Abstract
Kaempferia parviflora, also known as black ginger, is a popular medicinal plant among the Southeast Asian community. Recently, there has been increased interest in further exploring the potential health benefits and therapeutic applications of K. parviflora in Malaysia, Thailand's neighboring country. This review provides an overview of current literature regarding K. parviflora's traditional use and literature on efficacy and safety. We found that, traditionally, K. parviflora was used for gastrointestinal disorders, allergies, as sexual stimulants, and body nourishment. K. parviflora demonstrated antioxidant, anti-inflammatory, antiobesity, anticancer, vascular relaxation, and antimicrobial effects. In humans, there is evidence of its general benefits on physical fitness. In conclusion, K. parviflora is a plant with significant potential for various pharmacological applications. The rhizome is most popularly used and investigated, with polymethoxyflavones as the main bioactive phytoconstituent.
Introduction
Kaempferia parviflora, a herbaceous plant belonging to the family Zingiberaceae, is native to Southeast Asia and is also known as black ginger, Thai ginseng, or Krachai Dum to the locals. K. parviflora is a perennial herb that grows up to 90 cm in height. Its inflorescence is white with a purple tinge. Its rhizome is light to dark purple and may also be black. It has one to several leaves, 7 to 20 cm long. This herb carries a characteristic aromatic odor, with a slightly bitter taste. 1 K. parviflora is widely distributed throughout Southeast Asia, India, South China, Malaysia, Thailand, and Laos. 2 In Thailand, this herb is recognized as one of the top five spearheaded herbal products. Its widespread use and generated income for Thailand is based on anecdotal evidence of efficacy. 3
The rhizome of K. parviflora is the most important plant part for medicinal use due to its rich content of phytoconstituents. The therapeutic benefits of K. parviflora have been mostly attributed to the presence of methoxyflavones. Methoxyflavones, structurally defined as 5,7-dimethoxyflavone (DMF), 57,4′-trimethoxyflavone (TMF), and 3,5,7,3′,4′-pentamethoxyflavone (PMF), have been identified as the main components of K. parviflora rhizomes as shown in Figure 1.4,5
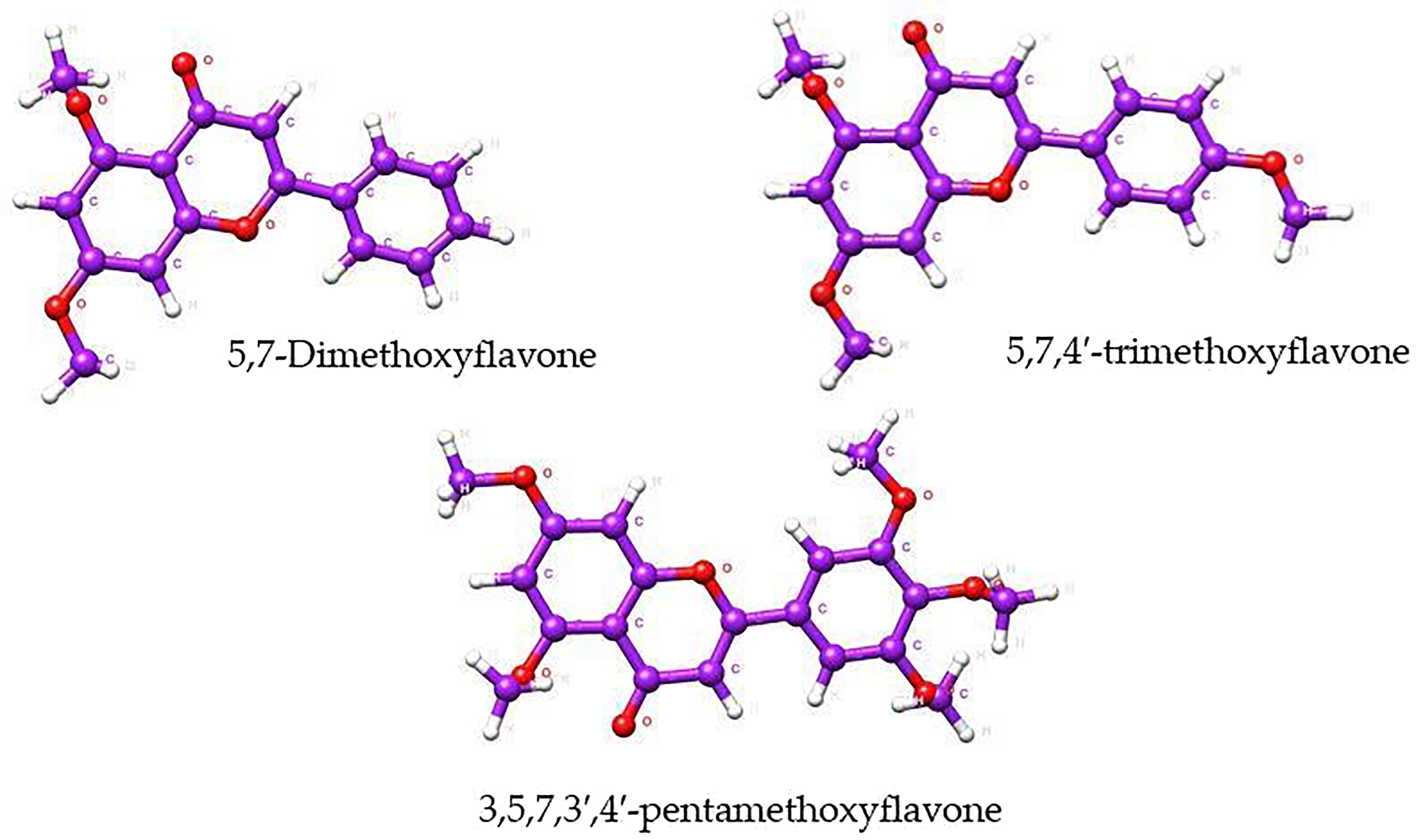
Structures of three major methoxyflavones of Kaempferia parviflora rhizome.
The DMF compound has been extensively studied for its biological activities, including anti-diabetes, 6 anti-obesity,7,8 and anti-inflammatory9,10; while TMF have potential vasodilator effects evidenced by its ability to elicit concentration-dependent vasorelaxation in the rat aorta, mediated via endothelium-derived nitric oxide, 11 which could contribute towards beneficial effects in exercise performance and general physical fitness. 12 The third methoxyflavone, the PMF, can act as a sexual stimulant in men. PMF demonstrated a relaxing effect on isolated human corpus cavernosum tissue via calcium mobilization. 13 It has also been identified as an important mediator of phosphodiesterase type 5 enzyme (PDE5) phosphodiesterase type 6 enzyme (PDE6) inhibition and vasorelaxation, suggesting potential benefit in the treatment of erectile dysfunction. 14
Hence, Malaysia has made continuous efforts in its local industry to develop herbal products that contain or are based on K. parviflora. There are numerous traditional medicine products containing K. parviflora available in Malaysia, as documented in the QUEST 3 + database managed by the National Pharmaceutical Regulatory Agency (NPRA). 15 Due to the growing interest among local stakeholders in further exploring the potential health benefits and therapeutic applications of K. parviflora in Malaysia, this review was conducted to map the current biomedical research landscape and identify the gaps related to K. parviflora.
Results and Discussion
Results
Traditional Uses of Kaempferia parviflora Rhizome
Most of the published information on the traditional uses of black ginger is centered on Thailand because of the nation's extensive use of the herb. K. parviflora is traditionally used for colic disorder, peptic and duodenal ulcers, relief of impotent symptoms, allergies, asthma, gout, gastrointestinal disorders (eg, diarrhea and dysentery), fungal infections, as well as a tonic for body strengthening and carminative agents.16–20
Health Benefits of Kaempferia parviflora
Summary of Included Studies
A total of 276 articles were identified from keyword search on published literature. After deduplication, 192 articles were screened based on the inclusion and exclusion criteria. A final 92 articles were included. The screening and article selection process is shown in Figure 2.

Preferred reporting items for systematic review and Meta-analysis (PRISMA) flow chart of articles inclusion.
Reported Medicinal Properties and Health Benefits
K. parviflora demonstrated various pharmacological properties including antioxidant, anti-inflammatory, antiobesity, anticancer, vascular relaxation, and antimicrobial effects, as well as displaying evidence for general benefits on physical fitness. Among the most studied efficacies include antiobesity, anti-aging in physical and physiological functions, antidiabetic, and benefits in male reproductive and urology (benign prostate hyperplasia systems). Although there were quite a few studies for anticancer properties, these were all in vitro cell studies. For skin health, only preclinical evidence was available.
Clinical Studies
Seven out of the 12 clinical studies included are randomized controlled trials (parallel design). The other study designs are randomized crossover trials (n = 3); open labeled, single-arm interventional trial (n = 1), and single arm pilot trial (n = 1). Most of the clinical studies were conducted on orally administered preparations, except for one which uses transdermal patch for anti-inflammatory effects. A total of eight studies investigated the rhizome plant part while the remaining four studies did not report clearly on the plant part used. Dosage used in clinical studies differs based on preparations and ranged from 90 mg to 360 mg for extracts, 1.35 g for dried powder in capsule, and 12 mg for purified PMF compound. The duration of the studies ranged from two to 12 weeks. The characteristics of included studies and key findings on efficacy are summarized in Table 1.
Details of Clinical Studies of K. parviflora.

Preclinical Studies
This review includes 25 animal studies (in vivo), almost all of which employed rodents as the study model (one study used healthy pigs as a model). A majority of the studies (n = 23) investigated rhizome as a standardized extract or in crude form. In terms of route of administration, K. parviflora was given orally in most studies (n = 21), followed by the intravenous (n = 2), and topical route (n = 1). K. parviflora was given for 25 min (single dose study) to 13 weeks, depending on the disease model studied. The characteristics of included animal studies and key findings on efficacy are summarized in in Table 2.
Details of in Vivo Studies of K. parviflora.

In vitro studies (n = 57) investigated 15 different potential biological effects of K. parviflora. Of these, anti-cancer (n = 11) and antiaging (n = 8) effects were most researched. Like clinical and animal in vivo studies, the rhizome of this herb was the most investigated plant part in in vitro studies (n = 48). As for formulation, more than half (n = 36) investigated standardized extracts of K. parviflora. Seventeen studies used specific compounds either extracted from the plant or commercially bought. Four studies used both the extract and compound. The characteristics of included in vitro studies and key findings on efficacy are summarized in Table 3.
Details of in vitro studies of K. parviflora.

DPPH: 2,2-diphenyl-1-picrylhydrazyl; CUPRAC: cupric reducing antioxidant capacity; FRAP: ferric reducing antioxidant power; NBT: Nitroblue Tetrazolium; MCA: metal chelating ability; PBD: phosphomolybdenum; ABTS: 2,2-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid); AChE: acetylcholinesterase; BChE: butyrylcholinesterase; RCT: randomized controlled trial; PGE2: prostaglandin E2; COX-2: cyclooxygenase-2; CFA: Complete Freund’s adjuvant; NF-κB: Nuclear Factor-kappa B; MAPK: mitogen-activated protein kinase; RBL-2H3: rat basophilic leukemia; DMF: 5,7-dimethoxyflavone; TMF: 5,7,4′-trimethoxyflavone; THP-1: human leukemia monocytic cell line; iNOS: inducible nitric oxide synthase; TNF-α: tumor necrosis factor alpha; IL: interleukin; NO: nitric oxide; NOS: nitric oxide synthase; P38: p38 mitogen-activated protein kinase; STAT: signal transducer and activator of transcription; AGS: adenocarcinoma gastric cell line; sHP: Src homology 2 domain-containing phosphatase; TeMF: 3,5,7,4 tetramethoxyflavone; BAT: activated brown adipose tissue; PMF: 3,5,7,3′,4′-pentamethoxyflavone; BMI: body mass index; KPEE: ethanolic extract of K. parviflora; PMFE: PMF-rich extract; EtOAc: ethyl acetate; TSOD: Tsumura Suzuki, Obese Diabetic; PPARγ: peroxisome proliferator- activated receptor gamma; UCP-1: uncoupling protein 1; β3AR: β-3 adrenergic receptors; ATGL: adipose triglyceride lipase; HSL: hormone-sensitive lipase; IV: intravenous; cGMP: cyclic guanosine 3’,5'-cyclic monophosphate (cGMP); L-NAME: L-NG-nitroarginine methyl ester; eNOS: endothelial nitric oxide synthase; CSE: cystathionine-γ-lyase; H2S: hydrogen sulphide; HDL: high density lipoprotein; ROS: reactive oxygen species; TJ: tight junction; sUV: solar wavelengths; VO2 max: maximal oxygen consumption; SIRT1: sirtuin-1/5’; AMPK: adenosine monophosphate-activated protein kinase; PGC-1a: peroxisome proliferator-activated receptor-gamma coactivator -1 alpha; PPARδ: peroxisome proliferator-activated receptor delta; LPS: lipopolysaccharide; DIC: disseminated intravascular coagulation; HMF: 5-hydroxy-7-methoxyflavone; SAMP1: senescence-accelerated mouse-prone 1; GLUT4: glucose transporter 4; MCT: monocarboxylate transporter 1; ATP: adenosine triphosphate; MRR: methoxyflavones-rich residue; CRE: cAMP response element; IIEF: International Index of Erectile Function; STZ: streptozotocin; UVB: ultraviolet B; MMP: matrix metalloproteinases; NHDFs: normal human dermal fibroblasts; HaCat: Human Adult Low Calcium High Temperature; TRP: tyrosine-related protein; COLIA1: collagen type I alpha 1; AP-1: activator protein 1; BPH: benign prostatic hyperplasia; ERK1/2: extracellular signal-regulated protein kinase 1 and 2; MEK: mitogen-activated protein kinase; MCP-1: monocyte chemoattractant protein-1; IκB: inhibitor kappa B; AKT: Protein kinase B; MCL-1: myeloid leukemia cell differentiation protein; PANC-1: human pancreatic cancer cell line; NDM: nutrient-deprived medium; DMEM: nutrient-rich medium; HPV: human papillomavirus; sEH: soluble epoxide hydrolase; EETs: epoxyeicosatrienoic acids; DHETs: hydroxyeicosatetraenoic acids; HUVEC: Human umbilical vein endothelial cell; FcεRI: high-affinity IgE receptor; MDCK: Madin–Darby canine kidney; IFN: interferon; C/EBP: CCAAT/enhancer-binding protein; mTOR: mechanistic target of rapamycin complex 1; SCN: suprachiasmatic nucleus; MPOA: medial pre-optic area of the hypothalamus; NPC: neural progenitor cells
Discussion
This review of current evidence shows that K. parviflora is a plant with potential for 16 different diseases and other various pharmacological applications, with the rhizome most popularly used traditionally and investigated, and PMF as the most studied bioactive phytoconstituent. A few effects were only demonstrated at the in vitro stage, including anticancer, antimicrobial, hepatoprotective effects, and potential benefits in ophthalmological diseases. Effects such as antioxidant, antidiabetic, cardiovascular, skin health, urology and immunomodulation have shown positive findings in preclinical studies only, at both in vivo and in vitro levels, thus needing further validation in clinical studies. K. parviflora shows conflicting findings in neuropsychiatry applications whereby an in vivo animal study demonstrated its ability to ameliorate cognitive impairments while no significant effect was observed in healthy adults in terms of changes in physical or psychological stress. Positive findings were consistently seen for anti-inflammatory and anti-obesity across the preclinical and clinical studies. Beneficial effects were also observed similarly via improvements in aging-physical and physiological functions and in the male reproductive system. Details of these general trends of research landscape and research gaps is in Supporting information: Supplemental Table S1. A recent meta-analysis which included 57 articles (both clinical and preclinical studies included) focusing on metabolic syndrome and erectile dysfunction suggested improvements in blood glucose, physical, and exercise performance, but not other metabolic or reproductive function parameters, following K. parviflora administration. 95 The variation in effects could be due to high heterogeneity of intervention including formulation, dose, duration, and study population.
A meaningful research approach for K. parviflora would be focusing on the potentially bioactive compounds. Based on published studies included in this review, the rhizome extracts of K. parviflora that have been explored include those from ethyl acetate, methanolic, ethanol, saline, aqueous, and dichloromethane. From these extracts, the phytochemical compounds of K. parviflora that have been studied include flavonoids (such as tetramethylkaempferol, tectochrysin, anthocyanidins, kaempferiaosides A and B, pentamethylquercetin, and trimethylkaempferol) and methoxyflavones (such as DMF, TMF, TeMF, and PMF). Additional phytochemical studies have identified other compounds within K. parviflora including di-O-methylpinocembrin, bisdemethoxycurcumin, aloe-emodin, 96 phenolic compounds, phenolic glycosides, kaempanosides, 97 and terpenes 45 Several researchers have taken the initiative to study the preclinical efficacies of the mentioned compounds,45,97 but product development or clinical studies related to these compounds are not yet available. Hence, these compounds represent potential areas for further research and product development based on K. parviflora.
Although K. parviflora has been used as a functional food and supplement in various forms for centuries, its safety remains to be ascertained, especially when used on a long-term basis. Sub-chronic toxicity (90 days) study on a standardized hydroalcoholic extract of K. parviflora extract (medium dose at 125 mg/kg body weight/day K. parviflora extract and high dose at 249 mg/kg body weight/day K. parviflora extract) in Sprague-Dawley rats showed no signs of toxicity without any meaningful histopathological changes in the collected organs. No abnormalities with insignificant changes to biochemical and hematological parameters were observed when compared to the control group. Genotoxicity tests using a bacterial reverse mutation assay using five different strains also exhibited no mutations up to 5000 µg/plate. 98
Another aspect of safety that should be considered is herb-drug interactions. An animal study showed herb-drug interaction between K. parviflora extract and paracetamol by decreasing the concentration of paracetamol through accelerated drug clearance and elimination rate in male Wistar rats for nine days. 99 K. parviflora extract was also found to interact with sildenafil, a PDE-5 inhibitor in rats. K. parviflora interfered with first-pass metabolism and absorption of sildenafil in the liver and intestine. This effect could be clinically important as K. parviflora has been investigated for use as a male sexual stimulant or reproductive enhancer, while slidenafil is a common drug used for erectile dysfunction. 100 This could be attributed to its inhibitory effect on p-glycoprotein and multidrug resistance-associated proteins, which are efflux ATP-binding cassette transporters in the intestines that affect drug absorption and oral bioavailability.99,100
Herb-drug interaction studies also showed that K. parviflora extract could affect CYP enzyme activity in rodents. In detail, K. parviflora extract could induce the enzymatic activity of several CYP enzymes, including CYP1A1, CYP1A2, CYP2B, and CYP2E1. 101 Studies on DMF of K. parviflora also showed it significantly reduced the expression of CYP3A11 and CYP3A25 in the liver. The CYP enzyme family is responsible for metabolizing many drugs, such as vitamin D3, dexamethasone and herbs, such as berberine.102–104 Consequently, the use of K. parviflora will eventually increase (when acting as an inhibitor) or decrease (when acting as an inducer) the blood concentration of drugs that are metabolized by CYP enzymes. 105 In addition to harms, herb-drug interactions that increase the bioavailability of the other drugs could potentially be lobbied for benefits instead. For example, K. parviflora extract can increase the oral absorption of certain anticancer drugs (eg paclitaxel), which therefore could be further investigated for its potential to improve the delivery route of the affected drug in a beneficial way (eg, from intravenous injection to oral). 106 Due to the potential interactions, co-administration of K. parviflora with conventional drugs needs to be carefully considered on a case-by-case basis.
Due to concerns for potential raw material sourcing issues, it might be beneficial for local researchers of industry to strategically seek alternative plants which carry the main bioactive phytoconstituents of K. parviflora, ie, the methoxyflavones. For example, DMF can be isolated from the aerial parts of Caesalpinia pulcherrima 107 and rhizomes of Boesenbergia pandurata. 108 Both C. pulcherrima (local name: Bunga Merak) and B. pandurata (local name: Temu Kunci) are plants that were cultivated locally or naturally occurring in Malaysia. 109 Traditionally, C. pulcherrima was used for diarrhea, distended stomach, fever and cough 110 while B. pandurata was used for various skin infections, including ringworm, impetigo, and abscesses; relieving joint pains and malarial symptoms. 111 Another potential local plant which can be further investigated is Orthosiphon aristatus, 112 locally known as misai kucing, which has been traditionally used for diuretic activity. 107 PMF extracted from the leaves of O. aristatus has shown anti-infective properties. 110
The main strength of this review is that we have covered both published (from several electronic databases) and gray literature, both local and international. This approach enabled the review team to collect a variety of literature to synthesize multi-angled and locally relevant views when considering the research landscape and gap for the local researchers and industries in Malaysia.
Review Limitations
A major limitation of this review is that searches, screening, and data extraction were performed by individual investigators instead of independent pair of researchers with cross-checking, which may have led to missing out on some literature. This review also focuses on efficacy rather than safety, which may warrant a separate review of its own in the future. Some considerations for safety aspects were discussed above. Lastly, due to the nature of narrative scoping reviews and our objective to map research landscape, we did not perform critical appraisal on the quality of included studies. Critical appraisal of study findings can be undertaken in future systematic reviews which focus on more specific outcomes instead.
Conclusions
In conclusion, K. parviflora is a plant with potential for various pharmacological applications, with the rhizome most popularly used and investigated and the PMF as the most studied bioactive phytoconstituent. Further research expanding on its traditional use for men's health could be beneficial. However, local research direction must appraise operational considerations, in particular raw material sourcing and sustainability.
Materials and Methods
This review was designed to provide an overview of the current published and gray literature regarding K. parviflora, including its traditional use and scientific literature on its efficacy and safety. This literature review can be broadly divided into two parts: (1) a review of traditional use; and (2) a review of published biomedical research on biological activities and health effects.
Searches on Traditional use
The search for information on traditional uses of K. parviflora was carried out by reviewing published journals, herbal monographs, books, proceedings, and pharmacopoeia accessible within our institution. Among the references reviewed were the ‘Thai Herbal Pharmacopeia 2018’ and ‘Khazanah Perubatan Melayu - Tumbuhan Ubatan (Volumes 1-3)’.1,114–116
Published Literature Review
A systematic search was conducted by investigators on electronic databases including MEDLINE, CENTRAL, and Web of Science for relevant articles published in the past 10 years until October 2022. The keywords combination used include “Kaempferia parviflora”, “Kaempferia rubromarginata”, “Stahlianthus rubromarginatus”,
Inclusion and Exclusion Criteria
The following inclusion and exclusion criteria were applied to identify primary articles that reported biological activities and health effects of K. parviflora.
A. Inclusion criteria
Primary literature documenting the biological activities and health effects of K. parviflora as a sole active ingredient, including its representative compounds in all forms (all plant parts and formulations) Both clinical and preclinical papers Articles on combination products and formulations
B. Exclusion criteria
No language restrictions were applied.
Data Extraction and Analysis
For the purpose of research gap analysis, the following data were extracted:
Plant part Formulation details Pharmacological activity Disease model Level of evidence (ie, type of clinical and preclinical study eg, randomized controlled trials, case report, in vivo, in vitro, in silico etc) Key efficacy findings
Descriptive analysis of the health benefits and biological activities of K. parviflora was performed. Analysis of efficacy focused on the pharmacological actions and level of evidence to identify research gaps and the potential of this plant.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241281615 - Supplemental material for Application of Kaempferia parviflora: A Perspective Review
Supplemental material, sj-docx-1-npx-10.1177_1934578X241281615 for Application of Kaempferia parviflora: A Perspective Review by Terence Yew Chin Tan, Xin Yi Lim, Puspawathy Krishnan, Siti Hajar Muhamad Rosli, Janice Sue Wen Chan, Yee Lin Voon, Ida Farah Ahmad, Thien Chen Siau, Norizah Awang and Ami Fazlin Syed Mohamed in Natural Product Communications
Footnotes
Acknowledgements
We would like to thank the Director General of Health Malaysia, Deputy Director General of Health Malaysia (Research & Technical Support), Director of Institute for Medical Research, and Head Centre of Herbal Medicine Research Centre for their support and permission to publish this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statements
Ethical Approval is not applicable for this article. This article does not contain any studies with human or animal subjects. There are no human subjects in this article and informed consent is not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
