Abstract
Background and Objectives
The genus Piper (family Piperaceae) includes aromatic plants widely used as spices and in traditional medicine. Essential oils from Piper species are known for their antimicrobial and pesticidal properties. This study aims to characterize the chemical profiles of the stem bark and leaf essential oils from two Vietnamese Piper species, Piper hainanense and P. thomsonii, and evaluate their biological activities.
Methods
Essential oil components were identified using gas chromatography-mass spectrometry (GC-MS). Antimicrobial activity was assessed using the broth microdilution method, while mosquito larvicidal activity was evaluated against fourth instar larvae of Aedes aegypti.
Results
The major constituents of P. hainanense essential oils were sabinene (14.4-15.9%), δ-selinene (6.7-14.6%), β-pinene (13.5-13.9%), β-selinene (5.9-12.4%), α-pinene (7.0-9.0%), and β-elemene (6.1-6.6%). In P. thomsonii stem bark essential oil, elemicin (23.8%), spathulenol (14.5%), and caryophyllene oxide (7.4%) predominated, while its leaf essential oil contained elemicin (23.0%), β-pinene (15.6%), and γ-elemene (13.6%). Antimicrobial assays revealed that P. thomsonii essential oils exhibited strong antifungal activity against Aspergillus niger (MIC = 32 µg/mL). P. hainanense essential oils demonstrated strong mosquito larvicidal activity, with 24-h LC50 values of 26.72–32.57 µg/mL and LC90 values of 34.15–43.48 µg/mL.
Conclusion
P. hainanense and P. thomsonii essential oils exhibit potential as natural agents for creating antimicrobial medications and mosquito-control strategies. These results serve as a foundation for additional investigation into the medicinal and insecticidal uses of essential oils from Piper.
Introduction
Piper, or pepper, is a genus of about 2000 herbaceous vines or small trees in the family Piperaceae. 1 They are frequently found in the understory of lowland tropical forests but also occur in clearings and higher-elevation life areas. 1 Due to its pungency and ability to lend a unique flavor to a variety of cuisines worldwide, pepper is the most often used spice and condiment in the world. Originally used primarily as a spice and medicinal plant, its uses have expanded to include fresh and processed vegetables, spices, dried forms, food coloring, ornamental plant breeding, and the creation of extracts for the pharmaceutical and cosmetics industries. 2 Among species, black pepper (P. nigrum L.) can be seen as the “King of spices”. 2 Alkaloid derivatives, together with flavonoids, amides, phenolic acids, lignans, and terpenoids, are the main secondary metabolites found in Piper crude extracts. 3
It was noted that Piper species are a good reservoir of essential oils, and monoterpene hydrocarbons, sesquiterpene hydrocarbons, and their oxygenated derivatives were the principal classes. 4 Essential oil from Cuban P. aduncum aerial parts were reported to contain camphor (17.1%), viridiflorol (14.5%), and piperitone (23.7%). 5 β-Caryophyllene achieved the highest percentages of 10.9–11.2% in essential oils of Malaysian P. officinarum leaves and stems. 6 Bicyclogermacrene (21.88%)/β-caryophyllene (20.69%) and myrcene (52.60%)/linalool (15.89%) were the main constituents in essential oil from the leaves of Brazilian P. cernuum and P. regnellii, respectively. 7 These oil samples showed antimicrobial activity against Staphylococcus aureus and Candida albicans with inhibition zones of 12–15 mm. 7 The leaf essential oils from other Brazilian Piper species P. permucronatum and P. hostmanianum showed larvicidal activity against third-instar larvae of Ae. aegypti mosquitoes with LC50 values of 36 and 54 µg/mL, respectively. 8 Piper essential oils have also been documented to possess antiprotozoal, antinociceptive, anti-inflammatory, and cytotoxic activities. 1
Piper hainanense Hemsl. is native to Hainan, China, and Vietnam.9,10 It grows to a height of 2–4 meters, has pink leaves, and a distinctive aroma. It was used in traditional medicine for headaches, dizziness, and shortness of breath. Phytochemical separation revealed that the main constituents of this species were cytotoxic benzenoids. 9 Piper thomsonii (C.DC.) Hook.f. is a climbing shrub, and grows primarily in the wet tropical biome of India, China, and Indo-China. 10 This communication briefly describes the chemical profiles of essential oils from the fresh stem barks and leaves of these two Piper species, collected from Central Vietnam. The studied essential oils have been investigated for their potential in antimicrobial experiments against the Gram bacteria, fungi, and yeast, and mosquito larvicidal activity against fourth instar larvae of Ae. aegypti mosquitoes.
Materials and Methods
Plant Materials
Piper hainanense fresh stem barks and leaves were collected in March 2023 from Chauly, Pu Huong Natural Reserve, Nghean, Vietnam (19°11′57″N, 105°60′14″E). P. thomsonii fresh stem barks and leaves were gathered in April 2022 from Dongvan, Pu Hoat Natural Reserve, Nghean, Vietnam (19°48′28″'N, 105°40′54″E). Plant identification was confirmed by co-author Dr Do Ngoc Dai, and voucher specimens (11-PH for P. hainanense and 967-PT for P. thomsonii) were deposited at Nghe An College of Economics.
Due to differences in plant metabolism, growth stages, and environmental factors, the essential oil content may be impacted by the collections’ March and April dates, which coincide with these species’ growing seasons in Vietnam. Temperature, humidity, soil composition, and sunlight levels during these times can all affect the yield and chemical makeup of the essential oils. The biosynthesis of terpenoids and other secondary metabolites can be greatly impacted by these environmental factors, even though thorough investigations of seasonal variation were not carried out.
Hydro-Distillation of Essential Oils
The fresh stem barks and leaves were carefully washed and sliced into small pieces. These sliced materials have been subjected to hydro-distillation using a Clevenger-type apparatus with a duration of 3.5 h. These actions resulted in essential oils, characterized by yellow color. The extraction yields were 0.24, 0.22, 0.31, and 0.27% v/w (fresh weight) for the stem barks and leaves of P. hainanense and P. thomsonii, respectively. To eliminate any remaining water content, the gathered essential oils were dried over Na2SO4. The oils were then meticulously kept at 5 °C to preserve their integrity for further examinations.
The GC-MS Analysis
The GC-MS analytical procedure was conducted using a Shimadzu Technologies GCMS-QP2010 Plus chromatograph equipped with a fused silica Equity-5 capillary column (30 m length, 0.25 mm diameter, 0.25 μm film thickness, Supelco, USA). 11 The analytical conditions were optimized to ensure accurate and efficient separation of essential oil components, which are primarily volatile and thermally sensitive compounds. The carrier gas helium was chosen for its inert nature and stable flow rate (1.2 mL/min) to maintain consistent chromatographic performance. The injector and interface temperatures were set at 280 °C to ensure efficient volatilization and transfer of analytes. The column temperature program, ranging from 60 °C to 280 °C with gradual increases (5 °C/min), was designed to provide sufficient resolution of both low- and high-boiling-point compounds while minimizing thermal degradation. The split ratio of 10:1 was applied to balance sample concentration and column performance, preventing column overload while ensuring adequate sensitivity. The MS parameters, including an ionization voltage of 70 eV and a scanning mass range of 50–500 amu, were selected to achieve reliable fragmentation patterns for compound identification. Retention indices (RI) were determined by co-injecting compounds with a homologous series of n-alkanes (C8–C38) for accurate peak identification. Compound identification was based on comparisons with reference libraries (NIST 11 and WILEY 7), the Adams book, 12 and the NIST Chemistry WebBook. 13 The relative percentages of identified compounds were calculated from GC peak areas without correction factors, ensuring a reliable quantitative profile.
Antimicrobial Assay
Ten microbial strains were selected from the ATCC (American Type Culture Collection, USA) based on their relevance as model organisms representing Gram-positive bacteria (Bacillus subtilis ATCC 5230, Staphylococcus aureus ATCC 33591, Clostridium sporogenes ATCC 7955), Gram-negative bacteria (Escherichia coli ATCC 8739, Pseudomonas aeruginosa ATCC 27853), fungi (Aspergillus brasiliensis ATCC 9642, A. niger ATCC 9587, Fusarium oxysporum ATCC 11739), and yeasts (Candida albicans ATCC 12354, Saccharomyces cerevisiae ATCC 4078). These strains were chosen due to their clinical and industrial significance, providing a comprehensive evaluation of the essential oils’ antimicrobial spectrum. Positive controls included standard antibiotics (streptomycin and tetracycline for bacteria, nystatin for fungi and yeasts) to validate assay reliability. Negative controls consisted of media without essential oil to account for background microbial growth. Each assay was conducted in triplicate.14,15
Mosquito Larvicidal Assay
The mosquito species Ae. aegypti was selected due to its global significance as a vector for diseases such as dengue, Zika, and chikungunya, making it a priority target for vector control strategies. 16 The reporting of this study conforms to ARRIVE 2.0 guidelines. 17 Mosquito eggs were hatched in tap water overnight, and the larvae were fed on a mixture of yeast and cat food (1:3 w/w). Daily, the water was replaced with fresh overnight tap water, and dead larvae were removed. Adult mosquitoes were fed on a 10% glucose solution and sucked blood from white mice. All stages of mosquito development were placed under laboratory conditions: temperature 25 °C, relative humidity 70 ± 5%, 12 h/12 h light/dark cycle. The larvicidal activity was assessed using a previously described protocol, where aliquots of essential oils were added to water containing 20 fourth-instar larvae. Permethrin served as the positive control (its detailed doses were in Table S1), while a separate set of controls consisting solely of ethanol functioned as the negative control. Mortality rates were recorded at 24-h and 48-h post-exposure, during which no dietary supplements were provided. The trials were conducted at a controlled temperature of 25 ± 2 °C. The test was performed with five different concentrations (100, 50, 25, 12.5, and 6 µg/mL). The dead larvae were enumerated, and the average percentage mortality was subsequently calculated.
All tests were conducted at a controlled temperature of 25 ± 2 °C to minimize variability. Five concentrations of essential oils (100, 50, 25, 12.5, and 6 µg/mL) were tested in triplicate, and average mortality rates were calculated. Mortality data were analyzed using log-probit analysis (Minitab® 19), providing robust estimates of LC50 and LC90 values with 95% confidence intervals to ensure statistical reliability.
Results
Chemical Profile of Essential Oils
The hydro-distillation of Piper hainanense stem barks produced a yellow essential oil with a yield of 0.24% v/w. A total of 40 compounds were identified, accounting for 99.6% of the oil (Table 1 and Fig. S1). Monoterpene hydrocarbons (46.8%) and sesquiterpene hydrocarbons (33.3%) dominated the chemical profile, followed by non-terpenic compounds (8.8%), oxygenated sesquiterpenes (6.7%), and oxygenated monoterpenes (4.0%). The major components were sabinene (14.4%), β-pinene (13.9%), α-pinene (7.0%), δ-selinene (6.7%), β-elemene (6.1%), and β-selinene (5.9%).
Chemical Compositions of the Studied Essential Oils (%).
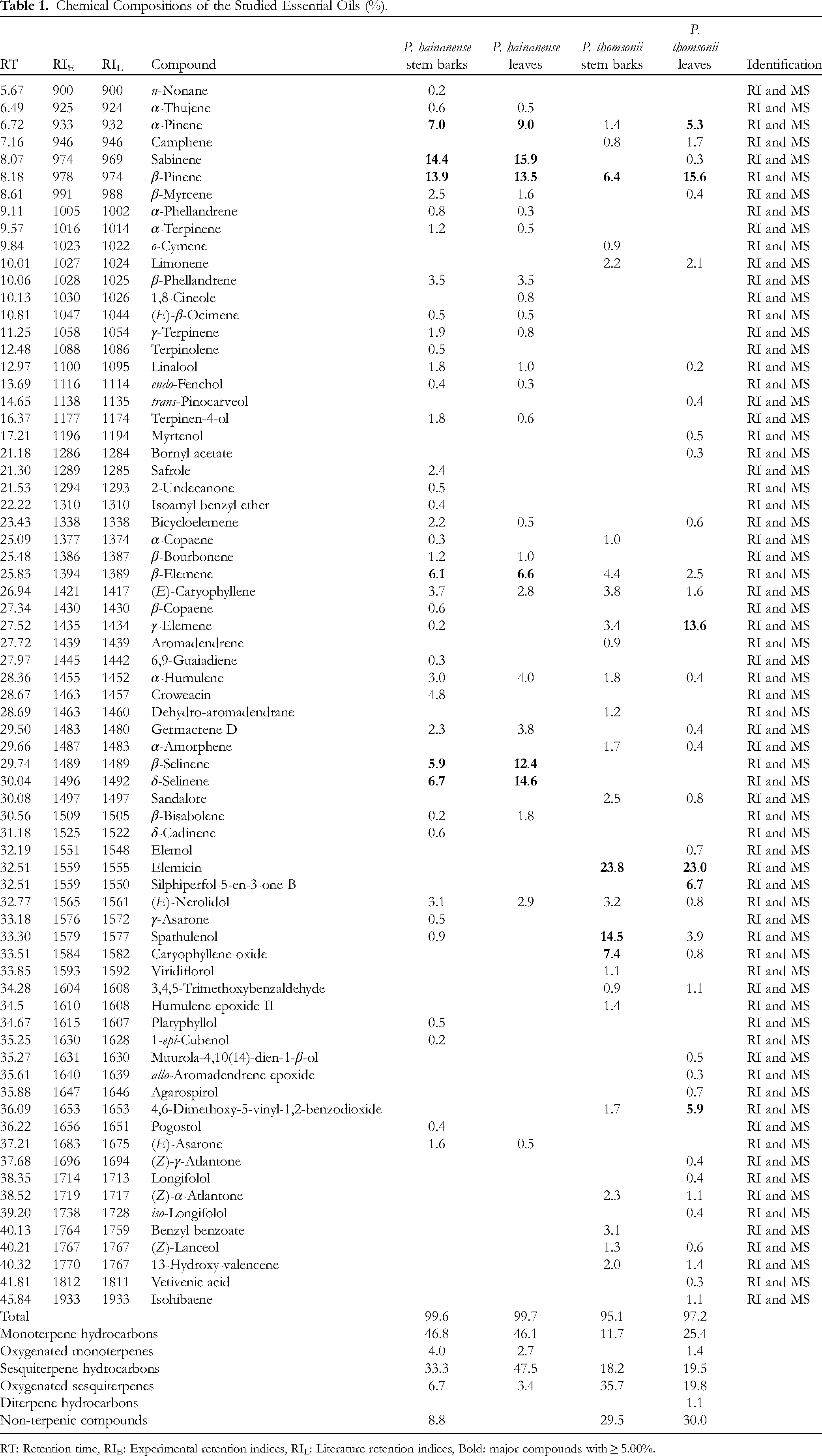
RT: Retention time, RIE: Experimental retention indices, RIL: Literature retention indices, Bold: major compounds with ≥ 5.00%.
The extraction of P. hainanense leaves also resulted in yellow essential oil, yielding 0.22% v/w. A list of 25 identified compounds was tabulated in Table 1 and Fig. S2, representing 99.7%. The main chemical classes were monoterpene hydrocarbons (46.1%) and sesquiterpene hydrocarbons (47.5%), whereas their oxygenated derivatives accounted for 2.7 and 3.4%, respectively. The studied sample was characterized by sabinene (15.9%), δ-selinene (14.6%), β-pinene (13.5%), β-selinene (12.4%), α-pinene (9.0%), and β-elemene (6.6%). Some other compounds were associated with significant percentages, such as α-humulene (4.0%), β-phellandrene (3.5%), (E)-nerolidol (2.9%), and (E)-caryophyllene (2.8%).
The GC-MS data of essential oil (yellow color, 0.31% yield, v/w) from P. thomsonii stem barks contained 26 identified compounds, corresponding to 95.1% (Table 1 and Fig. S3). Four chemical classes consisted of oxygenated sesquiterpenes (35.7%), non-terpenic compounds (29.5%), sesquiterpene hydrocarbons (18.2%), and monoterpene hydrocarbons (11.7%). Elemicin (23.8%), spathulenol (14.5%), caryophyllene oxide (7.4%), and β-pinene (6.4%) were the principal compounds.
The hydro-distillation of P. thomsonii fresh leaves produced a yellow essential oil with a yield of 0.27 v/w. From the GC-MS analysis, 37 compounds were identified, which represented 97.2% (Table 1 and Fig. S4). The main chemical classes comprised non-terpenic compounds (30.0%), monoterpene hydrocarbons (25.4%), oxygenated sesquiterpenes (19.8%), and sesquiterpene hydrocarbons (19.5%). Meanwhile, oxygenated monoterpenes were the remaining class with 1.4%. This leaf essential oil was characterized by elemicin (23.0%), β-pinene (15.6%), γ-elemene (13.6%), silphiperfol-5-en-3-one B (6.7%), 4,6-dimethoxy-5-vinyl-1,2-benzodioxide (5.9%), and α-pinene (5.3%). Some others were also significant, such as spathulenol (3.9%), β-elemene (2.5%), and limonene (2.1%).
Antimicrobial Activity
Four studied essential oils have been subjected to antimicrobial activity, and the result is outlined in Table 2. P. hainanense stem bark essential oil only showed moderate activity against the fungus A. niger with the MIC value of 128 µg/mL, but failed to inhibit the remaining microbial strains (MIC > 256 µg/mL). P. hainanense leaf essential oil controlled the growth of the Gram (+) bacterium B. subtilis and the yeast S. cerevisiae with the same MIC value of 64 µg/mL, compared to that of the standards streptomycin (MIC 4 µg/mL) and nystatin (MIC 8 µg/mL). This sample was found to inhibit the Gram (+) bacterium C. sporogenes, the Gram (–) bacteria E. coli and P. aeruginosa, the fungi A. niger, and F. oxysporum, and the yeast C. albicans with the same MIC value of 128 µg/mL, but it did not show activity against the Gram (+) bacterium S. aureus and the fungus A. brasiliensis.
Antimicrobial Activity of the Studied Essential Oils.
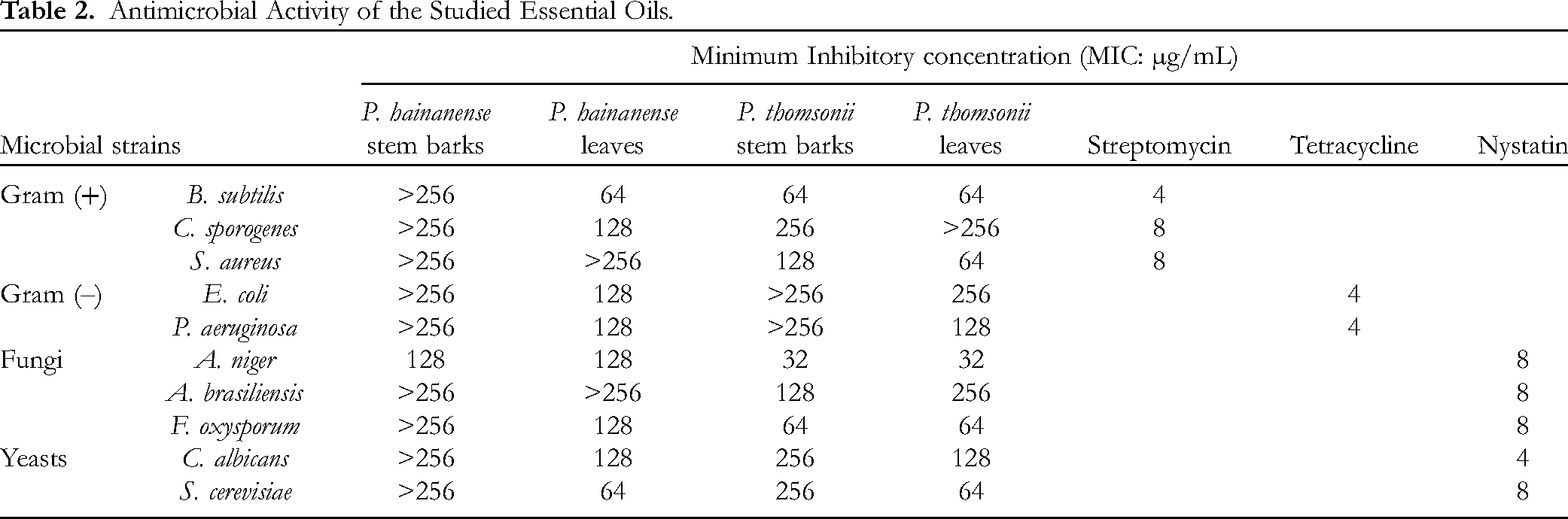
P. thomsonii stem bark essential oil suppressed the proliferation of the Gram (+) bacteria B. subtilis, C. sporogenes, and S. aureus with the corresponding MIC values of 64, 256, and 128 µg/mL, when the leaf essential oil exerted MIC values of 64, > 256, and 64 µg/mL, respectively. The stem bark essential oil was inactive against two tested Gram (–) bacteria, whereas the leaf essential oil exerted MIC values of 128–256 µg/mL. Both studied essential oils exhibited the same MIC values of 32 and 64 against the fungi A. niger and F. oxysporum, respectively. The stem bark sample outstripped the leaf sample in the antimicrobial assay against the fungus A. brasiliensis. In contrast, the leaf essential oil was superior to the stem bark essential oil against two yeasts C. albicans and S. cerevisiae (Table 2).
Mosquito Larvicidal Activity
The studied essential oils have been further taken into mosquito larvicidal consideration against Ae. aegypti fourth instar larvae. For Table 3, essential oils from P. hainanense stem barks and leaves were associated with 24-h LC50 values of 26.74–32.57 µg/mL and 24-h LC90 values of 34.15–43.38 µg/mL. In the meantime, the larvicidal activity of P. thomsonii stem bark and leaf essential oils was accompanied by 24-h LC50 values of 50.03–55.12 µg/mL and 24-h LC90 values of 77.08–80.13 µg/mL.
Mosquito Larvicidal Activity of the Studied Essential Oils (LC50 and LC90: µg/mL).

LC50: 50% Lethal concentration, LC90: 90% Lethal concentration.
Considering the 48-h treatment, LC50 values of 26.37–31.08 µg/mL and LC90 values of 34.34–41.35 µg/mL were assigned to P. hainanense stem bark and leaf essential oils. P. thomsonii stem bark and leaf essential oils were responsible for LC50 values of 48.94–51.54 µg/mL and LC90 values of 62.16–76.94 µg/mL.
Discussion
There is a difference in chemical profiles between essential oils from P. hainanense stem barks and leaves. Various compounds were found in one sample. For instance, non-terpenic compounds were only found in the stem bark essential oil. β-Selinene and δ-selinene in the leaf essential oil were higher two times than those of the stem bark essential oil. Regarding P. thomsonii essential oils, monoterpene hydrocarbons were higher in the leaf essential oil, whereas oxygenated sesquiterpenes in the stem bark essential oil were higher. In addition, oxygenated monoterpenes and diterpene hydrocarbons were only found in the leaf essential oil. Spathulenol and caryophyllene oxide were higher in the stem bark essential oil, while α-pinene, β-pinene, γ-elemene, and 4,6-dimethoxy-5-vinyl-1,2-benzodioxide predominated in the leaf essential oil.
This is the first time that essential oils from Vietnamese P. hainanense were chemically analyzed. Hao et al (2018) reported that β-elemene (10.49%), β-pinene (8.37%), (E)-β-farnesene (7.23%), and α-pinene (7.14%), were identified as the main compounds of P. hainanense leaf essential oil, collected from Hainan island, China. 18 The current study matches well with the previous publications since monoterpenes, sesquiterpenes, and their oxygenated derivatives were also predominant in other Central Vietnamese Piper essential oils. As an example, P. laosanum leaf essential oil collected from Thanhhoa, Vietnam, contained α-curcumene (12.0%), germacrene D (6.3%), sabinene (6.1%) and spathulenol (5.1%). 19 γ-Elemene (12.7%), valeranone (9.3%), and ishwarone (6.0%) were the major compounds in essential oil from P. betle f. densum leaves, another species derived from Pu Hoat Natural Reserve. 20
The dominance of compounds such as sabinene, elemicin, and β-pinene suggests that these species have evolved to produce specific metabolites with strong ecological and biological roles. The higher levels of oxygenated sesquiterpenes in P. thomsonii stem bark oil might be linked to its defensive strategies against microbial pathogens or herbivores. Significant findings, such as the presence of silphiperfol-5-en-3-one B, highlight the potential for discovering unique bioactive compounds in these essential oils, which could have implications for antimicrobial and pesticidal applications.
In general, P. thomsonii essential oils were better than P. hainanense essential oils in antimicrobial treatment, as well as the leaf essential oil is superior to the stem bark essential oil. Especially, P. thomsonii essential oils showed strong activity against the fungus A. niger. The current result matches well with previous reports since Piper essential oils were known for bacterial inhibitions. P. cernuum leaf essential oil, gathered from Brazil in autumn, generated the MIC value of 48 µg/mL against B. subtilis. 21 Essential oils from the aerial parts of Malaysian P. erecticaule oil exhibited the best activity on A. niger (MIC 31.3 μg/mL), followed by P. lanatum aerial part essential oil (MIC 62.5 μg/mL). 22 P. minutistigmum is a species found in Pu Mat National Park, Nghean, Vietnam. Its leaf and stem bark essential oils exhibited strong activity against three Gram (+) bacteria Enterococcus faecalis, S. aureus, and B. cereus with MIC values of 16–64 µg/mL. 23
A criterion can be applied to mosquito larvicidal activity: strong, LC50 ≤ 50 µg/mL; moderate, 50 < LC50 ≤ 100 µg/mL; weak, 100 < LC50 ≤ 750 µg/mL, and inactive, LC50 > 750 µg/mL. 24 By this means, essential oils from P. hainanense stem barks and leaves showed strong activity, whereas essential oils from P. thomsonii stem barks and leaves were moderate. Previously, there have many attempts using Piper essential oils in larvicidal activity. Essential oils from the aerial parts of P. caninum, P. longum, P. montium, and P. mutabile, collected from Central Vietnam, exhibited good larvicidal activity against Ae. aegypti with 24-h LC50 and 24-h LC90 values less than 10 µg/mL. 25 As mentioned above, the leaf essential oils of Brazillian P. permucronatum and P. hostmanianum showed larvicidal activity against third-instar larvae of Ae. aegypti mosquitoes with 24-h LC50 values of 36 and 54 µg/mL, respectively. 8 Besides the antimicrobial, antioxidant, and tyrosinase inhibitory activities, P. betle leaf essential oil originating from Taiwan was responsible for mosquito larvicidal activity against Ae. aegypti with 2-h and 24-h LD50 values of 86 and 48 ppm, respectively. 26 Collectively, the current study provides useful evidence of the use of Vietnamese Piper essential oils in two in vitro biological models. However, it still lacks in vivo molecular mechanisms of action. Chromatographic separation of the main compounds, and their biological assays are also encouraged.
Conclusions
The current study provides a detailed chemical profile of essential oils from two Vietnamese Piper species, highlighting the dominance of monoterpene hydrocarbons, sesquiterpene hydrocarbons, and their oxygenated derivatives, along with some non-terpenic compounds. The major compounds in P. hainanense essential oils included sabinene, δ-selinene, β-pinene, β-selinene, α-pinene, and β-elemene. In contrast, P. thomsonii essential oils were characterized by high levels of elemicin, spathulenol, caryophyllene oxide, γ-elemene, and silphiperfol-5-en-3-one B. Biological evaluations revealed that P. thomsonii essential oils outperformed P. hainanense in antimicrobial assays, with strong inhibition of A. niger. Meanwhile, P. hainanense essential oils demonstrated potent larvicidal activity against Ae. aegypti larvae, highlighting their potential for mosquito control applications. To investigate their contributions to the reported activities, future research should concentrate on extracting and purifying the main bioactive substances, such as elemicin, silphiperfol-5-en-3-one B, and sabinene. Confirming these substances’ and essential oils’ safety and effectiveness requires testing them in in vivo models. Further understanding of their patterns of activity may also be obtained by examining the molecular mechanisms that underlie their biological effects. The study's encouraging findings point to the possibility of using these essential oils to create natural antimicrobials or insect repellents. Partnerships with the pharmaceutical and agricultural sectors could further investigate commercial uses, such as environmentally friendly insecticides and antifungal therapies.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251324002 - Supplemental material for Piper hainanense Hemsl. and P. thomsonii (C.DC.) Hook.f.: Essential Oil Compositions, Antimicrobial and Mosquito Larvicidal Activities
Supplemental material, sj-docx-1-npx-10.1177_1934578X251324002 for Piper hainanense Hemsl. and P. thomsonii (C.DC.) Hook.f.: Essential Oil Compositions, Antimicrobial and Mosquito Larvicidal Activities by Le Thi Huong, Do Ngoc Dai, Nguyen Huy Hung, Nguyen Dinh Luyen, Ty Viet Pham, Nguyen Ngoc Linh and Ninh The Son in Natural Product Communications
Footnotes
Acknowledgements
The authors are grateful to the director boards of Pu Huong and Pu Hoat Natural Reserves for sample collections
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approach
This study was approved by the Duy Tan University Medical–Biological Research Ethics Council, number DTU/REC2020/NHH02.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

