Abstract
A new phenylpropane (
Introduction
The plant Pandanus tonkinensis Mart. ex B. Stone belongs to Pandanaceae family, which is widely distributed in the northern provinces of Vietnam, such as Hoa Binh, Vinh Phuc, Bac Giang, and Ninh Binh, and is used in folk medicine to treat liver diseases very effectively.1,2 However, up to now, there has been no publication on the chemical composition of this plant. In the program to search for antioxidant components from Vietnamese medicinal plants, the roots of P tonkinensis were selected for research. This paper reports the isolation and structure determination of 1 new and 9 known compounds from the methanol extract of the roots of this plant. In addition, the lipid peroxidation inhibitory effects of the isolated compounds were also evaluated by in vitro assay.
Results and Discussion
Compound

Chemical structures of compounds

Theoretical calculated ECD spectra of 2 possible stereoisomers and experimental CD for compound
The other compounds were identified as isorhapontigenin (
Phenolic compounds, including lignans, are a type of secondary plant metabolite exhibiting diverse structures, which exhibit potentially beneficial bioactive properties due to antioxidant activity.15–18 Therefore, all the isolates were tested for their lipid peroxidation inhibitory effects by in vitro assay.19,20 At a concentration as high as 100 µg/mL, compounds
Lipid Peroxidation Inhibitory Activity of Compounds
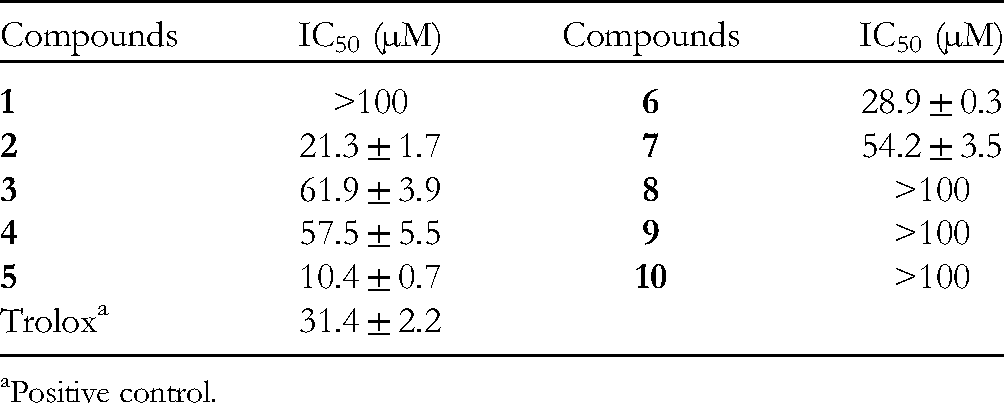
Positive control.
Materials and Methods
General Experimental Procedures
Optical rotation was measured on a Jasco P-2000 polarimeter, IR spectra on a Spectrum Two FT-IR spectrometer, CD spectra on a Chirascan spectrometer (Applied Photophysics), NMR spectra on a Bruker Avance NEO 600 MHz spectrometer, and HRESIMS on a SCIEX X500 QTOF LC/MS. Flash column chromatography was performed using either silica gel or reversed phase (RP-18) resins as adsorbent. The ratio between the amount of silica gel and fraction was 20/1 (w/w). A fraction collector was set by volume per tube (15 mL/tube). Fractionation was monitored by thin layer chromatography (TLC). Contents of test tubes showing a similar TLC pattern were combined. TLC was carried out on either precoated silica gel 60 F254 and/or RP-18 F254S plates. Compounds were visualized by UV irradiation (254 and 365 nm) and by spraying with H2SO4 solution (5%), followed by heating with a heat gun. HPLC was conducted on an Agilent 1100 system including quaternary pump, autosampler, DAD detector, and preparative HPLC column YMC J'sphere ODS-H80 (4 µm, 20 × 250 mm). An isocratic mobile phase with a flow rate of 3 mL/min was used in pre-HPLC.
Plant Material
The roots of Pandanus tonkinensis Mart. ex B. Stone were collected in Son Duong District, Hoa Binh Province, Vietnam, in April 2021, and identified by Dr Do Thi Xuyen, Head of Department of Botanic, Faculty of Biology, VNU University of Science. A voucher specimen (code HNU 024663) is kept at the Herbarium, Vietnam National University, Hanoi, Vietnam.
Extraction and Isolation
Dried powder of P tonkinensis roots (14 kg) was sonicated with methanol (3 times, each 25 L MeOH). After removal of solvent, the MeOH extract (40 g) was suspended in water and then partitioned with n-hexane, dichloromethane, and ethyl acetate to give the corresponding residues (PT1, 2.5 g, PT2, 3.7 g, PT3, 5.3 g), and water (PT4, 27.0 g). After checking by TLC, PT4 (26.5 g) was further chromatographed on a Diaion HP-20 column eluting with water to remove sugar, then with increasing concentration of methanol in water (25, 50, and 100%) to obtain 3 fractions, PT4A (2.4 g), PT4B (2.6 g), and PT4C (21.0 g). PT4C (20.0 g) was chromatographed on a silica gel column eluting with dichloromethane/methanol (v/v) 20:1, 5:1, 1:1, and 0:1 (each 2 L) to give 4 corresponding subfractions (PT4C1-PT4C4). PT4C1 (14.0 g) was further chromatographed on a YMC column eluting with acetone/water (1/3, v/v) to give 7 smaller fractions, PT5A-PT5G. PT5C (5.8 g) was fractionated by silica gel CC eluting with dichloromethane/methanol (7/1, v/v) to obtain 6 subfractions, PT6A-PT6F. PT6A (83.0 mg) was chromatographed by HPLC (J’sphere H-80 column, 250 mm length × 20 mm ID, eluting with 15% acetonitrile in water, a flow rate of 2.5 mL/min) to give compounds
(7S)-2,6-Dimethoxyphenyl-7,9-Propanediol-1-O-β-D-Glucopyranoside (1)
Colorless amorphous powder,
1H NMR (CD3OD, 600 MHz) δ (ppm): 6.71 (2H, s, H-2, H-6), 4.51 (1H, t, J = 7.2 Hz, H-7), 1.76 (2H, m, H-8), 0.94 (3H, d, J = 7.2 Hz, H-9), 4.85 (1H, d, J = 7.8 Hz, H-1′), 3.50 (1H, dd, J = 9.0, 7.8 Hz, H-2′), 3.43 (1H, dd, J = 9.0, 9.0 Hz, H-3′), 3.44 (1H, dd, J = 9.0, 9.0 Hz, H-4′), 3.22 (1H, m, H-5′), 3.68 (1H, dd, J = 12.0, 5.0 Hz, Ha-6′), 3.79 (1H, dd, J = 12.0, 5.0 Hz, Hb-6′), 3.89 (6H, s, 2 × OCH3). 13C NMR (CD3OD, 150 MHz) δ (ppm): 143.4 (C-1), 105.0 (C-2, C-6), 154.1 (C-3, C-5), 135.3 (C-4), 76.6 (C-7), 30.0 (C-8), 10.6 (C-9), 105.6 (C-1′), 75.8 (C-2′), 77.8 (C-3′), 71.4 (C-4′), 78.3 (C-5′), 62.6 (C-6′), 57.0 (2 × OCH3).
Acid Hydrolysis of Compound 1
Compound
Antioxidant TBARS assay19,20
Thiobarbituric acid reactive substances (TBARS) assay values are usually reported in MDA equivalents, a compound formed from the decomposition of polyunsaturated fatty acid lipid peroxides. Briefly, a 0.1 mL sample at difference doses was added to 1 mL of mice brain homogenate (10%) and 0.8 mL phosphate buffer in the presence of 0.1 mL Fenton reagent (FeSO4 0.1 mM: H2O2 15 mM at a ratio of 1:1). After incubating the mixture at 37 °C for 15 min, 1 mL of trichloroacetic acid 10% was added to each tube and the tubes were centrifuged at 12000 rpm for 5 min. The supernatant was then mixed with 1 mL of 0.8% thiobarbituric acid at a ratio of 2:1 and heated at 100 °C for 15 min. After cooling, the absorbance of the mixture was determined at 532 nm by using a microplate reader (BioRad). The percentage inhibition was calculated by the following formula:
Conclusions
Phytochemical study of the methanol extract of P tonkinensis roots led to the isolation of a new phenyl propane, (7S)-2,6-dimethoxyphenyl-7,9-propanediol-1-O-β-D-glucopyranoside (
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221088372 - Supplemental material for Lignans and Other Compounds From the Roots of Pandanus tonkinensis and Their Lipid Peroxidation Inhibitory Activity
Supplemental material, sj-docx-1-npx-10.1177_1934578X221088372 for Lignans and Other Compounds From the Roots of Pandanus tonkinensis and Their Lipid Peroxidation Inhibitory Activity by Dinh Thi Huyen Trang, Pham Hung Viet, Duong Hong Anh, Bui Huu Tai, Ngo Quoc Anh, Nguyen Xuan Nhiem and Phan Van Kiem in Natural Product Communications
Footnotes
Acknowledgments
The authors would like to thank Dr Do Thi Xuyen, Department of Botanic, Faculty of Biology, VNU University of Science for the identification of the plant sample, and Dr Ngo Duc Phuong, Institute of Vietnamese Medicinal Plants, Ministry of Health for collecting the plant sample.
Author Contribution
Research idea: PH Viet, DH Anh, NX Nhiem, PV Kiem. Isolation: DTH Trang. Structure elucidation and writing: BH Tai, DTH Trang, DH Anh, PV Kiem.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Science and Technology of Ho Chi Minh City.
Supplemental material
Supplemental material for this article is available online.
List of Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
