Abstract
Objective
To study the chemical constituents of Synotis solidaginea (SS) from Tibet. In our previous research, it was found that the extract of Synotis solidaginea stems has potential inhibitory activity against the proliferation of melanoma cells. Therefore, we hope to separate, extract and structurally identify the extracts of Synotis solidaginea stems, and determine the main components that exert potential inhibitory activity on the proliferation of melanoma cells.
Methods
The compounds were separated and purified using silica gel, polyamide, reversed phase C18 column chromatography, and preparative high performance liquid chromatography. The structure of each compound was identified based on the spectral data and literature comparisons. To verify the inhibitory activity of each component against melanoma cells, cell and enzyme assay were conducted.
Results
A total of 7 compounds were isolated from the dichloromethane extract of Synotis solidaginea. The irimofen lactone structure compound SS-1 and SS-5 were isolated from Synotis solidaginea for the first time. Among these, irimofin sesquiterpenoid compound SS-2 exhibited strong inhibitory activity against B16F10 of melanoma cells. Meanwhile, the iremorphene sesquiterpenoid SS-2 exhibited significant tyrosinase inhibitory activity, and the docking results explained the mechanism of its action.
Discussion
Solidaginea has laid the foundation for the prevention and treatment of melanoma with the active ingredients of Tibetan medicine.
Introduction
Synotis solidaginea Hand.-Mazz. (SS) is a traditional Tibetan medicinal herb with a long medicinal history, as recorded in Tibetan medicine texts such as “Tibetan Medicine Records”, “The Chinese Ethnic Medicinal Records”, and “Newly revised JingZhu BenCao”.1–7 Synotis solidaginea (SS) belongs to the genus Synotis in the family Asteraceae, and is primarily distributed in the high-altitude areas of eastern Tibet and western Sichuan in China. As a traditional herb, Synotis solidaginea is known for its cold in nature and bitter taste. It is believed to have therapeutic effects, such as clearing heat and detoxifying, treating ulcers, setting a bone, clearing liver and gallbladder heat. Additionally, it is used to treat various diseases, including acute conjunctivitis, dermatitis and wound inflammation. 8 In Tibetan folk medicine, Synotis solidaginea has been employed to treat ailments such as dermatitis, acute conjunctivitis, swelling, and traumatic injury. In our previous research, it was found that the extract of Synotis solidaginea stems has potential inhibitory activity against the proliferation of melanoma cells. Therefore, we hope to separate, extract and structurally identify the extracts of Synotis solidaginea stems, and determine the main components that exert potential inhibitory activity on the proliferation of melanoma cells, and develop targeted whitening agents. Given its medicinal properties, Synotis solidaginea holds significant potential for further development and utilization in the field of traditional medicine.
A series of compounds, including sesquiterpenoids, flavonoids, and phenolic acids, have been isolated from this plant. Traditional methods, such as thin-layer chromatography (TLC) and column chromatography, have been widely used to study the chemical basis of Synotis solidaginea, 9 as outlined in the “Sichuan Provincial Standards for Tibetan Medicinal Materials” (2020 Edition). 1 Recently, a multi-components qualitative and quantification strategy for the quality evaluation of Synotis solidaginea has been developed, incorporating techniques such as UHPLC-Q Exactive Orbitrap HRMS technique. This approach enables the quantification of 58 components in Synotis solidaginea, including flavonoids, organic acids, terpenoids, coumarin, and alkaloids, identified or tentatively characterized using authentic reference standards, accurate masses and characteristic fragment ions. 1
Modern pharmacological investigations have revealed that these compounds possess a variety of bioactivities, including anti-inflammatory, antiaging, antiviral, antibacterial, antioxidant, anti-tumor and anti-depressant properties,10–13 generating considerable interest in their biological potential. Among these, eremophilanes, a special class of sesquiterpenes, have recently attracted attention in drug discovery researchers due to their numerous biological activities.12,14 However, limited researches has focused on the comprehensive qualitative and quantitative analysis of the chemical compounds in Synotis solidaginea and their biological activity. As a result, eleven previously undescribed sesquiterpenoids were isolated from the methanol extract of Synotis solidaginea, and the majority of the new compounds possess eremophilanolide-type skeletons. The anti-inflammatory activities of selected isolates were evaluated. Here, we present the isolation, structural elucidation, and biological evaluation of these isolated and identified sesquiterpenoids.
Materials and Methods
Chemical Reagents and Materials
NMR spectra were recorded on a Bruker AV - 600 MHz spectrometer (Bruker, Germany). High - resolution mass spectra were obtained on a Waters Xevo G2 QTOF mass spectrometer (Waters Technology Co., Ltd.).A ZF - 20D type UV analyzer (Shanghai Jihui Scientific Analysis Instrument Co., Ltd, China) was used for dark - box triple - purpose UV analysis. Rotary evaporation was conducted using a RE52 - AA rotary evaporator (Shanghai Yarong Biochemical Instrument Factory, China).
High - performance thin - layer chromatography (HPTLC) was performed on precoated HSGF254 plates (8 ± 2 μm ≥ 80%, 0.4−0.5 mm, Yantai Jiangyou Silica Gel Development Co., Ltd.). Column chromatography was carried out with normal - phase silica gel (200-300 mesh, Qingdao Dingkang Silica Gel Co., Ltd.) and hydroxypropyl dextran gel (Qingdao Hailang Silica Gel Desiccant Co., Ltd.) using a gradient elution system. TLC spots were visualized using the UV analyzer at 254 nm or 365 nm, or by spraying with vanillin - sulfuric acid solution followed by heating. Common eluents including petroleum ether, ethyl acetate, dichloromethane, and methanol were purchased from Chengdu Kelong Chemicals Co., Ltd. All commercial reagents were obtained from Shanghai Aladdin Reagent Co., Ltd.
Plant Material
Chinese herbal medicine Synotis solidaginea, was collected in Nyingchi, Xizang (94.33647, 29.66594). It was authenticated as the western Synotis solidaginea by Professor Dongzhi Zhuoma, an expert in Tibetan medicine of Tibet University. At present, the sample is preserved in the Feng Gao research Group of Southwest Jiaotong University, with the number SWJTU-GF-024. The subject of this study is the extract from the stem.
Extraction Method
Extraction conditions: 6 kg of medicinal materials Synotis solidaginea were extracted twice with 80% ethanol . The first extraction used 12 times the material's weight, and the second used 10 times the weight, with each extraction lasting about 2 h. After filtration, the ethanol was recovered in a concentration tank under reduced pressure (with a cold trap configuration), and the solution was concentrated using rotary evaporation to yield approximately 3 kg (1.06-1.15) of the clearing paste, which is then ready for further use.
Separation Method
The concentrated extract was dispersed in water and extracted with petroleum ether, dichloromethane and ethyl acetate successively. The petroleum ether extract yielded 67 g, the dichloromethane extract yielded 78 g, and the ethyl acetate extract yielded 33 g, respectively. The extraction parts of dichlorom were separated according to the extraction method below. First, the extraction parts of dichlorom were passed through the chromatographic column with dichloromethane and methanol (ration 100:1-0:1), then silica gel chromatography column, gels chromatography column and reversed-phase chromatography column were applied and SS-1 to SS-7 were isolated and identified.
Cell Viability Assay
The B16F10 murine melanoma cells were purchased from the American Type Cultura Collection (ATCC, VA, USA) and cultured in DMEM containing 100 IU/Ml penicillin, 100 ug/mL streptomycin, and 10% FBS at 37 °C in a humidified atmosphere with 5% CO2. 15 The cell viability activity was tested by the MTT assay. Cells were seeded at a density of 2 × 104 cells per well in 96-well plates. After 24 h, the corresponding doses of drugs were added and incubated at 37°C for another 24 h. Then 20ul of MTT reagent solution was added to each well, the plates were shaken gently to mix, and they were incubated in the dark for 4 h. Remove the 96-well plates and discard the medium. 150ul of DMSO was added to each well, the plates were shaken for 10 min. and incubated at 37°C in the dark for an additional 30 min. Absorbance (OD) was measured at 490 nm using a ELISA Plate Reader (Tecan Infinite), and the activity and inhibition rate of the drug on the cells were calculated.16–18
Tyrosinase inhibition experimentSample solutions with different concentrations of 57 μL and 29 μL of tyrosinase were prepared by adding disodium hydrogen phosphate and citrate buffer (pH = 6.8) into 96-well plates. The mixtures were thoroughly mixed and incubated at 37 °C for 10 min. Following this, 114 μL of 1 mg/mL L-Dopa was added, and the solution was mixed well and incubated at 37 °C for an additional 5 min. Sample background group: 29 μL disodium phosphate - citrate buffer instead of 29 μL tyrosinase; Enzyme reaction group: 57 μL disodium phosphate - citrate buffer instead of 57 μL sample solution; Solvent background group: 57 μL disodium phosphate - citrate buffer was used to replace 57 μL sample solution, and 29 μL tyrosinase was replaced with 29 μL disodium phosphate - citrate buffer. Absorbance was determined at 475 nm wavelength, and arbutin was used as positive control19, 20. Inhibition rate = [1−(A1−A2)/(A3−A4)] × 100%, where A1 is the sample group, A2 is the sample background group, A3 is the enzyme reaction group, and A4 is the solvent background group
Statistical Analysis
Date were expressed as mean ± SD of three independent experiments and were analyzed using GraphPad Prism 8.3 software. P < .05 was considered statistically significant.19,20
Molecular Docking, Molecular Simulation
Firstly, the 3D structural formula of the compound was drawn by ChemDraw. 21 Molecular docking was performed using virtual screening software (Autodock, Schrodinger and Discovery) to assess the binding efficiency, and the docking result was visualized by visualization software.22,23 The crystal structures of tyrosinase and other relevant proteins was obtained from the protein database and the PDB format file was downloaded and imported into the virtual screening software. The structure of the target molecule was optimized by removing water molecules and adding hydrogenating atoms. The active docking site of the target protein was identified, and finally, the compound ligand was docked with the protein receptor. After docking, the binding energy was calculated to evaluate the binding effect between the ligand and the receptor. A binding energy value of less than 0 indicates a good binding ability between the compound and the target. Both the number of hydrogen bonds and the binding energy are important indicators of the binding strength. Generally, lower the binding energy indicates stronger affinity, and a higher number of hydrogen bonds corresponds to greater stability in the ligand-receptor interaction.
Results
Extraction and Separation Results
The main components of the plant include phenolic acids, terpenoids and flavonoid glycosides. At present, 8 compounds have been isolated, and the structures of 7 have been confirmed structure. Among these, 2 are steroids, 4 are Irimofen sesquiterpenoids, and 1 are aromatic compound.
Structural identification as follows
Compound SS-1, SS-2 and SS-5, with an irimofenolide structure, which have been isolated from the biflowering Senecium plant. The compound SS-3, Ligularenolide, an irimofen sesquiterpene glycoside, previously isolated from Senecium biflorum. Compound SS-4, aromatic ring substitute. Compound SS-6, sitosterol, a steroidal compound. The structure was determined by comparison with the hydrogen spectrum data available in the laboratory. The compound SS-7, carotenin, a steroid, previously isolated from the elderberry plant (Figure 1).

Compounds Isolated from Synotis solidaginea.
Cell Experiment Results
Melanin is formed through a series of oxidative reactions involving the amino acid tyrosine in the presence of the enzyme tyrosinase. The tyrosinase inhibition assay can characterize the quantity of melanin production, which has received the most attention in the development of skin whiteners.24,25
For reference, the IC50 range of arbutin against melanoma cells was 64-1550uM, with the B16F10 cell line showing an IC50 of 391.2 µM. Additionally, several compounds in Synotis solidaginea dichloromethane layer generally exhibited certain activity against melanoma cells B16F10, with SS-2 showing superior activity compared to other sesquiterpenoids (Table 1).
Activity of Compounds Extracted from Synotis solidaginea Against Melanoma Cells B16F10.

Tyrosinase Inhibition Experiment Results
As for tyrosinase inhibition variation tendency of SS-2 in different concentration, the result showed positive results. Namely, SS-2 had good tyrosinase inhibition rate at the concentration of 0.2–200 ug/ml when compared with Arbutin (Figure 2).
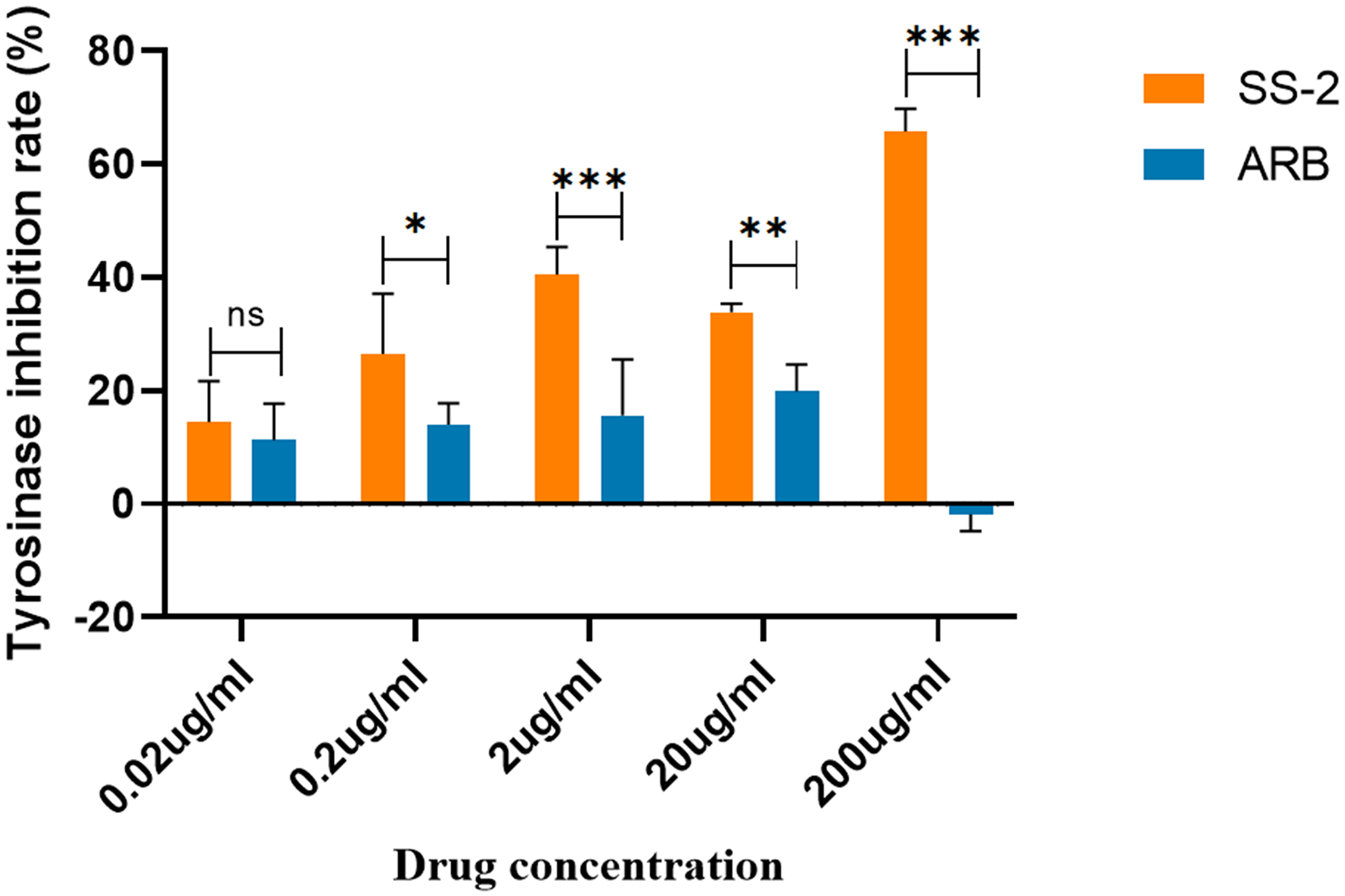
Tyrosinase Inhibition Rate Variation Trend. Results are Representative of Three Independent Experiments, and the Data are Shown as Means ± SD. The “ns” Means No Significant Statistical Difference, * p < .05, ** p < .01, *** p < .001.
Docking Result
Overall binding mode analysis diagram of the compound with tyrosinase protein (PDB ID: 7RK7); the right figure shows the analysis diagram of the best binding pose of the compound with tyrosinase. The tyrosinase model is displayed in cartoon form, and the compound is shown in stick form. The tyrosinase receptor is light blue, the carbon atoms in the compound are yellow, the oxygen atoms are red, and the nitrogen atoms are blue. The green dotted line represents hydrogen bonds. In the best docking result of Autodock Vina, the docking binding energy score based on the Affinity function is −5.4 kcal/mol. The compound forms a hydrogen bond interaction with the MET 6 amino acid residue of tyrosinase, which is indicated by the green dotted line and has a length of 2.5 Å26,27 (Figure 3).
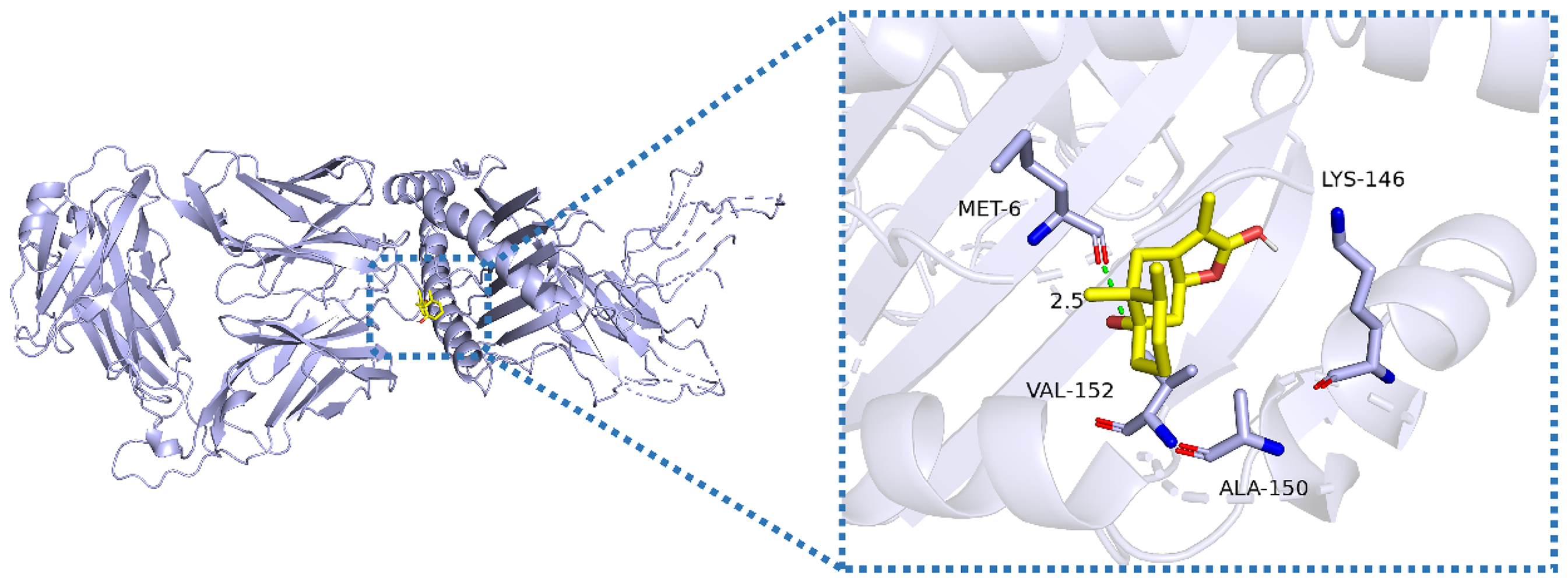
Docking Result.
Discussion
The common topical drug used in Tibetan medicine has a long history of medicinal use and is widely employed in clinic. 28 The plant is widely distributed and rich in resources; however, there are limited reports on its chemical constituents and pharmacological effects. Therefore, this study conducted a systematic investigation into the chemical composition of the plant, leading to the isolation and identification of a total of 7 compounds, ranging from SS-1 to SS-7. Notably, compound SS-2 and SS-5 were isolated from the genus Synthopsis for the first time. These findings expand the understanding of the plant's chemical composition and provide a foundation for future pharmacological research, development, utilization, and quality evaluation. Moreover, the compound SS-2 exhibited a good inhibitory activity in the production of melanin against B16F10 cells, which possesses a complete tyrosinase (TYR) pathway. And in our previous research, we found that it shares common targets with the tyrosine-related protein family. Therefore, it's hypothesize that SS-2 may rely on the tyrosinase-related pathway to exert the effect on inhibiting melanin production. In the subsequent work, we will conduct verification on the pathway proteins and related mechanisms. The identified exhibited inhibitory effects on melanin production, highlighting their potential in preventing melanoma and other skin diseases such as melasma, hyperpigmentation and solar dermatitis within Tibetan medicine.
Research Limitations
However, there are limited reports on its chemical constituents and pharmacological effects. This article need more animal and cell experiments.And pharmaceutical effect maybe have different targets, which has more evidence.
Footnotes
Acknowledgments
The authors have reviewed and edited the output and take full responsibility for the content of this publication.
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
Conceptualization, Zhuoma Basang, Feng Gao and Wangdui Suolang; methodology, Longwei Fang and Zhen Ni; software, Yanglei Shen.; validation, La Yang and Nanling Liu; formal analysis, Zhuoma Duoji; investigation, Longwei Fang and Dongzhi Zhuoma; data curation, Longwei Fang, Zhen Ni and Yanglei Shen; writing—original draft preparation, Longwei Fang; writing—review and editing, Zhen Ni and Yanglei Shen; visualization, Yanglei Shen; supervision, Zhuoma Basang and Feng Gao; project administration, Wangdui Suolang and Zhuoma Basang; funding acquisition, Wangdui Suolang and Zhuoma Basang. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Shigatse City 2025 Science and Technology Plan Project (RKZ2025ZD-003); Shigatse City 2024 Regional Collaborative Innovation Project (QYXTZX-RKZ2024-01-1); National Natural Science Foundation of China (82241023); 2023 Xizang Science and Technology Department high-tech social development field “Hang Gang” project (JBGS2023000004);2023 Science and Technology Department of Tibet Autonomous Region Project (XZ202303ZY0008G); 2024 Project Co-funded by the Science and Technology Department of Tibet Autonomous Region (Research on Early Screening Model and Diagnosis and Treatment System of High Altitude Pulmonary Hypertension (High Altitude Heart Disease)) (XZ202403ZY0006); Graduate High Level Talent Training Program of Xizang University (2021-GSPS052); National Natural Science Foundation of China (82241023); Basang Zhuoma Discipline Leading Talents Program (00060695/003);2024 Annual Funding Project of the Lhasa Science and Technology Bureau (LSKJ202466); Discipline Leading Talents Program of Xizang University (0061243/047).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
