Abstract
Objective/Background
Cinnamomum insularimontanum Hayata, an endemic Taiwanese tree species, has leaves and fruits rich in essential oils that have been traditionally used for medicinal purposes. This study aimed to analyze the chemical composition of essential oils extracted from the leaves, twigs, and fruits, and to evaluate their in vitro antimicrobial, antioxidant, and tyrosinase-inhibiting activities for potential applications in natural preservation and health-related industries.
Methods
Essential oils were extracted through hydrodistillation and analyzed using GC-FID and GC-MS. Antibacterial, antifungal, antioxidant (β-carotene bleaching, DPPH, ABTS, CUPRAC), and tyrosinase inhibition activities were assessed. Major components were further examined for their contributions to bioactivity.
Results
The highest yield was obtained for the fruit essential oil (4.52 ± 0.03 mL/100 g), which also showed the strongest bioactivity. It exhibited strong antibacterial activity against Bacillus cereus, Staphylococcus aureus, and S. epidermidis (MIC = 32 μg/mL) and antifungal effects against Aspergillus flavus, A. niger, and Chaetomium globosum (MIC = 64-128 μg/mL). The fruit oil also had the highest antioxidant activity in the β-carotene–linoleic acid bleaching assay, with an IC50 value of 138.9 ± 1.5 μg/mL, indicating strong inhibition of lipid peroxidation. In the DPPH and ABTS assays, it showed IC50 values of 92.6 ± 0.5 and 105.2 ± 0.5 μg/mL, respectively. In the CUPRAC assay, it had an A0.50 value of 203.2 ± 0.5 μg/mL, indicating high reducing power. Additionally, it exhibited strong tyrosinase inhibition (IC50= 137.4 ± 4.1 μg/mL). Citral, the main component of the fruit essential oil (52.2%), was identified as the key cause of these effects.
Conclusion
Cinnamomum insularimontanum fruit oil exhibits strong antimicrobial, antioxidant, and tyrosinase-inhibiting effects, mainly due to its high citral content. These findings suggest its promising potential as a natural preservative and functional ingredient in the food, cosmetic, and pharmaceutical industries. Further studies on their mechanisms of action and formulation potential are warranted.
Introduction
With the intensification of global warming, people are increasingly seeking methods to harmoniously coexist with nature. Among these efforts, the development of pharmaceuticals, food products, and cosmetics from substances found in nature has become a key trend that aligns with the principles of sustainable forestry management. Essential oils, which are extracted from aromatic plants, are natural volatile compounds primarily comprising monoterpenes, sesquiterpenes, and their oxygenated derivatives. These oils are antibacterial, contain antioxidants, and have various other biological effects. Our previous studies have confirmed that the essential oils of many tree species possess excellent bioactive properties.1-5 Therefore, extracting essential oils from plant parts—such as leaves, twigs, and fruits—and developing diverse products on the basis of these oils may contribute to the sustainable development of forestry.
Recently, public concern for health and food safety has increased, and preserving food and maintaining the quality of food crops have become global concerns. Currently, synthetic antimicrobial agents, antioxidants, and tyrosinase inhibitors are widely used in the food industry to extend products’ shelf life; however, long-term consumption of these synthetic compounds may cause allergies and gastrointestinal disorders and even pose carcinogenic risks.6,7 Consequently, the development of natural antimicrobial, antioxidant, and tyrosinase-inhibiting agents has become an essential direction of research, with essential oils emerging as optimal agents because of their natural bioactive components.
Taiwan, located in a subtropical region, provides a favorable climate for the growth of Lauraceae species. Cinnamomum insularimontanum Hayata, a broadleaf tree endemic to Taiwan, is widely distributed in mid-altitude mountainous areas. 8 Research has indicated that the essential oils extracted from this tree's leaves and fruits have excellent anti-inflammatory properties,9,10 whereas its leaf extracts have been demonstrated to have antiviral activity. 11 Additionally, actinodaphnine, an alkaloid isolated from the roots of C. insularimontanum, has been found to possess antihepatocarcinoma activity. 12 However, research into C. insularimontanum remains incomplete. Despite limited studies, existing reports have revealed that C. insularimontanum contains several bioactive compounds with promising physiological effects, indicating its potential for further development in functional applications.
This study aimed to comprehensively evaluate the chemical composition and biological activities of essential oils extracted from the leaves, twigs, and fruits of C. insularimontanum. Specifically, we assessed their antibacterial, antifungal, antioxidant, and tyrosinase-inhibiting activities. Among these, tyrosinase inhibition was included due to its relevance in both the cosmetic and food industries. Tyrosinase is a key enzyme in melanin biosynthesis, and its inhibition is critical in the development of natural skin-whitening agents. In the food sector, tyrosinase activity is also associated with enzymatic browning, which affects the quality and shelf life of produce. Therefore, tyrosinase inhibitors are widely sought after for applications in cosmetics and skin-whitening formulations, as well as for preventing enzymatic browning in food products. Given the increasing demand for natural and safe tyrosinase inhibitors, and the traditional medicinal use of C. insularimontanum, investigating its tyrosinase-inhibitory potential was considered both timely and valuable. This study represents the first report to simultaneously examine the essential oils from three distinct plant parts of this endemic species, providing novel insight into their potential for use in natural preservation, skincare, and functional product development.
Materials and Methods
Plant Materials
In September 2024, fresh leaves, twigs, and fruits of C. insularimontanum were collected at the Pilu Experimental Station of the Lienhuachih Research Center, Taiwan Forestry Research Institute (TFRI). This collection site is located at 24°22′69″ N, 121°31′00″ E and has an altitude of 2100 m. Plants were taxonomically identified by Dr Hsu Chun-Kai from the TFRI. To serve as a reference for future studies, a voucher specimen (HCCLH-101) was carefully preserved and deposited in the Wood Utilization Division of the TFRI.
Chemicals, Standards, and Reagents
The following chemicals and reagents were used in this study and purchased from Sigma-Aldrich (St. Louis, MO, USA): alkane standard solution (C8–C30), 2,2-diphenyl-1-picrylhydrazyl (DPPH, 97%), 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid diammonium salt, ABTS), β-carotene (≥95%), linoleic acid (≥98%), copper(II) chloride (≥99%), neocuproine (≥98%), ammonium acetate (≥99%), kojic acid (≥98.5%), tyrosinase (from mushroom), 3,4-dihydroxy-L-phenylalanine (L-DOPA), α-tocopherol (≥95.5%), butylated hydroxyanisole (BHA, ≥ 99%), Mueller-Hinton agar (MHA), potato dextrose agar (PDA), nystatin (≥95%), and amoxicillin (≥95%).
The essential oil reference standards used in GC–MS analysis, also purchased from Sigma-Aldrich, included: α-pinene (98%), camphene (95%), sabinene (75%), β-pinene (≥99%), 6-methyl-5-hepten-2-one (≥99%), myrcene (≥99%), α-phellandrene (≥95%), α-terpinene (≥95%), p-cymene (≥99%), limonene (96%), 1,8-cineole (≥99%), β-ocimene (≥90%), γ-terpinene (97%), terpinolene (≥95%), linalool (≥97%), myrtenol (≥95%), camphor (96%), citronellal (≥95%), borneol (97%), terpinen-4-ol (≥95%), α-terpineol (≥95%), citronellol (≥95%), nerol (≥97%), geraniol (≥97%), piperitone (≥95%), bornyl acetate (≥95%), geranyl acetate (≥97%), β-elemene (≥95%), (E)-caryophyllene (≥95%), elemol (≥90%), (E)-nerolidol (≥95%), and caryophyllene oxide (≥99%).
Citral (≥99%), T-muurolol (≥99%), and α-cadinol (≥99%) were previously isolated and identified in our laboratory according to the methods described in references.13-15
Isolation of Leaf, Twig, and Fruit Essential Oils
Hydrodistillation was used to obtain essential oils from the leaves, twigs, and fruits of C. insularimontanum. A total of 10 kg of each type of plant part was used, and 6-h distillation was conducted three times by using a Clevenger apparatus. The water-to-plant material ratio was maintained at 3:1 (v/w), and the distillation rate was controlled at 2.5 mL/min. The obtained essential oils were dried over anhydrous sodium sulfate to remove residual moisture, collected in sealed sample vials, and stored at 4 °C to preserve their stability and quality. Essential oil yields and related experimental data were derived from three independent replicates and are reported as mean ± standard deviation.
Essential Oil Analysis
To analyze the composition of the extracted essential oils, we conducted Gas chromatography–flame ionization detection (GC-FID) and Gas chromatography–mass spectrometry (GC-MS) analysis. In the GC-FID analysis, a Hewlett–Packard Model 6890 gas chromatograph equipped with a flame ionization detector was employed. The instrumental parameters were as follows. The detector was connected to a DB-5 fused silica capillary column, consisting of 5% phenyl and 95% methylpolysiloxane, with a column length of 30 m, an internal diameter of 0.25 mm, and a film thickness of 0.25 μm. The GC oven temperature was initially set at 50 °C, held for 2 min, and then increased at a rate of 5 °C/min to reach 250 °C. The injection port temperature was maintained at 270 °C, whereas the detector temperature was maintained at 250 °C. Hydrogen was used as the carrier gas at a flow rate of 1.0 mL/min and a split ratio of 1:60. Before injection, a 1 μL sample was prediluted with ethyl acetate at a 1:100 (v/v) ratio. Linear retention indices (LRIs) were then determined by comparing the retention times of the samples with those of C8 to C30 n-alkane standards. The relative content of each essential oil component was quantified through the electronic integration of peak areas and was expressed using the normalization method, without any correction factors applied.
For the GC-MS analysis, a DB-5 fused silica capillary column was employed in conjunction with a Hewlett Packard Model 6890/5973 GC-MS system. The carrier gas used was high-purity helium (99.995%), which flowed at a rate of 1 mL/min, and the same analytical parameters as those used for GC-FID were applied. Data were acquired in full scan mode, covering a mass-to-charge ratio range of 30–500 (m/z).
Component Identification
Compounds were identified through an analysis and comparison of their LRIs, retention times, and mass spectra. In this process, data from the NIST and Wiley mass spectral databases, authentic reference standards, and pertinent literature16-18 sources were used for comparison.
Antibacterial Activity of Essential Oils Against Foodborne Pathogens
The following foodborne pathogenic bacteria were employed in this study: three Gram-positive bacteria—Bacillus cereus (ATCC 11778), Staphylococcus aureus (ATCC 6538P), and S. epidermidis (ATCC 12228)—and three Gram-negative bacteria—Escherichia coli (IFO 3301), Enterobacter aerogenes (ATCC 13048), and Vibrio parahaemolyticus (ATCC 17803). These bacterial strains were sourced from the Culture Collection and Research Center of the Food Industry Research and Development Institute, Hsinchu City, Taiwan.
The antibacterial properties of the extracted essential oils were examined using the paper disk diffusion method. Their minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) values were also determined. Additionally, the antibacterial effects of the primary compound isolated from the fruit essential oil was determined using the paper disk diffusion method, as reported by Bauer et al. 19
Each bacterial inoculum was applied to a Mueller Hinton agar (MHA) plate to ensure even growth on the plate. Filter paper discs with a diameter of 6 mm and containing 10 μL of undiluted oil were placed on the inoculated plates. After 24 h of incubation at 37 °C, the inhibition zones (IZs) were measured, with the measured values inclusive of the 6-mm diameter of the disc. Amoxicillin was used as the positive control. Each test was conducted in triplicate.
MIC values were determined through a microdilution susceptibility test, which was performed in accordance with the guidelines of the National Committee of Clinical Laboratory Standards 20 and as previously reported. 21 After the essential oils were diluted to a concentration of 1.024 mg/mL in dimethyl sulfoxide, a series of twofold dilutions (ranging from 16 to 1024 μg/mL) were tested.
MBC values were determined using the method described by Chikezie. 22 Aliquots (100 μL) of each dilution were spread on MHA plates, which resulted in no visible growth. These plates were incubated at 37 °C for 24 h, as were the control plates. After incubation, colony counts were performed, and the results were compared with those for the control plates. The MBC was defined as the lowest concentration that killed >99.9% of the initial bacterial inoculum. All experiments were performed in triplicate to ensure reproducibility and consistency.
Antifungal Activity of Essential Oils Against Foodborne Pathogens
An antifungal assay developed by Su et al 23 was conducted. The following foodborne pathogenic fungi were obtained from the Culture Collection and Research Center of the Food Industry Research and Development Institute, Hsinchu City, Taiwan: Aspergillus flavus (ATCC 1007), A. niger (ATCC 6275), Chaetomium globosum (ATCC 6205), Penicillium citrinum (ATCC 9849), and Trichoderma viride (ATCC 8678). All assays were conducted in triplicate, and the data were averaged. Different concentrations of the essential oils and the main constituents of the fruit oil (concentrations of 16-1024 μg/mL) were added to sterilized potato dextrose agar. The test Petri dishes were then incubated at 27 °C. Once the mycelium of the fungi reached the edge of the control plate, the anti–foodborne fungal pathogen index (%) was calculated as (1 − Da/Db) × 100, where Da and Db represent the mycelium diameter (in centimeters) in the experimental and control groups, respectively. Nystatin was used as the positive control. Each test was conducted five times, and the data were averaged. The half-maximal inhibitory concentration (IC50) values were calculated in probit analysis.
Antioxidant Activity
β-Carotene–Linoleic Acid Bleaching Assay
A β-carotene–linoleic acid bleaching assay was conducted as previously described, with minor adjustments. 24 In addition, a correlation plot between sample concentration and antioxidant efficacy (percentage inhibition) was drawn to determine each sample's IC50 value (in micrograms per milliliter).
DPPH Free Radical Assay
A DPPH assay was conducted using the method described by Ho and Su. 25 The procedure involved mixing 50 μL of essential oil at a certain concentration with 5 mL of methanol solution containing 0.004% DPPH. After a 30-min incubation period, the solution's absorbance was measured at a wavelength of 517 nm by using a Jasco 7800 spectrophotometer. Each experiment was conducted in triplicate, with α-tocopherol and BHA serving as positive controls. Each sample's DPPH scavenging activity (%) was calculated using the following formula: (A0 − A1)/A0 × 100, where A0 and A1 represent the absorbance of the control group and the sample, respectively. The IC50 values were determined by plotting percentage scavenging activity against sample concentration.
ABTS Cation Radical Scavenging Assay
This assay was conducted through spectrophotometry, following the method described by Re et al 26 The IC50 value, representing the sample concentration required to inhibit 50% of ABTS•+ radicals, was determined by plotting inhibition percentage against sample concentration (in micrograms per milliliter).
CUPRAC Assay
A CUPRAC assay was conducted using the method proposed by Apak et al, 27 and the A0.50 value was determined. The A0.50 value, the sample concentration required to achieve absorbance of 0.500, was determined by plotting CUPRAC absorbance against sample concentration (in micrograms per milliliter).
Tyrosinase Inhibition Assay
We adopted the method proposed by Masuda et al 28 to evaluate the tyrosinase inhibitory activity of the extracted essential oils and the active compounds of the fruit essential oil, with kojic acid used as the positive control. These inhibitory effects are expressed as inhibition rate (%), with test concentrations ranging from 31.25 to 1000 μg/mL.
The inhibition rate was calculated using the following formula:
The IC50 value was determined by conducting a nonlinear regression analysis on the dose–response curve.
Statistical Analysis
All experiments were conducted in quintuplicate, and the results are expressed as mean ± standard deviation (SD). Data were analyzed using one-way ANOVA, followed by Tukey's honestly significant difference (HSD) multiple-range test. 29 All statistical analyses were performed using PASW Statistics version 18. 30 IC50 values were calculated based on dose–response curves generated using GraphPad Prism, plotting the percentage of inhibition (%) against essential oil concentration.
Results
Yields of Essential Oils
Yellow essential oils were extracted through 6-h hydrodistillation from C. insularimontanum biomass—leaves, twigs, and fruits. The essential oil yield, calculated on the basis of dry weight, was 3.31 ± 0.02, 0.31 ± 0.01, and 4.52 ± 0.03 mL/100 g for leaves, twigs, and fruits, respectively. Thus, the highest and lowest yields were obtained for fruits and twigs, respectively.
Components of Essential Oils
GC and GC-MS were used to analyze the components of the essential oils and determine their concentrations. The identified compounds and their concentrations and LRI values are summarized in Table 1, with the compounds arranged in this table in accordance with their order of elution on a DB-5 capillary column.
Chemical Composition of Essential Oils Extracted from the Leaves, Twigs, and Fruits of C. insularimontanum.

Classification: MH = monoterpene hydrocarbons; OM = oxygenated monoterpenes; SH = sesquiterpene hydrocarbons; OS = oxygenated sesquiterpenes; DI = diterpenes; OT = others.
LRILit = LRI values from a previous study. 16
LRIExp = calculated LRI values obtained for a mixture of a continuous series of n-alkane hydrocarbons (C8–C30) in a DB-5 capillary column.
Identification through MS = comparison of the NIST and Wiley mass spectral libraries; LRI = linear retention index same as the previous findings;16-18 CO-ST = co-injection/comparison with the LRI and MS standards.
Not detected.
The essential oil extracted from the leaves of C. insularimontanum contained 36 identifiable compounds. The compounds with the highest concentrations were camphor (34.6%), 1,8-cineole (20.2%), α-pinene (6.6%), sabinene (5.5%), and limonene (5.2%). These compounds were classified into five categories: oxygenated monoterpenes (68.4% of the total content), monoterpene hydrocarbons (26.8% of the total content), oxygenated sesquiterpenes (2.7% of the total content), sesquiterpene hydrocarbons (1.9% of the total content), and diterpenes (0.1% of the total content).
In this study, 41 compounds were identified in the twig essential oil, with α-pinene (21.1%), linalool (12.1%), camphene (7.6%), and β-pinene (5.4%) being the major constituents. The twig essential oil consisted of 47.6% monoterpene hydrocarbons, 30.2% oxygenated monoterpenes, 12.1% oxygenated sesquiterpenes, and 9.8% sesquiterpene hydrocarbons, with no diterpenes detected.
Analysis of the fruit essential oil's components identified 41 compounds. The main component was citral (52.2%), which comprised neral (22.0%) and geranial (30.2%). Other notable compounds included citronellal (9.6%), isoneral (5.5%), and camphor (4.9%). The identified compounds were classified into five major categories: oxygenated monoterpenes (90.9%), monoterpene hydrocarbons (4.5%), oxygenated sesquiterpenes (1.9%), and sesquiterpene hydrocarbons (1.4%); again, no diterpene compounds were detected.
Antibacterial Activity of Essential Oils Against Foodborne Pathogens
Foodborne pathogens pose significant threats to public health and food safety, often leading to food spoilage and economic losses. The rise of antibiotic-resistant bacteria has further increased the need for natural antimicrobial alternatives. Despite the aromatic properties of C. insularimontanum, its antimicrobial potential—particularly that of essential oils extracted from its leaves, twigs, and fruits—remains underexplored. Therefore, to evaluate the antibacterial activity of C. insularimontanum against foodborne pathogens, six bacterial strains were tested: B. cereus, S. aureus, S. epidermidis, E. coli, E. aerogenes, and V. parahaemolyticus. Antibacterial activity was assessed using the disk diffusion method and serial dilution method. Finally, the IZ, MIC, and MBC of the essential oils were compared.
The results indicated that the fruit essential oil exhibited the strongest antibacterial activity (Table 2), showing potent effects against all six tested bacterial strains. For Gram-positive bacteria (B. cereus, S. aureus, and S. epidermidis), the fruit essential oil achieved 100% inhibition, with an inhibition zone measuring 85 mm in diameter. The MIC and MBC values were 32 and 64 μg/mL, respectively.
Antibacterial Effects of C. insularimontanum Essential Oils and Citral on Foodborne Pathogens.
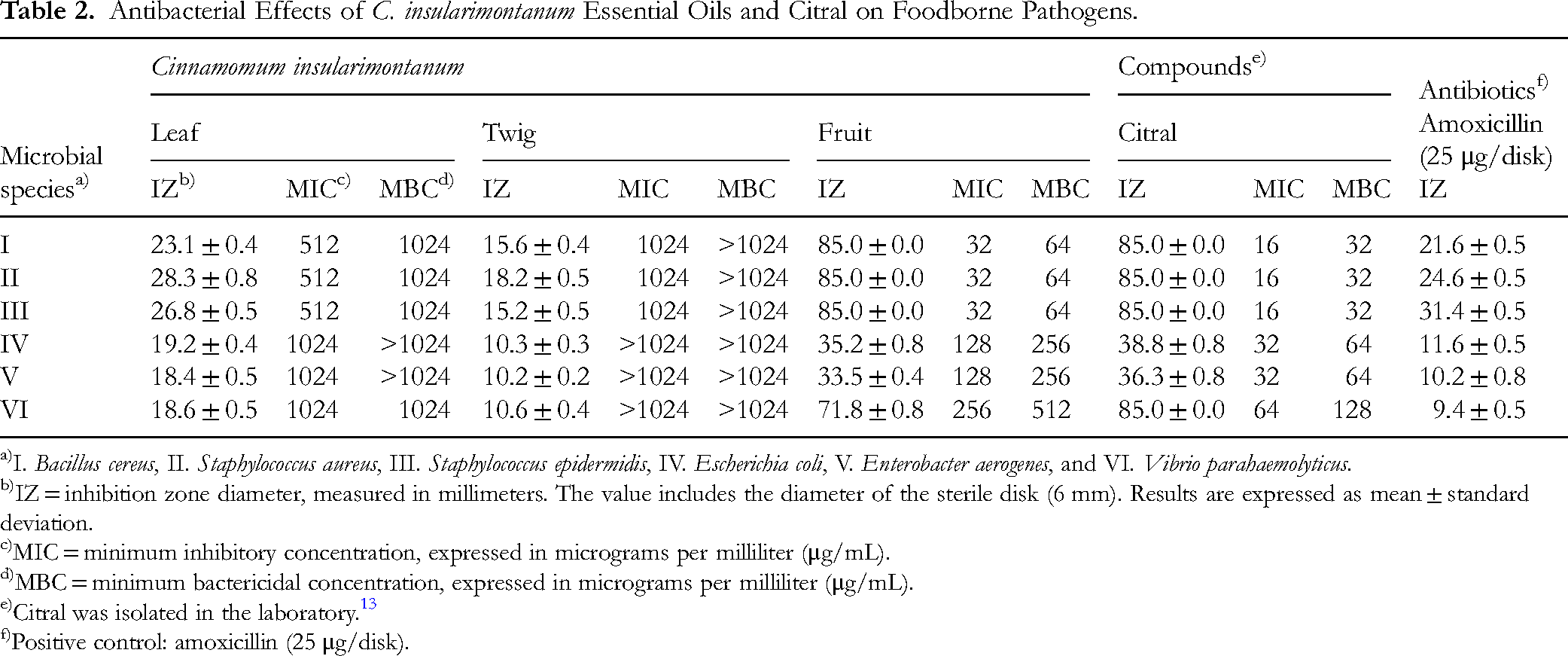
I. Bacillus cereus, II. Staphylococcus aureus, III. Staphylococcus epidermidis, IV. Escherichia coli, V. Enterobacter aerogenes, and VI. Vibrio parahaemolyticus.
IZ = inhibition zone diameter, measured in millimeters. The value includes the diameter of the sterile disk (6 mm). Results are expressed as mean ± standard deviation.
MIC = minimum inhibitory concentration, expressed in micrograms per milliliter (μg/mL).
MBC = minimum bactericidal concentration, expressed in micrograms per milliliter (μg/mL).
Citral was isolated in the laboratory. 13
Positive control: amoxicillin (25 μg/disk).
To further investigate the active components responsible for this bioactivity, the major constituent of the fruit essential oil, citral, was also evaluated. As shown in Table 2, citral demonstrated strong antibacterial activity across all tested strains, supporting its key role in the observed effects of the fruit essential oil.
Antifungal Activity of Essential Oils Against Foodborne Pathogens
To assess the antifungal activity of C. insularimontanum essential oils against foodborne pathogenic fungi, five fungal strains were tested: A. flavus, A. niger, C. globosum, P. citrinum, and T. viride. The oils’ MIC and IC50 values were determined.
As shown in Table 3, the fruit essential oil exhibited the strongest antifungal activity, followed by the leaf and twig essential oils. For the fruit essential oil, the MIC values against the five aforementioned fungal strains were 128, 128, 64, 256, and 512 μg/mL, respectively. This high antifungal activity of the fruit essential oil was primarily attributable to its high citral content. At a concentration of 256 μg/mL, citral was found to completely inhibit the growth of all five fungal strains.
MIC and IC50 Values (μg/mL) of C. insularimontanum Essential Oils and Citral Against Five Foodborne Pathogenic Fungi.

MIC = minimum inhibitory concentration, expressed in micrograms per milliliter (μg/mL).
IC50 = half-maximal inhibitory concentration, expressed in micrograms per milliliter (μg/mL).
Citral was isolated in the laboratory. 13
Positive control: Nystatine.
Antioxidant Activity
The antioxidant properties of essential oils extracted from C. insularimontanum leaves, twigs, and fruits were evaluated using four in vitro assays: β-carotene–linoleic acid bleaching assay, 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assay, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) radical cation scavenging assay, and cupric-ion-reducing antioxidant capacity (CUPRAC) assay. In all assays, α-tocopherol and butylated hydroxyanisole (BHA) served as reference antioxidants.
The β-carotene–linoleic acid bleaching assay was used to assess the antioxidant potential of natural extracts. The results indicated that the fruit essential oil had the strongest inhibitory effect on lipid peroxidation, with an IC50 value of 138.9 ± 1.5 μg/mL (Table 4). The leaf and twig essential oils exhibited markedly lower antioxidant activity, with IC50 values exceeding 1000 μg/mL. These findings suggested that the fruit essential oil had stronger free-radical-scavenging and lipid-peroxidation-inhibiting effects compared with the leaf and twig oils.
Antioxidant Effects of C. insularimontanum Essential Oils and Citral.

Citral was isolated in the laboratory. 13
Positive control: α-Tocopherol, BHA.
abcde Means in a row without a common superscript letter different (P ≤ .05; Tukey's HSD test).
To further assess free-radical-scavenging capacity, DPPH and ABTS assays were conducted. The results indicated that the fruit essential oil had considerably stronger radical-scavenging effects than did the leaf and twig oils, with IC50 values of 92.6 ± 0.5 μg/mL in the DPPH assay and 105.2 ± 0.5 μg/mL in the ABTS assay. By contrast, the leaf and twig essential oils exhibited minimal scavenging activity, with IC50 values again exceeding 1000 μg/mL in both assays.
A CUPRAC assay was used to evaluate the reducing power of the essential oils. The results indicated that the fruit essential oil exhibited a moderate electron-donating ability, reflected by an A0.50 value of 203.2 ± 0.5 μg/mL. However, the leaf and twig essential oils had a low reducing capacity, with A0.50 values exceeding 1000 μg/mL.
Citral was identified as the primary compound contributing to the antioxidant effect of the fruit essential oil. This compound exhibited strong antioxidant activity in all four assays, yielding IC50 values of 86.2 ± 0.5, 66.7 ± 0.5, and 69.5 ± 0.2 μg/mL in the β-carotene–linoleic acid bleaching, DPPH, and ABTS assays, respectively. In the CUPRAC assay, citral demonstrated an A0.50 value of 103.5 ± 0.2 μg/mL. These results confirmed that citral was the primary contributor to the antioxidant properties of the fruit essential oil.
Tyrosinase-Inhibiting Activity
Tyrosinase is a key enzyme involved in melanin synthesis, and its overactivity is associated with hyperpigmentation and oxidative stress–related conditions. Therefore, tyrosinase inhibitors are widely sought after for applications in cosmetics and skin-whitening formulations, as well as for preventing enzymatic browning in food products. In this study, tyrosinase-inhibiting activity was evaluated by measuring the conversion of L-tyrosine to L-DOPA and its subsequent oxidation to DOPA quinone. The rate of this enzymatic reaction reflects the inhibitory effect of the tested samples.
Essential oils extracted from the leaves, twigs, and fruits of C. insularimontanum were assessed for their tyrosinase-inhibiting activities. As shown in Figure 1, the fruit essential oil exhibited the strongest inhibitory effect, with inhibition increasing in a concentration-dependent manner. The IC50 value of the fruit oil was determined to be 137.4 ± 4.1 μg/mL. Citral, the major constituent of the fruit essential oil, was also tested and demonstrated a significantly stronger inhibitory effect, with an IC50 value of 65.8 ± 2.1 μg/mL. These results suggest that citral plays a central role in the tyrosinase-inhibiting activity of the fruit essential oil.
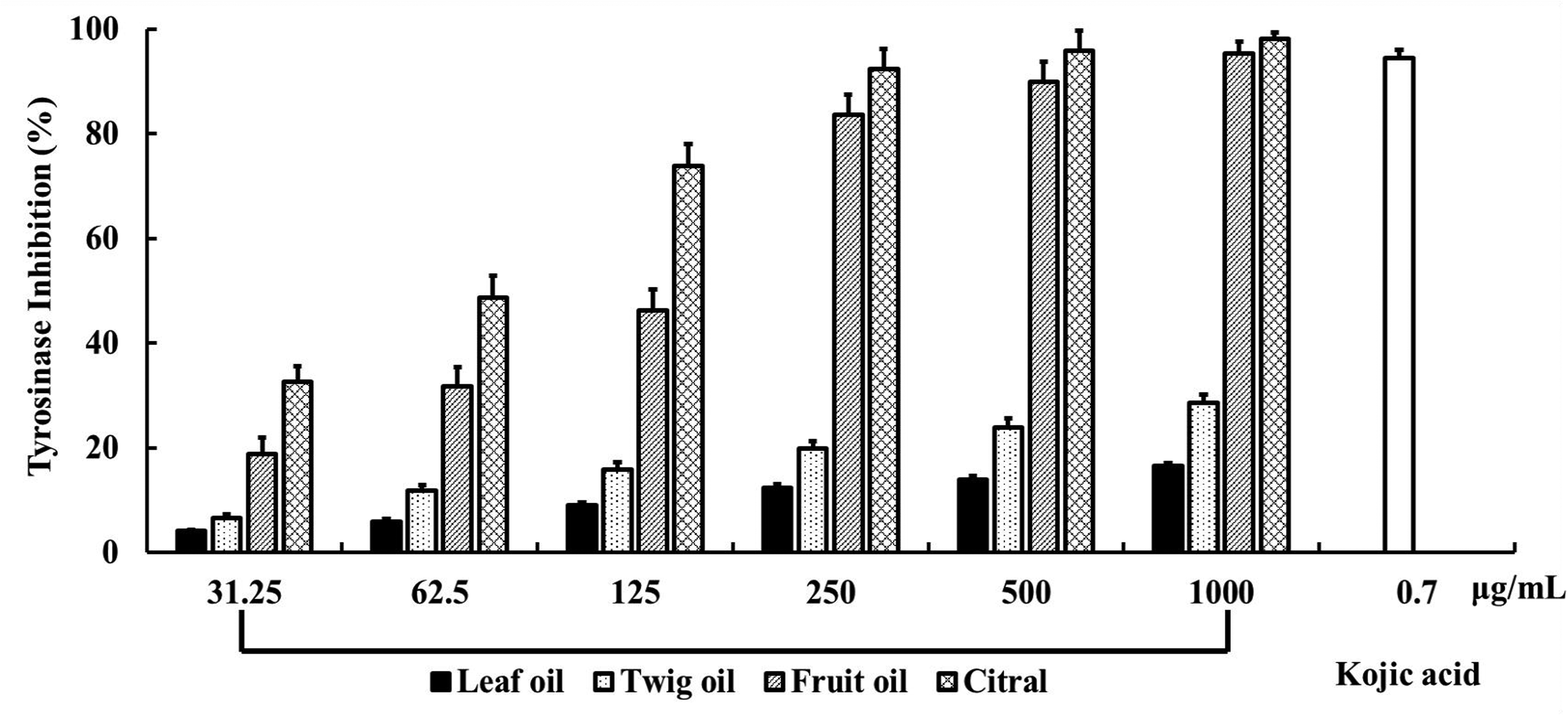
Tyrosinase-Inhibiting Activity of C. insularimontanum Essential Oils and Citral. Note: Kojic acid served as the positive control.
Discussion
Components of Essential Oils
Firstly, regarding the chemical composition of C. insularimontanum leaf essential oil, Luo 31 reported α-pinene (28.2%), camphene (11.4%), β-pinene (8.8%), linalool (10.9%), l-bornyl acetate (9.1%), and caryophyllene oxide (8.6%) as the major constituents, whereas Chen et al 9 identified 1,8-cineole (35.94%), α-pinene (7.55%), α-eudesmol (6.17%), sabinene (5.06%), and isobornyl acetate (4.81%) as the predominant compounds. These discrepancies may be attributed to geographic variation, environmental factors, or differences in plant chemotypes.
In contrast, for the twig essential oil, a thorough review of the literature revealed no previous reports on the chemical composition of C. insularimontanum twigs, indicating that the present study is the first to characterize its volatile profile.
As for the fruit essential oil, to the best of our knowledge, only Lin et al 10 have examined its chemical composition. They identified citral (35.89%), citronellal (24.64%), citronellol (16.78%), α-pinene (9.45%), β-pinene (4.30%), limonene (1.76%), and camphene (1.70%) as the major components. Our findings are generally consistent, as both studies identified citral as the predominant constituent. Based on this compositional analysis, the high citral content in the fruit essential oil prompted further investigation into its biological activities, beginning with its antimicrobial potential.
In addition, this study compared the essential oil compositions extracted from the leaves, twigs, and fruits of C. insularimontanum with those of other Cinnamomum species based on available literature (Table 5). For the leaf essential oil, only C. camphora,32-34 in which camphor was reported as a major constituent, exhibited partial similarity in composition to that identified in this study. In contrast, other species such as C. glaucescens, 35 C. micranthum, 36 C. subavenium, 37 C. tamala, 38 C. verum, 39 and C. zeylanicum40-42 showed markedly different chemical profiles. Similarly, the twig essential oil composition of C. insularimontanum was notably different from those previously reported for C. cassia, 43 C. micranthum, 36 and C. osmophloeum. 44 As for the fruit essential oil, it also exhibited significant differences from those of C. camphora, 32 C. glaucescens,34,45 and C. zeylanicum. 46
Comparison of Major Essential Oil Compositions from Various Plant Parts of Cinnamomum insularimontanum with Other Cinnamomum Species.
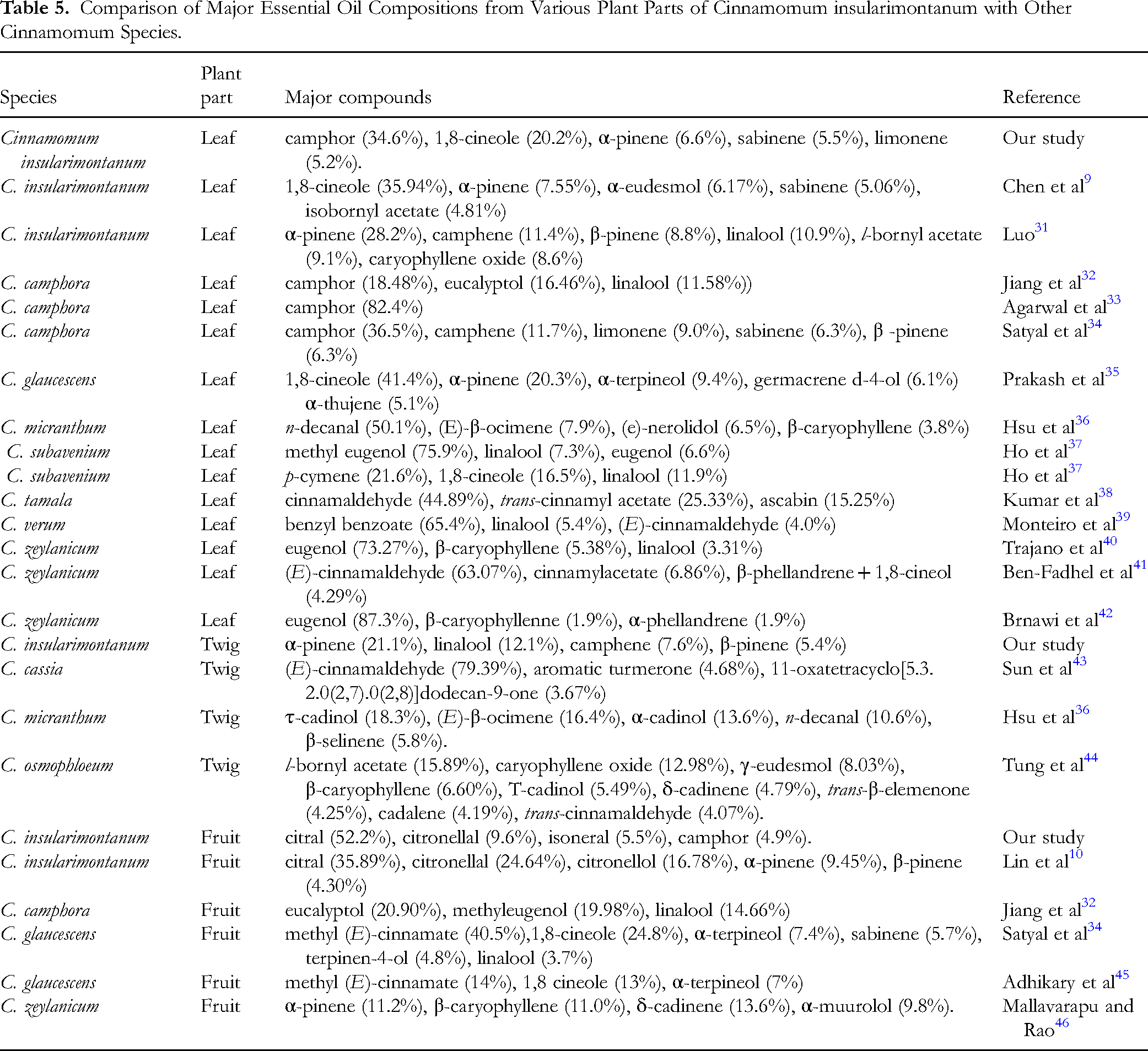
Although this comparative analysis is limited to currently available literature, the essential oils derived from the three parts of C. insularimontanum consistently demonstrated distinct chemical characteristics compared to other reported Cinnamomum species. These findings suggest that C. insularimontanum may represent a unique chemotype within the genus, with promising potential for applications in natural antimicrobial agents, skincare formulations, and other functional product development.
Antibacterial and Antifungal Activities
Among the essential oils extracted from C. insularimontanum, the fruit essential oil showed the most potent antimicrobial activity against both foodborne bacteria and fungi. Further investigation confirmed that this effect was largely due to its high concentration of citral.
Previous research has demonstrated that citral, a monoterpene aldehyde, exerts antimicrobial effects through multiple mechanisms; it cross-links amino groups in bacterial structures, inhibits thiol-containing enzymes, and disrupts the integrity of membranes, leading to intracellular leakage and cell death.47-49 At high concentrations, citral induces cytoplasmic coagulation, disrupts metabolism, and causes oxidative DNA damage, compromising cell survival.50,51 It also alters the lipid composition of bacterial membranes, increasing the concentration of saturated fatty acids and reducing membrane fluidity and stability. 26 Notably, unsaturated α,β-aldehydes exhibit broad-spectrum antibacterial activity against both Gram-positive and Gram-negative bacteria. 52
Regarding antifungal effects, citral's activity has been attributed to a combination of synergistic mechanisms, including disruption of fungal cell membranes, the induction of oxidative stress, the inhibition of key enzyme activity, and the interference with fungal gene expression.53-55
Antioxidant Activity
Given citral's characteristic α,β-unsaturated aldehyde structure and well-documented redox activity, we further investigated the antioxidant potential of the essential oils. The results showed that the fruit essential oil of C. insularimontanum had a considerably higher antioxidant capacity than the leaf and twig essential oils did, primarily because of its high citral content.
Previous studies have shown that citral exerts antioxidant effects through multiple mechanisms, including free radical stabilization, metal ion reduction, inhibition of lipid peroxidation, and disruption of oxidative chain reactions.56-58 Citral effectively stabilizes DPPH radicals, superoxide anions, and hydroxyl radicals by donating hydrogen atoms. Additionally, its conjugated double bonds enhance resonance stabilization, further improving free radical stability.56,59
Moreover, citral reduces transition metal ions such as iron and copper, thereby inhibiting the formation of metal-catalyzed free radicals and reducing oxidative stress.56,60 By disrupting free radical chain reactions, citral limits the continuous generation and propagation of reactive species, which in turn mitigates oxidative damage and lowers the risk of chronic diseases such as cancer, cardiovascular disorders, and neurodegenerative conditions.61,62
In summary, the antioxidant properties of C. insularimontanum fruit essential oil and citral support their potential application as natural antioxidants in food preservation and health-related industries.
Tyrosinase-Inhibiting Activity
Since oxidative stress is closely associated with melanin production, we further evaluated the tyrosinase-inhibiting activity of the essential oils and citral.
Tyrosinase inhibitors have gained increasing attention for their potential applications in cosmetic whitening products and the treatment of hyperpigmentation-related disorders.63,64 In the present study, we demonstrated that the fruit essential oil of C. insularimontanum possesses superior tyrosinase-inhibiting activity compared with other reported plant-derived essential oils. For example, the twig oil of Taiwan incense cedar (Calocedrus macrolepis var. formosana) showed an IC50 of 168.5 ± 3.2 μg/mL, 15 and that Eucalyptus camaldulensis flower oil exhibited 30% inhibition at 26 mg/mL. 65 Compared with these oils, the fruit essential oil of C. insularimontanum exhibited stronger inhibition of melanin synthesis, emphasizing its potential as a natural depigmenting agent.
In particular, citral was identified as the principal compound responsible for the tyrosinase-inhibiting activity observed in the fruit essential oil. This result aligns with the findings of Yang et al, 66 who investigated Litsea cubeba fruit essential oil and attributed its inhibitory activity to citral. Mechanistically, citral is proposed to inhibit tyrosinase via its reactive aldehyde group, which interacts with nucleophilic residues—such as thiol, amine, or hydroxyl groups—near the enzyme's active site. These interactions may lead to Schiff base formation and steric hindrance, thereby impairing enzyme–substrate binding and ultimately reducing tyrosinase activity.
Study Limitations
This study provides valuable insights into the chemical composition and bioactivity of essential oils extracted from C. insularimontanum, underscoring their potential as natural antimicrobial, antioxidant, and tyrosinase-inhibiting agents. However, certain limitations should be acknowledged. First, the antibacterial and antifungal assessments were conducted on a limited range of foodborne pathogens and fungi, and the results may therefore not fully represent all potential microbial threats. Second, although the fruit essential oil exhibited strong tyrosinase-inhibitory activity, the precise mechanism underlying this effect remains unclear. It is hypothesized that the high citral content may contribute significantly to this activity, potentially by binding to the active site of tyrosinase or chelating copper ions essential for its catalytic function. These interactions may inhibit the hydroxylation of tyrosine and subsequent oxidation of L-DOPA, key steps in melanin biosynthesis. However, the contributions of minor constituents and potential synergistic effects warrant further investigation. Third, the stability and long-term efficacy of the essential oils—particularly under various environmental conditions of temperature, humidity, and storage duration—were not evaluated. Additionally, the interactions between the major active compound, citral, and the other essential oil constituents were not fully explored, leaving gaps in the understanding of potential synergistic or antagonistic effects. Future studies should involve a broader microbial spectrum, investigate the long-term stability of essential oils, and explore innovative methods for delivering these oils, such as microencapsulation, to enhance their effectiveness in various applications, including food preservation, cosmetics, and pharmaceuticals. In particular, further research is warranted to elucidate the detailed molecular mechanisms of tyrosinase inhibition, as well as to conduct comprehensive toxicity evaluations to assess safety for human use.
Conclusion
In this study, we extracted essential oils from the leaves, twigs, and fruits of C. insularimontanum by using hydrodistillation and analyzed their chemical composition and bioactivity. The results indicated that the major constituents of the essential oils varied by plant part: camphor and 1,8-cineole were dominant in the leaves; α-pinene and linalool were dominant in the twigs; and citral was dominant in the fruits. Among all three essential oils, the fruit essential oil exhibited the highest bioactivity and was found to have strong antibacterial effects against foodborne pathogens and strong antifungal effects, as well as favorable antioxidant and tyrosinase-inhibiting properties. Citral was identified as the key contributor to these effects. Overall, these findings suggest that C. insularimontanum essential oils, particularly fruit essential oils, have potential applications as natural preservatives and functional ingredients in food, cosmetics, and pharmaceuticals. Future studies should focus on increasing the stability of essential oils, developing formulations that have high efficacy, and assessing their long-term effectiveness in practical use.
Footnotes
Acknowledgment
The authors would like to thank the Taiwan Forestry Research Institute.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.

