Abstract
Objectives
This study aimed to evaluate the anti-inflammatory potential of anthocyanin-rich bilberry extract by investigating its antioxidant activity and effects on rat peritoneal macrophage inflammatory responses. The extract's ability to modulate key inflammatory markers such as nitric oxide and TNF-α, along with its predicted molecular interactions and lipoxygenase inhibition, was assessed to elucidate underlying mechanisms.
Methods
Present study examined in vitro antioxidant and enzyme inhibiting activity of chemically characterized bilberry extract obtained from wild growing ripe fruits. Also, the effects of extract on peritoneal macrophage inflammatory response has been studied in order to potentially reveal the underlining mechanism of action.
Results
The strong predicted interaction of the extract's main constituents with CD38, a key regulator of redox balance and immune signaling, supported the evaluation of antioxidant activity, macrophage response, and LOX inhibition by the bilberry extract. The extract exhibited strong antioxidant potential in two assays for studying total antioxidant potential and prevention of lipid peroxidation, but showed weak lipoxygenase inhibitory activity. Cells viability following bilberry extract treatment were found not to be affected in concentration up to 120 mg/mL. At the same time macrophages exposed to inflammatory stimulation in the presence of bilberry extract produced less nitric oxide and TNF-α, compared to the control.
Conclusion
The bilberry extract affects inflammatory response by potentially influencing several molecular targets which could underlay the inflammatory response. This multitarget approach might be the most effective in preventing inflammation without affecting normal body function.
Introduction
Bilberry (Vaccinium myrtillus L. Ericaceae), a wild growing dwarf shrub, has been well-known for its health-promoting effects.1–3 Owing to the numerous health benefits, the use of Vaccinium species fruits is increasing and its usage is suggested for different conditions such as diabetes, metabolic diseases, rheumatoid arthritis, cardiovascular and neurological disorders and even cancer. 3 Many of the health benefits are due to the main group of bioactive constituents, polyphenols, particularly anthocyanins. 2 Despite their limited stability, water-soluble pigments anthocyanins have attracted interest in recent years due to their diverse biological activities and potential therapeutic advantages. In vitro experiments, animal studies and clinical trials implied that anthocyanin exert antioxidant, anti-inflammatory, anti-diabetic, anti-hypertensive and anti-dyslipidemic effects and its health promoting effect is primarily associated with their antioxidant properties.3,4 Antioxidants from the diet could prevent inflammation-related pathologies and exerting anti-inflammatory effect. Phenolic antioxidants modulate the activities of lymphocytes and macrophages, and among them anthocyanins are known to impact various signaling pathways involved in the inflammatory response. 5 Antitumor activity is also attributed to anthocyanins since they show anti-proliferating and antiangiogenic properties.6–8 Anthocyanins are known to suppress lipid peroxidation, stabilize DNA, modify adipocyte gene expression, improve insulin secretion and sensitivity, and have anti-carcinogenic, anti-inflammatory, and antibacterial effects. 3 Veljkovic et al 9 proved that bilberry diet (100 mg/kg/day) for 15 days protected rats against gentamicin-induced nephrotoxicity by increasing the activity of antioxidant enzymes.
Inflammation is beneficial in most cases because it defends the body from external invaders. In certain cases, it can be harmful and can cause damage to the host tissue(s). Inflammation can cause pain in patients with symptoms of rubor (calor), tumor (swelling), fever, pain, and dysfunction. Although the current anti-inflammatory therapy is designed to relieve the suffering of patients, new drugs from natural origins with polypharmacological properties are desirable and needed. 10 In moderate amounts, free radicals can be beneficial to the immune system response, cell signaling and health maintenance. Nevertheless, excessive production or insufficient elimination of free radicals gives rise to oxidative stress which can harm the way organism functions and result in numerous diseases such as heart disease, diabetes, and cancer. 3
Inflammatory mediators that are the culprits for shift from acute inflammation into a chronic one are: nitric oxide (NO), interleukin-1(IL-1), prostaglandin E2 (PGE2), interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-α). 11 Recently, anti-inflammatory effects of bioactive constituents from functional food have been put into the focus of research lessening symptoms of chronic inflammatory disorders. Since bilberry is listed in functional food, it is documented to have numerous compounds with health promoting effects. These compounds include polyphenols, vitamins, and minerals.12,13 Particularly well studied bilberry constituents are anthocyanins, which have anti-inflammatory, antioxidant, anti-diabetic, anti-cancer, cardio-protective, anti-Alzheimer, antimicrobial, and anti-obesity effects. 11
This study aims to investigate some of the mechanisms responsible for anthocyanins-rich bilberry extract anti-inflammatory effects by performing: (i) in silico target prediction, (ii) estimation of in vitro antioxidant activity, (iii) impact on peritoneal macrophages viability and inflammatory response under the impact of LPS, (iv) inhibition of lipoxygenase.
Materials and Methods
In Silico Target Prediction
SwissTargetPrediction (http://www.swisstargetprediction.ch/) was used to accurately predict the targets of previously identified constituents 9 based on a combination of 2D and 3D similarity measures with known ligands. 14
Drugs and Chemicals
Roswell Park Memorial Institute Medium-1640 (RPMI-1640) consisted of 20 mM HEPES and L-glutamine, supplemented with 10% fetal bovine serum and antibiotic and antimycotic solution were obtained from Sigma-Aldrich (St. Louis, Missouri, USA). 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) was purchased from AppliChem (Darmstadt, Germany) while all other chemicals were obtained from Sigma-Aldrich (St. Louis, Missouri, USA). All solvents (HPLC grade) were purchased either from Sigma-Aldrich (St. Louis, Missouri, USA) or Fisher Chemical (Pittsburgh, USA).
Bilberry Extract
Ripe fruits of wild bilberries (Vaccinium myrtillus L.) picked in the woods of Koroška and Škofja Loka, Slovenia were frozen (at −20 °C) for 1 week and then extracted by deoxygenated methanol according to Može et al 15 method. Briefly, berries were homogenized in methanol, extracted by shaking on a magnetic stirrer and vacuum filtered to obtain the extract. The extraction was repeated three times and all three filtrated were finally pooled and evaporated under nitrogen. Liquid extract (DER 3:20) was used for this study.
Antioxidant Testing
Antioxidative properties of bilberry extract were tested through two test methods: ferric reducing/antioxidant power assay Ferric Reducing Antioxidant Power (FRAP) as a measurement of total antioxidant capacity 16 and in β-carotene bleaching assay for estimation of ability to inhibit lipid peroxidation in β-carotene–linoleic acid in vitro system. 17 In the FRAP assay to a 3000 μL FRAP reagent (10 mmol/l TPTZ in 40 mmol/l HCl, sodium acetate buffer (300 mmol/l, pH 3.6) and FeCl3×6H2O (20 mmol/l) in a ratio of 10:1:1 (v/v/v)) a 100 μL of extract, in ascending concentrations, was added. The absorbance of the mixture was measured immediately and after 5 min of incubation at 593 nm. Freshly prepared β-carotene-linoleic acid emulsion (0.2 mg:25 μL) was added to the extract in ascending concentrations in a 96-well microtitration plate. The reactions absorbance at 450 nm was measured immediately and after 120 min of incubation at 55 °C. Results are expressed as concentration of Fe2+ in µmol/l reduced by certain dilution of bilberry extract or the % of bleaching β-carotene inhibition caused by certain concentration of bilberry extract, respectively.
Animals and Housing
Adult male Wistar rats (200-250 g) were housed according to standard laboratory conditions at Vivarium of Faculty of Medicine University of Niš with free access to tap water and commercial solidified food for rats. All experimental procedures with animals complied with the rules of the European Union Normative (86/609/EEC) adopted by the Ethical Committee of Republic of Serbia (No. 323-07-06862/2016-05/2).
Preparation of Peritoneal Macrophages
Healthy male and female Wistar rats, used for the isolation of peritoneal macrophages (Mφs) were harvested using by peritoneal lavage with 10 mL of cold phosphate buffered saline (PBS). The obtained lavage fluid was transferred to a glass centrifuge tube and centrifuged for 15 min at 1200 rpm. Supernatant was discarded and cells were washed once with PBS, after which they were suspended in RPMI and maintained at 37 °C in a humidified incubator under 5% CO2 atmosphere. Before placing them in the plates cell suspension was set at either 2.5 × 106 cell/mL of RPMI (for MTT assay and MPO assay) or 1 × 106 cell/mL of RPMI (for LPS stimulation) using Trypan blue staining protocol. After 1.5 h cells were washed, and fresh medium was added. 18
Cytotoxicity Evaluation in Peritoneal Mφs
Bilberry extract was diluted with RPMI medium in order to obtain the final concentrations in the range from 0.012 to 120 mg/mL. Isolated Mφs were allowed to adhere to 96-well microtiter plates for 1.5 h and afterward, the wells were filled with fresh RPMI medium containing different concentrations of cisplatin (CP, 1 μM), bilberry extract in different concentrations, and further incubated for 24 h at 37 °C under 5% (V/V) CO2. 19 In the case when cells were used for the viability and myeloperoxidase determination they were seeded, 100 μL/well, from the suspension containing 2.5 × 106/mL of Mφs. In the case when the cells were used to examine their response to LPS in the presence of extract they were seed, 100 μL/well, from the suspension containing 1 × 107/mL of Mφ. Each experiment was done in triplicate and repeated three times.
Cell Viability Determination
After 24 h incubation period, the content of all wells was aspirated, cells were washed, MTT reagent (10 μL, 5 mg/mL) was added, and the plates were incubated for additional 4 h. The formed formazan crystals were dissolved in acidified isopropanol (100 μL), and the optical density was measured at 540 nm (Multiskan Ascent ELISA (enzyme-linked immunosorbent assay) reader). The results were expressed as % of viability based on the formed formazan crystals relative to the one produced by the RPMI medium-cultured cells. Cell viability (%) = OD control – OD sample / OD control x 100. All experiments were performed in triplicate and repeated two times. 18
Determination of Myeloperoxidase (MPO) Activity
Upon 24 h exposure to the extract, MPO activity was measured in the survived cells. The reaction was initiated by the addition of o-phenylenediamine, dissolved in citric acid buffer, completed with H2O2 and Triton X-100. 18 The reaction was stopped by the addition of 2 M H2SO4 and the OD was determined immediately at 540 nm. The results were presented as OD × 1000.
Inhibition of NO Production Induced by LPS
Peritoneal Mφs were incubated with the bilberry at different concentrations and CP (1 μM) in the presence of 1 μg/mL of LPS for 24 h. After the time elapsed the supernatants were collected and the NO concentration in supernatants was estimated using the Griess reaction as described previously. 18 In this method equal amounts of supernatant were mixed with Griess reagent (sulfanilamide and N-(1-naphthyl)ethylenediamine) and shortly incubated at room temperature prior to measuring the absorbance. The absorbance of the reaction mixture was measured at 540 nm using microplate reader, and the NO concentrations were calculated using a standard curve method. The results were expressed as % of inhibition of NO production relative to the untreated, control cells. All experiments were performed in triplicate and repeated two times.
Measurement of TNF-α Concentrations
Supernatants obtained after 24 h of cultivation of bilberry extract and LPS were used for the TNF-α determination. TNF-α was quantified using sandwich ELISA following the manufacturers’ instructions (Rat TNF alpha ELISA kit, Abcam, USA). The assay range was between 18.75 and 1200 pg/mL. 18
Determining LOX Inhibitory Activity
Lypoxygenase (LOX) inhibition activity of bilberry extract was determined using a modified procedure from Gunathilake et al 20 Sodium linoleate (0.01 M) solution was prepared by homogenizing 70 mg of linoleic acid and 70 mg of Tween 20 in 4 mL of water; 0.5 mL of 0.5 M NaOH was added, and the obtained solution was diluted to 25 mL. Lipoxygenase isolated from soybeans (8000 U/ml, 100 μL) and bilberry extract (final concentration in range between 1.0 and 6.7 mg/mL, 1 mL of solution in borate buffer) were incubated with 2 mL of sodium borate buffer (pH 8.8, 0.1 M) for 5 min. Sodium linoleate (20 μL, 0.01 M) was added, and the absorbance of the mixture was measured over time at 234 nm. Borate buffer was used as the control. Same procedure was used to determine the LOX inhibition activity of diclofenac (concentration range covered: 2.5-25 μg/mL).
Statistical Analysis
All quantitative values are expressed as mean ± standard deviation (SD). Statistical differences between two groups were examined by One Way Analysis of Variance (ANOVA) followed by Tuckey's post hoc test using SPSS version 17.0. Probability values (p) less than 0.05 were considered to be statistically significant.
Results
Target Prediction
To explore the potential molecular mechanisms underlying the anti-inflammatory and antioxidant properties of the bilberry extract, we conducted an in silico target prediction analysis using SwissTargetPrediction for 15 major anthocyanin constituents (compounds 1-15, Table 1, see Veljkovic et al 9 ). The predicted targets and their associated probabilities are summarized in Table 1. Only targets with scores ≥ 0.1 were shown, as these scores may indicate interesting leads. Among all predicted protein targets, lymphocyte differentiation antigen CD38 emerged with the highest overall probability values, ranging from 0.43 to 1.00 across all compounds. Notably, compounds 1–6 (the major constituents) and 11 exhibited particularly strong predicted affinities (≥0.76). A second cluster of predicted targets, including nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 4, aldose reductase, and several carbonic anhydrase isoforms (II, IV, VII, XII), exhibited consistent probability scores in the range of 0.12-0.15. Predictions also included several enzyme and receptor targets with lower confidence scores (0.10-0.12), such as cyclooxygenase-2 (COX-2), tumor necrosis factor alpha (TNF-α), interleukin-2 (IL-2), and the adenosine A1 receptor. While the low probabilities suggest a reduced likelihood of direct interaction, these targets remain relevant due to their well-established roles in inflammatory signaling.
Potential Targets for Compounds 1-15, Identified by SwissTargetPrediction. The Values Represent the Probabilities to Target Specific Protein.

*1: delphinidin 3-galactoside; 2: delphinidin 3-glucoside; 3: delphinidin 3-arabinoside; 4: cyanidin 3-galactoside; 5: cyanidin 3-glucoside; 6: cyanidin 3-arabinoside; 7: petunidin 3-glucoside; 8: malvidin 3-glucoside; 9: peonidin 3-glucoside; 10: petunidin 3-galactoside; 11: petunidin 3-arabinoside; 12: malvidin 3-galactoside; 13: malvidin 3-arabinoside; 14: peonidin 3-galactoside; 15: peonidin 3-arabinoside.
Antioxidant Activity of Bilberry Extract
All tested dilution of bilberry extract possesses notable in vitro antioxidant activity in two different assays: FRAP and β-carotene/linolenic acid assay. No statistically significant difference was found in the percentage bleaching inhibition of β-carotene (from 80.55 ± 1.4% to 86.76 ± 0.16%) suggesting a strong potency of extract in preventing degradation. At the same time FRAP value (from 196.55 ± 35.23 µmol Fe2+ to 591.88 ± 17.89 µmol Fe2+ mmol/L) was found to be concentration dependent with the highest concentration being able to scavenge the most Fe2+ (Figure 1A and B).

Results of Antioxidant Testing: Inhibition of β-Carotene Bleaching (A) and FRAP Assay (B). FRAP: Ferric Reducing Antioxidant Power.
Effect of Bilberry Extract on Macrophage Viability
Exposure of peritoneal macrophages (Mφs) to bilberry extract in different descending concentrations did not produce noticeable toxicity in MTT assay (Figure 2). The highest tested concentration of bilberry extract produced the most pronounced decrease in Mφs viability causing a decrease in cell viability by almost 20%. Other tested concentrations ranging from 12 to 0.12 mg/mL led to an almost identical slight decrease in cell viability by roughly 10%. Finaly the lowest tested concentration had no meaningful decrease in cell viability (Figure 2). At the same time standard drug cisplatin (CP) in concentration of 1 μM statistically significantly reduced cell viability by around 40% (Figure 2).

Effect of Bilberry Extract on Isolated Rat Macrophage Viability Estimated Using MTT test. Data are Presented as Mean ± SD and Further Compared Using One-Way ANOVA followed by Tuckey's post hoc Test (n = 6). **P < .01 Versus Control (Untreated Cells).
Effect of Bilberry Extract on Macrophage Myeloperoxidase Activity
Incubation of isolated Mφs with bilberry extract did not produce any significant decrease in myeloperoxidase (MPO) activity (Figure 3). The most pronounced decrease in MPO activity, although non-significant, was seen in cells exposed to the highest concentration of the extract. On the other hand, CP application produced a significant decrease in the studied enzyme activity.

Effect of Bilberry Extract on Isolated rat Macrophage Myeloperoxidase Activity. Data are Presented as Mean ± SD and Further Compared Using One-Way ANOVA Followed by Tuckey's post hoc Test (n = 6). **P < .01 Versus Control (Untreated Cells).
Effect of Bilberry Extract on NO Production in Stimulated Macrophages
Exposure of Mφs to lipopolysaccharide (LPS) led to a significant increase in NO production, estimated using Griess method, leading to more than 10-fold increase in NO content (Figure 4). Coincubation of bilberry extract with LPS produced concentration dependent inhibition of NO production, with the most pronounced effect observed in cells exposed to the highest extract concentration. In the case when cells were treated with extract in the concentration of 0.012 mg/mL no significant inhibition of NO production was noticed. Interestingly the most extensive inhibition was seen in cells treated with CP and LPS during 24 h (Figure 4).
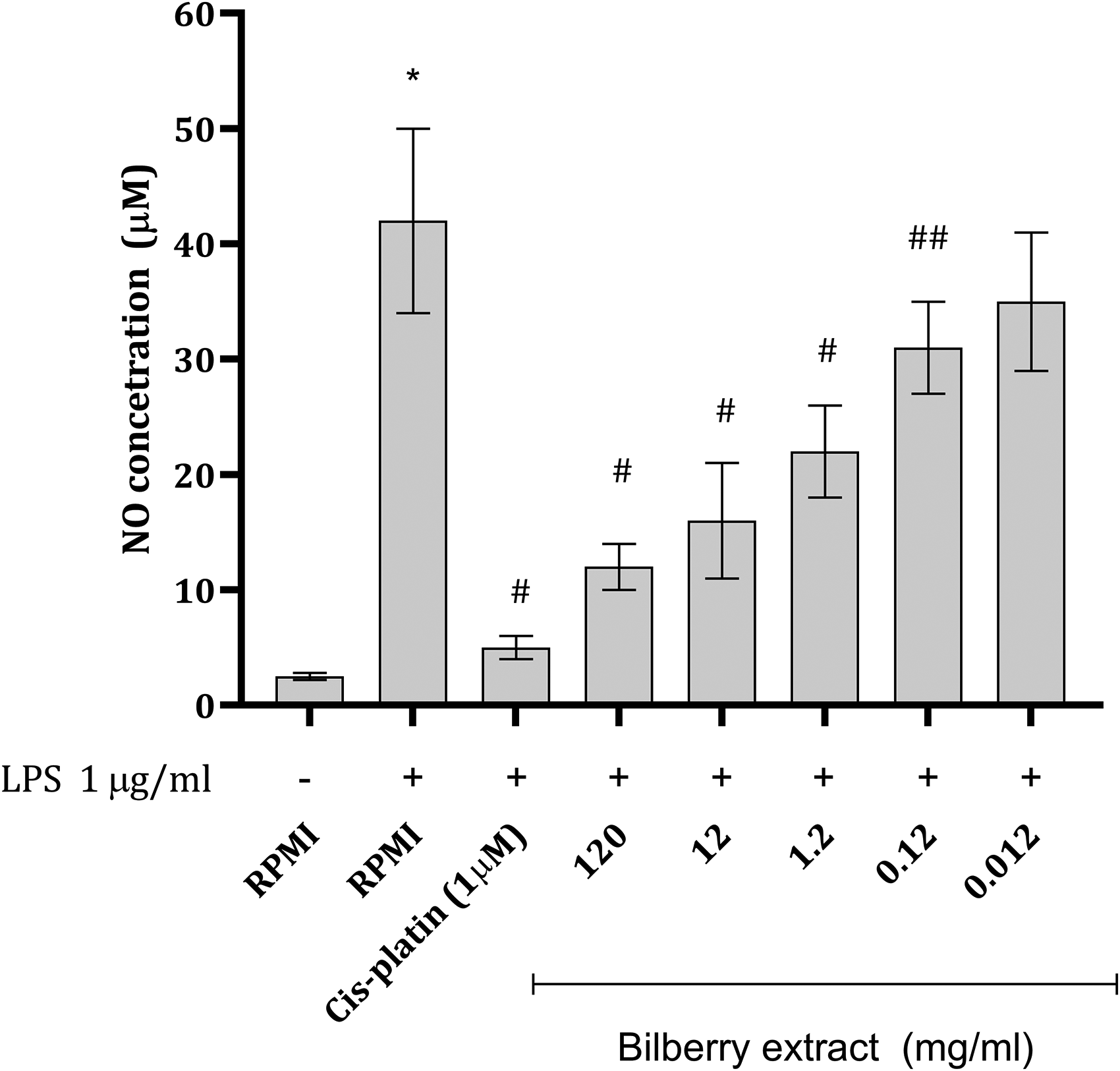
Concentration of NO (µmol/l) in Mφ Supernatants Exposed to Different Concentrations of Bilberry Extract in the Presence of LPS. Data are Presented as Mean ± SD and Further Compared Using One-Way ANOVA Followed by Tuckey's post hoc Test (n = 6). *P < .001 versus RPMI Treated Cells (Control); ## P < .01; # P < .001 Versus RPMI + Lipopolysaccharide.
Effect of Bilberry Extract on TNF-α Production in Stimulated Macrophages
In the cell medium of Mφs exposed to LPS a significant increase in TNF-α content was detected, which is 5-fold increase from the basal value (Figure 5). Coincubation of bilberry extract with LPS produced a decrease in TNF-α production, with the most prominent action seen in cells exposed to 120 and 12 mg/mL of bilberry extract (Figure 5). In the case when cells were incubated with 1.2, 0.12 and 0.012 mg/mL almost identical response was observed. Finaly cells incubated with CP and LPS during 24 h had almost unaltered levels of TNF-α (Figure 5).

Concentration of TNF-α in Mφ Supernatants After Exposure to LPS and Bilberry Extract for 24 h. Data are Presented as Mean ± SD and Further Compared Using One-Way ANOVA Followed by Tuckeys Post hoc Test. ## P < .01; #P < .001 Versus RPMI + Lipopolysaccharide.
Effect of Bilberry Extract on Lipoxygenase Inhibition in Vitro
In the lipoxygenase inhibition test the result (IC50 value that denotes concentration of the sample required for inhibition of 50% in test system) obtained for bilberry extract for bilberry extract and diclofenac were 2.16 ± 0.24 mg/mL and 7.1 ± 0.6 µg/mL, respectively.
Discussion
According to the Committee on Herbal Medicinal Products of the European Medicinal Agency (EMA/HMPC) both dried and fresh bilberry fruit are considered herbal medicinal substances based on their long-standing use. Dried fruit is used for preparation of herbal tea for the treatment of minor inflammation of the oral mucosa and mild diarrhea due to tannins’ astringent and antimicrobial properties. 20 Fresh bilberries are suggested to be beneficial in minor venous circulatory disturbances and cutaneous capillary fragility due to anthocyanins ability to protect/repair blood vessels from/after damage and improve blood circulation by relaxing and widening blood vessels. 21
In silico studies showed several potential targets for the most abundant compounds in the extracts. The high predicted affinity of bilberry anthocyanins for CD38—a key regulator of immune cell function, redox balance, and inflammatory signaling—provides a mechanistic rationale for evaluating the extract's biological effects. CD38 activity is closely linked to oxidative stress and macrophage-mediated inflammation, supporting our investigation into antioxidant potential and modulation of peritoneal macrophage responses. Additionally, as CD38 influences NADPH-dependent pathways, its modulation may indirectly affect enzymes such as lipoxygenase, further justifying LOX inhibition assays. Although some scores represent moderate confidence, the fact that they were predicted for nearly all compounds suggests potential secondary or supportive roles in redox balance and oxidative stress pathways. In particular, NADPH oxidase 4 and quinone reductase 2 are directly involved in the generation and scavenging of reactive oxygen species, aligning well with the extract's antioxidant potential.
In traditional medicine, bilberry had found its purpose in curing skin ulcers, hemorrhoids, nausea, vomiting, diarrhea, mucosal inflammation and improving eyesight. 4 Bilberry reduces inflammation by downregulating the expression of pro-inflammatory factors (TNF-, IL-1β, and IL-6) and enzymes (iNOS, COX-2), altering signaling pathways (NF-kβ and Janus kinase (JAK)-STAT), and reducing reactive oxigen species (ROS) levels in cell culture and in experimental animals.1,22 The anti-inflammatory action of different formulations of bilberries has also been confirmed in several clinical studies.23–28
According to the monograph in the European Pharmacopoeia 29 the fresh bilberry fruits (Myrtilli fructus recens) should contain minimal 0.3% of anthocyanins (expressed as cyanidin 3-O-glucoside chloride) and dried fruits (Myrtilli fructus siccus) minimal 1.0% of tannins expressed as pyrogallol. The phytochemical profile of liquid bilberry extract (used in this study), previously determined, 9 revealed high levels of total polyphenols, tannins, anthocyanins, and hydroxycinnamic acid derivatives. HPLC analysis identified delphinidin and cyanidin glycosides as the most abundant anthocyanins, while glycosides of malvidin, peonidin, and petunidin were present in smaller concertation in bilberry extract. 9 Previous reports from other research groups also suggest that the most abundant anthocyanins in bilberry are delphinidin, cyanidin, petunidin, peonidin and malvidin.11,30,31
Neutralization of superoxide anion and free 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical, reduction of reactive oxygen and nitrogen radicals, decrease in production of H2O2 and reduction of oxidative damage to DNA, proteins and lipids are some of the demonstrated antioxidant effects of bilberry fruit extracts.9,32,33 Since previous study pointed out excellent antioxidant activity of the here examined bilberry extract in DPPH radical scavenging and inhibition of lipid peroxidation in liposomes, 9 two additional methods for different ROS scavenging were included in the present study. FRAP assay is a measure of a total antioxidant capacity in the sample, while the inhibition of lipid peroxidation by β-carotene–linoleic acid in vitro system mimics in vivo oxidation/protection of valuable biological fatty acids present in cell membranes. 34 According to the results of these two assays bilberry extract might be consider possessing strong antioxidant activity even in low tested concentrations (extract in concertation ranging from 0.011 to 0.22 mg/mL)(Figure 1).
Macrophages are found in tissues and belong to both innate and adaptive immune response. They have a crucial impact on sustaining local homeostasis and are fundamental in the initiation and maintenance of numerous inflammatory diseases. These cells interact with pathogens and further phagocyte them. Also, they could act as antigen presenting cells and play a role in tissue repair. 35 Some drugs exerting anti-inflammatory action might be associated with a decrease in cell viability, thus providing false positive results. Namely, a decrease in cell viability would reflect the extent of inflammatory response. The cells exposed to standard cytostatic drug, cisplatin, were found to be less viable than those in the control (RPMI) wells (Figure 2). In the present study exposure of peritoneal Mφs to increasing concentrations of bilberry extract did not cause any significant decrease in their viability. Interestingly the highest tested concentration of the extract (120 mg/mL) led to a more pronounced decrease in cell viability with large SD. This kind of large SD can be potentially associated with different toxic effects of compounds present in the extract, in cases when the bilberry extract concentration (mg/mL) exceeds the toxicity limit. Also, the disturbance in osmotic pressure due to the presence of various molecules could lead to cell damage.
When stimulated with LPS peritoneal Mφs could be used to examine the production of a variety of inflammatory mediators including nitric oxide (NO), TNF-α, etc18,36 under in vitro conditions. Thus, this allows the evaluation of potential mechanism of action of an anti-inflammatory agents. In the present study anti-inflammatory properties of wild bilberry extract were determined in the described model.
Myeloperoxidase is a constitutive enzyme expressed in both Mφs and neutrophils. Its activity leads to the formation of hypochlorous acid in the presence of H2O2 generated by ROS. This acid, known for its highly aggressive oxidizing properties, diffuses into target cells, inducing oxidative stress. 36 MPO is involved in the development of numerous chronic inflammatory disorders such as arteriosclerosis, neurodegenerative disease, lung disease, arthritis, cancer, and kidney diseases. 37 Interestingly, applied bilberry extract during 24 h incubation period did not produce any changes in MPO activity in isolated rat peritoneal Mφs (Figure 3). These data point to the fact that cell viability is not decreased by the extract and that the resident MPO activity is not altered either. On the other hand, cell viability decrease produced by CP (Figure 2) can be directly corelated with a decrease in MPO activity (Figure 3). Although this might not be an adequate positive control in this experimental setting, the obtained results provide some correlation between viability and MPO activity. More adequate positive control in the mentioned experimental setting would be an anti-inflammatory drug such as non-steroid anti-inflammatory drug or corticosteroid based drug.
Since NO could be considered as more of a pro-inflammatory mediator the present work examined the capacity of bilberry extract to inhibit NO release from activated macrophages. 38 Application of LPS causes inducible nitric oxide synthase (iNOS) upregulation and thus consequently higher NO production. Coincubation of bilberry extract with LPS in Mφs led to a concentration dependent decrease in NO production (Figure 4). Bilberry extract administration in previous studies also significantly decreased liver malondialdehyde and NO contents. 1 The observed activity might arise from the inhibition of iNOS expression as previously pointed out.1,27 Also, a decrease in NO concentration might arrive from the ability of bilberry extract constituents to scavenge NO radicals. Pomari et al 39 assessed the effect of bilberry extract on the viability of RAW264.7 cells using H2O2 (200 µM) during different time points. Exposed to H2O2 Mφs express high amounts of COX2, TNF-α, IL-1, iNOS, which is all associated with the activation of NF-kβ. Examined extracts downregulated COX2, TNF-α, IL-1, iNOS, and which was observed in the present study through the estimated levels of NO (product of iNOS activity) and TNF-α (Figure 4 and 5). In the prediction study one of the targets for the action of the bilberry extract constituents was found to be TNF-α (Table 1). TNF-α can up-regulate the expression of IL-6 and IL-1β, which are also considered important inflammatory cytokines. 1 Previously anti-inflammatory activity was found to be highly dependent on aglycon structure (hydroxylation and methylation pattern) and the sugar moiety. 39 The cyanidin-3-O-glycosides themselves appear to be responsible for the anti-inflammatory effect, as they are completely stable in cell medium for up to 3 h, 40 while the breakdown products and/or metabolites may contribute to anti-inflammatory potential. Poor stability under cell culture conditions (eg protocatechuic acid) must generally be considered in in vitro research 40 and deserve further focus.
Lipoxygenase (LOX), belonging to a family of lipid peroxidizing enzymes, 41 is a rate-limiting enzyme in the process of arachidonic acid metabolism into several active metabolic products, leukotrienes, which mediates the occurrence of inflammation and a lot of diseases. For instance, diabetes, cardiovascular diseases, hypertension, renal diseases, and neurological conditions have the imbalance of leukotrienes in its origin. 10 Kuhn and O’Donnel 41 have suggested that 12/15-LOX may play an important role in the development of atherosclerosis, diabetes, and neurodegenerative disease. Thus, the inhibition of LOX reduces leukotriene production and thereby lessens the inflammatory process and reduces disease progression. The extract demonstrated poor LOX inhibition, with an IC50 value of 2.16 ± 0.24 mg/mL. This value is above the IC50 values reported for plant extracts of other Ericaceae family members in the literature, ranging from 277 to 900 μg/mL for extracts of two Gaultheria species. 42 It is worth noting that the IC50 value for Diclofenac, the standard drug in this study, was 7.1 ± 0.6 μg/mL, and this result aligns well with the published value of 5.29 μg/mL. 43
The substrate for LOX reaction, arachidonic acid, can be obtained from damaged cell membranes (liberated). 44 Thus, potential cell death and their destruction could give the substrate for this reaction. However, since here tested bilberry extract did not produce any significant cell death, we could speculate that the activity could be also from substrate limitation in in vivo conditions. Zhu et al 45 have shown that monocyte 12/15-LOX mediates the oxidative modification of low-density lipoproteins, thereby influencing the process of atherosclerosis. The utilized cells in this experiment belongs to a class of monocytes which are transformed into peritoneal Mφs and the impact of bilberry extract on their function could be indirectly associated with the impact of bilberry extract on the process of atherosclerosis.
The immune response on the cellular levels upon which LOX acts is carefully orchestrated by signal transduction pathways such as the NF-kβpathway. 46 Bilberry, through its anti-inflammatory activity decreases NF-kB which might be one of the mechanisms through which the activity of this enzyme is regulated. LTB4, which is made through the action of LOX has an important function as chemo-attractant and is also involved in the formation of ROS through NF-kβ pathway, 46 further explaining the ability of bilberry extract to prevent ROS generation. Our study supports this by pointing out that LOX inhibitory effect of bilberry extract is comparable with activity of diclofenac.
Conclusions
The initial step, the in-silico study, revealed that the compounds present in the extract might interact with several potential molecular targets associated with inflammation and oxidative tissue damage. This study further revealed in several in vitro systems involving chemically generated radicals that the extract has antioxidant potential. Furthermore, the evidence suggests that extract bilberry extract has anti-inflammatory potential proven on both resident and stimulated isolated macrophages. Finaly, the extract exerted moderate lipoxygenase inhibitory action providing yet another evidence of its potential in preventing inflammation signaling. Having in mind the obtained results the consumption of bilberries could be beneficial in the treatment of many inflammatory diseases. However, more research is needed to fully understand the chemical mechanisms underlying these benefits and to investigate potential new applications for these molecules. In addition, future studies have confirmed the traditional use of Vaccinium species in the treatment of inflammatory diseases, which may contribute to a better understanding of the role of these edible fruits.
Footnotes
Acknowledgements
This work was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (Grant No: 451-03-47/2023-01/200113).
Ethics Approval
Ethical approval for this study was obtained from the Animal Ethical Committee of Faculty of Medicine, University of Niš, Serbia (decision number: 323-07-00,278/2017-05/2). All experiments were performed in compliance with the legislation covering the use of animals for scientific purposes, the declaration of Helsinki and EU Directive 2010/63/EU for animal experiments (EU Directive 2010/63/EU For Animal Experiments. Legislation for the Protection of Animals Used for Scientific Purposes. ![]() ; Accessed August 20, 2021.).
; Accessed August 20, 2021.).
Author Contributions
Conceptualization, D.S., N.S. and D.P.; methodology, N.S., M.V., A.D, and D.Z.; software, N.S. and D.Z; validation, D.S., D.P. and D.S.; formal analysis, N.S. and A.D.; investigation, N.S., A.D. and D.Z.; resources, D.S.; data curation, N.S., A.D., D.Z. and M.V.; writing—original draft preparation, M.V., A.D., D.S. and N.S.; writing—review and editing, D.S., D.Z. and D.P.; visualization, N.S.; supervision, D.S., D.P., and D.S.; project administration, D.S.; funding acquisition, D.S. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Ministry of Education, Science and Technological Development of the Republic of Serbia (Grant No: 451-03-65/2024-03/200113).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The authors confirm that the data supporting the findings of this study are available upon request from the corresponding author.
