's ability to neutralize or eliminate their harmful effects through antioxidant defenses. ROS are highly reactive molecules that can damage cellular components, including DNA, proteins, and lipids. Such damage can contribute to a wide range of health problems, including inflammation, aging, and the development of various diseases such as cancer, cardiovascular diseases, neurodegenerative disorders, and metabolic syndrome.67,68 Oxidative stress is a significant contributing factor to the pathogenesis and progression of gastric ulcers.69,70 Antioxidants are molecules that can delay or inhibit the oxidation of various substrates by preventing the initiation or propagation of oxidative chain reaction. 71 The gastroprotective effects of antioxidants, mediated through their antioxidant mechanisms, have been demonstrated.72,73 DPPH is a stable free radical with a maximum absorption wavelength of 515–517 nm and a characteristic purple color. Antioxidants neutralize DPPH by donating hydrogen atoms, resulting in a color change of the reaction solution from purple to yellow. This change is quantified by measuring the absorbance of the solution at 515–517 nm. The DPPH method is a rapid, simple, accurate and inexpensive assay for measuring the antioxidant activity of different compounds or extracts. 74 The results suggested that mixture A1, with a Cur/Ber/Rot ratio of 50:10:40, may exert a synergistic antioxidant effect. The combination of curcumin with other compounds such as piperine, resveratrol, and gallic acid has previously been reported to exert synergistic antioxidant effects.64,75,76 Indeed, the synergistic antioxidant and anti-inflammatory activities of mixture A1 were further supported by a combination index (CI) value of less than 1, as calculated using the Chou–Talalay method.77,78
Based on screening results for anti-inflammatory and antioxidant activities, mixture A1 with a Cur/Ber/Rot ratio of 50:10:40 was selected for the fabrication of nanofibers loaded with the active compounds. As mentioned in the Introduction, the polymers CS, PEG, and PLA are all biocompatible, biodegradable, non-toxic, and commonly used as drug carriers. Among them, PLA, with its good mechanical strength and excellent fiber-forming ability, was used as the primary polymer. CS, known for its antibacterial and anti-inflammatory properties, was incorporated to enhance the biological activity of the obtained nanofibers. The presence of CS has previously been reported to reduce fiber diameter and increase the specific surface area. PEG, a hydrophilic polymer, was used to improve the interaction between CS and PLA, as well as to enhance the hydrophilicity of the nanofibers, thereby promoting drug release in biological environments. The electrospun nanofibers CS/PEG/PLA loaded with 9% of mixture A1 (CS/PEG/PLA/Cur/Ber/Rot 9%) were prepared and investigated for their morphological, chemical, mechanical, and wettability properties, as well as drug release behavior and anti-ulcer effects.
Tensile strength is generally believed to be proportional to the thickness of the material. 79 The relatively low tensile strength of the obtained nanofibers may be attributed to the presence of chitosan, a polymer known for its low mechanical strength. 80 Therefore, improving the mechanical properties of the nanofibers may be possible by increasing their thickness during fabrication.
The nanofibers were found to be hydrophilic with a contact angle of 73.9 ± 1.1°. Wettability is a fundamental property that significantly impacts drug release mechanisms, dissolution rate, and overall therapeutic efficacy. Thus, understanding and controlling wettability is essential for the development of effective pharmaceutical formulations.81,82
Curcumin is a poorly water-soluble active ingredient, both in distilled water and in phosphate-buffered saline (PBS). Therefore, the in vitro drug release study of the nanofibers was conducted in PBS containing 30% ethanol (pH = 7.4) at 37 °C, which mimics the pH of the terminal ileum and plasma, as well as the physiological body temperature, in accordance with previous studies.54,55 There are three primary mechanisms by which drugs can be released from nanofibers: (1) desorption of the drug from the fiber surface, (2) diffusion of the drug through the solid polymer matrix, and (3) degradation of the fibers in vivo. 83 Incomplete drug release may result from the active ingredient being entrapped within the polymer matrix, whereas complete drug release can be achieved through the gradual biodegradation of the nanofiber. 83 The in vitro drug release profiles of curcumin, berberine, and rotundine from the CS/PEG/PLA nanofibers exhibited a biphasic pattern, characterized by an initial burst release within the first 4 h, followed by a sustained release lasting up to 72 h. This release behavior is typical of electrospun drug-loaded nanofibers, as previously reported.10,13,54,55 Notably, statistically significant differences were observed in the release rates among the three active ingredients. By 72 h, rotundine demonstrated the highest cumulative release (86%), followed by curcumin (79%), while berberine showed the lowest release (59%). Several possible mechanisms may account for these differences in release profiles. First, differences in chemical structure may result in varying interactions between the active ingredients and the polymer matrix, which can influence their release behavior. 84 Second, different concentrations of the active ingredients loaded into the fibers may also contribute to the variation. It is well established that higher drug loading leads to a greater concentration gradient between the fiber and the release medium, thereby promoting increased diffusion and enhancing the release rate of the active compound. This is consistent with the previous report by Shababdoust et al, in which 75% to 81% of curcumin was released from nanofibers loaded with 5% curcumin, and 79% to 86% was released from those loaded with 10% curcumin over 432 h. 55 Third, the solubility of the active ingredients in the release medium also plays a crucial role.85,86 Although the curcumin content loaded in the nanofibers higher than that of rotundine, its poor solubility in PBS buffer resulted in a lower release rate compared to rotundine.
Ethanol-induced gastric ulcer is a widely used experimental for the preclinical study of agents with potential anti-ulcer activity, as it mimics several features of acute human peptic ulceration. It is characterized by pathophysiological changes, such as hemorrhage, edema, inflammatory infiltration and epithelial cell loss. 60 To the best of our knowledge, this is the first study to report the anti-ulcer effect of electrospun nanofibers loaded with a combination of three active ingredients: curcumin, berberine, and rotundine. While the anti-ulcer properties of curcumin and berberine have been previously documented, the inclusion of rotundine and its synergistic combination with the other two agents in nanofibers has not been reported before.
Oxidative stress and apoptosis have been implicated in the pathogenesis of gastric ulcers. Moreover, there is a clear correlation among pro-inflammatory mediators, oxidative stress, and apoptosis in the development of gastric ulcers. Increasing evidence suggests that agents with antioxidant and anti-inflammatory properties can exert anti-gastric ulcer effects. 87 The following mechanisms may contribute to the anti-ulcer activity of the nanofibers CS/PEG/PLA/Cur/Ber/Rot 9%. Curcumin promotes the prevention and healing of gastric ulcers by inducing angiogenesis in the granulation tissue of the ulcers. Curcumin exerts its anti-ulcer effects not only by modulating oxidative stress and enhancing total antioxidant capacity, but also by inhibiting IL-6 secretion and preventing apoptosis.88–90 Berberine has been reported to significantly improve the pathological morphology of gastric ulcers, increase the macroscopic healing score and healing rate, reduce serum levels of NO, IL-6, and PGE2, and elevate serum IL-10 levels, thereby effectively alleviating gastric ulcer severity in rats. 91 Rotundine, with its potential anti-inflammatory and analgesic effects,36,37 may also contribute to the anti-ulcer activity of the nanofibers loaded with the triple active mixture. Preliminary results evaluating the gastroprotective and anti-ulcer effects of nanofibers loaded with Cur/Ber/Rot mixture have shown their potential application in the prevention and treatment of gastric ulcers. Further studies will be conducted to elucidate the underlying mechanisms of the nanofibers’ anti-ulcer activity in the ethanol-induced gastric ulcer rat model.
The acute toxicity study demonstrated the safety of CS/PEG/PLA/Cur/Ber/Rot 9% nanofibers in animals. At a dose of 5000 mg/kg body weight, no mortality or observable signs of toxicity were detected in mice during the 14-day observation period. This dose can serve as a reference for dose calculation in future pharmacological studies. Converting animal doses to human equivalent doses (HED) is a crucial step to ensure safety and efficacy in both preclinical and clinical research. The HED can be calculated based on the animal dose and the correction factor (Km) as follows:
92

In this case, the animal dose is 5000 mg/kg, the Km factor for mice is 3, and the Km factor for humans is 37. Therefore, the calculated human equivalent dose (HED) is approximately 405 mg/kg.
In summary, the results obtained in the current study are promising. However, it should be acknowledged that the study has certain limitations. Although electrospinning is a technique with many advantages, its large-scale application with consistent stability remains a challenge. Moreover, the use of some organic solvents in the preparation of electrospun nanofibers poses a potential risk of residual solvents affecting human health. In this study, the drug release profile of the nanofibers was evaluated in PBS buffer containing 30% ethanol (pH = 7.4), which does not fully replicate the physiological conditions of the human digestive tract. Therefore, the findings of this paper provide preliminary evidence for the potential application of the nanofibers CS/PEG/PLA/Cur/Ber/Rot 9% in the treatment of gastric ulcers. Further investigations into the mechanism of action and safety profile of the nanofibers are needed to provide sufficient scientific data for their clinical application in gastric ulcer treatment.
Conclusion
In the present study, a mixture of three active ingredients curcumin, berberine, and rotundine was prepared at a ratio of 50:10:40, respectively. This formulation exhibited synergistic anti-inflammatory and antioxidant effects. The electrospun nanofibers composed of CS, PEG, and PLA loaded with 9% of the active mixture were successfully fabricated. The nanofibers were investigated for their morphological, chemical, mechanical, and wettability properties, as well as their drug release property and anti-ulcer effect. The nanofibers (CS/PEG/PLA/Cur/Ber/Rot 9%) at doses of 250 and 500 mg/kg/day revealed significant anti-gastric ulcer effects. These findings suggested the nanofibers may serve as a promising candidate for the treatment of gastric disorders.
Footnotes
Acknowledgements
Authors would like to thank Vietnam Ministry of Science and Technology for providing funding to carry out this research.
Ethical Approval
This study was approved by the Scientific Council of Institute of Biotechnology, VAST, Vietnam. All animal procedures were performed in strict accordance with the guideline of the Scientific Council of Institute of Biotechnology, VAST, Vietnam and Vietnamese ethical laws for the care and use of laboratory animals.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was performed under the financial support of Vietnam-Czech Republic joint research project (Grant No. NĐT/CZ/23/07).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.