Abstract
In our survey on the chemical composition of Chinese folk medicines, nine compounds were isolated from methanol extract of the leaves of
Introduction
Chinese herbal medicine is a unique medicine, which has been used for thousands of years in China. Because plant medicine accounts for the majority of traditional Chinese medicine, traditional Chinese medicine is also called Chinese herbal medicine. At present, there are over 5000 kinds of traditional Chinese medicines used in various places, and there are countless prescriptions formed by the combination of various kinds of traditional Chinese medicines, which play an important role in the fight against various diseases, such as Corona Virus Disease 2019 (COVID-19).1,2 Chinese herbal medicine is an important part of medicine. Although herbal medicine plays an important role in the treatment of various diseases, it is difficult to widely accept Chinese herbal medicine because of its complex chemical composition, unclear pharmacodynamic components and mechanism. Thus, it is necessary to elucidate the chemical constituents and investigate action mechanism of Chinese herbal medicines.
Among numerous Chinese herbal medicines,
The current study aims to investigate the chemical constituents in the leaves of
Results and Discussion
After extraction, the petroleum ether- and ethyl acetate-soluble fractions of the methanol extract derived from the leaves of
Compound

1H-1H COSY and key HMBC and NOESY correlations of compound 1.

Structures for compounds 1–9.
The configuration of
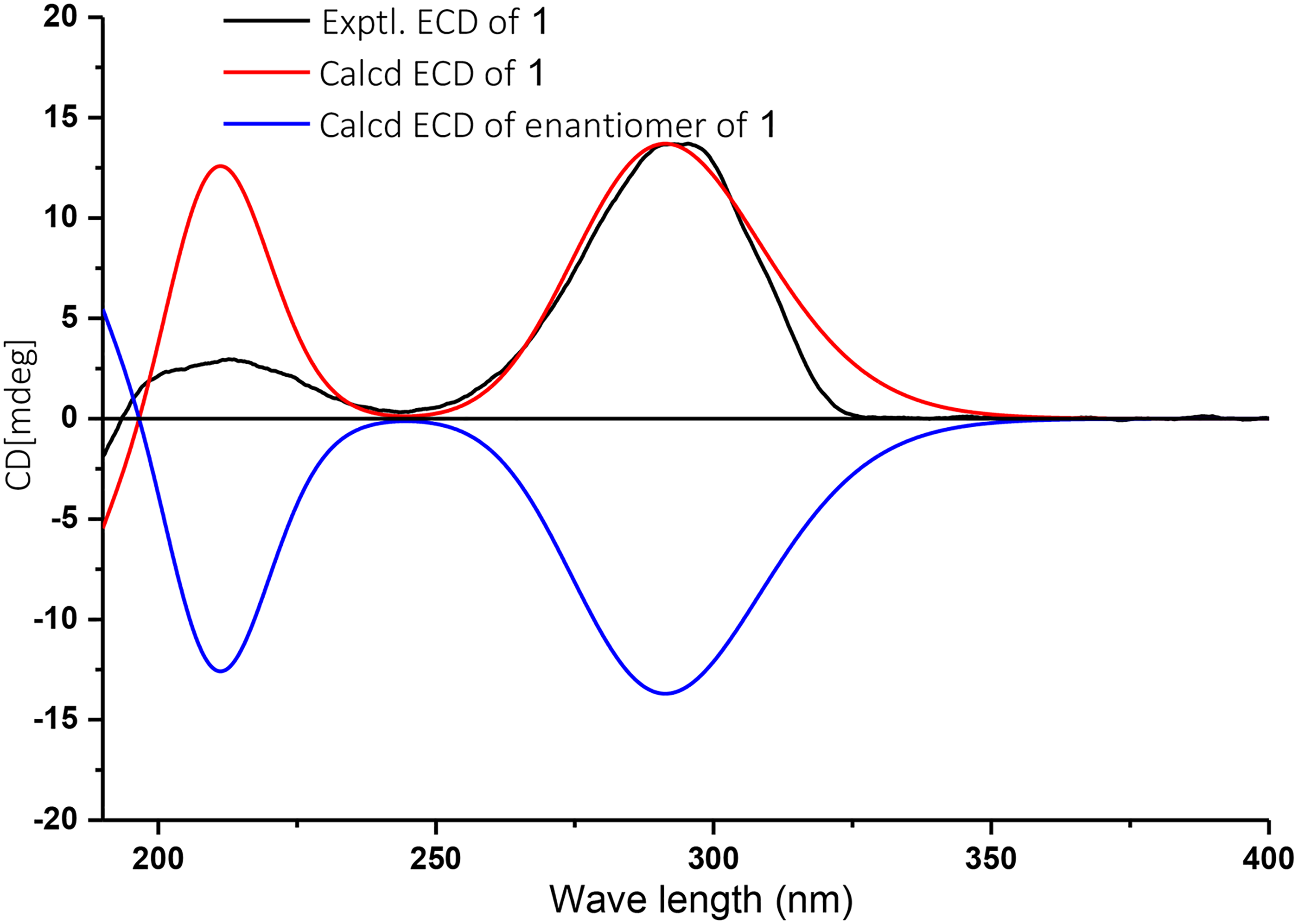
The ECD spectra of compound
In addition to a new compound, torriangulate A, eight known compounds were also isolated from the leaves of
In conclusion, the current chemical composition survey on the traditionally folk medicine led to the isolation of nine chemical constituents from the leaves of
Experimental
General
Optical rotations were measured by InsMark IP120 automatic optical rotator (InsMark Co.Ltd, Shanghai, China). ECD data were measured by JASCO J-715 CD spectrometer (JASCO Co.Ltd, Japan). IR spectrum was recorded on a Bruker Tensor 37 FT-IR spectrometer with KBr discs (Bruker Co. Ltd, Germany). All NMR tests were performed on a Bruker AV 400 instrument (Bruker Co. Ltd, Germany) with TMS as an internal standard at room temperature. High resolution spectra obtained on IonSpec 7.0T FTICR (IonSpec Co. Ltd, Canada) mass spectrometer. HPLC separations were conducted on a CXTH system, equipped with a Shodex RI-102 detector (Showa Denko Co., Ltd, Tokyo, Japan) and a YMC-pack ODS-AM (20 × 250 mm) column (YMC Co. Ltd, Kyoto, Japan). Medium pressure liquid chromatography (MPLC) was run on a P0100 pump with an ultraviolet (UV) detector (Huideyi Co., Beijing, People's Republic of China) and a column (40 × 400 mm) filled by octadecylsilyl (ODS, 50 μm, YMC Co., Ltd). Silica gel (200–300 mesh) used for column chromatography was purchased from Qingdao Haiyang Chemical Group Co., Ltd (Qingdao, People's Republic of China). Chemical reagents (analytical grade) for isolation were of analytical grade and were provided by Tianjin Chemical Reagent Co. (Tianjin, China) and Sigma Co., respectively.
Plant Material
The leaves of
Extraction and Isolation
The air-dried leaves (6.0 kg) of
The ethyl acetate-soluble portion was fractionated by silica gel column chromatography (silica gel, 700 g; column, 7 × 50 cm), using a gradient solvent system of petroleum ether-acetone (100: 0, 100: 2, 100: 4, 100: 6, 100: 9, 100: 13, 100: 19, 100: 27, 100: 35, 21 L for each gradient elution), to afford nine fractions (F1−F9) according to the TLC analysis. F5, F6, and F7 were subjected to MPLC and eluted with a step gradient of 64 − 92% MeOH in H2O to give subfractions F5-1−F5-6, F6-1−F6-8, F7-1−F7-8. Subsequenly, HPLC separations were carried out for some of these subfractions. The purification of F6-1 (45% MeOH in H2O) led to the isolation of compounds
The petroleum ether-soluble portion was subjected to silica gel column chromatography (silica gel, 700 g; column, 7 × 50 cm), using ethyl acetate as eluent to give one fraction A1. Then, A1 was separated by MPLC to obtain nine subfractions A1-1−A1-9. The purification of A1-1 (70% MeOH in H2O) resulted in the obtainment of compounds
Dimethyl 2-(hydroxymethyl)-5-oxocyclohexane-1,4-dicarboxylate (
Methyl succinate (
5-Hydroxymethyl-2-furfuraldehyde (
7-Hydroxy-6-methoxycoumarin (
Loliolide (
(8
Methyl malate (
Griselinoside (
Methyl linoleate (
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The project was supported by the Natural Science Foundation of Guizhou (No. QKHJC[2020]1Y390) and Qiannan Medical College for Nationalities Research Fund Project (No.qnyz202034), China.
Ethical Approval
Not applicable, because this article does not involve any animal experiments.
Statement of Human and Animal Rights
Not applicable, because this article does not involve any animal experiments.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
