Abstract
Background
Bladder cancer (BCa) is the most common malignant tumor of the urinary system, and there remains an urgent need for more effective therapeutic strategies. Xiaozheng Decoction has been reported to prevent postoperative recurrence of BCa; however, its specific pharmacological effects and molecular mechanisms remain inadequately defined.
Objective
This study aimed to investigate the inhibitory effects of Xiaozheng Decoction on BCa progression and elucidate its underlying molecular targets.
Methods
CCK-8, colony formation, as well as FCM assays showed the effects on bladder cancer cell growth. Transwell assays showed the effects on cell motility. qPCR as well as Immunoblot assays showed the effects on miR-21/ SMAD7 axis. Luciferase activity assay, quantification PCR, and Immunoblot assays showed the effects on the interact between miR-21 and SMAD7. Tumor growth assays were performed in mice.
Results
The results demonstrated that Xiaozheng Decoction suppressed the growth and induced apoptosis of BCa cells, and also significantly reduced their cell migration. Mechanistically, Xiaozheng Decoction could downregulate the expression of miR-21 and its downstream target SMAD7. Further analysis confirmed that miR-21 directly binds to and suppresses SMAD7 expression. In vivo experiments showed that Xiaozheng Decoction also inhibited tumor growth in a xenograft model of BCa.
Conculsion
Taken together, these findings indicate that Xiaozheng Decoction exerts anti-tumor effects in BCa by modulating the miR-21/SMAD7 signaling axis, thereby suppressing both proliferation and migration of BCa cells.
Introduction
Bladder cancer (BCa) is the most common malignant tumor of the urinary system and ranks among the top ten most frequently diagnosed cancers worldwide, with incidence and mortality rates in men being approximately four times higher than in women. 1 In China, both the incidence and mortality of BCa have shown a rising trend in recent years. Clinically, 70% to 80% of newly diagnosed BCas are non-muscle-invasive tumors, for which the standard treatment involves transurethral resection combined with intravesical chemotherapy.2,3 However, the efficacy of first-line cisplatin-based intravesical chemotherapy remains suboptimal, with recurrence rates reaching up to 60%. Recurrent disease may progress to muscle-invasive BCa, contributing to increased malignancy and drug resistance.4,5 Thus, there is an urgent need for more effective therapeutic strategies and preventive interventions to combat this disease.
The empirical formula “Xiaozheng Decoction,” developed by the urologist Youfang Liu, has been reported to prevent recurrence of BCa after surgery. 6 This traditional Chinese herbal formulation consists of coix seed, Astragalus, Polygonatum (yellow essence), Hedyotis diffusa, Poria, Curcuma zedoaria (Zedoary), and Lophatherum gracile (flower stem). 6 Previous clinical studies have indicated that Xiaozheng Decoction, when combined with intravesical perfusion, may prolong disease-free survival and reduce metastatic spread in patients with BCa. 7 In addition to being used to treat BCa, Xiaozheng Decoction has also demonstrated therapeutic potential in treating endometriosis associated with blood stasis and in suppressing the growth of hepatocellular carcinoma through anti-inflammatory mechanisms. 8 Moreover, recent network pharmacology analyses suggest that its anti-cancer activity may be mediated by modulation of the GSK3β/β-catenin signaling pathway.7,9 Nevertheless, the precise pharmacological mechanisms and molecular targets through which Xiaozheng Decoction exerts its effects on BCa remain incompletely understood.
This study aimed to investigate the inhibitory effects of Xiaozheng Decoction on BCa progression and to elucidate its molecular mechanism of action. The results demonstrated that Xiaozheng Decoction could suppress the proliferation and migration of BCa cells by modulating the miR-21/ SMAD7 signaling axis, thereby supporting Xiaozheng Decoction as a promising therapeutic candidate for the treatment of BCa.
Methods
Cell Culture
Human BCa T24 cells were obtained from the Chinese Academy of Sciences. Cells were cultured in RPMI-1640 complete medium under standard conditions. For treatment, T24 cells were incubated with Xiaozheng Decoction extract (MXD, available in our laboratory) at concentrations of 0, 50, 100, or 200 μg/mL for 24 h. Cells treated with Bropirimine (100 ng/mL; purchased from Sigma-Aldrich) served as positive control. The effects of MXD on T24 cells were subsequently validated and used for downstream experiments.
Immunoblotting
Total protein was extracted from T24 cells using RIPA lysis buffer and quantified using a BCA protein assay. Equal amounts of protein were separated by SDS-PAGE and transferred onto PVDF membranes. The membranes were then incubated with primary antibodies against SMAD7 (ab216428, 1:500, Abcam), AP-1 (#701702, 1:1000, Invitrogen), Bcl-2 (ab32124, 1:500, Abcam), Nanog (ab109250, 1:1000), Maspin (ab182785, 1:500), NF-κB (ab288751, 1:500), c-Myc (ab32072, 1:1000), and β-actin (ab8226, 1:3000). After incubation with the appropriate HRP-conjugated secondary antibodies, immunoreactive bands were visualized using a chemiluminescence detection system. All reagents, unless otherwise specified, were obtained from Wuhan Google Co., Ld
Cell Viability Assays
For MTT assays, T24 cells were seeded in 96-well plates and incubated at 37 °C. After treatment, MTT reagent was added and incubated for 4 h. The resulting formazan crystals were dissolved in 150 µL of DMSO, and absorbance was measured at 450 nm to assess cell viability at 24, 48, and 72 h post-treatment.
For colony formation assays, cells were seeded in 6-well plates and cultured for 10 days. Colonies were then fixed with paraformaldehyde for 20 min and stained with 0.2% crystal violet for 10 min. Plates were photographed for qualitative analysis of colony formation.
Quantitative PCR (qPCR) Assay
Total RNA was extracted from the indicated cells using TRIzol reagent (Invitrogen, Shanghai, China), following the manufacturer's instructions. Complementary DNA (cDNA) was synthesized using the PrimeScript™ Reverse Transcription Reagent Kit (Takara Biotechnology Co., Ltd, Dalian, China). Quantitative real-time PCR was performed using a SYBR Green-based detection system (Takara) on a suitable real-time PCR platform. The gene-specific primers used are as listed below:

Transwell-Migration or Invasion Assays
Transwell assays were conducted using BD Falcon cell culture inserts as the upper chambers and 24-well plates as the lower chambers. For invasion assays, the upper chambers were pre-coated with 100 μL of Matrigel (diluted 1:3 with serum-free medium), while uncoated inserts were used for migration assays. A total of 1 × 105 cells were suspended in a serum-free medium and seeded into the upper chamber. After incubation, cells that had migrated or invaded the lower surface of the membrane were fixed with 4% paraformaldehyde, stained with 0.2% crystal violet, and imaged under a light microscope.
Cell Apoptosis Assay
Apoptosis was assessed using an Annexin V-FITC/PI apoptosis detection kit (Beyotime, Beijing, China) according to the manufacturer's protocol. Briefly, T24 cells were washed twice with PBS and subsequently fixed in 70% ethanol at −20 °C for 2 h. After fixation, the cells were incubated with propidium iodide (PI) and fluorescein isothiocyanate (FITC)-conjugated Annexin V at 4 °C in the dark. Apoptotic cell populations were then analyzed using flow cytometry.
Luciferase Assay
For luciferase reporter assays, T24 cells were co-transfected with miR-21 mimics (Riobio, China) and either wild-type or mutant SMAD7 reporter plasmids using Lipofectamine 3000 (Invitrogen), following the manufacturer's protocol. Each transfection included 0.2 μg of luciferase reporter plasmid and 2.5 ng of SV-Renilla luciferase plasmid as an internal control. After incubation, cells were harvested and lysed, and luciferase activity was measured using the Dual-Luciferase® Reporter Assay System (Promega). Relative luciferase activity was normalized to Renilla luciferase activity.
In Vivo Tumor Growth Assay
6-8 weeks old female nude mice were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd (Beijing, China). For tumor growth assay, cells were treated with Xiaozheng Decoction extract (MXD, available in our laboratory) at concentrations of 200 mg/mL in the above description. About 1 × 106 cells were implanted into athymic nude mice, and the tumor size was monitored. All animal experiments were approved by the Ethics Committee of Nanjing Hospital of Chinese Medicine affiliated to Nanjing University of Chinese Medicine for the use of animals and conducted in accordance with the National Institutes of Health Laboratory Animal Care and Use Guidelines.
Immunohistochemical
The sections were first fixed by the use of 4% PFA for 30 min. The sections were subsequently blocked using 2% BSA for another half an hour. The blocked sections were then incubated through the use of antibodies for 2 h, and subsequently incubated with secondary antibodies for another 1.5 h. Then the chromogen substrate was used.
Statistical Analysis
All experiments were conducted in triplicate unless otherwise specified. Quantitative data are expressed as mean ± standard deviation (SD). Statistical comparisons between two groups were performed using an unpaired Student's t-test, while multiple group comparisons were analyzed using one-way analysis of variance (ANOVA) followed by Tukey's posthoc test. A P-value < .05 was considered statistically significant. All statistical analyses and graphical presentations were conducted using GraphPad Prism version 5.0.
Results
Xiaozheng Decoction Inhibited the Growth and Induced Apoptosis of BCa Cells
To evaluate the effect of Xiaozheng Decoction on BCa cell growth, T24 cells were treated with varying concentrations of Xiaozheng Decoction for 24 h. Bropirimine (30 ng/mL) was included as a positive control. MTT assays revealed that Xiaozheng Decoction treatment significantly reduced the viability of T24 cells across all tested concentrations, as indicated by decreased OD570 values (Figure 1a). Consistently, colony formation assays demonstrated a marked reduction in the number of colonies formed in the presence of Xiaozheng Decoction, further supporting its inhibitory effect on cell proliferation (Figure 1b and c).

Xiaozheng Decoction Inhibited the Growth and Induced Apoptosis of Bladder Cancer Cells. a) MTT Assays Showing the Viability of T24 Bladder Cancer Cells Treated with Xiaozheng Decoction (XZD; 50, 100, or 200 μg/mL) or Bropirimine (30 ng/mL) for 24 h. Cell Viability was Assessed by OD570 Measurement. (b-c) Colony Formation Assays Showing the Effect of XZD or Bropirimine on T24 Cell Proliferation. Representative Images are Shown (b), and Colony Numbers Were Quantified (c). (d-e) Flow Cytometry Assays Assessing Apoptosis of T24 Cells Following Treatment with XZD or Bropirimine for 24 h. Representative dot Plots are Shown (d), and Quantification of Apoptotic Cells is Presented (e). *P < .05, **P < .01, ***P < .001. XZD, Xiaozheng Decoction.
Flow cytometry analysis revealed that treatment with Xiaozheng Decoction at 50, 100, and 200 μg/mL led to a significant decrease in the proportion of apoptotic cells compared to the control group (Figure 1d and e), indicating that Xiaozheng Decoction induced apoptosis in BCa cells. Together, these results suggest that Xiaozheng Decoction suppresses both the proliferation and survival of BCa cells.
Xiaozheng Decoction Inhibited BCa Cell Motility
To further explore the effect of Xiaozheng Decoction on cell motility, Transwell assays were performed to assess cell migration and invasion. Xiaozheng Decoction treatment significantly impaired the migratory capacity of T24 cells, as evidenced by the decreased number of cells migrating through the uncoated membranes (Figure 2a and b). Similarly, Xiaozheng Decoction reduced the number of invading cells in Matrigel-coated chambers, indicating suppression of invasive behavior (Figure 2c and d). Bropirimine (30 ng/mL) treatment also inhibited both migration and invasion of T24 cells, consistent with the observed effects of Xiaozheng Decoction. These findings indicate that Xiaozheng Decoction effectively reduces the motility of BCa cells.

Xiaozheng Decoction Inhibited Bladder Cancer Cell Migration. (a–b) Transwellmigration Assays Showing the Migratory Capacity of T24 Cells Treated with XZD (50, 100, or 200 μg/mL) or Bropirimine for 24 h. Representative Images are Shown (a), and Migrated Cells were Quantified Per Field (b). (c-d) Transwell Invasion Assays Showing the Invasive Ability of T24 Cells Under the Same Treatment Conditions. Representative Images are Shown (c), and Invasive Cells were Quantified Per Field (d). ***P < .001. XZD, Xiaozheng Decoction.
Xiaozheng Decoction Down-Regulates the Expression of miR-21 and its Downstream Target SMAD7
To investigate the molecular mechanism underlying the antitumor activity of Xiaozheng Decoction, we assessed the expression of miR-21 and its downstream targets. qPCR analysis showed that treatment with either Xiaozheng Decoction or Bropirimine significantly reduced miR-21 mRNA levels in T24 cells (Figure 3a). To further examine downstream targets of miR-21, we evaluated the expression of several related proteins, including AP-1, Bcl-2, Nanog, Maspin, NF-κB, c-Myc, and SMAD7. Treatment with 200 μg/mL Xiaozheng Decoction led to a downregulation of both mRNA and protein levels of these targets in T24 cells (Figure 3b and c), indicating that Xiaozheng Decoction suppresses the miR-21 signaling axis. These results suggest that Xiaozheng Decoction exerts its inhibitory effects in part through downregulation of miR-21 and subsequent modulation of SMAD7 and related downstream effectors.

Xiaozheng Decoction Down-Regulates the Expression of miR-21/ SMAD7. (a) qPCR Assays Showing the mRNA Levels of miR-21 in Bladder Cancer Cells upon the Treatment of XZD at Concentration of 50, 100, and 200 μg/mL or Bropirimine for 24 h. (b) qPCR Assays Showing the mRNA Levels of AP-1, Bcl-2, Nanog, Maspin, NF-KappaB, c-Myc, and SMAD7 in Bladder Cancer Cells upon the Treatment of XZD at Concentration of 50, 100, and 200 μg/mL or Bropirimine for 24 h. (c) Immunoblot Assays Showed the Protein Levels of AP-1, Bcl-2, Nanog, Maspin, NF-kappaB, c-Myc, and SMAD7 in Bladder Cancer Cells upon the Treatment of XZD at the Concentration of 50, 100, and 200 μg/mL or Bropirimine for 24 h. *P < .05, **P < .01, ***P < .001. XZD, Xiaozheng Decoction. ***P < .001, miR-21 Mimic Versus NC Mimic, XZD, ###P < .001, miR-21 Mimic + XZD Versus XZD. XZD, Xiaozheng Decoction.
miR-21 Binds to SMAD7 and Inhibits its Expression
To investigate whether miR-21 directly regulates SMAD7 expression in BCa cells, we first analyzed the predicted binding site of miR-21 within the 3'-UTR of SMAD7 mRNA (Figure 4a). Dual-luciferase reporter assays revealed that overexpression of miR-21 significantly suppressed the luciferase activity of the wild-type SMAD7 reporter. In contrast, this inhibitory effect was abolished when the miR-21 binding site was mutated, indicating a specific interaction between miR-21 and SMAD7 (Figure 4a). Subsequent qPCR analysis demonstrated that miR-21 overexpression in T24 cells led to a marked increase in miR-21 levels and a concomitant reduction in SMAD7 mRNA expression (Figure 4b). Consistent with these findings, immunoblot analysis confirmed that miR-21 overexpression downregulated SMAD7 protein levels (Figure 4c).
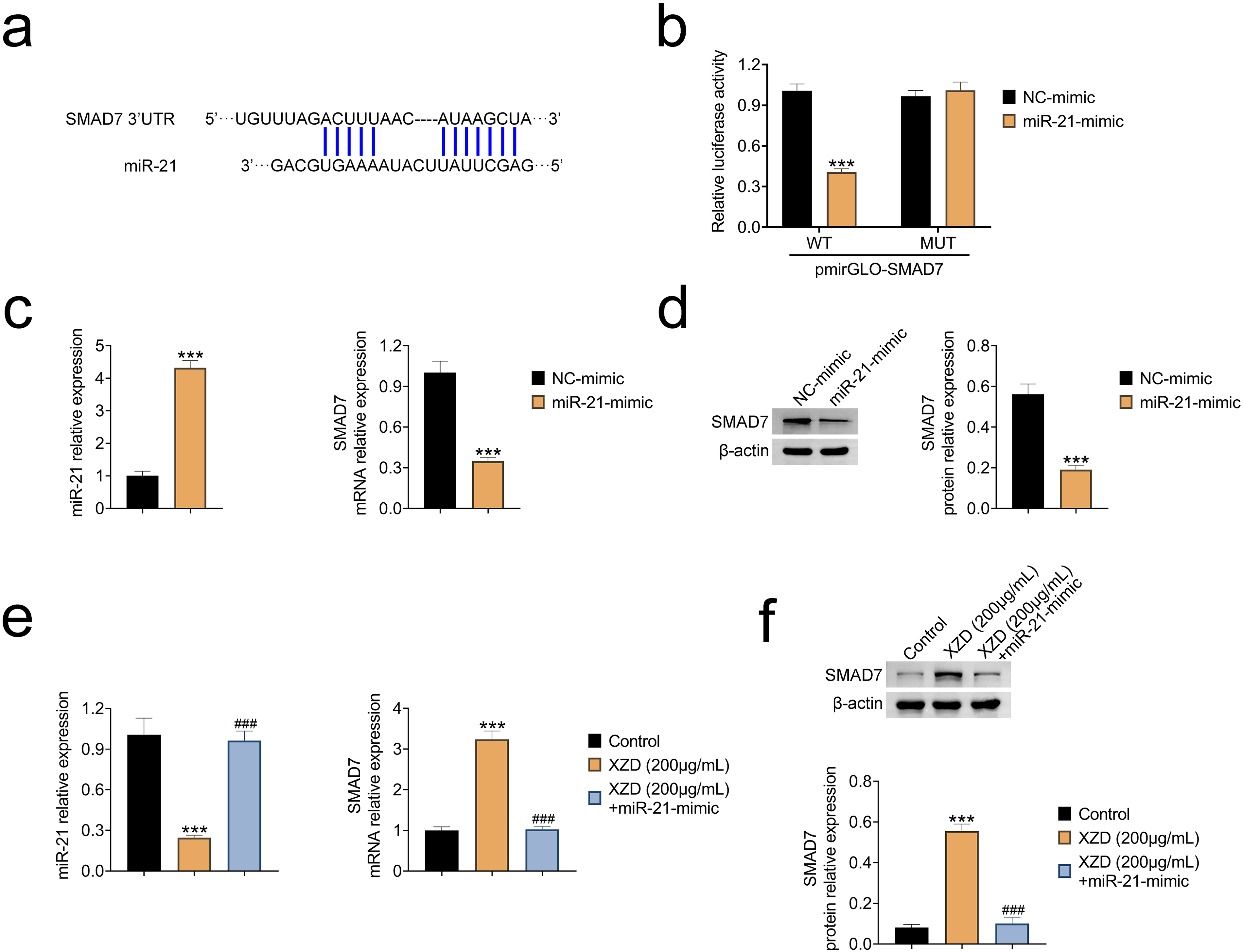
miR-21 Binds to SMAD7 and Inhibits its Expression. (a) Predicted Binding Site of miR-21 in the 3′-UTR of SMAD7. (b) Luciferase Reporter Assay Showing the Effect of miR-21 overexpression on Wild-Type and Mutant SMAD7 Promoter Activity. (c) qPCR Analysis of miR-21 and SMAD7 mRNA Levels in T24 Cells Following Transfection with miR-21 Mimics or Controls. (d) Immunoblot Analysis of SMAD7 Protein Expression under the Same Conditions. (e) qPCR Analysis of miR-21 and SMAD7 mRNA Levels in T24 Cells Treated with XZD with or without miR-21 Mimic Transfection. (f) Immunoblot Analysis of SMAD7 Protein Expression Under Corresponding Treatment Conditions.
Interestingly, treatment with MXD significantly decreased miR-21 expression in T24 cells (Figure 4d). However, when miR-21mimics were transfected into MXD-treated cells, miR-21 levels increased, resulting in reduced SMAD7 mRNA expression despite the presence of MXD (Figure 4d). Immunoblot analysis further confirmed that miR-21 overexpression reduced SMAD7 protein levels even in the context of MXD treatment (Figure 4e and f). These results indicate that miR-21 directly binds to and negatively regulates SMAD7 expression, and that the suppressive effects of Xiaozheng Decoction on miR-21 contribute to SMAD7 upregulation.
Xiaozheng Decoction Inhibits Tumor Growth of BCa Cells in Vivo
The antitumor efficacy of Xiaozheng Decoction was further evaluated in vivo using a nude mouse xenograft model. Intratumoral injection of Xiaozheng Decoction significantly suppressed the growth of T24-derived tumors, as evidenced by reduced tumor volume and weight (Figure 5a). Similar inhibitory effects were observed in the Bropirimine-treated group. qPCR analysis of tumor tissues revealed that both Xiaozheng Decoction and Bropirimine treatments significantly decreased miR-21 expression while increasing SMAD7 mRNA levels (Figure 5b). Immunohistochemical analysis showed reduced Ki-67 staining and increased TUNEL-positive cells in Xiaozheng Decoction-treated tumors, indicating decreased proliferative activity and enhanced apoptosis (Figure 5c). These in vivo findings further support the conclusion that Xiaozheng Decoction exerts antitumor effects in BCa by modulating the miR-21/SMAD7 signaling axis and promoting apoptotic responses.

Xiaozheng Decoction Inhibits Tumor Growth of Bladder Cancer Cells in Vivo. (a) Representative Tumor Images, Tumor Weights, and Growth Curves of T24-Derived Xenografts in Nude Mice Treated with XZD or Bropirimine. (b) qPCR Analysis of miR-21 and SMAD7 mRNA Levels in Excised Tumor Tissues. (c) Immunohistochemical Staining for Ki-67 and TUNEL in Tumor Sections from Each Group. Scale bar, 200 μm. **P < .01, ***P < .001. XZD, Xiaozheng Decoction.
Discussion
BCa can be divided into local progressive BCa and metastatic BCa. 10 For patients with localized disease, radical cystectomy remains the standard of care, often supplemented by adjuvant chemotherapy. In contrast, systemic chemotherapy is the main treatment for metastatic cases.11,12 Therapeutic strategies for BCa typically include chemotherapeutic agents, targeted therapies, and immune checkpoint inhibitors. 11 Treatment selection depends on tumor staging, overall patient health, and individual clinical profiles. In cases of early-stage disease, tumors may be completely resected through minimally invasive approaches, and at this stage, traditional Chinese medicine (TCM) may serve two complementary functions: first, enhancing the patient's general condition and aiding postoperative recovery; and second, reducing the high recurrence rate characteristic of superficial BCa.13–15 In advanced-stage disease, TCM may contribute to tumor suppression and enhancement of systemic immunity. 14 In the present study, we demonstrated that Xiaozheng Decoction effectively inhibited the proliferation and migration of BCa cells, indicating its potential as a therapeutic candidate for BCa treatment.
Our in vivo experiments demonstrated that Xiaozheng Decoction significantly downregulated miR-21 expression while upregulating SMAD7 in tumor tissues, consistent with our in vitro results and further substantiating the conclusion that modulation of the miR-21/SMAD7 axis plays a pivotal role in mediating the anti-tumor effects of Xiaozheng Decoction. Histological analysis revealed an increase in TUNEL-positive cells, indicative of enhanced apoptosis, along with a marked reduction in Ki67 expression, reflecting decreased cellular proliferation in response to Xiaozheng Decoction treatment. These tissue-level changes corroborate the anti-proliferative and pro-apoptotic effects observed in cultured BCa cells, thereby reinforcing the therapeutic potential of Xiaozheng Decoction and its mechanistic action through regulation of miR-21/SMAD7 signaling in BCa.
In recent years, accumulating evidence has highlighted the anti-tumor properties of various traditional Chinese medicines (TCMs) and their bioactive constituents, positioning them as potential therapeutic strategies in oncology. In the context of BCa, these natural compounds have shown considerable promise by inhibiting cancer cell proliferation and enhancing chemosensitivity. For instance, Qicao Sanling Decoction has been reported to suppress glutamine metabolism and inhibit BCa cell growth via downregulation of c-Myc expression. 16 Similarly, orientin has been shown to inhibit the proliferation and promote apoptosis of T24 BCa cells by targeting the NF-κB and Hedgehog signaling pathways. 16 According to TCM theory, the pathogenesis of BCa is associated with a deficiency of vital qi and the accumulation of damp-heat. Consistent with this framework, our findings demonstrate that Xiaozheng Decoction effectively suppresses the proliferation, motility, and survival of BCa cells in vitro and inhibits tumor growth in vivo. These results suggest that Xiaozheng Decoction may serve as a promising therapeutic agent for BCa and further support the potential utility of traditional Chinese medicine in modern oncological treatment.
S SMAD7 is a nuclear protein that interacts with the E3 ubiquitin ligase Smurf2. Upon binding, the SMAD7–Smurf2 complex translocates to the cytoplasm, where it associates with transforming growth factor-β receptor 1 (TGFBR1), promoting the ubiquitination and subsequent degradation of TGFBR1 and its associated signaling proteins.17,18 TGFBR1 also induces the expression of SMAD7, and variants in this gene have been implicated in susceptibility to colorectal cancer type 3 (CRCS3). 19 In BCa, the role of SMAD7 has been extensively studied, with evidence indicating that it regulates cell proliferation, motility, and apoptosis.20,21 Several compounds and proteins have been reported to modulate tumor progression via SMAD7-dependent mechanisms. miR-21 is a highly conserved microRNA known to be upregulated in numerous malignancies, where it functions as an oncogene by targeting tumor suppressor genes.22,23 Its involvement in promoting cancer cell proliferation and resistance to apoptosis has been well characterized. In the present study, we demonstrated that miR-21 directly binds to SMAD7 and suppresses its expression in BCa cells. Moreover, treatment with Xiaozheng Decoction led to downregulation of miR-21 and upregulation of SMAD7, thereby inhibiting tumor cell growth.
Notably, a previous study by Zhuang et al suggested that Xiaozheng Decoction may exert anti-BCa effects by modulating the GSK3β/β-catenin signaling pathway, based on network pharmacology analysis and limited experimental validation. 24 In contrast, our present study focused on the miR-21/SMAD7 axis and provided comprehensive experimental evidence—including both in vitro and in vivo analyses—supporting its critical role in mediating the anti-tumor effects of Xiaozheng Decoction. Therefore, our findings complement and extend the existing literature by identifying an additional molecular mechanism through which Xiaozheng Decoction may exert its therapeutic activity against BCa.
Despite these promising results, this study has several limitations. First, the in vivo experiments were performed using a subcutaneous xenograft model in immunodeficient nude mice, which may not fully recapitulate the complex tumor microenvironment of human BCa. Second, while we identified the miR-21/SMAD7 axis as a key regulatory pathway influenced by Xiaozheng Decoction, we cannot exclude the possibility that other signaling pathways or molecular targets are also involved and merit further investigation. Third, the translational relevance of our findings remains to be validated in clinical settings, and future studies involving patient-derived samples or clinical trials are essential to confirm the therapeutic applicability of Xiaozheng Decoction.
Nevertheless, by elucidating the molecular mechanism of Xiaozheng Decoction through regulation of the miR-21/SMAD7 axis and demonstrating its consistent anti-tumor effects in both cellular and animal models, our study provides a strong foundation for future translational research. These results support the potential clinical application of Xiaozheng Decoction as a complementary therapeutic strategy for BCa.
Conclusion
In summary, Xiaozheng Decoction suppresses the proliferation and migration of BCa cells by targeting the miR-21/SMAD7 signaling axis. These findings highlight its potential as a promising therapeutic candidate for the treatment of BCa and warrant further investigation in preclinical and clinical studies.
Footnotes
Ethical Considerations
Ethical approval was obtained from the Ethics Committee of Nanjing Hospital of Traditional Chinese Medicine Affiliated to Nanjing University of Traditional Chinese Medicine. The approval file were provided in the supplementary.
Author Contributions
Gang Yang and Xiaoyu Zhu designed the study, completed the experiment and supervised the data collection, Changxiao Ye, Yi Wu and Fangze Tao analyzed the data, interpreted the data, Yanchao Xu, Jin Zhao, Ting Huang, Wu Wei and Xuelai Yin prepare the manuscript for publication and reviewed the draft of the manuscript. All authors have read and approved the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the Nanjing Chinese Medicine Youth Talent Training Program, (grant number ZYQ20018).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article. No additional datasets were generated or analyzed.
