Abstract
Introduction
The role of natural products in drug discovery is paramount, given their distinctive features that set them apart from conventional synthetic molecules, offering both advantages and challenges to the drug discovery process. 1 Natural products, sourced from diverse environments such as plants, animals, microorganisms, and marine organisms, encompass a broad spectrum of metabolites, serving as intermediate products in biological systems’ metabolic processes.2,3 Mucilage, a high molecular weight gelatinous substance, widely produced by various plant species, has garnered attention. Composed primarily of polysaccharides with trace amounts of proteins, minerals, and lipids, mucilage has been investigated for its monosaccharide composition and polysaccharide structure. Recently, there has been a shift towards understanding the functional properties of mucilage in connection with its underlying chemistry. 4 Plant-derived mucilage, recognized for its diverse health and food-related benefits, including anticancer, enzyme inhibition, immune-stimulation, and anti-diabetic activity, is a valuable active ingredient in pharmaceutical, functional, and nutraceutical product formulations. Structurally, mucilage presents as a complex of polymeric polysaccharides, featuring highly branched structures with monomer units such as L-arabinose, D-xylose, D-galactose, L-rhamnose, and galacturonic acid. Glycoproteins, along with a variety of bioactive compounds like tannins, alkaloids, and steroids, contribute to its composition. Commonly extracted from the seed coat of various plant species, mucilage's structural variations have been linked to biotic/abiotic parameters and changes in plant physiology, as noted in the literature.5–7 Understanding these structural characteristics is crucial, as multiple studies suggest that the applications and effects of polysaccharides are intricately tied to their specific structural features. 7
Exploring the flora of arid environments is crucial for unraveling the biological traits and biogeographical distribution of plant species. While floristic studies offer insights, numerous aspects of plant biology, taxonomy, and ecology remain poorly understood. 8 The specialized plant community thriving in severe climatic conditions of arid steppic rangelands, covering 15 million hectares in the Algerian steppe, holds significant ecological importance. 9 Despite the Algerian Sahara constituting approximately 80% of the total surface area, limited research has been dedicated to understanding the biological resources unique to this region, which are representative of arid environments. 10 The septentrional Sahara boasts a notably scarce flora, a consequence of harsh conditions and limited resources that challenge the spontaneous survival of living organisms. 11 The Saharan flora exhibits distinct modes of adaptation to drought, categorized into ephemeral plants that emerge after rainfall, completing their vegetative cycle before the soil dries, and permanent plants. 12 Within the extensive Fabaceae family of angiosperms, 13 Astragalus, the largest genus, stands out for its diverse bioactive metabolites, commonly utilized in pharmaceutical and food industries. Found predominantly in Mediterranean climatic regions of Europe and North Africa, the Sahara Desert in Algeria is home to fifteen documented Astragalus species. 14 Notably, Acruciatus holds particular renown intraditional North African medicine. 15
Disorders related to hyperglycemia, prominently diabetes, pose a significant global public health. 16 While various therapeutic strategies, including insulin replacement, enhancing insulin sensitivity, and oral antidiabetic drugs, have been employed to manage diabetes, inhibiting crucial enzymes such as α-amylase and α-glucosidase remains a popular approach to delay glucose release from dietary starch. 17 Inhibiting α-amylase disrupts starch hydrolysis into oligosaccharides, while inhibiting α-glucosidase suppresses disaccharide hydrolysis. 18 The clinical inhibitor of α-glucosidase, acarbose, is associated with gastrointestinal side effects, including flatulence and diarrhea. 19 These adverse effects are attributed to increased abnormal fermentation of partially digested carbohydrates by gut microbiota, often leading to treatment discontinuation. This emphasizes the necessity for searching new inhibitors with improved safety profiles. 20 Plant polysaccharides have demonstrated an inhibitory effect on carbohydrate digestion enzymes, offering a promising alternative to existing inhibitors with fewer associated side effects. 21
To our knowledge, Astragalus cruciatus seeds have not been studied for their polysaccharide composition or biological activities in any previous research. This study is the first to investigate the potential of A. cruciatus seed polysaccharides, setting it apart from other, more extensively explored species within the Astragalus genus. Our work expands the understanding of plant-based polysaccharides and their bioactivities. This study aims to isolate novel polysaccharides from A. cruciatus seeds using a comprehensive analytical approach, including colorimetric assays, UV-Vis scanning, Fourier Transform Infrared spectroscopy (FT-IR), X-ray diffraction (XRD), Scanning Electron Microscopy (SEM), Thin Layer Chromatography (TLC), High Performance Liquid Chromatography (HPLC), and Gas Chromatography-Mass Spectrometry with Electron Ionization (GC-MS-EI) for structural characterization. Furthermore, we assess the inhibitory effects of the extracted mucilage on α-amylase and α-glucosidase, as part of this investigation.
Materials and Methods
General Experimental Procedures
Spectrophotometer (the device is Shimadzu UV-1800, UV–Vis Spectrophotometer) controlled by Agilent Scan software. Fourier transform infrared spectrometer (FT-IR, the Agilent Cary 630 type (Agilent Technologies)). X-Ray Powder Diffraction (PROTO AXRD Benchtop). Scanning Electron Microscope (SEM Zeiss Evo 10). The HPLC system consisted of a Shimadzu LC-9A isocratic pump (Kyoto) equipped with a refractive index detector, the HPLC system was fitted with a ShodexAsahipak NH2P-50 4E column. TLC analyses were carried out on the precoated silica gel GF254 plates (Qingdao Marine Chemical Co., Ltd, P.R.). GC/MS-EI system was an Agilent 6890 GC / 5973 Network Mass Selective Detector, equipped with an OPTIMA-1MS Accent column (Macherey-Nagel; 30 m, 0.32 mm, 0.25 μm). α-amylase (E.C.3.2.1.1) and α-glucosidase (E.C.3.2.1.20) and all the solvents used for isolation were of analytical grade and the solvents used for HPLC were of HPLC grade.
Plant Material
The botanical specimen (A. cruciatus Figure 1) was carefully collected from Hassani Abdelkarim, El-Oued (septentrional Algerian Sahara) (GPS:6.53205N33.27159E) during the months of March and April in 2019 and was identified through a combination of morphological and taxonomic analyses, including examination of key features such as leaf shape, flower morphology and seed characteristics by Pr. SLIMANI Noureddine (Faculty of Biology, University Echahid Hamma Lakhdar, El-Oued, Algeria).

(a) Astragalus cruciatus Link. Plant (b) Astragalus cruciatus Link. Seeds (April 2019).
Extraction and Isolation of the Mucilage
The mucilage component of A. cruciatus was isolated using our previously established method, 22 involving hot maceration of seeds (10 g) in distilled water (20%w/v) at 70 °C for 2 h with moderate stirring (450 rpm). The resulting highly viscous dispersion was subjected to filtration through a fine filter to remove residual debris and the process was repeated three more times, followed by centrifugation (4000 rpm/15 min). The macerate was then concentrated by rotary evaporation at 65 °C to a volume of one-third of its original amount and precipitated by the addition of three volumes of cold ethanol (96%), followed by refrigeration for 24 h at 4 °C. The resulting pellet was recovered via centrifugation (4000 g, 4 °C, 15 min) and washed multiple times with acetone. The resulting mucilage fractions called (PSAC) were then dried at 50 °C for 48 h and pulverized into a fine powder with a mechanical blender with a particle size less than 3 mm.
Chemical Determination
The mucilage (PSAC) composition was analyzed using colorimetric assays. Total sugars were quantified via the phenol-sulfuric acid method with glucose as the standard, 23 while neutral sugars were measured using the 1,3-dihydroxybenzene method with glucose as the standard. 24 The content of uronic acids was determined using the m-hydroxydiphenyl assay, with galacturonic acid as the standard. 25 The protein content was estimated using the Bradford assay with bovine serum albumin as the reference. 26 The total phenolic compounds were quantified by means of the Folin–Ciocalteu method with gallic acid as the reference compound. 27
UV-Visible Spectrum Scanning
The extract (PSAC) was dissolved in distilled water at a concentration of 2.5 mg/mL and analyzed using a UV-visible spectrophotometer in the 200–500 nm wavelength range at a temperature of 25 °C.
FT-IR Spectroscopy
The identification of organic functional groups and primary structure of PSAC was performed by analyzing the FT-IR spectrum in the spectral range of 400–4000 cm−1.
XRD Characterization
The crystal structure of the extract was determined using single crystal X-ray diffraction analysis. Diffraction data was collected at room temperature with Cu Kα radiation (λ = 0.154281 nm) in 2θ ranging from 10° to 90° with a determinate scanning speed of 0.5°/min.
SEM Imaging
The microstructure of the powder was analyzed using scanning electron microscopy (SEM) at magnifications ranging from ×5 to ×500.
Chemical Hydrolysis
To initiate preliminary investigations, PSAC was subjected to acid hydrolysis using 2 M trifluoroacetic acid (TFA) at 100 °C for 4 h. The hydrolyzed product was then analyzed using thin layer chromatography (TLC) and high-performance liquid chromatography (HPLC). 28
Monosaccharide Compositions by Thin Layer Chromatography (TLC)
TLC analysis of the hydrolysate was performed using two different mobile phases. System 1 consisted of a mixture of ethyl acetate, pyridine, water, n-butanol and acetic acid in the proportions 5/4/4/10/2. 29 While System 2 consisted of a mixture of Chloroform, n-butanol, methanol, acetic acid and water in the proportions 4.5/12.5/5/1.5/1.5. 30 To visualize the separated compounds, the plates were developed using the nigrum as revelator and the Rf values for the separated spots were calculated and compared with the Rf values of pure standards.
Monosaccharide Compositions by High Performance Liquid Chromatography (HPLC)
In this study, the sample was analyzed using high-performance liquid chromatography (HPLC) according to a modified method of. 31 The mobile phase consisted of acetonitrile: water (80:20, v/v), and was retained for 15 min at a flow rate of 1.0 mL/min. The sample injection volume was 10 μL and the column temperature was maintained at 35 °C. The HPLC analysis allowed for the separation and quantification of the monosaccharide compositions present in the sample, based on their retention times and refractive index signals.
Monosaccharide Compositions by gas Chromatography-Mass Spectrometry with Electron Ionization
The monosaccharide composition analysis using GC/MS-EI was conducted following the method described by.32,33 Polysaccharides weighing ten milligrams were hydrolyzed using 2 M trifluoroacetic acid (TFA) at a temperature of 120 °C with stirring for 90 min. The resulting hydrolysates were then derivatized by trimethylsilylation using BSTFA: TMCS (N, O bis[trimethylsilyltrifluoroacetamide] with 1% trimethylchlorosilane) in a ratio of 99:1. Afterward, the samples were evaporated under dry nitrogen and dissolved in dichloromethane. The same procedure was applied to the standards containing the monosaccharidesAra (Arabinose), Glc (Glucose), Gal (Galactose), GalA (Galacturonic acid), Rha (Rhamnose), and Xyl (Xylose). During the GC/MS-EI analysis, the following parameters were employed: target ion range of 40–800 m/z, injector line temperature set to 250 °C, trap temperature at 150 °C, split ratio of 50:1, helium pressure of 8.8 psi, and helium flow rate of 2.3 mL/min. Ionization was performed at 70 eV. The temperature program involved initially starting at 100 °C for 3 min, followed by an increase of 8 °C/min until reaching 200 °C, which was maintained for 1 min. Finally, the temperature was further increased at a rate of 95 °C/minupto215 °C.
Inhibition of α-Amylase Activity
To estimate the inhibition of α-amylase activity, a modified method of Kumar et al 34 and Kajaria et al 35 was employed. Briefly, dry tubes were prepared with 180 µL of each dilution of the PSAC sample, ranging from 0.1 to 5 mg/mL, along with acarbose as a positive control or phosphate buffer solution (PBS) as a negative control. To each tube, 90 µL of α-amylase solution (5IU/L) was added, and the reaction mixtures were pre-incubated for 15 min at 37 °C. Following this, 500 µL of 2-chloro-p-nitrophenylα-D-maltotrioside (CNPG3) solution (0.5 mg/mL) was added with gentle stirring and the reaction mixtures were incubated for an additional 10 min at 37 °C. The absorbances were then measured at a wavelength of λ = 405 nm to determine the degree of α-amylase inhibition.
Inhibition of α-Glucosidase Activity
To estimate the inhibition of α-glucosidase activity, a modified method of Bisht et al 36 and Qian et al 37 was employed. Briefly, dry tubes were prepared with 500 µL of α-glucosidase solution (2IU/L) and 100 µL of each dilution of the PSAC sample, ranging from 0.1 to 5 mg/mL, along with acarbose as a positive control or phosphate buffer solution (PBS) as a negative control. Then the reaction mixtures were pre-incubated for 15 min at 37 °C. Following this, 100 µL of p-nitrophenyl α-Dglucopyranoside (p-NPG) solution (4 mM) was added with gentle stirring and the reaction mixtures were incubated for an additional 20 min at 37 °C. The absorbances were then measured at a wavelength of λ = 405 nm to determine the degree of α- glucosidase inhibition.
The inhibition of both α-amylase and α-glucosidase activities was determined and the results were expressed using the following equation
38
:
All the experiments were done in triplicate.
Statistical Analysis
The data obtained from the experiments were analyzed using Microsoft Excel 2007 and origin Pro8.
Results
Chemical Composition of PSAC
The mucilage fraction extracted from A cruciatus seeds appeared as a white powder and its extraction yield is presented in the Table 1, ie, 128.7 g/kg (12.87% w/w). The high proportion (85.47 ± 0.2%) of total sugar in the mucilage confirms that it is predominantly composed of carbohydrates.
Biochemical Characterization of Polysaccharides from Astragalus cruciatus Link. Seeds.

Biochemical analysis of the extracted mucilage revealed that it is primarily composed of neutral sugars (64.13 ± 1.6%) and uronic acid (19.97 ± 1.48%). The extract was found to contain detectable levels of both protein (4.84 ± 0.16%) and polyphenolic compounds (2.72 ± 0.37%).
UV-Visible spectrum Scanning
The Figure 2 displays the UV-visible absorption spectra of the (PSAC) extract, which showed distinct peaks at 210 nm characteristic of polysaccharides and between 250 and 300 nm is indicative of lower protein and/or nucleic acid impurities. Additionally, there was no absorption observed at 620 nm, indicating complete removal of the pigment.

UV-Vis Absorption Spectra of PSAC.
FT-IR Spectroscopy Analysis
Analysis of PSAC structure was conducted using FT-IR in the frequency range of 400 cm−1 to 4000 cm−1 the (Figure 3), which allowed for the identification of key absorption peaks. Results revealed the presence of broad absorption peak at approximately 3421.72 cm−1 that suggest the presence of hydroxyl group. The peak observed in the 2800 to 3000 cm−1 regions may be attributed to the vibrational motion of the methyl group –CH. The absorption peak observed at 1647.21 cm−1 is likely attributed to the presence of bound water in the sample. The peak observed at 1373.32 cm−1 is indicative of the deformation of the C–H group. The spectral features observed in the 900–1200 cm−1 region correspond to the C–C–O, C–OH and C–O–C groups. The absorption peaks observed at 1029.99 cm−1 and 1149.57 cm−1 are indicative of the constituent sugar cycles in the PSAC, specifically belonging to the pyranose cycle. These peaks are generated by the vibrational motion of the COC ether bond. The absorption peaks observed at 815–820 cm−1 and 873–878 cm−1 are indicative of the presence of α and β anomeric configurations and glycosidic bonds in the sample, attributed to α-D-galactopyranose and β-D-mannopyranose, respectively.

FT-IR Spectra of PSAC.
XRD Characterization
The x-ray diffraction (XRD) patterns obtained the (Figure 4) confirm the amorphous nature of the PSAC, as indicated by the presence of a broad peak around 2θ = 20°, which is characteristic of amorphous structures.

XRD Pattern of PSAC.
SEM Imaging
The Figure 5 displays the SEM findings of PSAC. At a magnification of 500x and 100x, the biopolymer appears to exhibit distinct layers. Increasing the magnification to 20x reveals a smooth topography with a uniform composition, suggesting homogeneity. At 5x, the smooth and glittering surface of the galactomannan biopolymer becomes even more apparent, indicating its structural integrity.

SEM Imaging of PSAC.
Thin Layer Chromatography (TLC)
The hydrolysis of the PSAC extract in an acidic environment resulted in the liberation of individual monosaccharide units, which were then subjected to characterization through the use of thin layer chromatography (TLC) utilizing two different systems. The first system (Figure 6a) revealed two distinct spots in the sample's migration pattern, Rf1 = 0.43 and Rf2 = 0.54 which corresponded to the migration patterns of galactose and mannose, respectively. In the second system (Figure 6b) four spots were observed in the sample's migration pattern, Rf1 = 0.18, Rf2 = 0.22, Rf3 = 0.45 and Rf4 = 0.531 which corresponded to the migration patterns of galacturonic acid, glucuronic acid, galactose and mannose, respectively. These findings are in agreement with the results obtained previously from the colorimetric analysis of neutral sugars and uronic acids. The outcome of the thin layer chromatography (TLC) separation of the extract using the first system indicated the presence of a galactomannan-type polysaccharide in the sample. On the other hand, the use of the second system suggested the presence of galactomannan and pectin-type polysaccharides in the extract.

Chemical Characterization of PSAC (a) TLC Chromatograms of PSAC by the first system. (b) TLC Chromatograms of PSAC by the Second System (A. Gal : Galacturonic Acid, A. Glc: Glucuronic Acid, Ara: Arabinose, Gal: Galactose, Glc: Glucose, Man: Mannose and Rha: Rhamnose).
High Performance Liquid Chromatography (HPLC)
The chromatogram in Figure 7 displays the monosaccharide composition of the pure standards (galactose and mannose) and the extract. Which reveals the separation of various sugars present in the PSAC hydrolysat. The detailed information for each sugar is summarized in Table 2. It is important to note that the total percentage for neutral sugar did not add up to 100% The total amount of neutral sugar obtained by HPLC was approximately 49.556%, which is lower than the value obtained from the colorimetric assay for neutral sugar analysis, where the result showed 64.13%. The monosaccharide analysis of PSAC revealed that the extract contained approximately 71.82% mannose and 28.16% galactose, which suggests the presence of galactomannan. The ratio Man/Gal of the galactomannan in the present study was (2.54) and this value can significantly impact its properties.
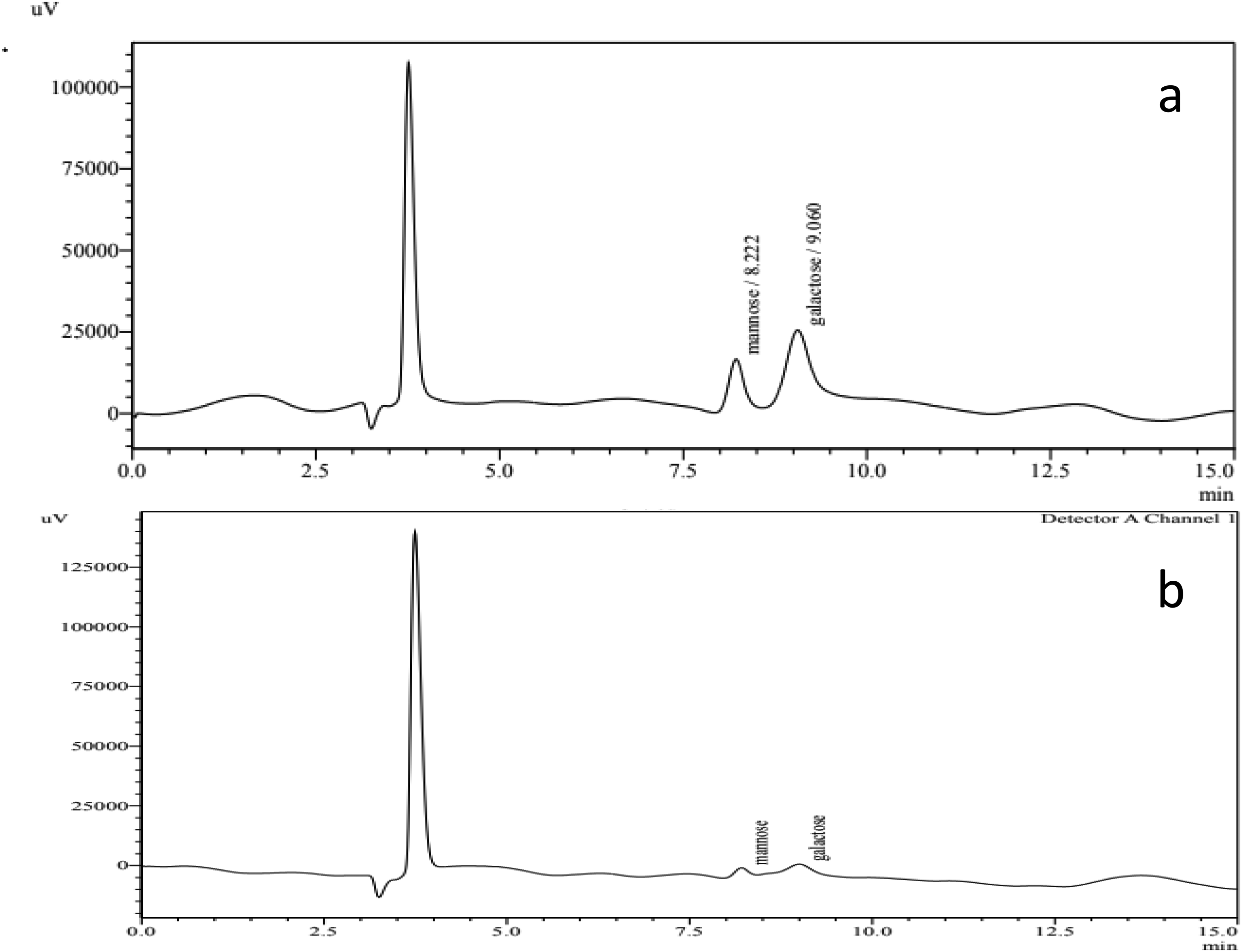
HPLC Chromatogram (a) The Pure Standards (Galactose and Mannose). (b) Monosaccharide Compositions of PSAC from Astragalus Cruciatus Link.
Monosaccharide Composition of PSAC from Astragalus cruciatus Link. Seeds.

Monosaccharide Compositions Bygas Chromatography-Mass Spectrometry with Electron Ionization (GC-MS-EI)
The determination of the monosaccharide composition of the plant extract is confirmed by GC-MS-EI analysis after trimethylsilylation (Figure 8 and Table 3). It is important to note that the total percentage of neutral sugars did not reach 100% due to the presence of uronic acids, proteins, phenolic compounds and other constituents still present in the mucilage fraction even after the extraction process. The total amount of neutral sugar present in the extract was approximately 71.18% for PSAC, suggesting the presence of galactomannans. The monosaccharide composition for PSAC clearly demonstrates the predominant presence of galactomannan with 74.22% D-mannose and 24.37% D-galactose, with a Man/Gal ratio of 3.04, Small traces of pectic derivatives (visible between 10-12 min) and a small trace of D-glucose were also observed.
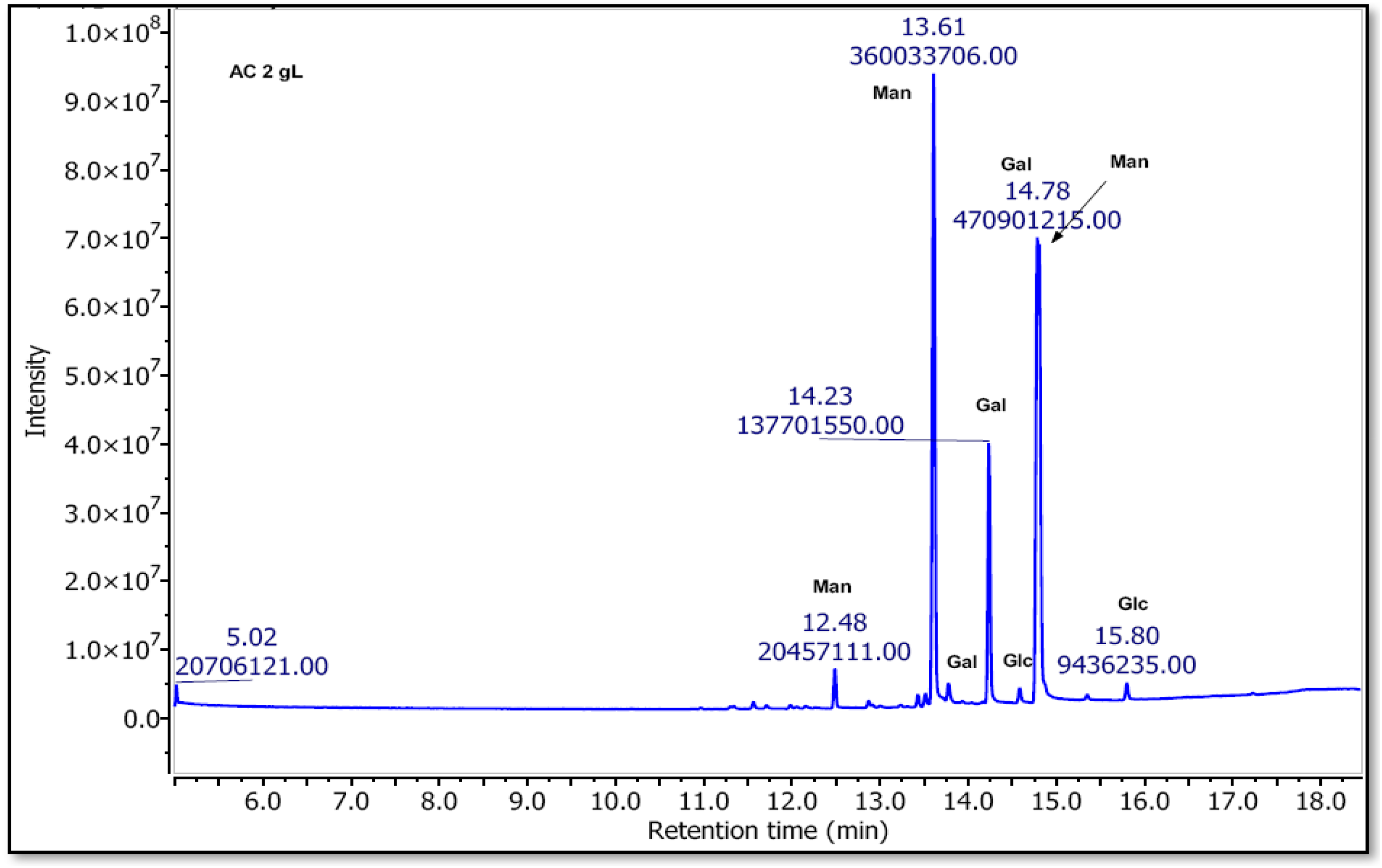
GC-MS-EI profile after Trimethylsilyl-O-Glycosides Derived from Polysaccharides of Astragalus cruciatus Link. seeds.
GC-MS-EI Profile After Trimethylsilyl-O-Glycosides Derived from Polysaccharides of Astragalus cruciatus Link. Seeds.

However, The HPLC and GC-MS-EI analyses revealed differences in monosaccharide composition, with [mannose and galactose] being identified as major components. These discrepancies may be attributed to the sensitivity and specificity of the methods employed. HPLC, relying on aqueous-based separations, primarily detects neutral and charged monosaccharides, whereas GC-MS, involving derivatization, may yield varying results due to incomplete derivatization, selective ionization, or differences in volatility of the monosaccharides. Additionally, variations in sample preparation (eg, acid hydrolysis) could influence the detectable monosaccharide profile.
Evaluation of Antihyperglycemic Activity
The mucilage fraction of A. cruciatus seeds was evaluated for its in vitro antihyperglycemic properties by quantifying its inhibitory effect on the activities of α-amylase and α-glucosidase and comparing it with acarbose, a positive control (as illustrated in Table 4).
IC50Value of PSAC and Acarbose against α-amylase and α-glucosidase.

The data presented in Table 4 demonstrated that PSAC exerts inhibitory effect on α-amylase with an IC50 value of 2.58 ± 0.56 mg/mL, which is superior to that of acarbose (0.295 ± 0.006 mg/mL). Additionally, The extract exhibited a weak inhibitory effect on α-glucosidase.
Discussion
The mucilage fraction extracted from A cruciatus seeds is predominantly composed of carbohydrates. Compared to other Astragalus species seeds, such as A. cicer (5.9%), 39 A. lehmannianus (4.8%), 40 A. sericeocanus (3.6%), 28 A. danicus (3.4%), 41 A. alpinus (0.6%), and A. tibetanus. 42 the carbohydrate yield from the seeds of the studied plant was notably higher. It also surpassed the carbohydrate content of seeds from endemic plant species in the septentrional Algerian Sahara, such as A. armatus (4.21%) 43 and A. gombo (6.8%), 44 but was lower than A. gyzensis seeds (20.69%), 22 This suggests the potential of A. cruciatus seeds as a rich source of dietary carbohydrates.
Allocation of resources to carbohydrate storage in plants is a key trait influencing ecological and economic strategies. Species storing water-soluble poly- and oligosaccharides as primary carbon storage compounds often exhibit a slow economic strategy, characterized by conservative resource use and long-term persistence under stressful conditions, especially drought. 45 Modifications in structural sugars, including those forming part of cell wall components, play a crucial role in conferring drought tolerance to plants. The major constituents of the plant cell wall, such as cellulose, hemicelluloses (mannan, xyloglucan, xylan, and mixed-linked glucan), and lignin, provide mechanical strength and support to the plant cell. Changes in the biosynthesis and deposition of these cell wall components can enhance a plant's resistance to drought stress by strengthening the cell wall and improving water retention. 46
According to, 47 Plant polysaccharides can be classified into intracellular polysaccharides, extracellular polysaccharides and cell wall polysaccharides. Different methods are used to extract these polysaccharides from different plant tissues and the extraction efficiency can be improved by optimizing the extraction conditions. Studies have shown that the extraction rate of polysaccharides is particularly sensitive to temperature, with optimal extraction rates observed at temperatures below 92 °C. However, the use of certain acidic extraction and purification methods, such as trichloroacetic acid (TCA), can lead to the degradation of certain polysaccharides. Therefore, careful selection of extraction and purification methods is critical for obtaining high-quality plant polysaccharides. 48 The neutral sugar content is consistent with the typical biochemical composition of other polysaccharides extracted from the seeds of Astragalus species.22,28,39–43 The uronic acid content of the extracted polysaccharides was found to be consistent with that of polysaccharides from A. gyzensis seeds, 22 and notably higher than the values reported for polysaccharides extracted from other types of seeds. A. armatus and A. gombo.43,44 The observed differences in biochemical composition can likely be attributed to variations in both the plant species and the harvest area. The soil type, climatic conditions and geographical location can all impact the biochemical composition of plant tissues, including polysaccharides. These findings suggest that the mucilage extracted from this species could have similar physicochemical properties and potential applications as other Astragalus-derived polysaccharides, such as in the food, pharmaceutical and cosmetic industries. Efforts to fully remove the protein component were unsuccessful, possibly due to the presence of polysaccharide conjugates challenging to separate. 47 Caution is warranted in using protein separation techniques, as they may lead to polysaccharide degradation and loss of native structure due to the covalent linkage between protein and polysaccharide moieties within glycoproteins. 49
To ensure an accurate structural analysis of the mucilage, it is important to first determine its purity by estimating the proportion of its polysaccharide components and detecting the presence of impurities. The term “total sugars” encompasses monosaccharides, oligosaccharides and polysaccharides and distinguishes them from other non-sugar components (such as proteins, lipids, aromatic compounds, minerals, etc) that may be present. 50
The peak at around 190 nm is characteristic of polysaccharides, while the peak appear around 280 nm is indicative of lower protein and/or nucleic acid impurities.50,51 Additionally, there was no absorption observed at 620 nm, indicating complete removal of the pigment. 47 These findings are consistent with previous results obtained through colorimetric assays.
In the field of polysaccharide structural analysis, IR spectroscopy has emerged as a potent technique. This approach is highly sensitive to the position and anomeric configuration of glycosidic bonds, enabling the identification and characterization of various polysaccharides. 52 FT-IR displayed the characteristic bands that have been frequently reported in the literature for galactomannans. 43 as reported by other studies.43,53–57
The x-ray diffraction (XRD) patterns are consistent with previous studies that have reported similar XRD patterns related to amorphous galactomannan extracted from Delonixregia seeds. 58 Polysaccharides are known to interact with water molecules, leading to structural transitions that can affect their molecular mobility and functional properties, particularly in relation to amorphous-crystalline transitions. 59
SEM is a powerful tool used in scientific research to observe the surface morphology and structure of materials at very high magnifications. It utilizes electron beams to scan the surface of a sample and generate detailed images. 60 The finding is consistent with other studies.60,61
Thin layer chromatography (TLC) is in agreement with the results obtained previously from the colorimetric analysis of neutral sugars and uronic acids. The outcome of the thin layer chromatography (TLC) separation of the extract using the first system indicated the presence of a galactomannan-type polysaccharide in the sample. On the other hand, the use of the second system suggested the presence of galactomannan and pectin-type polysaccharides in the extract. Pectin, which is mainly composed of D-Galacturonic, is a predominant component in many plant tissues. D-Glucuronic, a key component of various natural polysaccharides, is a vital element in metabolic processes that occur within living organisms. 62 The findings of our study exhibit a resemblance to the thin layer chromatography (TLC) analysis of a hydrolyzate of guar galactomannans using two monophasic solvent systems: first, n-butanol/ethanol/water (4/1/1) and subsequently, n-butanol/acetic acid/water (4/1/1). The obtained results from the TLC revelation demonstrated the presence of two distinct spots that corresponded to D-mannose and D-galactose. 63 The hydrolysis of the polysaccharide isolated from unripe pods of Caesalpinia spinosa in an acidic medium liberated free monosaccharide units, which were subsequently characterized using thin layer chromatography (TLC) techniques. The results of the TLC analysis revealed that the hydrolyzed galactomannan sample migrated with the same Rf value as a 1:1 mixture of galactose and mannose monosaccharide units. 64
The HPLC finding suggests that the solubility of galactomannan in water decreases as the number of galactose side chains decreases. 65 Varying ratios have been reported in numerous plant species, such as locust bean gum (approximately 4:1), tara gum (about 3:1), guar gum (about 2:1) and fenugreek gum (about 1:1), according to several studies. 66
GC-MS-EI analysis is consistent with the presence of galactomannan found in polysaccharide extracts from Fabaceae seeds. The monosaccharide composition of the seed polysaccharides and the ratio between galactose and mannose are characteristic of Fabaceae galactomannans. Molecular, chemical and structural variations leading to differences in functional behavior and distribution contribute to the wide variety of plant polysaccharides that exist. 67 The Man/Gal ratio of seed galactomannan is close to that of tara gum. Compared to other galactomannans extracted from seeds of different leguminous plants, the Man/Gal ratio was higher than those estimated for Astragalus gombo Bunge (1.7), 44 Astragalus armatus (1.6) 43 and Alhagi maurorum Medik. (2.2). 68
In terms of bioactivity, the galactomannan with a higher Man/Gal ratio may possess a more rigid structure, allowing for more effective interactions with enzymes like α-amylase, which could lead to inhibitory effects. This structural stability could facilitate the formation of stable enzyme–polysaccharide complexes, thereby impairing enzyme function and contributing to the observed biological effects. Moreover, the relationship between the Man/Gal ratio and the inhibition of α-amylase and α-glucosidase has been explored in various studies. For instance, polysaccharides with a higher mannose content, such as those found in tara gum (Man/Gal ratio of about 3:1), have been shown to exhibit inhibitory effects on α-amylase. 69 Similarly, our findings may suggest that the galactomannan extracted from Astragalus cruciatus could demonstrate comparable or even enhanced inhibitory effect due to its higher mannose content. The varying ratio of galactose and mannose influences the degree of enzymatic inhibition, where a higher mannose content could enhance the polysaccharide's ability to disrupt the activity of α-amylase and α-glucosidase, critical enzymes involved in carbohydrate digestion and glucose regulation.
The findings reported in another study observed that guar galactomannan exhibits a direct noncompetitive inhibitory effect on α-amylase, suggesting the possibility of direct binding between galactomannan and the enzyme. The resultant formation of a galactomannan–α-amylase complex may render the enzyme inactive, leading to the observed inhibitory effect. 69 Additionally, the fenugreek seed endosperm galactomannan demonstrated dose-dependent inhibition of both α-amylase and α-glucosidase, with IC50 values of 21.08 ± 0.085 and 67.17 ± 5.15 μg/mL, respectively. 70 In contrast, polysaccharides extracted from Astragalus gyzensis Bunge exhibited a weak inhibitory effect on α-glucosidase, while showing a antihyperglycemic effect due to the inhibition of α-amylase (IC50 = 0.8 mg/mL). 22 Our findings are inconsistent with a previous study reporting a α-glucosidase inhibitory effect of an APS extract from dried Radix Astragalus, which may be due to differences in the chemical composition of the polysaccharides. 71 It is important to note that the weak inhibition of α-glucosidase observed in our study does not negate the antidiabetic benefits of the galactomannan. In fact, the modest α-glucosidase inhibitory effect may still contribute to the overall antidiabetic activity, as even a low level of inhibition can delay glucose absorption and modulate postprandial hyperglycemia. Moreover, polysaccharides like galactomannan may work through multiple mechanisms that extend beyond enzyme inhibition, including modulation of glucose uptake. In an in vitro study, fenugreek galactomannan was shown to enhance glucose uptake by hemidiaphragm. 72 Furthermore, the administration of Adenanther apavonina L. galactomannan at concentrations of 1% and 2% over 21 days resulted in significant glycemia reduction compared to diabetic control groups 73 This evidence suggests that while the α-glucosidase inhibition might be weak, the overall therapeutic potential of galactomannan for managing diabetes could involve a combination of factors, including α-amylase inhibition, glucose uptake enhancement, and other as-yet-uncertain mechanisms.
Although this study provides valuable insights into the physicochemical properties and biological activities of galactomannan from Astragalus cruciatus seeds, some limitations should be acknowledged. The study was conducted on a limited sample size, which may affect the generalizability of the findings. Additionally, the biological activity was assessed in vitro, and further in vivo studies are required to confirm its therapeutic potential. Future research should also explore the long-term stability and formulation potential of this galactomannan in pharmaceutical applications.
Conclusion
In conclusion, our research has successfully extracted and identified a novel galactomannan from the mucilage fraction of A. cruciatus seeds, with a mannose to galactose (Man/Gal) ratio of 3.04. In vitro biological assays revealed its antihyperglycemic effects, primarily through the inhibition of α-amylase activity. Despite showing limited inhibition of α-glucosidase, these findings highlight the potential of this galactomannan as an active ingredient in antidiabetic formulations. The results of this study provide a foundation for future research exploring the full therapeutic potential of this galactomannan. Further studies will focus on the purification and comprehensive characterization of the polysaccharide to better understand its structural features and their relationship to bioactivity. In particular, in silico approaches will be utilized to model its interaction with key enzymes such as α-amylase and α-glucosidase, enabling a more precise elucidation of its inhibitory mechanisms. In vitro and in vivo studies will also be crucial in assessing its efficacy and safety as a therapeutic agent for managing hyperglycemia and diabetes. Moreover, given the promising bioactivity of this galactomannan, future research could explore its synergistic effects with other natural compounds to enhance its therapeutic outcomes. Investigating its role in modulating glucose metabolism, reducing postprandial hyperglycemia, and assessing its in diabetic animal models will be key steps in moving toward clinical applications. Finally, evaluating the long-term safety and biocompatibility of this galactomannan in chronic disease models will be essential for its development as a viable and sustainable alternative in diabetes management.
Footnotes
Acknowledgements
The author would like to thanks Pr. Lanez Touhami the director of the VTRS Lab El Oued Algeria.
Ethical Considerations
This study is not applicable to both human and/oranimal studies that require ethical approval.
Author Contributions
All authors contributed to the study AT: corresponding author, conception and design, methodology development, data acquisition, analysis and data interpretation, english revision. ZB: study supervision. AEC, AM, AVA, MM: conception revision. NAR, MB, NH: data acquisition. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that supports the findings of this study are available within the article.

