Abstract
Background
Medicinal plants have long been recognized for their importance in treating serious illnesses. In recent years, the incorporation of metallic nanoparticles into plant-based therapies has emerged as a promising strategy for combating malaria.
Objective
This study investigated the therapeutic efficacy of Indigofera oblongifolia extract combined with silver nanoparticles (IOLE AgNPs), along with chloroquine (CQ, 10 mg/kg), in mitigating lung inflammation caused by Plasmodium chabaudi infection in a murine model.
Materials and Methods
A total of fifty female C57BL/6 mice were randomly assigned to five groups: a healthy control group, an IOLE AgNPs-only group, P. chabaudi-infected group, an infected group treated with IOLE AgNPs, and an infected group receiving both IOLE AgNPs and chloroquine. Lung tissues were examined through hematoxylin-eosin staining and morphometric evaluation.
Results
Treatment with IOLE AgNPs led to a marked improvement in lung tissue architecture among infected mice. The intervention significantly reduced alveolar damage, septal thickening, edema, and necrotic changes in comparison to untreated infected lungs. Despite these improvements, some signs of epithelial hyperplasia and fibrosis persisted. In comparison, treatment with chloroquine at a dose of 10 mg/kg resulted in moderate restoration of lung tissue structure. This group showed partial alleviation of bronchiolar and alveolar wall thickening, along with better airway expansion. Nonetheless, signs of tissue damage such as septal and alveolar congestion, accumulation of hemosiderin, fluid buildup (edema), and cellular necrosis were still evident. Additionally, the thickness of the interstitial tissue remained greater than that observed in the group treated with IOLE AgNPs.
Conclusions
Both IOLE AgNPs and chloroquine (10 mg/kg) demonstrated potential in alleviating pulmonary inflammation, though IOLE AgNPs appeared more effective in reducing structural damage.
Introduction
The lungs play a central role in pulmonary circulation, primarily due to their unique position in facilitating gas exchange and converting oxygen-poor blood into oxygen-rich blood. 1 Structurally, the lung's respiratory units consist of bronchioles, alveolar ducts, and alveolar sacs, terminating in the alveoli, which serve as the primary sites of respiration. 2 These alveolar structures perform the essential function of gas exchange, establishing them as the core functional components of the lungs. 3 Both the conducting and respiratory portions of the respiratory system collaborate to support not only gas exchange but also air filtration and conditioning. 4
Malaria has long been recognized as one of the most widespread infectious diseases globally, primarily caused by various Plasmodium species. 5 As resistance to standard antimalarial drugs has become increasingly problematic, contemporary research has turned to medicinal plants and their integration with metallic nanoparticles as a novel therapeutic approach. 6
Malaria remains one of the most devastating infectious diseases globally, Plasmodium infections causing an estimated 247 million cases and over 600,000 deaths annually. 7 While cerebral malaria and severe anemia dominate clinical concerns, pulmonary complications including acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) are increasingly recognized as critical determinants of mortality. 8 These conditions constitute a cascade of inflammation caused by parasite sequestration and endothelial cell activation, leading to alveolar damage and impaired CO2 and O2 exchange. 9 Despite advances in chloroquine therapies, drug resistance has emerged as a dire threat, underscoring the urgent need for novel therapeutic agents.
Historically, medicinal herbs have played a vital role in healthcare and in managing serious illnesses. A substantial portion of pharmaceutical drugs originates from plant-derived compounds. 10 These natural products are known for their broad therapeutic properties, and recent advances have identified new bioactive compounds that have significantly enhanced medicinal efficacy. 11
Indigofera oblongifolia, commonly known as Indigofera, is a species in the Fabaceae family that has been traditionally used for its healing properties. This plant commonly grows in disturbed areas such as roadsides, abandoned agricultural land, and sandy or gravelly plains. 12 It is widely recognized as a perennial shrub adapted to such environments. 12 Various parts of I. oblongifolia have historically been used to treat conditions like hepatomegaly and splenomegaly, largely due to their anti-inflammatory effects. 13
Recent investigations have provided extensive evidence supporting the pharmacological potential of I. oblongifolia. In experimental models of P. chabaudi infection, extracts from the plant have shown promising results in reducing inflammation and enhancing antioxidant activity. 14 Specifically, administration of leaf extracts has been found to lower leukocyte infiltration and reduce the number of apoptotic cells in the liver, suggesting a protective role in malaria-associated inflammation. 15 Histomorphometric evaluation is essential for examining tissue-level changes resulting from infection and inflammation. Through these analyses, researchers can gain deeper insights into how I. oblongifolia confers protection to lung tissues compromised by P. chabaudi. This study investigates the plant's impact on lung structure, immune cell infiltration, and overall tissue preservation, offering a valuable understanding of its therapeutic capacity.
Materials and Methods
This study adhered to the ARRIVE 2.0 guidelines, ensuring the proper design, conduct, and reporting of animal-based experiments. 16 All procedures involving animals received approval from the Animal Ethics Committee, part of the Institutional Animal Care and Use Committee at the Department of Zoology, College of Science, King Saud University, Riyadh, Saudi Arabia. The experiments were carried out in both national and institutional standards for the ethical treatment of animals in research.
Preparation of Indigofera oblongifolia Leaf Extract
The preparation of the leaf extract followed a previously described protocol. 17 I. oblongifolia leaves were collected from the Jazan region in Saudi Arabia. A 70% methanolic extract was prepared by first drying and pulverizing the leaves. The powdered material was immersed in methanol for 24 h at 4 °C with continuous stirring. The mixture was then filtered using Whatman filter paper, and the resulting extract was concentrated using a Yamato RE300 rotary vacuum evaporator (Tokyo, Japan).
Synthesis and Characterization of Silver Nanoparticles (AgNPs)
The biosynthesis of silver nanoparticles was performed as outlined earlier. 18 To synthesize AgNPs, the IOLE was mixed with silver nitrate in a methanol solution at a 1:9 ratio. This mixture was heated at 50 °C for one hour until the color changed to dark brown, indicating nanoparticle formation. The presence and properties of AgNPs were confirmed through UV-Visible spectroscopy. Further analysis of particle size and morphology was conducted using transmission electron microscopy (TEM) with a JEOL JEM-2100 (JEOL Ltd, Tokyo, Japan).
Experimental Design and the Inoculum Preparation
A total of 50 female C57BL/6 mice, aged 9 ± 2 weeks and weighing approximately 22 ± 3 grams, were generously provided by the Research Unit at King Faisal Hospital in Riyadh, Saudi Arabia. The P. chabaudi strain used in this study was obtained from the Parasitology Laboratory, College of Science, King Saud University. P. chabaudi cryopreserved blood stages have been passed through C57Bl/6 mice and injected intraperitoneally (i.p.) into experimental mice with 100 µL of phosphate-buffered saline (PBS) containing 105 infected red blood cells. For the calculation of 105 parasitized erythrocytes, the Neubauer chamber was used. Giemsa-stained smears from the blood of the mouse tail were prepared to ensure the parasitemia.
In Vivo Antimalarial Evaluation
Animals were divided into five groups. Each group consisted of 10 mice. For 7 days, we administered PBS orally to the first control non-infected group. We administered 50 mg/kg of IOLE AgNPs orally to the second group every day for 7 days. We injected 105 parasitized erythrocytes of P. chabaudi intraperitoneally into the third (Not processed for comparison), fourth, and fifth groups. One hour later, the fourth group received 50 mg/kg of IOLE AgNPs daily for 7 days, while the fifth group orally received 10 mg/kg chloroquine phosphate (Sigma-Aldrich, St. Louis, MO) daily for four days. 19
Histological Assessment
Collection of Samples
On the seventh day following the Infection, all mice were euthanized by CO2 asphyxiation and then dissected to collect lung tissues. The lung was dissected and sectioned for pathological examination. The tiny fragments were immersed in a neutral buffered formalin solution (10%) for histological analysis.
Lung Histopathology
Lung tissue samples collected from both control and experimental groups were fixed in 10% neutral buffered formalin, followed by paraffin embedding to preserve the specimens. 20 Sections were cut at a thickness of 6 μm and stained with hematoxylin and eosin (H&E) for microscopic examination. Morphological parameters, including alveolar wall thickness, necrotic intensity, and structural irregularities, were assessed using ImageJ software. These evaluations aimed to identify histoarchitectural disruptions resulting from P. chabaudi infection.
Data were collected from five experimental groups to evaluate the therapeutic role of IOLE AgNPs post-infection. Statistical methods were employed to calculate the mean (M) and standard error (S.E.) for each measured parameter.21,22 The data analysis was conducted using SPSS software to ensure accurate comparisons across the four main study groups. Histological slides were scored following previously established protocols.23,24
To semi-quantitatively assess tissue alterations, pathological features were graded on a scale from 0 to 3, where 0 indicated normal architecture and 3 indicated severe damage, based on predefined criteria outlined in Table 1. A cumulative score was then generated by summing the values for five distinct parameters, yielding a maximum score of 10.
The Scoring System Used to Calculate the Histological Score in Lung Tissues.

Statistical Analysis
Data analysis was performed using SPSS software version 20 and GraphPad Prism version 5. One-way analysis of variance (ANOVA) followed by appropriate post hoc tests was employed to evaluate statistical differences among the experimental groups. The Bonferroni range test was specifically applied to assess pairwise comparisons between the mean values of the five groups. Results were presented as the mean ± standard error of the mean (SEM). A p-value of less than 0.05 was considered indicative of a statistically significant difference between groups.
Results
This study investigated the therapeutic potential of IOLE-conjugated silver nanoparticles (IOLE AgNPs) in mitigating lung inflammation caused by P. chabaudi, a malaria-inducing parasite. Morphometric and histopathological assessments were conducted across five experimental groups: healthy control lungs, lungs treated with IOLE AgNPs alone, lungs infected with P. chabaudi, infected lungs subsequently treated with IOLE AgNPs, and infected lungs treated with chloroquine (CQ) at a dose of 10 mg/kg. The findings demonstrated notable restoration in lung tissue structure, particularly in the group treated with IOLE AgNPs post-infection, highlighting its promising pharmacological effect in reducing malaria-induced pulmonary damage.
Histomorphometric Observations of the Control Lung
The structure of a healthy lung includes two primary bronchi that divide into secondary and tertiary branches, eventually forming bronchioles. These bronchioles typically maintain a diameter of 581.68 ± 6.22 µm2 and a wall thickness of approximately 19.60 ± 0.27 µm, which differentiates them from larger bronchial passages (Figures 1: C & D). Histological examination revealed that the bronchiolar walls were lined with stratified columnar epithelium interspersed with goblet cells, and encased externally by smooth muscle fibers (Figures 2: A & B).

Bar Charts Showing the Morphometric Analysis of Histopathological Parameters of Lung Tissue Which A: Alveolar Expansion, B: Alveolar Wall Thickness, C: Bronchiolar Dimensions, D: Bronchiolar Wall Thickness, E: Hemosiderin Intensity, F: Diameter of the Interstitium, G: Alveolar Congestion, H: Septal Congestion, I: Intensity of Necrosis, and J: Edema among Five Groups: Control; IOLE AgNPs Group; Infected Group by P. chabaudi, Infected Group by P. chabaudi and 50 mg/kg of IOLE AgNPs and the Administrated Group by P.chabaudi and CQ 10 mg/kg. Values are Represented as Mean ± STDV & n = 10 Animals. Means Within the Same Parameter and not Sharing a Common Superscript Symbol(s) Differ Significantly at p < 0.05, and Values That are Recorded with a non-Significant Difference (n.s).

Photomicrograph of Histological Sections of Lung Tissue of the Control Group (A& B) Showing Dispersion of Bronchiolar Airway Passage (Br) with its Thickened Wall (Brw) Encircled by a Layer of Smooth Muscle Fiber (Sm). Its Wall was Constituted of Stratified Columnar Epithelium (Stc) Pervaded by Goblet Cells (Gc). Numerous Alveoli (Av) were Separated from Each Other by Alveoli Septum (Av sp) and were Composed of Two Types of Cells: Pneumocystis Type I (TI) and a Different Type, Pneumocystis Type II (TII). The Interstitium (int) Regions were Infiltrated by Various Endothelial Cells (en). Aggregated Alveoli were Thickened (Th Av) from the Alveolar sac (Av sc). The Blood Vessels (Bv) Spread Over the Tissue. (H&E X, A: 100 µm & B: 400 µm).
As the bronchioles taper off, they lead into alveolar ducts, which in turn open into the pulmonary alveoli. The current study recorded standard measurements for alveolar expansion and wall thickness, reported at 200.47 ± 2.54 µm2 and 10.82 ± 0.46 µm, respectively (Figures 1: A & B). Microscopic observations showed that the alveoli were cup-shaped and lined by a delicate layer of two epithelial cell types. The dominant type was squamous epithelial cells, which formed a thin barrier essential for efficient gas exchange between the alveolar air and blood. The second, less common type consisted of cuboidal cells scattered among the squamous cells (Figures 2: A & B).
Additionally, normal spreading of interstitial tissue among the alveolar sacs and alveoli was observed, along with a regular distribution of blood vessels, measuring 128.40 ± 2.65 µm2 in density. This connective tissue network was composed primarily of capillary endothelial cells. Owing to the preservation of normal histological architecture in this group, the histopathological score was low, indicating minimal tissue alteration, with a recorded intensity of 2.23 ± 0.21%, significantly lower than other experimental groups (Figures 3).

the bar Graph Shows the Scoring of the Histopathological Alterations. * p < 0.05 **, p < 0.01, and ***p < 0.001,
Histomorphometric Observations of the I. oblgofola AgNPs Lung
Microscopic analysis of lung tissue treated with IOLE AgNPs demonstrated a marked improvement (p < 0.001) in overall histological structure, notably with an increased diameter in both the bronchioles (701.70 ± 5.08 µm2) and alveolar ducts (214.62 ± 1.33 µm2) compared to the control group (Figures 2: A & C). The bronchiolar walls retained their typical architecture, featuring stratified columnar epithelium interspersed with goblet cells, and an outer layer of smooth muscle fibers. Likewise, the alveolar walls maintained the normal composition of the two pneumocyte types, while the alveolar sacs appeared as open spaces containing multiple alveoli (Figures 2: C & D).
However, certain necrotic changes were significantly more pronounced (p < 0.001) in the IOLE AgNP-treated lungs than in controls. These included thinning of the alveolar and bronchiolar walls, measuring 7.81 ± 0.58 µm and 15.78 ± 0.50 µm, respectively. Additionally, increased infiltration of immune cells within the interstitial tissue suggested an enhanced inflammatory response, leading to a thickening of this tissue to 219.06 ± 1.63 µm2 compared to controls. Edema intensity also rose to 2.84 ± 0.44%, accompanied by epithelial cell necrosis (1.92 ± 0.02%) and a wider distribution of hemosiderin granules (Figures 2: B, D, E, F, 1 & J). Consequently, histopathological scoring indicated a significantly higher value (13.18 ± 2.77%) relative to control lungs (Table 2 & Figures 3).
Shows the Morphometric Analysis of Histopathological Parameters of Lung Tissue, Which Includes Alveolar Expansion, Alveolar Wall Thickness, Bronchiolar Dimensions, Bronchiolar Wall Thickness, and Diameter of the Interstitium among Five Groups: Control; Iole Agnps Group; Infected Group by P. Chabaudi, Infected Group by P. Chabaudi and 50 mg/kg of Iole Agnps and the Administrated Group by P.Chabaudi and Cq 10 mg/kg.
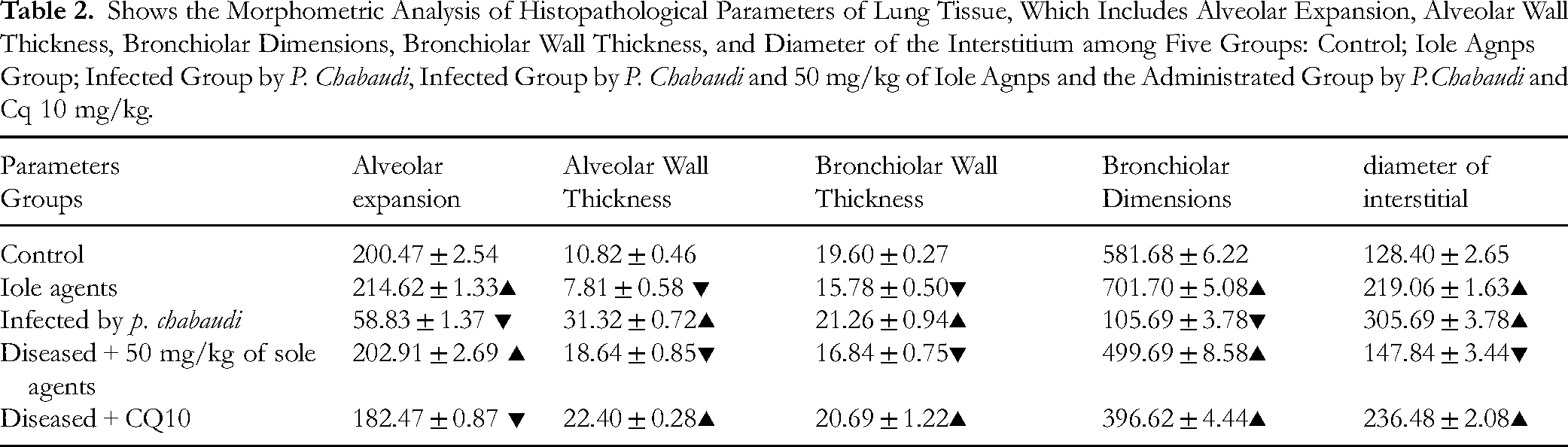
Values are represented as Mean ± STDV & n = 10 animals. Means within the same parameter and not sharing a common superscript symbol(s) differ significantly at p < 0.05, and values that are recorded with a non-significant difference (n.s).
Histomorphometric Observations of the P. chabaudi Lung
Figures 1E and 1F depict extensive histological damage following P. chabaudi infection, with both bronchiolar and alveolar diameters showing marked shrinkage, measuring 105.69 ± 3.78 µm2 and 58.83 ± 1.37 µm2, respectively, a significant reduction (p < 0.001) compared to normal values. The inflammation caused by P. chabaudi disrupted normal tissue architecture by significantly thickening the alveolar walls (31.32 ± 0.72 µm) and bronchiolar walls (21.26 ± 0.94 µm) beyond standard measurements (Figures 1: A, B, C & D). Hemorrhagic leakage was evident within the alveolar spaces and interstitial tissue, resulting in elevated hemosiderin levels due to red blood cell destruction and subsequent iron accumulation. Consequently, the inflamed lungs exhibited a notably higher hemosiderin concentration (20.57 ± 0.63%) compared to healthy lungs (p < 0.001) (Figure 1E).
Furthermore, congestion was observed prominently in the pulmonary structures, specifically in the alveolar septa (septal congestion) with an intensity of 35.66 ± 1.39%, and within the alveolar lumens (alveolar congestion) reaching 69.42 ± 2.72%, which was paralleled by congestion in the bronchiolar lumen (Figures 1: G & H). Epithelial hyperplasia was also detected in the alveolar and bronchiolar ducts, manifesting as lesions composed of cuboidal cells with oval to round nuclei, considered potential precursors to neoplastic transformation at the ductal termini.
The findings further revealed cellular damage characterized by epithelial cell degeneration and widespread necrosis. Degeneration was evidenced by cytoplasmic vacuolation, cellular swelling, and nuclear condensation (pyknosis). Necrotic cells were shed into the bronchiolar lumen and interstitial connective tissue, forming necrotic debris with an intensity of 14.84 ± 0.65%, exceeding that seen in normal tissue. Collagen-rich fibrotic tissue also developed within the extracellular matrix. The interstitial connective tissue exhibited significant expansion (P < 0.001), with a diameter of 305.69 ± 3.78 µm2, attributable to edema accumulation (42.53 ± 1.70%) and immune cell infiltration in response to Plasmodium infection (Figures 1: F, 1 & J). As a result, this group demonstrated markedly elevated histopathological scores, registering 183.02 ± 27.09%, significantly higher than other experimental groups (Table 3 and Figure 3).
Shows the Morphometric Analysis of Histopathological Parameters of Lung Tissue, Which Hemosiderin Intensity, Alveolar Congestion, Septal Congestion, Intensity of Necrosis, and Edema among Five Groups: Control; IOLE AgNPs Group; Infected Group by P. Chabaudi, Infected Group by P. Chabaudi and 50 mg/kg of IOLE AgNPs, and the Administrated Group by P. chabaudi and CQ 10 mg/kg.
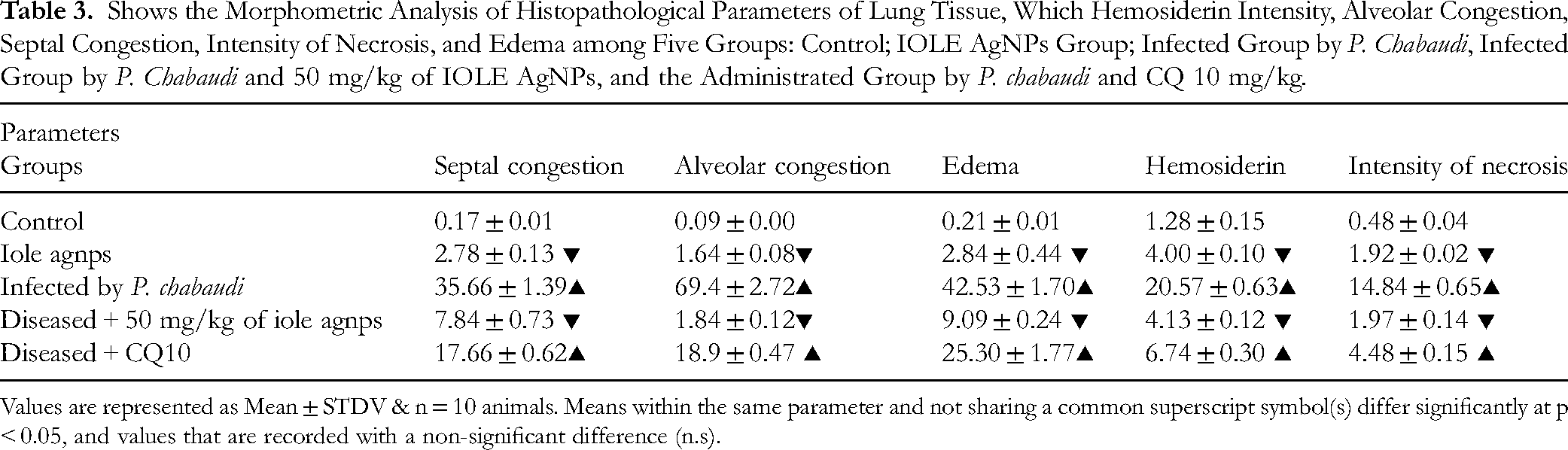
Values are represented as Mean ± STDV & n = 10 animals. Means within the same parameter and not sharing a common superscript symbol(s) differ significantly at p < 0.05, and values that are recorded with a non-significant difference (n.s).
Histomorphometric Observations of the Treated Lung by IOLE AgNPs After Infection
Based on the current findings in the treated lung, administration of I. oblongifolia loaded with AgNPs demonstrated a marked improvement in the damaged histological features caused by infection. The bronchiolar wall thickness showed significant recovery, reaching 16.84 ± 0.75 µm, which was comparable to normal values with no significant difference (p > 0.05). In contrast, the alveolar wall thickness exhibited a statistically significant improvement (p < 0.001), surpassing the normal control at 18.64 ± 0.85 µm. Meanwhile, bronchiolar diameter significantly increased (p < 0.001) to a typical size of 499.69 ± 8.58 µm2, while alveolar expansion displayed a non-significant restoration (P > 0.01), closely resembling the control measurement of 202.91 ± 2.69 µm2 (Figures 1: A, B, C & D). Notably, congestion levels in the alveolar (1.84 ± 0.12%) and septal (7.84 ± 0.73%) regions decreased significantly (p < 0.001) compared to the infected group, returning to near-normal values (Figures 1: G & H). Furthermore, edema (9.09 ± 0.2%) and necrosis (1.97 ± 0.14%) were significantly reduced (p < 0.001) relative to the infected lung (Figures 1: 1 & J). These improvements contributed to a significant decrease in the overall histopathological score (24.87 ± 5.35%) compared to diseased tissue (Table 4 & Figure 3).
The Table Shows the Scoring of the Histopathological Alterations Versus the Control Group Using the METAVIR Scoring Test among Five Experimental Groups: Control, IOLE AgNPs-Infected Lung; Infected Lung, Infected +50 mg/kg IOLE AgNPs, and CQ 10 mg/kg.

However, epithelial hyperplasia lesions persisted in the treated lungs, particularly around the alveolar and bronchiolar terminals, and were accompanied by scattered fibrotic tissue (Figures 2: G & H). Despite this, reductions in fibrosis, immune cell infiltration, edema, and hemosiderin deposition (down to 4.13 ± 0.12%) were observed, significantly lower than in inflamed lungs. This correlated with a decrease in interstitial tissue thickness to 147.84 ± 3.44 µm2 (Figures 2: G & H).
Histomorphometric Observations of the Treated Lung by CQ10 mg/kg After Infection
Similarly, chloroquine (CQ) at a dose of 10 mg/kg was administered to lungs infected with P. chabaudi. This treatment resulted in moderate improvement in the overall histological structure, though it was less pronounced than the recovery seen with I. oblongifolia treatment. The data revealed a slight, non-significant decrease (p > 0.001) in bronchiolar wall thickness (20.69 ± 1.22 µm), while alveolar wall thickness showed a significant reduction (22.40 ± 0.28 µm) compared to the infected group (Figures 1: A & B). Meanwhile, bronchiolar (396.62 ± 4.44 µm2) and alveolar (182.47 ± 0.87 µm2) diameters increased significantly (P < 0.001) relative to infected lungs.
Microscopic evaluation of lungs treated with CQ (10 mg/kg) indicated persistent septal and alveolar congestion with significant intensity (p < 0.0001) when compared to diseased tissue. Hemosiderin accumulation remained elevated (6.74 ± 0.30%) and close to infected levels (Figures 1: E, G & H). Additionally, alveolar epithelial hyperplasia was prominent alongside fibrotic tissue and necrotic cells (Figures 2: 1 & J). The interstitial tissue diameter was enlarged (236.48 ± 2.08 µm2), attributed to moderate edema (25.30 ± 1.77 µm2), fibrosis, and necrotic debris. Morphometric analysis revealed significant differences (p < 0.001) between the two treatment groups regarding interstitial thickness and edema levels (Figure 1: F), as well as notable differences compared to the infected group in nearly all measured parameters. These observations were supported by the elevated histopathological score (73.11 ± 13.31%) in this treatment group (Table 4 & Figure 3).
Correlation Analysis
The heatmap demonstrates varying correlation levels, reflecting how different treatments influence lung structure. For example, alveolar expansion showed a significant negative correlation with wall thickness (r = −0.85, P < 0.01), implying a compensatory adjustment following tissue damage. Similarly, bronchiolar diameter correlated negatively with its wall thickness (r = −0.59, P < 0.01), indicating airway narrowing during inflammatory responses. Additionally, the sizes of airways, including alveolar and bronchiolar ducts, negatively correlated with several inflammatory indicators such as septal congestion (r = −0.926, P < 0.01), alveolar congestion (r = −0.981, P < 0.01), edema (r = −0.891, P < 0.01), hemosiderin deposition (r = −0.967, P < 0.01), and necrosis intensity (r = −0.972, P < 0.01), reflecting airway constriction associated with congestion from trapped red blood cells.
On the other hand, interstitial diameter was strongly positively correlated with septal congestion (r = 0.858, P < 0.01), alveolar congestion (r = 0.847, P < 0.01), edema severity (r = 0.85, P < 0.01), hemosiderin accumulation (r = 0.869), and necrosis (r = 0.869, P < 0.01), which suggests an increase in inflammatory cell infiltration and deposition of hemosiderin within interstitial tissue. Notably, a strong positive correlation was also found between alveolar congestion and edema (r = 0.931, p < 0.01), indicating a direct link between vascular leakage and fluid build-up in alveolar spaces (Table 5 & Figure 4).

Triangle Heatmap Representing the Pearson Correlation Coefficients between Lung Morphometric Parameters (Alveolar Expansion, Alveolar Wall Thickness, Bronchiolar Wall Thickness, Bronchiolar Dimensions, Interstitial Diameter, Septal Congestion, Alveolar Congestion, Edema, Hemosiderin Deposition, and Necrosis Intensity). The Heatmap Displays Correlations Across Five Experimental Groups: Control, IOLE AgNPs-Infected, Infected, Infected + 50 mg/kg IOLE AgNPs, and CQ10-Treated. The Strength and Direction of Correlations: Positive Correlations are Represented in Purple, While Negative Correlations are in orange. The Color's Intensity Reflects the Correlation's Strength, With Darker Shades Indicating Stronger Associations.
Pearson Correlation Matrix of Lung Morphometric Parameters, Including Alveolar Expansion, Alveolar Wall Thickness, Bronchiolar Wall Thickness, Bronchiolar Dimensions, Interstitial Diameter, Septal Congestion, Alveolar Congestion, Edema, Hemosiderin Deposition, and Necrosis Intensity. the Correlations are Analyzed Across Five Experimental Groups: Control, IOLE AgNPs-Infected, Infected, Infected + 50 mg/kg IOLE AgNPs, and CQ10-Treated.

The heatmap displays the strength and direction of correlations. Statistical significance is denoted as follows: p < 0.05 (*), p < 0.01 (**).
Discussion
This study assessed the pharmacological effects of IOLE AgNPs on malaria caused by the P. chabaudi parasite. Morphometric and histopathological analyses were performed on lung tissues from five groups of mice. Treatment with IOLE AgNPs in control lungs improved respiratory function by widening the conducting airways, specifically the bronchiolar and alveolar ducts, as evidenced by increased diameters. The bronchiolar duct's normal branching and distribution create a transitional zone between the conducting and respiratory regions. 25 Previous research highlights that alveolar function in gas exchange depends on the diversity of pneumocyte cell types forming the alveoli.1,26
In infected lung tissue, significant disruptions in normal histological architecture were observed. The alveolar ducts showed notable narrowing due to increased interstitial space filled with inflammatory cells, edema, and hemosiderin deposits. Wall thickness was significantly greater than controls,27,28 likely due to fibrotic collagen fiber accumulation as part of the immune response to malaria-induced inflammation. Conversely, treatment led to improved diameters and thickness of alveolar and bronchiolar ducts, with bronchiolar and alveolar ducts showing significantly larger diameters than controls. Similar protective effects of I. oblongifolia have been reported in rheumatoid arthritis treatment and appetite restoration.29,30 Moreover, I. oblongifolia demonstrated hepatoprotective effects against carbon tetrachloride toxicity. 31
Chronic inflammation from Plasmodium infection caused smooth muscle contraction around bronchiolar ducts, contributing to bronchoconstriction and significantly thicker walls than seen in control or treated groups. Edema accumulation in peribronchiolar spaces contributed to this thickening. Similar observations were reported in cerebral cortex tissue infected with vivax malaria.31,32 Another study described severe malaria's cytotoxic effects on vascular and pulmonary ducts, including increased alveolar and septal congestion, edema accumulation, hemosiderin deposits, and necrosis-all findings consistent with the current analysis of infected lungs. 33
The combined anti-inflammatory and antioxidant properties of I. oblongifolia with the antimicrobial and anti-inflammatory effects of AgNPs provided an effective treatment against P. chabaudi induced lung inflammation. Previous work documented that this combination reduced spleen and liver enlargement and helped overcome multidrug resistance in malaria.29,34 Consequently, bronchiolar and alveolar ducts exhibited significant recovery in diameter and wall thickness, approaching normal values. Nanoparticles combined with plant extracts have been shown to reduce inflammatory cell infiltration and edema, preserving airway and alveolar structures.35,36
In contrast, treatment with the conventional antimalarial CQ10 mg/kg resulted in modest histological improvements relative to IOLE AgNPs. Despite CQ10 mg/kg's antioxidant activity and ability to lower oxidative stress and lipid peroxidation, some pathological signs persisted, including epithelial hyperplasia, congestion, and interstitial edema. This study found that IOLE AgNPs offered superior recovery compared to CQ10 mg/kg treatment. The enhanced therapeutic effect of I. oblgofola combined with metallic nanoparticles is attributed to improved tissue penetration due to their nanosize, targeting inflammation sites more efficiently than larger molecules like CQ10 mg/kg. The broad-spectrum antiparasitic and antimicrobial activity of AgNPs further boosts the effectiveness of IOLE AgNPs compared to conventional drugs. 34
Correlation analyses illustrated in the heatmap confirm severe lung pathology from P. chabaudi infection and demonstrate rapid tissue recovery following IOLE AgNPs treatment. Comparable correlations were reported during acute lung injury treatment with albumin nanoparticles. Conversely, the CQ10 mg/kg group showed stronger correlations between necrosis and bronchiolar narrowing, indicating less airway protection. Similar observations were made when comparing CQ with quercetin nanogel therapy, which exhibited superior efficacy.
Conclusion
Finally, the IOLE AgNPs and CQ10 mg/kg showed therapeutic properties in treating lung inflammation induced by P. chabaudi. Nevertheless, using IOLE AgNPs yielded exceptional outcomes in restoring the typical tissue structure and diminishing inflammatory indicators. Additional study is required to understand the processes by which these medicines work completely and to enhance their therapeutic effectiveness for lung inflammation caused by malaria.
Footnotes
Acknowledgment
Thank you, Researchers Support Project No. (ROF2025R1081), at King Saud University, Riyadh, Saudi Arabia.
Ethical Approval
The research was conducted by the “Guide for the Care and Use of Laboratory Animals.” The research complied with the institutional guidelines for animal usage at King Saud University and met the standards set by the National Committee of Bio-Ethics (NCBE) in Saudi Arabia. The Royal Decree numbered M59 was issued on 14/9/1431H. The Research Ethics Committee of King Saud University (Approval No. KSU-Se-19-107) sanctioned all experimental techniques, guaranteeing the rights and welfare of animals, stress, and preventing harm or suffering.
Author Contributions
The study was conceived and designed by Mutee Murshed. Jameel Al-Tamimi and Saleh Al-Quraishy analyzed the data. Mutee Murshed prepared the first draft of the manuscript. Mutee Murshed, Jameel Al-Tamimi, and Saleh Al-Quraishy collected and interpreted the data. The other researchers contributed the data and critically revised the work to update it with significant intellectual substance. The authors read the final version of the manuscript and approved it.
Funding
This work was supported and funded by the Researchers Supporting Project number (ORF-2025R1081), King Saud University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available within the article.
Statement of Human and Animal Rights
No research involving humans or animals is included in this article.
Statement of Informed Consent
This is not applicable because no human subjects are in this article.
