Abstract
Introduction
Asparagus (Asparagus officinalis L.), known as Shídiāobǎi in Chinese, is a perennial herbaceous plant belonging to the Liliaceae family.1,2 Asparagus has achieved global prominence due to its dual utility in gastronomy and traditional medicinal systems. The plant's tender shoots are recognized as a nutritionally dense vegetable source, characterized by their rich content of essential vitamins (including A, C, and K), minerals (such as iron and calcium), and diverse bioactive compounds. 3 This exceptional phytochemical profile has led to its commercial designation as the “king of vegetables” in international horticultural markets. 1 From an ethnopharmacological perspective, asparagus holds particular therapeutic value in Traditional Chinese Medicine (TCM) practice, where it has been systematically incorporated into herbal formulations for centuries. 4 The species’ medicinal applications extend beyond its established culinary uses, demonstrating the plant's unique position as a functional food-phytomedicine continuum. Current phytopharmacological research continues to investigate the mechanistic basis of its historical employment in TCM protocols, particularly regarding its putative adaptogenic and diuretic properties. This synergetic combination of nutritional bioavailability and therapeutic potential underscores asparagus’ significance in both food science and complementary medicine disciplines. Historical records, including the Shennong's Classic of Materia Medica (Shennong Ben Cao Jing), classify it as a superior-grade medicinal herb, with long-term consumption purported to enhance vitality and longevity. The Compendium of Materia Medica further documents the therapeutic applications of its roots, which exhibit bitter, pungent, and mildly warm properties, traditionally employed to tonify Qi (enhance vital energy), resolve heat, eliminate phlegm, and regulate gastrointestinal function. Contemporary pharmacological investigations have substantiated its multifunctional bioactivities, including antitumor, immunomodulatory, hypolipidemic, anti-aging, and anti-fatigue effects. 1 Notably, modern pharmacopoeias incorporate asparagus-derived formulations, exemplified by Ru Ning Tablets, a TCM prescription for managing mammary hyperplasia and thymus disorders.
Generally, asparagus plant can be divided into three portions – edible portion, hard-stem byproducts, and roots. The edible portion that starts from crown is also called spear or cladophylls. The spear account only 23.5% of the whole plant and the remaining 76.5% accounts for hard-stem byproducts and roots. 5 During the post-harvest processing of edible asparagus, approximately 70%–80% of the plant biomass, including peelings, basal cuttings, and foliar components, is typically discarded as agricultural waste. This practice not only represents a significant underutilization of valuable phytochemical resources but also contributes to environmental pollution through improper waste management. Despite undergoing rigorous post-harvest processing, commercial asparagus exhibits significant susceptibility to textural degradation, particularly through rapid lignification and fiber development. This physiological deterioration frequently necessitates the removal and disposal of the basal portion during culinary preparation, resulting in additional product loss and reduced edible yield. (Figure 1A). According to statistical data, global asparagus production in 2020 reached 8.613 million metric tons. During harvesting and processing operations, approximately 40%–50% of the total stem biomass is annually discarded as agricultural by-products, representing a substantial loss of potentially valuable resources.

Harvest Processing and Reprocessing of asparagus Before Cooking. One of the Most Critical Steps in Preparing asparagus is Determining Which Part to cut off, a Decision That can Significantly Impact the Flavor, Texture, and Overall Dining Experience. A: After asparagus is Harvested from the Field, the Woody Parts at the Bottom are cut off as Commercial asparagus Shoots (Group B) and Agricultural Waste (Group C). B: to Prepare Fresh asparagus for Cooking, Remove and Discard the Tough Woody Ends Before Cooking. the Bottom of the asparagus Spear Tends to be Woody and Tough, This Fibrous and Woody Section is Unpleasant to eat, the Widespread Practice of Cutting off the Bottoms of asparagus Stems from a Desire for Optimal Texture and Flavor. A Harvest Processing of asparagus; B Reprocessing of asparagus Before Cooking.
Current scholarly investigations have yet to establish a systematic framework for examining phytochemical heterogeneity across distinct anatomical segments of Asparagus officinalis. A comprehensive characterization of spatial distribution patterns governing bioactive constituents, with particular emphasis on their non-edible fractions, could provide a theoretical framework for optimizing the valorization of these underutilized biomass components.
Materials and Methods
Materials
Experimental samples were collected from Enyang District, Bazhong City, Sichuan Province, China. The plant material consisted of two distinct morphological groups: young asparagus stems designated as Group B and mature asparagus stems designated as Group C (Figure 1B). Each experimental group comprised six biologically independent replicates to ensure statistical validity in subsequent analytical procedures. These specimens were systematically processed and preserved according to standardized protocols to maintain tissue integrity for downstream investigations.
Tissue specimens (100 mg) were subjected to cryogenic pulverization in liquid nitrogen and subsequently homogenized in pre-cooled 80% methanol using vigorous vortex agitation. Following a 5-min incubation on ice, homogenized samples were centrifuged at 15,000 × g for 20 min at 4 °C. An aliquot of the supernatant was diluted with LC-MS grade water to attain a final methanol concentration of 53%. The diluted mixture was transferred to fresh microcentrifuge tubes and recentrifuged under identical parameters (15,000 × g, 4 °C, 20 min). The clarified supernatant was finally analyzed by LC-MS/MS.
UHPLC-MS/MS Analysis
Liquid chromatography-tandem mass spectrometry (LC-MS/MS) analyses were conducted using a Vanquish UHPLC system (Thermo Fisher Scientific, Germany) coupled with a Q Exactive™ HF mass spectrometer (Thermo Fisher Scientific, Germany). Chromatographic separation was achieved on a Hypersil Gold analytical column (100 mm × 2.1 mm, 1.9 μm particle size) maintained at a constant flow rate of 0.2 mL min−1. The mobile phase consisted of eluent A (0.1% formic acid in water) and eluent B (methanol) with the following gradient program: initial 2% B (0-1.5 min), linear increase to 85% B (1.5-4.5 min), followed by gradient ascent to 100% B (4.5-10.5 min), immediate return to initial conditions (100%–2% B at 10.5-10.6 min), and column re-equilibration at 2% B (10.6-12 min).
Mass spectrometric detection was performed in both positive and negative ionization modes with the following optimized parameters: electrospray ionization voltage set at ±3.5 kV, capillary temperature maintained at 320 °C, sheath gas flow rate of 35 psi, auxiliary gas flow rate of 10 L/min, S-lens RF level at 60%, and auxiliary gas heater temperature of 350 °C.
Data Processing and Metabolite Identification
The raw data files generated by UHPLC-MS/MS were processed using the Compound Discoverer 3.3 (CD3.3, Thermo Fisher) to perform peak alignment, peak picking, and quantitation for each metabolite. The main parameters were set as follows: peak area was corrected with the first QC; actual mass tolerance, 5 ppm; signal intensity tolerance, 30%; and minimum intensity, 100000. After that, peak intensities were normalized to the total spectral intensity. The normalized data was used to predict the molecular formula based on additive ions, molecular ion peaks and fragment ions. And then peaks were matched with the mzCloud (https://www.mzcloud.org/), mzVault and Mass List database to obtain the accurate qualitative and relative quantitative results. Statistical analyses were performed using the statistical software R (R version R-3.4.3), Python(Python 2.7.6 version) and CentOS (CentOS release 6.6), When data were not normally distributed, standardize according to the formula: sample raw quantitation value/(The sum of sample metabolite quantitation value/The sum of QC1 sample metabolite quantitation value) to obtain relative peak areas; And compounds whose CVs of relative peak areas in QC samples were greater than 30% were removed, and finally the metabolites’ identification and relative quantification results were obtained. These metabolites were annotated using the KEGG database (https://www.genome.jp/kegg/pathway.html), HMDB database (https://hmdb.ca/metabolites) and LIPIDMaps database (http://www.lipidmaps.org/).
Statistical Analysis
The methods include principal component analysis (PCA), orthogonal partial least squares discriminant analysis (OPLS-DA), and Cluster Heatmap Analysis. PCA uses MetaX software to preprocess the raw peak area data by logarithmic transformation and standardization. Through orthogonal transformation, high-dimensional metabolite variables are reduced to the principal component space, which helps assess overall metabolic differences between groups, intra-group variation, and the clustering of quality control (QC) samples. The tight clustering of QC samples is used to verify the stability of the experimental system and the reliability of the data. OPLS-DA uses a supervised learning approach to construct a paired comparison model between Group B and Group C, eliminating system variations unrelated to grouping through orthogonal signal correction techniques, and screening for differentially metabolized substances between groups. Cluster Heatmap Analysis performs unsupervised hierarchical clustering based on the accumulation pattern of metabolites, showing the distribution differences of the top 20 metabolites in different samples. The clustering results are used to evaluate the heterogeneity of the metabolic accumulation patterns between groups and the consistency of biological replicates. Metabolites with FC > 1, P < .05, and VIP ≥ 1 were selected as differentially expressed metabolites (DEMs). The KEGG database was utilized to annotate the DEMs.
Results
Classification of the Overall Metabolite Situation
As can be seen in Figure 2, the biochemical composition of asparagus (Asparagus officinalis L.) exhibits distinct quantitative patterns among major phytochemical classes. The species demonstrates a predominant accumulation of lipid derivatives, with fatty acyls and lipid-like molecules collectively constituting 39.24% of its total metabolic profile (See Supplemental materials for details). This substantial lipid fraction likely corresponds to essential structural components and energy storage molecules characteristic of plant tissues. Secondary metabolites show differential representation, as phenylpropanoid derivatives and polyketides emerge as the second most abundant category at 16.24%, reflecting their established role as key secondary metabolites in plant defense mechanisms and pigmentation processes. Notably, nitrogen-containing compounds exhibit markedly lower representation, with alkaloids and their derivatives demonstrating limited accumulation at merely 0.42% of total composition. This pronounced compositional disparity between lipid-derived substances and specialized metabolites underscores potential metabolic prioritization in asparagus biosynthesis pathways, possibly reflecting evolutionary adaptation to environmental stressors and ecological requirements.
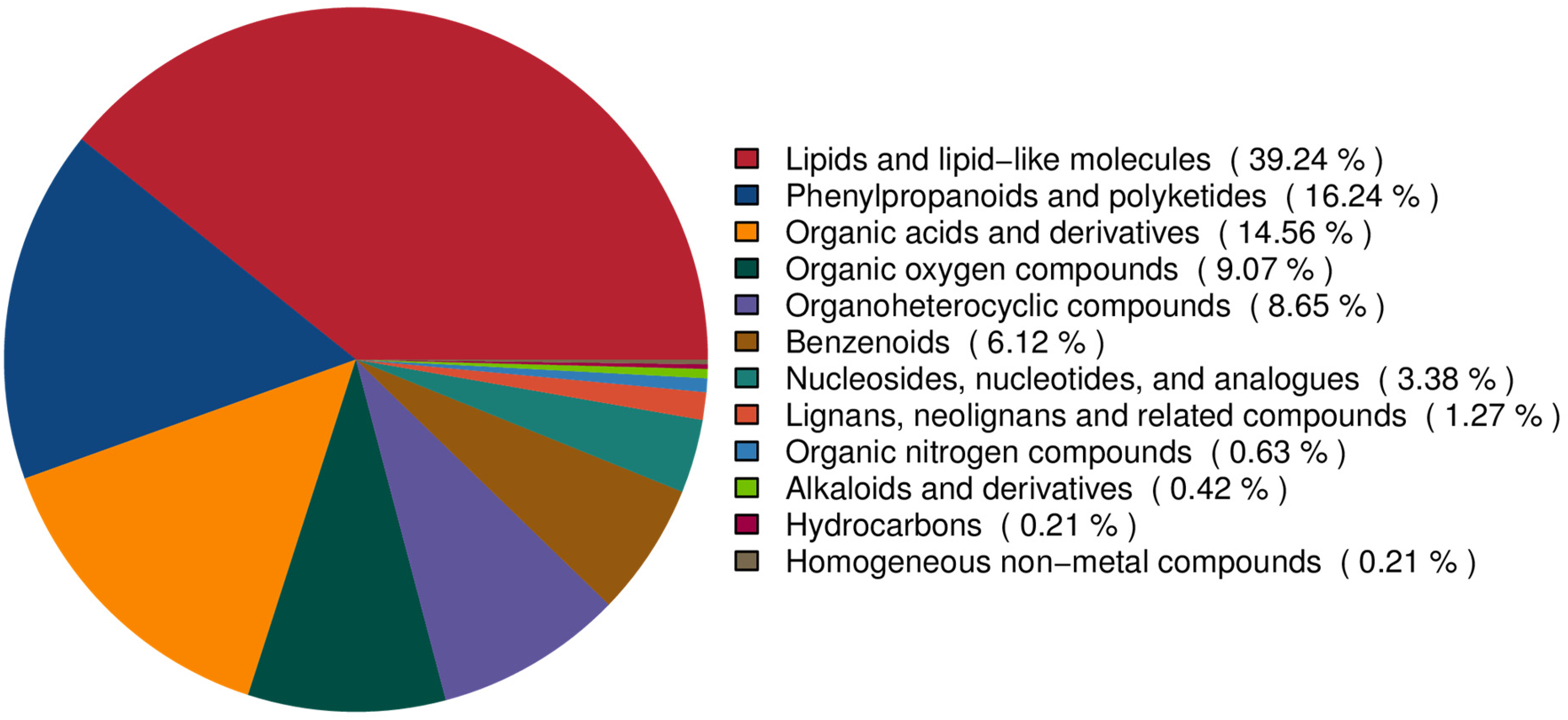
Pie chart of metabolite classification. The chemical classifications of the metabolites identified in this study were counted and pie charts were plotted to reflect the classifications of the metabolites detected and the number contained in each classification.
Overview of the Differential Metabolites
The dataset underwent logarithmic transformation and standardization procedures utilizing MetaX software prior to multivariate statistical analysis. Principal Component Analysis (PCA) was systematically applied to both experimental samples and Quality Control (QC) samples to evaluate methodological stability and data quality. The reduced variance observed among QC samples demonstrates enhanced system stability and superior data reproducibility, as evidenced by their tight clustering pattern in the PCA score plot. This analytical approach operates through dimensionality reduction, extracting n orthogonal principal components that preserve essential characteristics of the original dataset while enabling effective visualization of intra-group consistency and inter-group differentiation.
As can be seen in Figure 3, the PCA results revealed two critical findings: Firstly, the overlapping distribution of QC sample replicates confirmed minimal technical variability, thereby validating the methodological reliability and analytical precision. Secondly, distinct metabolic profiling was observed between Group B (young stems) and Group C (old stems), as manifested by their clear separation along principal component axes. This spatial differentiation indicates statistically significant compositional differences in metabolic constituents between the two biological groups. The first principal component (PC1) explained 43.26% of total data variance, representing the predominant source of metabolic variation within the analyzed system.

PCA Analysis of the Total Sample the Horizontal Axis PC1 and the Vertical Axis PC2 in the Figure Represent the Scores of the First and Second Principal Components, Respectively. the Scatter Points in Different Colors Indicate Samples from Different Experimental Groups, and the Ellipses Represent the 95% Confidence Intervals.
Detection of Substances
The LC-MS-based metabolomic investigation conducted in positive ionization mode successfully characterized 886 metabolites in asparagus stem tissues across developmental phases (Figure 4). Notably, hierarchical clustering analysis revealed pronounced compositional divergence in metabolite profiles between ontogenetic stages, with nitrogen-containing compounds constituting the predominant biochemical category. Peptides maintained their dominance throughout stem development, accounting for 82.88% and 73.92% of identified metabolites in juvenile and mature stems respectively. Nucleic acid derivatives consistently represented the secondary major class, maintaining over 13% relative abundance in both developmental phases. Multivariate statistical analysis demonstrated significant inter-stage metabolic disparities: juvenile stems exhibited preferential biosynthesis and accumulation of peptide derivatives, while mature stems displayed significant accumulation of three distinct metabolic groups – vitamin/cofactor derivatives (particularly phylloquinone and riboflavin analogs), carbohydrate-associated metabolites (including glycosides and sugar alcohols), and steroid derivatives (predominantly brassinosteroid-related compounds). This stage-specific metabolic signature suggests the activation of coordinated biochemical reprogramming mechanisms during stem maturation, potentially associated with structural lignification processes and developmental phase transition in asparagus. The observed metabolic shifts provide molecular evidence for the physiological differentiation between vegetative growth and maturation phases in perennial stem tissues.
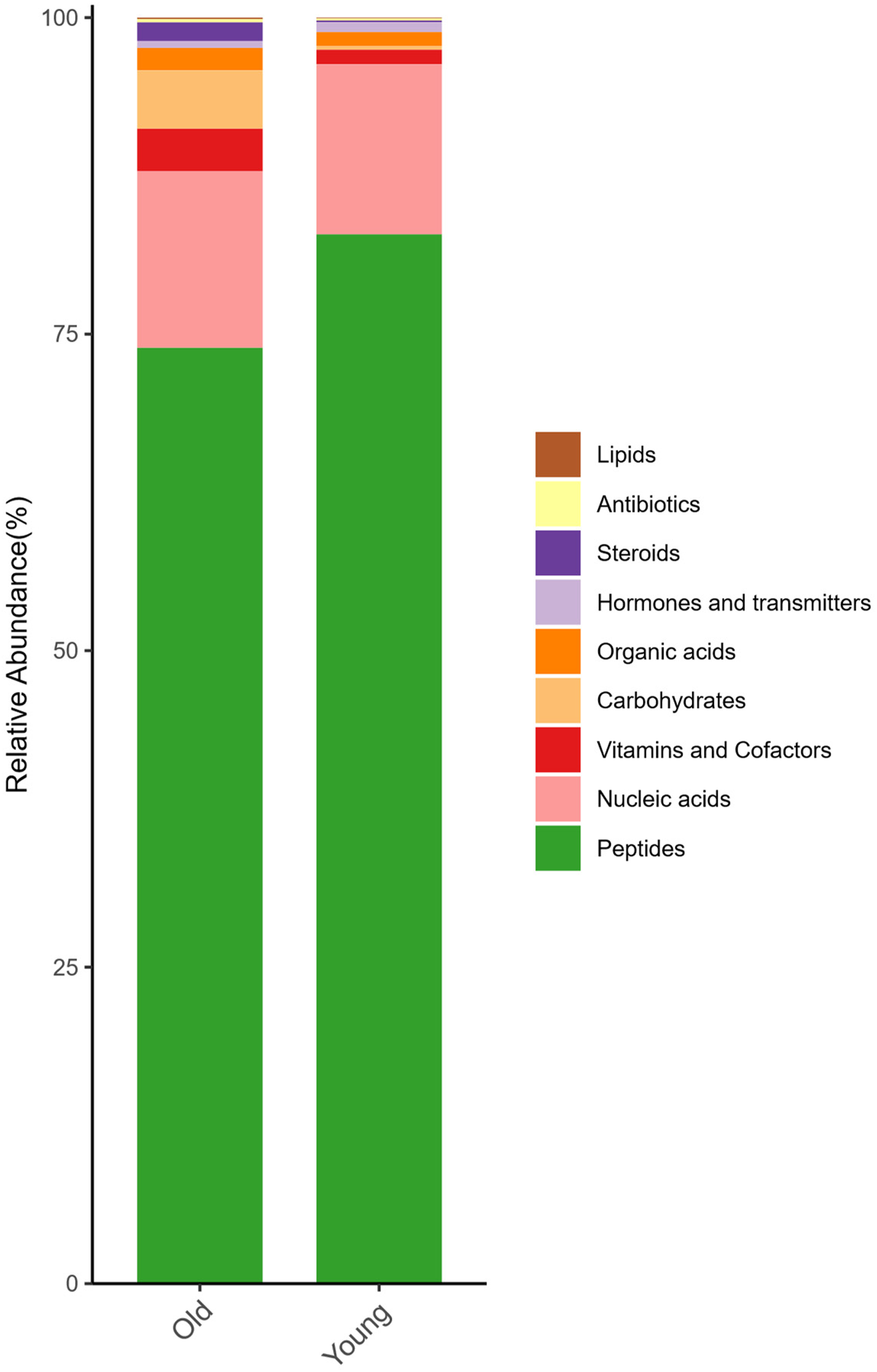
Overall metabolite differences between the two samples. Left: old stems; Right: young stems. The horizontal coordinates are the sample names, sorted according to the grouping order, while the different grouped samples are marked with different colours. Vertical co-ordinates indicate the percentage content of each metabolite, and the order of the columns corresponding to the metabolites from top to bottom is consistent with the legend.
Comparative analysis of the top 20 differentially abundant metabolites revealed a marked disparity in metabolomic composition between juvenile and mature stem tissues (Figure 5). The juvenile stem exhibited significant enrichment in proteinogenic amino acids, particularly L-Norleucine, L-phenylalanine, and Valine, which serve as essential precursors for protein biosynthesis and function as key mediators in nitrogen metabolism and intercellular signaling pathways. This metabolic signature aligns with the heightened biosynthetic demands characteristic of actively growing plant tissues. Conversely, mature stem tissues demonstrated preferential accumulation of long-chain fatty acid amides, including Oleamide and Hexadecanamide. These lipid derivatives are potentially associated with secondary metabolic processes that mediate environmental stress responses and cellular maintenance mechanisms in developed plant structures. The observed metabolic shift from primary nitrogenous compounds in juvenile tissues to specialized amides in mature tissues suggests an age-dependent reprogramming of metabolic priorities, possibly reflecting differential physiological requirements during distinct developmental stages.

Percentage Stacked bar Chart of Metabolites in the top 20 Contents. Left: Young Stems; Right: old Stems. the Horizontal Coordinates are the Sample Names, Sorted According to the Grouping Order, While the Different Grouped Samples are Marked with Different Colours. Vertical Coordinates Indicate the Percentage Content of Each Metabolite, and the Order of the Columns Corresponding to the Metabolites from top to Bottom is Consistent with the Legend.
Screening for Differentially Expressed Metabolites (DEMs)
The differential metabolites were systematically screened through an integrated approach combining univariate statistical analysis (fold change and p-value) with partial least squares-discriminant analysis (PLS-DA). Metabolites demonstrating maximal discriminatory power during multivariate modeling were prioritized based on their variable importance in projection (VIP) scores, with VIP >1 serving as the critical threshold indicating substantial contributions to intergroup differentiation. This dual-criterion selection process simultaneously considered both statistical significance (adjusted p-value <.05 after multiple testing correction) and multivariate discriminatory capacity. R2X = 0.464, R2Y = 0.982, and Q2 = 0.96. These values indicate the variance explained by the model and its predictive power, respectively. The relatively high Q2 value and the lower R2Y value suggest that the model has strong predictive capabilities. Both pR2Y and pQ2 are less than 0.05, indicating that the significance levels of R2Y and Q2 are high, thus confirming the model's stability and reliability.
The resultant candidate biomarkers, visualized within the designated significance region of the model (yellow highlighted area Figure 6), represent metabolites exhibiting both statistically robust differential expression and strong discriminative power across subgroups. Through this rigorous analytical framework, 332 metabolites were ultimately identified as significantly differentiated between the older and younger stem tissues of asparagus, warranting prioritization for subsequent functional characterization and biological interpretation. These findings were subsequently verified by OPLS-DA (R2X = 0.455, R2Y = 0.982, Q2 = 0.96) to better ensure that the results would not be subject to overfitting risks, and similar results were obtained.

PLS-DA analysis of the total sample.
Metabolomic profiling was performed to investigate quantitative variations in metabolic composition between old and young stem tissues of asparagus plants (Figure 7). Statistical analysis employed a dual-parameter approach combining fold change (FC) calculations with significance testing (p-value) to identify differentially abundant metabolites. The FC values were calculated using the reference group (young stems) as baseline, with positive values indicating up-regulation and negative values representing down-regulation in comparative groups. This analytical framework identified 362 significantly differentiated metabolites (p < .05) between developmental stages, comprising 258 up-regulated and 104 down-regulated compounds in old stems relative to their younger counterparts.
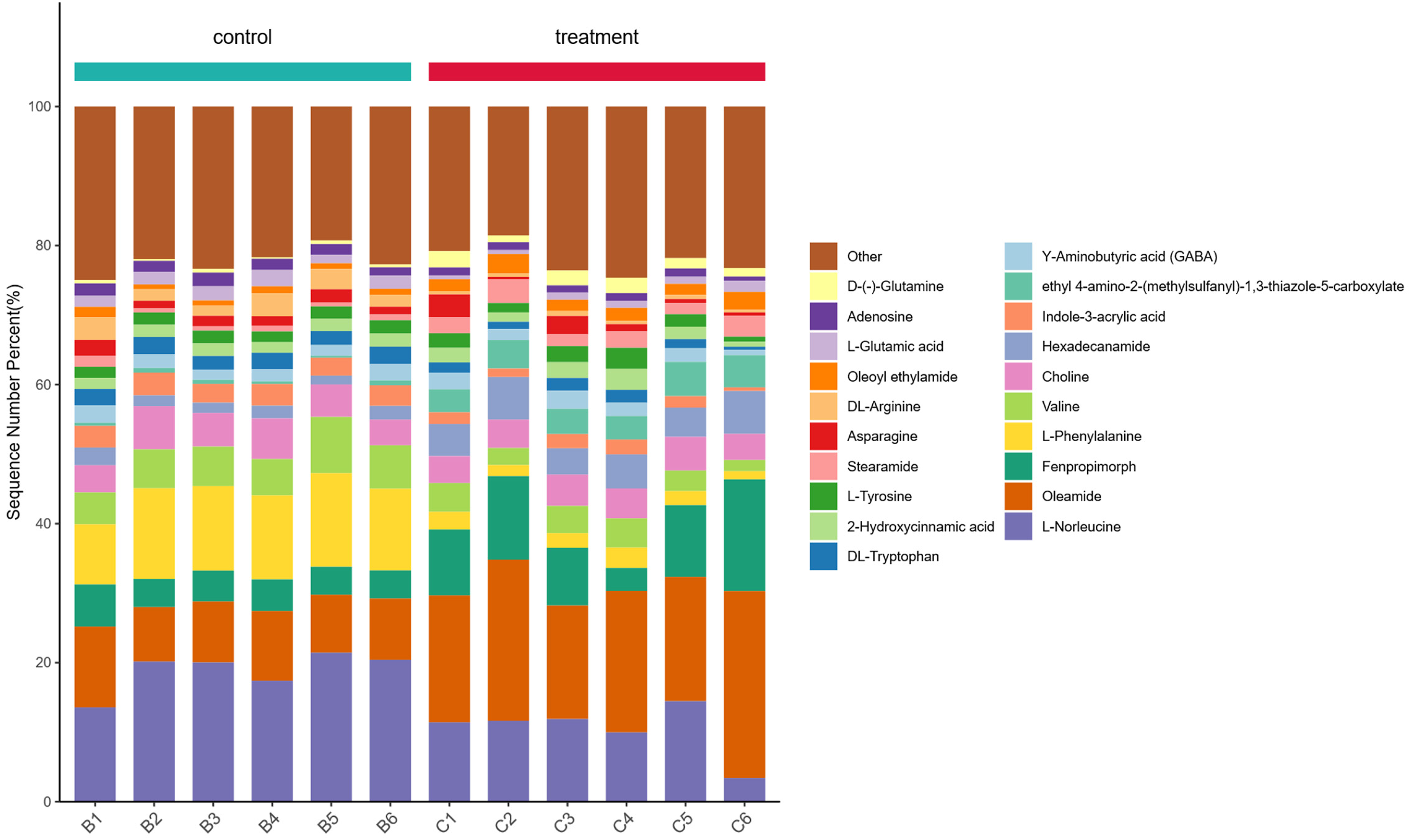
Histogram of Percentage Stacking of Differential Metabolites. Left: B1-B6, Young Stems; Right: C1-C6, old Stems. the Horizontal Coordinates are the Sample Names, Sorted According to the Grouping Order, While the Different Grouped Samples are Marked with Different Colours. the Vertical Coordinates Indicate the Percentage Content of Each Metabolite, and the Order of the Columns Corresponding to the Metabolites from top to Bottom is Consistent with the Legend.
Distinct metabolic signatures emerged from the comparative analysis (Figure 8). Young stem tissues demonstrated significant enrichment of nitrogen-containing compounds and secondary metabolites, including N’,N''-diferuloylspermidine, cis, cis-muconic acid, sedanolide, and asparagine. These metabolites are potentially associated with primary growth processes and cellular maintenance functions characteristic of developing plant tissues.

Heat map analysis of representative differential substances. B1-B6, young stems; C1-C6, old stems.
In contrast, old stems exhibited marked accumulation of fatty acid derivatives and specialized metabolites. This tissue type showed elevated levels of hexadecanamide, 2-aminoadipic acid, coclaethylene, nicotinic acid, oleoyl ethylamide, and oleamide. Particularly noteworthy was the dramatic age-dependent accumulation of steroidal sapogenins. Timosaponin A1 (sarsasapogenin 3-O-β-D-galactoside) demonstrated a FC value of 1468.8 in old stems, while its aglycone precursor sarsasapogenin showed a FC of 213.1 compared to young stems.
Metabolite Correlation Analysis
In the current investigation, a systematic correlation analysis was performed to delineate interrelationships among diverse metabolic compounds. The analysis revealed a robust positive correlation network involving four key metabolites: N’,N''-DiFeruloylspermidine, cis,cis-Muconic acid, Sedanolide, and Asparagine. These compounds exhibited mutual positive correlations, with each metabolite demonstrating statistically significant associations (p < .05) with all other members of this group (Figure 9).

Volcanic map of Differential Metabolites.

Heatmap of Pearson correlation.
Furthermore, extended correlation assessments were conducted between this core metabolite cluster and six additional compounds (Figure 10): Hexadecanamide, 2-Aminoadipic acid, Cocaethylene, Nicotinic acid, Oleoyl ethylamide, and Oleamide. Intriguingly, inverse correlation patterns were identified between the primary metabolite group and eight distinct compounds. Specifically, N’,N''-DiFeruloylspermidine, cis,cis-Muconic acid, Sedanolide, and Asparagine showed significant negative correlations (p < .05) with Sarsasapogenin, Timosaponin A1, Hexadecanamide, 2-Aminoadipic acid, Cocaethylene, Nicotinic acid, Oleoyl ethylamide, and Oleamide.
The observed dichotomy in correlation patterns suggests potential metabolic pathway interactions and regulatory mechanisms that warrant further investigation. These findings provide valuable insights into metabolic network dynamics, particularly regarding the coordinated regulation of functionally distinct compound groups.
Metabolic Pathway
Metabolic profiling of asparagus identified 55 pathways, with seven demonstrating significant pathway impact (Impact >0.1, P < .05). The affected pathways and their associated differential metabolites were systematically characterized as follows (Table 1): (1) Flavone and flavonol biosynthesis pathway showed three down-regulated metabolites: kaempferol, quercetin-3-β-D-glucoside, and quercetin; (2) Biosynthesis of amino acids pathway contained 10 differentially expressed metabolites, with shikimic acid, D-erythrose 4-phosphate, 3-methyl-2-oxobutanoic acid, and ribulose-5-phosphate being up-regulated, while argininosuccinic acid, N-acetylornithine, L-glutamic acid, and O-phospho-L-serine were down-regulated; (3) Linoleic acid metabolism pathway exhibited two down-regulated components: 13-HPODE and linoleic acid; (4) Nucleotide metabolism pathway demonstrated six down-regulated metabolites: uracil, GMP, guanine, xanthine, adenine, and adenosine; (5) Glutathione metabolism pathway displayed mixed regulation patterns with L-pyroglutamic acid and reduced glutathione being up-regulated, versus L-glutamic acid and oxidized glutathione showing down-regulation; (6) Arginine biosynthesis pathway featured three down-regulated metabolites: argininosuccinic acid, N-acetylornithine, and L-glutamic acid; (7) Tropane, piperidine and pyridine alkaloid biosynthesis pathway contained two differentially expressed metabolites, with tropine up-regulated and L-phenylalanine down-regulated.
Metabolic Pathway Analysis.
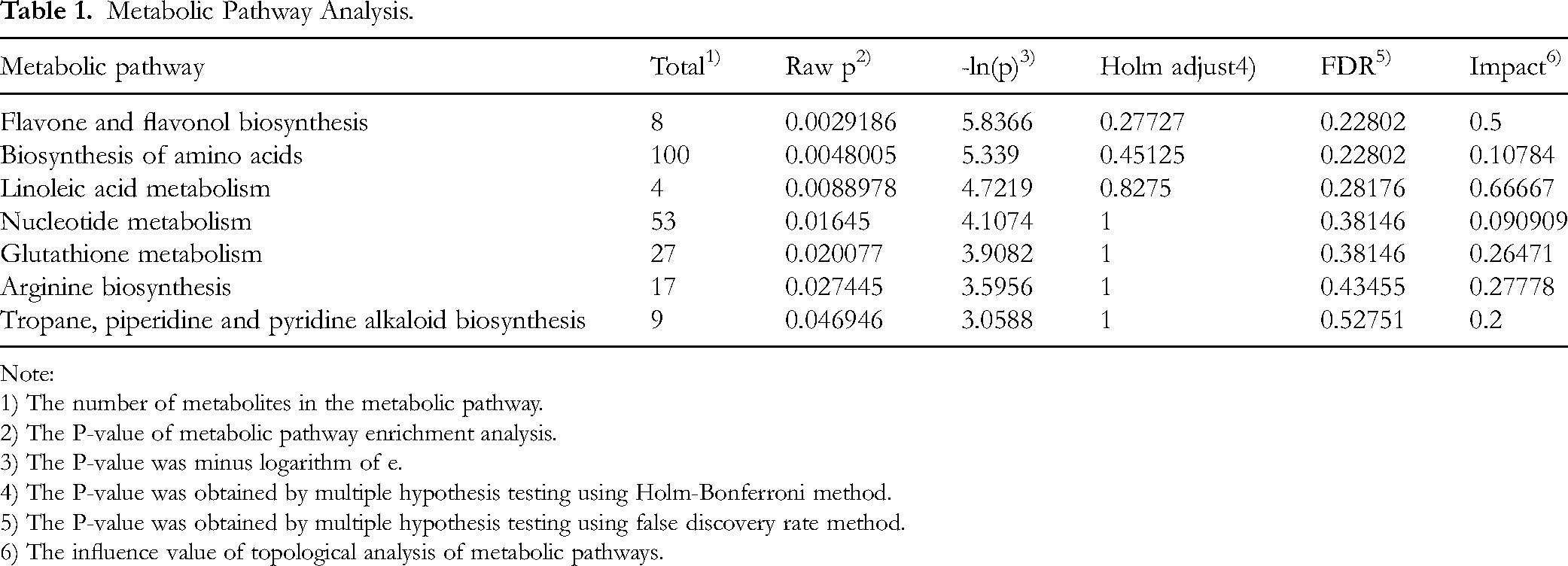
Note:
1) The number of metabolites in the metabolic pathway.
2) The P-value of metabolic pathway enrichment analysis.
3) The P-value was minus logarithm of e.
4) The P-value was obtained by multiple hypothesis testing using Holm-Bonferroni method.
5) The P-value was obtained by multiple hypothesis testing using false discovery rate method.
6) The influence value of topological analysis of metabolic pathways.
Discussion
Asparagus (Asparagus officinalis L.), a perennial herbaceous plant valued for its nutritional and medicinal properties, exhibits significant compositional variations in its stalks across different developmental stages, which fundamentally determine their distinct applications in both culinary and therapeutic contexts.5, 6 Notably, juvenile asparagus stalks demonstrate superior organoleptic characteristics characterized by tender texture and palatability, coupled with elevated concentrations of dietary fiber. 7 Comparative phytochemical analysis reveals that immature stalks contain lower purine alkaloids and their metabolites compared to mature counterparts, rendering them a safer dietary option for populations requiring purine intake moderation, particularly individuals with hyperuricemia or gout predisposition. The abundant dietary fiber complex in juvenile stalks manifests dual physiological functions: (1) enhancing gastrointestinal motility through mechanical stimulation of intestinal mucosa, and (2) modulating appetite regulation via prolonged gastric retention time, thereby conferring synergistic benefits for intestinal microbiota homeostasis and metabolic syndrome management. These structure-function relationships underscore the developmental-stage-dependent bioactivity of asparagus stalks, providing scientific rationale for their differentiated utilization in nutraceutical applications.
Phytochemical studies have shown the presence of various bioactive compounds as valuable secondary metabolites. The major bioactive substances in old asparagus stalks are flavonoids. In addition, the old stem of asparagus has significant pharmacological effects such as antioxidant, antimicrobial pharmacological effects, antiviral, anticancer, anti-inflammatory and antidiabetic actions. 8 Compared with young stems, old stems of asparagus have significant advantages in medicinal use. Older stems contain more saponin-like constituents, which are widely used in traditional Chinese medicine. These components have a variety of pharmacological activities, such as anti-inflammatory, antioxidant, anti-tumour, etc, which are potentially efficacious for the treatment of certain diseases. Studies have shown that the ethanol extract and water extract of asparagus stem have obvious effects on lowering blood lipid in mice with hyperlipidemia.1,5,9 In addition, the saponin of asparagus stem has significant preventive and therapeutic effects on hyperlipidemia in mice, and has obvious hypoglycemic effect on diabetic rats.10-12 And studies have shown that the growth inhibitory activity of asparagus sprouts and rhizomes is significantly stronger than that of above-ground parts, and the nitrogen and potassium content (especially nitrogen) of the remaining stems and leaves of asparagus are far higher than that of conventional green manure. Combined with the characteristics of high rutin and low inhibitory activity, it is proved that the disease-free above-ground parts can be used as an efficient organic green manure resource.13-15 In particular, the content of Sarasapogenin and its derivative Timosaponin A1, AIII in the old stems of asparagus was extremely high, 1468.8, 213.1 and 11.9 times higher than that in the young stems, respectively. Our metabolomic results such as containing more saponin content in the roots than in the young stems are similar to those previously studied by other researchers. 16
Sarsasapogenin exhibits multifaceted pharmacological properties including anti-inflammatory, anticancer, antidiabetic, anti-osteoclastogenic, and neuroprotective activities. Emerging evidence highlights its therapeutic potential in modulating immune responses and metabolic pathways through distinct molecular mechanisms. In neuroinflammatory contexts, sarsasapogenin promotes functional recovery post-spinal cord injury by regulating the immune microenvironment via suppression of the MAPK/NF-κB signaling pathway. 17 Its neuroprotective efficacy extends to diabetic models, where it ameliorates Alzheimer's disease-like pathology through undefined mechanisms potentially linked to metabolic regulation. 18 In metabolic disorders, sarsasapogenin demonstrates dual efficacy in attenuating adipose tissue inflammation and improving insulin sensitivity in high-fat diet-induced obese C57BL/6J mice, suggesting therapeutic value for obesity-related metabolic syndromes. 19 This metabolic modulation may involve interference with pathological glycolysis, as evidenced by its capacity to suppress fibroblast-like synoviocyte proliferation in rheumatoid arthritis through PKM2 downregulation. 20 Furthermore, its inhibitory effects on Alzheimer's disease pathogenesis extend to targeting key enzymatic pathways implicated in disease progression. 21 These collective findings position sarsasapogenin as a promising pleiotropic agent for neuroregeneration, immunomodulation, and metabolic syndrome management, warranting further investigation into its molecular targets and translational potential.
Timosaponin, especially Timosaponin AIII (TAIII) has emerged as a multifunctional phytochemical with significant potential in cancer management. Multiple studies demonstrate its broad pharmacological activities, including anti-cancer, anti-inflammatory, and neuroprotective effects, with particular efficacy observed in hepatocellular and breast cancers22 2.4 Recent mechanistic studies reveal TAIII's ability to induce ferroptosis through distinct pathways across different malignancies. In colorectal cancer, it promotes lipid peroxidation via Rab7-mediated lipophagy, 23 while in non-small-cell lung cancer, it facilitates HSP90-dependent GPX4 ubiquitination and degradation. 24 Notably, research on glioma models indicates that autophagy inhibition enhances TAIII-induced apoptosis both in vitro and in vivo, 25 suggesting context-dependent mechanisms of action. These findings collectively position TAIII as a promising candidate for targeted cancer therapy through programmed cell death modulation.
The lignified stems of asparagus exhibit considerable potential for application in traditional Chinese medicine formulations. Through targeted extraction and refinement of their pharmacologically active constituents, these structures may be developed into specialized therapeutic agents capable of addressing specific pathological conditions, thereby facilitating clinical rehabilitation processes.
Notably, asparagus tissues inherently contain measurable quantities of asparagine and aspartic acid. Recent studies have shown that aspartic acid has a role in cancer induction, especially lung cancer transformation.26-28 This biochemical characteristic underscores the necessity for strategic cultivar selection aimed at reducing these amino acid concentrations through agricultural biotechnology approaches.
Both young and mature stems of asparagus contain γ-aminobutyric acid (GABA), a natural compound with sedative properties that can help alleviate tension and improve sleep quality. Analytical data reveal significantly higher GABA concentrations in older stem tissues compared to younger segments, suggesting that controlled consumption of mature asparagus stalks and their processed products may offer therapeutic potential for sleep disorders, particularly insomnia. As an endogenous neurotransmitter, GABA has been scientifically demonstrated to possess marked anxiolytic and sleep-regulating effects.29,30 The observed elevation of GABA levels in mature stem tissues versus young meristematic tissues indicates that standardized intake of mature asparagus derivatives could serve as a viable nutritional intervention for sleep dysregulation syndromes, especially chronic insomnia. The underlying mechanisms of this bioactivity require systematic pharmacological investigation to validate its therapeutic applications, warranting further exploration of its potential in nutraceutical interventions.
Nevertheless, current limitations include insufficient clinical validation of nutraceutical efficacy, unclear dose-response relationships for bioactive compounds (particularly GABA and saponins), and lack of long-term safety assessments regarding chronic consumption (especially for saponin-rich extracts and aspartic acid derivatives). Further research should address cultivar optimization to balance therapeutic components while mitigating potential risks. And the studies on the enrichment of GABA in foods mainly focus on the simple process modification and optimization. Compared with the addition of the purified GABA as an additive to foods to meet the daily requirement for adults, especially for elderly people, enrichment of GABA in food ingredients or foods is a more promising approach due to the higher acceptance of natural foods by humans and the lower risk of the possibility of overdose. 31
In conclusion, both young and old stems of asparagus are rich in nutrients and beneficial substances. Comparatively speaking, the young stem contains more amino acids, while the old stem contains more amides and more saponins. These results reveal that different parts of asparagus can be treated separately according to the type and amount of their components, and provide a certain scientific basis for the use of the old stem of asparagus in medicine. Whereas young stems are more suitable for consumption as a vegetable, the possible risks associated with excessive amino acid intake, such as excess uric acid, etc, need to be considered.
Supplemental Material
sj-xls-1-npx-10.1177_1934578X251362085 - Supplemental material for Comparative Analysis of Bioactive Metabolites from Different Parts of Asparagus officinalis L
Supplemental material, sj-xls-1-npx-10.1177_1934578X251362085 for Comparative Analysis of Bioactive Metabolites from Different Parts of Asparagus officinalis L by Chen Deng, Haobin Chen, Yao Zhou, Jiayi Wang, Hui Zheng, Zhen Li, Yinyin Chen, Dongbei Xu, Dongju Feng, Wei Wu and Kai Hou in Natural Product Communications
Supplemental Material
sj-xls-2-npx-10.1177_1934578X251362085 - Supplemental material for Comparative Analysis of Bioactive Metabolites from Different Parts of Asparagus officinalis L
Supplemental material, sj-xls-2-npx-10.1177_1934578X251362085 for Comparative Analysis of Bioactive Metabolites from Different Parts of Asparagus officinalis L by Chen Deng, Haobin Chen, Yao Zhou, Jiayi Wang, Hui Zheng, Zhen Li, Yinyin Chen, Dongbei Xu, Dongju Feng, Wei Wu and Kai Hou in Natural Product Communications
Footnotes
Ethical Approval
Ethical Approval is not applicable for this article.
Author Contributions
Conceptualization, D.C. and H.K.; methodology, H.K and W.W.; formal analysis, Z.Y., W.J. and C.H.; investigation, H.K.; resources, H.K., C.Y., X.D., F.D. and Z.H.; data curation, X.W.; writing—original draft preparation, D.C.; writing—review and editing, H.K.; visualization, L.Z. and Z.Y.; supervision, H.K.; project administration, W.W.; funding acquisition, H.K. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the cooperation of Sichuan Agricultural University–Bureau of Agriculture and Rural Development, Enyang District, Bazhong City, and also funded by State Administration of Traditional Chinese Medicine, the Fourth National Census of Traditional Chinese Medicine Resources in China (No. GZY-KJS-2018-004); Chengdu Science and Technology Bureau, Key R&D support plan (NO. 2018-YFO9-00046-SN); 2022 Sichuan ISO International Standard Procurement Project, Sichuan Provincial Administration of Traditional Chinese Medicine.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
links to publicly archived datasets analyzed or generated during the study.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Materials
Nomenclature
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
