Abstract
Aim The effects of Atractylodes macrocephala Koidz. (Baizhu) and Citrus aurantium L. (Zhishi) (ZhiZhu pill, ZZP) on the defecation function, intestinal bacteria, and short-chain fatty acid metabolism of slow transit constipation (STC) rats were observed, and the mechanism of ZZP was initially understood.
Keywords
Introduction
Constipation is a common chronic functional disorder of the digestive system and is prevalent in the elderly, with clinical manifestations such as hard stools, difficult discharge, and reduced frequency of bowel movements.1,2 However, the prevalence of constipation has increased due to various factors such as fast-paced life, stressful work, irregular diet, and late nights, and there is a trend of younger people suffering from constipation. 2 Clinical symptoms of constipation are classified into three types: outlet obstruction constipation, slow transmission constipation (STC), and mixed constipation. 3 Among them, STC is the most common one, accounting for about 10%-45% of all constipated patients, 4 and long-term constipation, if not treated promptly and effectively, can eventually lead to colon cancer, which seriously affects people's physical and mental health. At present, western medicine treatment of chronic transmission type constipation is mostly based on improving gastrointestinal dynamics and relieving diarrhea, but the effect is not ideal, and it is easy to occur drug resistance, dependence, and other characteristics. 5 Therefore, it is necessary to find medications for chronic constipation.
Traditional Chinese medicine can affect constipation by adjusting intestinal flora. 6 Flax seeds can improve the defecation situation of elderly patients with chronic constipation, change the intestinal microecological structure, and be used as an effective dietary supplement for the treatment of chronic constipation. 7 Studies of Chinese herbs in the field of constipation have been reported extensively, and Atractylodes macrocephala Koidz. (Baizhu) and Citrus aurantium L. (Zhishi) are commonly used in the treatment of constipation, and the two are often used in combination to study functional constipation. 8 Simotang enhances gastrointestinal motility, motilin and cholecystokinin expression in chronically. 9 Additionally, Simotang Alleviates the Gastrointestinal Side Effects of Chemotherapy by Altering Gut Microbiota. 10 Previous studies have shown that Atractylenolide III, the active ingredient in Atractylodes macrocephala Koidz., can improve TNBS-induced intestinal inflammation in mice by reducing oxidative stress and modulating intestinal microbiota. 11 The combination of Atractylodes macrocephala Koidz. and Citrus aurantium L. alleviated compound diphenoxylate tablets-induced STC in rats through intestinal microbiota. 12 In addition, clinical studies have confirmed the therapeutic effect of Atractylodes macrocephala Koidz. and Citrus aurantium L. on functional dyspepsia. 13 ZhiZhu pill (ZZP) is made from fried Citrus aurantium L. and fried Atractylodes macrocephala Koidz. to strengthen the spleen and eliminate food. Our group has been devoted to the research of Atractylodes macrocephala Koidz. and Citrus aurantium L. on functional constipation, and the research shows that ZZP is beneficial to STC, but there is still a lack of systematic research on the therapeutic effect of ZZP on STC, and the potential mechanism of ZZP to regulate intestinal motility to improve constipation is unclear. It is necessary and urgent to study the therapeutic effects and mechanisms of ZZP on STC to provide new ideas for the treatment of STC.
In recent years, studies have gradually shown that intestinal microbiota plays a very important role in the pathogenesis of chronic constipation. 6 Nearly 1 × 1014 microorganisms are present in the human digestive system and these microorganisms significantly influence the immune function, metabolic function, and intestinal barrier function of the host and together participate in various important life activities of the host.14,15 Also, metabolomics is commonly used in STC studies, where disruption of the intestinal microbiota interferes with endogenous metabolites, leading to reduced excitability of intestinal neurons and reduced intestinal transit capacity.16,17
Based on the above background, loperamide hydrochloride was selected to replicate STC rats in this study to investigate the therapeutic effect of ZZP on STC. It is proposed to study the effect of ZZP on the pathophysiology of STC by intestinal motility and colonic histology. Serum GAS (gastrin) and SP (substance P) were used to test the effect of ZZP-regulated gastrointestinal hormones on intestinal motility. In addition, 16S rRNA gene high-throughput sequencing was used to monitor the structure and composition of the gut microbiome to determine the changes in species abundance caused by ZZP. LC-MS-targeted metabolomics was used to analyze the changes in short-chain fatty acids (SCFAs) in rat feces. The present study further elucidates the therapeutic mechanism of STC and provides more detailed data to support the therapeutic role of ZZP.
Material and Methods
Preparation of ZZP
ZZP is a classic prescription with a clinically proven high safety profile. ZZP was prepared by North Sichuan Medical College. The ZZP was made from fried Citrus aurantium L. and fried Atractylodes macrocephala Koidz. in a ratio of 1:2. The ZZP was ground into a powder and used for subsequent studies. Atractylodes macrocephala Koidz. was produced in Jiangsu Province. Citrus aurantium L. was produced in Sichuan Province.
Animals and Grouping
36 male SD rats without specific pathogens (SPF) were purchased from Chengdu Dossy Laboratory Animal Co., Ltd (Chengdu, Sichuan, China), body weight 200-220 g, and animal license number SCXK (Chuan) 2020-030. The animals were housed in an SPF-grade laboratory at North Sichuan Medical College and fed and watered ad libitum. All experimental protocols of the present study were approved by the Animal Ethics Committee of North Sichuan Medical College (IRB number: 20230508001). The reporting of this study conforms to ARRIVE 2.0 guidelines. 18 After 1 week of adaptive feeding, the rats were randomly divided into a control (Ctl) group, model group, low-ZZP group, medium-ZZP group, high-ZZP group, and positive drug group (n = 6). Loperamide was chosen to establish the STC rat model based on previous research reports. 19 Rats in control group received 0.2 mL saline by gavage for 7 consecutive days. The rats in model, low-ZZP, medium-ZZP, high-ZZP, and positive drug group received 8 mg/kg/d of loperamide (MFCD00058581, Merck, German) orally administered to establish the STC model for 7 consecutive days. After successful modeling, the control group was gavaged with saline. For ZZP treatment group, 10, 20, and 40 mg/kg ZZP and CMC-Na (S6703, Selleck, USA) were used to prepare suspensions for the treatment of STC rats in the low-ZZP group, medium-ZZP group, high-ZZP group. After the STC rat model was established, 3 mg/kg of mosapride (HY-B0189, MCE, USA) was gavaged in the positive group. For 7 consecutive days, the gavage volume was 0.125 mL/10 g via oral gavage once daily. Fecal pellet number, water content, and serum GAS and SP were used to initially evaluate the establishment of the STC rat model.
The rat colon tissue was collected for H&E staining, and the rat plasma was collected and separated to obtain serum for ELISA assay. The rat feces of the control group, model group, and 40 mg/kg ZZP group were collected for gut flora and metabolomic assay. Experimental rat feces were obtained from rat colorectum under sterile conditions for sequencing.
Measurement of Faecal Output and Water Content
Before assessing the characteristics, rat stool pellets were collected and counted for 24 h. Manure pellets were dried in a 60 °C incubator. The faecal water content was calculated using the following formula: Water content (%) = [(wet weight - dry weight)/wet weight] × 100%.
Measurement of Intestinal Transport Function
The rats were anesthetized 30 min after gavage with activated carbon suspension, and the entire intestine was separated by dissection, and the length of the intestine and the carbon ink advancement were measured. From the pylorus to the end of the colon, the distance from the pylorus to the front of the black semi-solid paste and the distance to the end of the colon were measured and photographed, and the intestinal propulsion rate was calculated. Intestinal transmission rate = carbon ink advancement distance (cm)/whole intestinal length (cm) × 100%.
ELISA Assay
The rats were sacrificed after the last administration, and plasma was collected from each group of rats. The plasma was centrifuged at 1500 rpm for 15 min at 4 °C, the supernatant was collected, and the levels of gastrin (D731177-0048, SanGon Biotech, China) and substance P (D751030-0048, SanGon Biotech, China) in the serum were determined as directed by the kit.
H&E Staining Assay
After dissection of the rats, 1 cm of the colonic segment was removed, and the removed tissues were immediately fixed in 4% paraformaldehyde, embedded and sectioned as usual, stained with hematoxylin-eosin (C0105S, Beyotime, China), and the pathological changes of the colonic tissues were observed under the microscope.
DNA Extraction of Rat Gut Microbiota, 16S rRNA Sequencing, and Related Bioinformatics Analysis
The total genomic DNA of each group of fecal samples was extracted according to the kit (51604, QIAGEN, Germany) procedure, and the extracted total DNA was examined by agarose gel electrophoresis. The universal primers (343 F: 5′-TACGGRAGAGCAG-3′; 798 R: 5′-AGGGATCTAATCCT-3′) were used for PCR amplification under Quant-iT PicoGreen dsDNA Assay Kit (Q33120, thermofisher, USA) instructions. Polymerase chain reaction (PCR) amplification of the V3-V4 high variant region of the bacterial 16S rRNA gene was performed in a 25 μL reaction system, and the obtained PCR products were sequenced and analyzed on a high-throughput sequencer. The sequencing library was prepared by Illumina's TruSeq Nano DNA LT Library Prep Kit. In this project, the Illumina MiSeq/NovaSeq platform was used for double-end (Paired-end) sequencing of community DNA fragments. Based on the valid data obtained from 16S rRNA gene amplicon sequencing, OTUs (Operational taxonomic units) clustering, and species classification analysis were performed, and the Rarefaction curve and Rank-abundance curve were prepared to check the sequencing depth. Meanwhile, the OTUs were analyzed by Alpha diversity calculation and Venn diagram (Venn) to obtain information on species abundance and diversity within samples, common and unique OTUs among different samples or subgroups.
Multiple sequence comparisons of differences among dominant species of different taxa were performed using Mafft software (V7.310) to investigate the phylogenetic relationships of different OTUs. The OTUs abundance information was normalized according to the ordinal number criterion corresponding to the smallest sequence sample. The alpha diversity method was used to analyze the diversity complexity of the samples using 3 indicators Chao1, Simpson, and Shannon. Greengeens annotated QIIME (version 1.8.0) PCoA and cluster analysis were used to calculate alpha and beta diversity. The result was plottedusing the ggbiplot package in R-3.4.4.
Metabolomic Study of Fecal Samples-Determination of Short-Chain Fatty Content
Gas chromatography conditions20,21: The GC analysis was performed on trace 1300 gas chromatograph (Thermo Fisher Scientific, USA). The GC was fitted with a capillary column Agilent HP-INNOWAX (30 m × 0.25 mm ID × 0.25 μm) and helium was used as the carrier gas at 1 mL/min. Injection was made in split mode at 10:1 with an injection volume of 1 μL and an injector temperature of 250°C. The temperature of the ion source and interface were 300°C and 250°C, respectively. The column temperature was programmed to increase from an initial temperature of 90°C, followed by an increase to 120°C at 10°C/min, and to 150°C at 5°C/min, and finally to 250°C at 25°C/min which was maintained for 2 min. In metabolomics research based on mass spectrometry technology, to obtain reliable and high-quality metabolomics data, quality control (QC) is required. 22
Mass spectrum conditions20,21: Mass spectrometric detection of metabolites was performed on ISQ 7000 (Thermo Fisher Scientific, USA) with electron impact ionization mode. Single ion monitoring (SIM) mode wasused with the electron energy of 70 eV.
Metabolites with more than 50% missing data in the samples were given a deletion treatment. Principal component analysis (PCA) and orthogonal partial least squares discriminant analysis (OPLS-DA) were performed to for modeling, dimensionality reduction and data visualization of the sample data using R, 23 respectively. In addition, the unit of sampling volume for solid samples of short-chain fatty acids is mg, and the unit of quantification is μg/g; the unit of sampling volume of liquid samples is μL, and the unit of quantification is μg/mL. Draw the content of SCFAs in each group through prism8.0 “Cloum”.
Statistical Analysis
SPSS21.0 statistical software was applied for data analysis, and the data were expressed as (
Results
The Analysis of Active Ingredients of ZZP
Results in supporting materials display the chromatograms of positive and negative ions of ZZP. For preprocessing, containing peak extraction, noise removal, deconvolution, and peak alignment, the raw data of LC-MS were put into MS-DIAL 4.60 software. We detected some flavonoids, glycosides, and sesquiterpenes: neohesperidin, cimicifugc acid, 2,7-dihydroxy-4-dimethoxyphenanthrene, odoratin-7-O-D-glucopyranoside, costunolide, baicalin-7-diglucoside, narirutin, malvalic acid.
ZZP Improves Constipation and Colon Histopathology in Rats
To study the effect of ZZP on rats with functional constipation, defecation, intestinal transit capacity, and structural changes in intestinal tissues were observed. As shown in Figure 1A, the number of 24-h stools was significantly reduced in the model group compared with the control group; the number of stools increased in the medium- and high-dose ZZP and positive drug groups compared with the model group. In addition, loperamide-induced rats (model group) showed a decrease in fecal water content, while ZZP high concentration and positive drug improved some differences (Figure 1B). The intestinal transit rate was able to characterize functional constipation, with a shorter distance traveled by activated charcoal in the intestine (lower intestinal transit rate) in the loperamide group compared to the control group; ZZP significantly increased the intestinal transit rate in a dose-dependent manner compared to the model group (Figure 1C and D). Intestinal neurotransmitters regulate intestinal secretion and motility and play an important role in the normal function of the intestine. Compared with the control group, GAS (gastrin), and SP (substance P) were reduced in the model group; ZZP increased serum GAS and SP in a dose-dependent manner, and medium and high doses of ZZP had similar effects as the positive drugs (Figure 1E and F). Then, we investigated the effect of ZZP on structural alterations of colonic tissue using H&E staining. As shown in Figure 1G, in the model group, the structure of colonic tissue was disrupted, including necrosis and detachment of mucosal epithelial cells, infiltration of inflammatory cells in the lamina propria, mucosal epithelial cells and infiltration by inflammatory cells, and degeneration of superficial intestinal glands. Fortunately, the colonic tissue structure of the rats in the ZZP-treated group gradually returned to the level of the control group, and the inflammatory cell infiltration in the colonic tissue was reduced (Figure 1H). Figure 1G shows the actual pictures of Atractylodes macrocephala Koidz. and Citrus aurantium L. Taken together, these results suggest that the administration of ZZP improves colonic motility and defecation in constipated rats.

ZZP-Dependently Improved Constipation and Colonic Tissue Pathology in Rats. (A) the Fecal Pellet Number was Recorded in 24 h. (B) Water Content was Measured Following Drying Stools in a 60 °C Oven for 12 h. (C) Intestinal Transit Rate was Detected. (D) Advancement of Activated Charcoal in the Intestine of Rats in Each Group. Elisa Assay for serum GAS (E) and SP (F). (G) H&E-Stained Colonic Tissue After Administration of ZZP (10, 20, and 40 mg/kg) Were Observed at 100× and 400×. (H) Pictures of Atractylodes macrocephala Koidz. (Baizhu) and Citrus aurantium L. (Zhishi). Data are Mean ± SEM, * Compared with ctl Group, *P < 0.05 and **P < 0.01, # Compared with Model Group, #P < 0.05.
ZZP Improves Intestinal flora Disorder
To investigate the changes of ZZP on the intestinal microbiota of STC rats, we analyzed the feces of each group of rats using 16S rRNA high-tfhroughput sequencing. The chao1 index can reflect microbial community richness, and the results showed differences in the Chao1 index between the control and model groups. The Chao1 index was greater in the ZZP group than in the model group, indicating that ZZP recovered from the decline in species number caused by loperamide. Shannon and Simpson indices can reflect the diversity of microbial communities. The results showed that the Shannon and Simpson indices were lower in the model group compared to the normal group. Compared with the model group, the Shannon and Simpson indices were higher in the ZZP group, indicating that the alpha diversity of the intestinal microbiota was reduced in the model group compared with the normal group, whereas ZZP promoted the alpha diversity of the intestinal microbiota in the STC model rats (Figure 2A). Community analysis using Venn diagrams revealed 33.2%, 27.13%, and 26.33% unique intestinal microbiota in the control, model, and ZZP groups. The control, model, and ZZP groups shared 611 identical bacteria. These flora changes indicated that the composition of the intestinal microbiota changed after the STC model was established. The same changes also occurred after ZZP treatment, but some pathogenic bacteria and therapeutic bacteria may not overlap (Figure 2B). Using Ramette's Classical Multidimensional Scaling (cMDScale) to analyze Principal coordinates analysis (PCoA). 24 The results of the β-diversity showed that the intestinal microbiota of between groups was different, and the distance between the control and ZZP groups was shorter than that between the model and control group, indicating that the flora of the control and ZZP groups were more similar (Figure 2C). The top 20 phylum level results indicated that Fruticutes and Bacteroidetes were the main dominant flora (Figure 2D). The analysis of the intestinal microbiota at the genus level showed that compared with the control group, the abundance of Escherichia-Shigella, Clostridium, and Desulfovibrio increased, and the abundance of Butyricimonas, Bifidobacterium, and Lactobacillus decreased. Compared with the model group, the abundance of Escherichia-Shigella, Clostridium, and Desulfovibrio decreased under the action of ZZP, while the abundance of Butyricimonas, Bifidobacterium, and Lactobacillus increased. Alistipes and Prevotellad frontal regulation by ZZP does not seem to be obvious (Figure 2E).

Bacterial Flora Analysis in Rat Feces. (A) Alpha Diversity Chaol, Simpson, and Shannon. (B) OUT Venn Diagram. (C) PCoA Diagram, Each Point in the Diagram Represents a Sample. (D) Abundance Map of Intestinal Microbiota at the Phylum Level. (E) Differential Abundance of Gut Flora at the Genus Level. Data are Mean ± SEM, * Compared with ctl Group, *P < 0.05 and **P < 0.01, # Compared with Model Group, #P < 0.05, ##P < 0.01.
Assessment of Intestinal Microbiota
The unweighted pair-group method with arithmetic means (UPGMA) cluster analysis can use any distance to evaluate the similarity between samples. As shown in Figure 3A, there were obvious clustering differences between the control and model groups, and the distance between the model and ZZP groups was the farthest, indicating that ZZP affected changing the structure of the intestinal microbiota in STC rats. Heat map of gut flora composition at the genus level with a color gradient from red to blue indicating high to low abundance. As shown in Figure 3B, Helicobacter, Sutterella, Clostridium, Acinetobacter, Shigella, Epulopiscium, Barnesiella, Ruminococcus, and Bacteroides in the model group compared to the control group. Butyricimonas, AF12, Prevotella, Parabacteroides, Phascolarctobacterium, Blautia, and Dorea increased in relative abundance. In addition, ZZP decreased compared to the model group for Helicobacter, Sutterella, Clostridium, Acinetobacter, Shigella, Epulopiscium, Barnesiella, Ruminococcus, Bacteroides, Butyricimonas, AF12, Prevotella, Parabacteroides, and the relative abundance of Phascolarctobacterium. The relative abundance at the genus level was close between the control and ZZP groups. The above experimental results indicate that these modulated intestinal bacteria could be used as markers for STC in rats treated with ZZP.

Effect of ZZP on the Composition of the Intestinal microbiota. (A) UPGMA Clustering Tree Based on the Sample Distance Matrix. (B) Heat map of species Composition at the Genus Level for Species Clustering.
Metabolome Study of SCFAs in Feces
To deeply investigate the relationship between intestinal microbiota and host diseases, it is mostly explored with the help of metabolomic techniques. Metabolites are the main mediators linking intestinal bacteria-host interrelation, and the analysis of metabolomics is also essential. After quality control (QC), 22 cluster heat maps showed that the relative abundance of metabolites differed between groups. Compared with the control group, the levels of SCFAs in the model group were reduced, which recovered after ZZP treatment (Figure 4A). As shown in figure 4B the fecal samples were well clustered within the control and ZZP groups, and the samples were more discrete within the model group. In addition, a trend of separation among the model and control groups and the fecal samples of the ZZP group can be observed, indicating that loperamide caused changes in the metabolic profile of STC rats. Orthogonal Projections to Latent Structures Discriminant Analysis (OPLS-DA) is a commonly used method in metabolomics data analysis. 25 There was a tendency of back-regulation toward the control group after ZZP administration, indicating that ZZP administration caused changes in the SCFAs metabolic profile of rats. All model Q2 values were low R2 values and all substitutions had Q2 less than 0, showing no overfitting, indicating that the model has high reliability and predictability (figure 4C). From the metabolite pathway analysis, it can be obtained that Acetic acid can be involved in the most pathway, Protein digestion and absorption, Carbohydrate digestion, and absorption, and Propanoate metabolism enriched the amount of more SCFAs (Figure 4D). As shown in Figure 4E, the levels of Isobutyric acid, Propionic acid, Acetic acid, Butyric acid, Isovaleric acid, Valeric acid, and Caproic acid were all decreased to different degrees when compared with the control group. The levels of all these SCFAs were increased in the ZZP group compared with the model group. The fecal metabolomics results further suggest that ZZP may play a role in regulating intestinal SCFAs in STC rats.
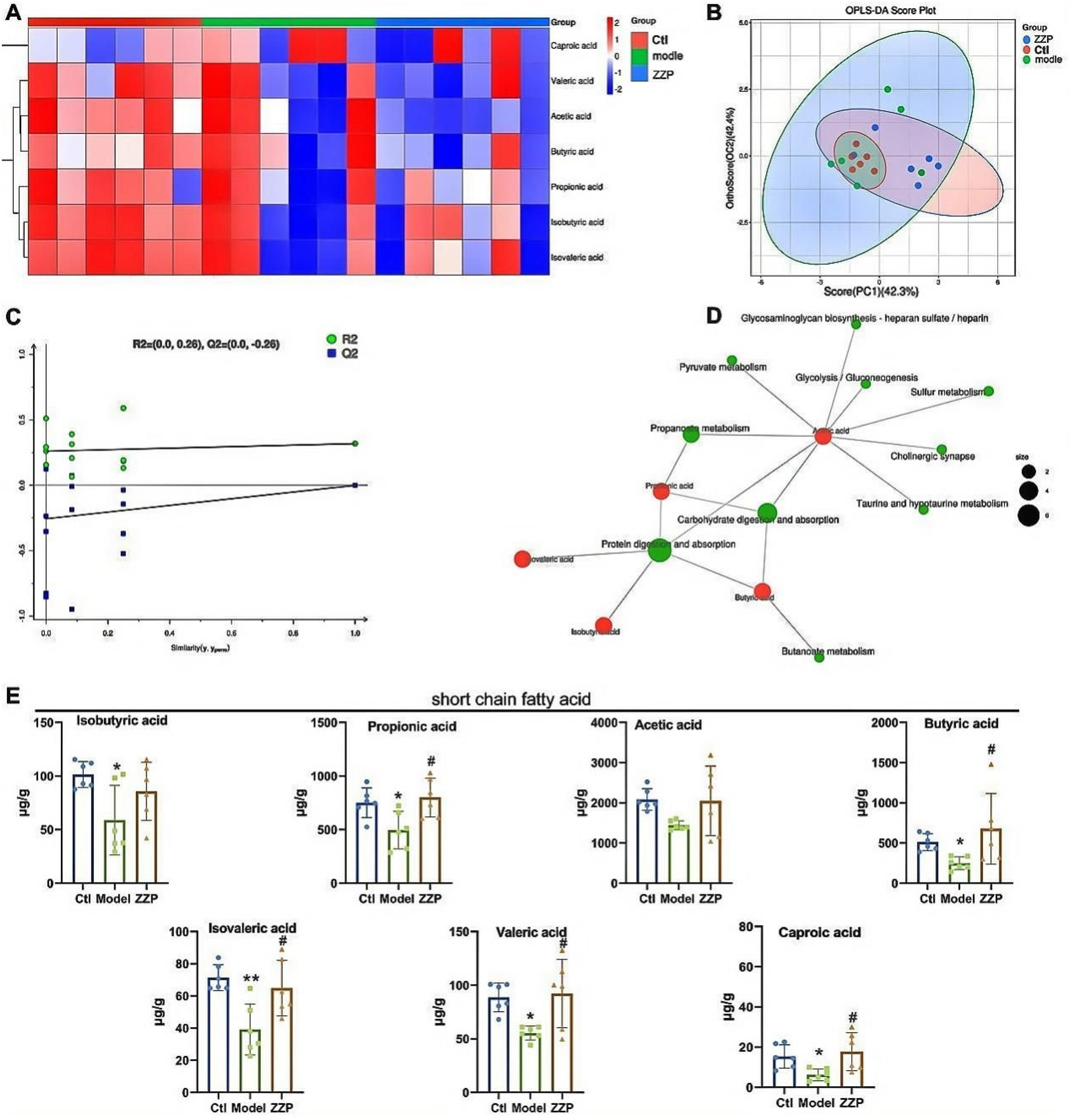
Effect of ZZP on the Metabolism of SCFAs. (A) Overall Metabolite Cluster Heatmap. (B) OPLS-DA Score Plot. (C) OPLS-DA Permutation Test Plot. (D) Molecular Network Diagram of KEGG Metabolites. (E) Differences in SCFAs Isobutyric Acid, Propionic Acid, Acetic Acid, Butyric Acid, Isovaleric Acid, Valeric Acid, and Caproic Acid Among the Groups. Data are Mean ± SEM, * Compared with ctl Group, *P < 0.05 and **P < 0.01, # Compared with Model Group, #P < 0.05, ##P < 0.01.
Correlation Analysis Between SCFAs and Intestinal Microbiota
Pearson correlation analysis was used to analyze the relationship between SCFAs and intestinal microbiota genus levels in the control group, model group, and ZZP group. As shown in Figure 5, Lactobacillus and Butyricimonas were significantly positively correlated with isobutyric acid, acetic acid, butyric acid, valeric acid, and caproic acid. Clostridium was significantly negatively correlated with isobutyric acid, acetic acid, butyric acid, valeric acid, and caproic acid. Butyric acid is the classical metabolite studied in STC, and our scatterplot analysis showed that Lactobacillus and Butyricimonas were positively correlated with butyric acid content, and Clostridium was negatively correlated with butyric acid content (Figures 5B-D). In addition, we also found that there is a definite relationship between bacteria and therapeutic effect, Lactobacillus and Butyricimonas are positive for the curative index, but Clostridium is negative for the therapeutic effect (Figure 5E). The above experimental results indicated that both intestinal microbiota and SCFAs were involved in the treatment of STC by ZZP.

Pearson Correlation Analysis Between Intestinal Microbiota and SCFAs. (A) The Heatmap was Used to Intuitively Display the Relationships Between Differential SCFAs and Intestinal Microbiota. (B). Scatter Plot of Correlation Analysis Between Lactobacillus and Butyric Acid, Y = 0.01457*X-0.3904, P = 0.38. (C) Scatter Plot of Correlation Analysis Between Butyricimonas and Butyric Acid, Y = 6.279e-006*X-0.001953, P = 0.043. (D) Scatter Plot of Correlation Analysis Between Clostridium and Butyric Acid, Y = -2.411e-006*X-0.004116, P = 0.048. (E) Correlation Between Lactobacillus, Butyricimonas, and Clostridium and Defecation in STC Rats. Significant Differences were Indicated: *p < 0.05, **p < 0.01, ***p < 0.001.
Discussion
Traditional Chinese medicine emphasizes the overall concept and regards the body as an organic whole. In this study, the overall physiology, intestinal microbial composition, and short-chain fatty acid metabolism of STC rats were observed in an attempt to understand the therapeutic mechanism of ZZP. Constipation is a common and persistent clinical disorder, which not only leads to different degrees of rectal prolapse and abnormalities of perianal tissue and other functions but also induces insomnia, irritability, depression, anxiety, and other mental and psychological disorders, which seriously affects patients’ daily life and work. 26 STC is a chronic gastrointestinal disease characterized by disturbances in colonic dynamics and slowed intestinal transit. 27 Loperamide is a common method to simulate constipation, whose main symptoms include difficult bowel movements, reduced frequency of bowel movements, dry and hard stools, and prolonged gastrointestinal emptying time. 28 The loperamide-induced constipation model develops mainly by inhibiting intestinal motility and affecting intestinal water metabolism and the enteric nervous system. 19 Mosapride is a gastrointestinal motility drug, which can promote the peristalsis of the entire digestive tract, thereby improving the symptoms of constipation. 29 Therefore, loperamide was chosen as the modeling drug, and mosapride was chosen as the positive control in this study. In addition, the defecation volume and fecal water content of rats in the model group were significantly lower than those in the control group, the propulsion speed of activated charcoal became slower, and there was a certain degree of colonic pathological damage, indicating the successful establishment of the STC model.
ZZP, as a common herbal formula, is beneficial in the treatment of constipation. In this study, ZZP was found to significantly improve intestinal motility and reduce colonic injury in the STC rat model. In addition, ZZP significantly increased the levels of excitatory gastrointestinal peptide hormones SP and GAS in STC rats. Experimental studies in recent years have demonstrated that Atractylodes macrocephala Koidz. and Citrus aurantium L. can improve the abnormal changes in intestinal tissue structure in STC model animals and restore their original barrier and secretion functions. 12 Atractylodes macrocephala Koidz. and Citrus aurantium L. also have a therapeutic effect on abnormal motility of the gastrointestinal tract, restoring its original motility pattern and improving the motility and emptying function of the stomach and intestines (6, 8). The levels of substance P (SP) and gastrin (GAS) were reported to be positively correlated with the time to first defecation of black feces in animal models of constipation. 30 These imply that ZZP can enhance intestinal motility and effectively improve defecation function in STC rats.
Some studies have shown that there is intestinal dysbiosis involved in the development of STC in patients with STC. The intestinal microbiota is significantly altered in the constipated population, and the number of some probiotics in the body is significantly reduced, while the number of some pathogenic bacteria is significantly increased. 31 The balance of the intestinal microflora is disturbed, further affecting the occurrence of STC, and leading to pathophysiological and other processes.31,32 Our study found that the intestinal microbiota varied greatly within the loperamide STC rat model group, with reduced α-diversity and β-diversity. Interestingly, Yutao Chen's 33 study reported an increase in α-diversity in STC models, which may be related to differences in experimental animals and inducing drugs. ZZP could promote the recovery of α-diversity and β-diversity, and the flora structure was highly similar within the ZZP group. Consistent with previous studies, the Fruticutes and Bacteroidetes continued to be the more abundant species in the STC model. 31 Moreover, Zoppi 34 studied the intestinal microbiota of 28 children with STC and 14 healthy normal children using bacterial culture methods and found that Clostridium was significantly higher in the stools of children with STC than in healthy normal children. Khalif 35 cultured stools from 57 adults with STC and found significantly lower levels of both Bifidobacterium and Lactobacillus and elevated levels of potentially pathogenic bacteria. Similar to the results of Zoppi 34 and Khalif, 35 Clostridium, Bifidobacterium, and Lactobacillus were regulated by ZZP to return to close to the control group. Intestinal bacterial imbalance affects the physiological state of the intestine and the colonization of intestinal bacteria, resulting in abnormal intestinal bacteria, accompanied by an increase in the abundance of harmful bacteria Escherichia coli, Desulfovibrio, and Salmonella Proteus, and the beneficial bacteria Lactobacillus, Bifidobacteria, Clostridium butyricum, and intestinal commensal bacteria decreased in abundance.36–38 ZZP reversed the species richness of intestinal microbiota, up-regulated the abundance of beneficial bacteria Lactobacillus and Butyromonas, and down-regulated the relative abundance of pathogenic bacteria Escherichiai-Shigella and Desulfovibrio. In addition, Lactobacillus can secrete bacteriocins to a variety of gram-negative bacteria pathogenic bacteria, including Escherichia coli, and Shigella, has a better antibacterial effect.39,40 Combined with previous studies we can determine that ZZP can restore flora abundance, upregulate probiotics and inhibit harmful flora.
Intestinal microorganisms are closely related to the metabolism of nutrients in the organism, and the metabolites and the associated enzymes that act on them are in turn involved in the regulation of the intestinal microecological balance together with the intestinal microbiota. 41 Generally, intestinal microorganisms under normal conditions can regulate their metabolism of peptides, carbohydrates, and some proteins that cannot be absorbed by the small intestine in the gastrointestinal tract, and perform a series of fermentations to produce metabolites beneficial to the intestine, mainly bile acids (BAs) and SCFAs. 42 This study focused on the metabolism of SCFAs. Loperamide reduced fecal SCFAs of STC in rats, and there was a large difference within the group. After the treatment of ZZP, the difference between groups of SCFAs in STC rats was reduced, and it was closer to that of the control group. SCFAs has important physiological regulatory effects, and an increase in intestinal acetic acid concentration can lead to an increase in intestinal osmotic pressure and water content, which stimulates intestinal motility. 43 Propionic acid can affect hepatic glucose and cholesterol metabolism. 44 Butyric acid provides energy, regulates immune cells, and enhances intestinal immunity. 45 In the present study, loperamide caused acetic acid, propionic acid, and butyric acid content reduction to be back-regulated by ZZP. In addition, Zhuang et al 41 gave a dietary fiber diet to constipated patients and found an increased spectrum of butyric acid-rich SCFAs, inhibited the growth of Desulfovibrio (an important endotoxin-producing bacterium in constipated patients), and ultimately accelerated gastrointestinal transit and thickened mucosal layer, which may be related to the secretion and enhanced expression of SCFAs by triggering colonic hormones. Anaerobic intestinal bacteria produce SCFAs through glycolysis, including formic acid, acetic acid, propionic acid, butyric acid, isobutyric acid, valeric acid, isovaleric acid, hexanoic acid and isohexanoic acid. 46 Our study found a reduction in butyric acid in the model group, along with a reduction in the abundance of anaerobic Butyricimonas, again suggesting a direct link between intestinal microbiota and metabolites. In addition, Lactobacillus and Butyricimonas were positively correlated with SCFAs, and Clostridium was negatively correlated with SCFAs. The results of these analyses suggest that ZZP is involved in the metabolism of SCFAs by intestinal microbiota and thus affects the defecation function of STC rats.
Conclusions
In conclusion, the present study showed that ZZP could improve bowel function and intestinal motility in rats with STC by regulating intestinal microbiota, decreasing the abundance of pathogenic bacteria such as Esherichia Shigella and Desulfovibrio, promoting the abundance of beneficial bacteria Lactobacillus and Bifidobacterium, and regulating the content of SCFAs, and influencing the production of metabolites through the interaction between intestinal microbiota and host to improve intestinal homeostasis. This study will provide insights into ZZP for the treatment of constipation. This multi-angle view helps to comprehensively evaluate the efficacy and mechanisms of botanical drugs. However, the main limitation is that we did not investigate the changes in proteins and signaling pathways during the ZZP treatment of STC. Therefore, considerably more work will be needed to explore the protein changes and signaling pathways involved in the ZZP treatment of STC.
Supplemental Material
sj-doc-1-npx-10.1177_1934578X251346009 - Supplemental material for Exploring the Therapeutic Effects of the ZhiZhu Pill on Rats with Chronic Transmission Constipation Based on Intestinal Microbiota and Metabolomics
Supplemental material, sj-doc-1-npx-10.1177_1934578X251346009 for Exploring the Therapeutic Effects of the ZhiZhu Pill on Rats with Chronic Transmission Constipation Based on Intestinal Microbiota and Metabolomics by Lijuan Du, Shi-yu Tang, Yu Zhan, Ge Yin and Xue gui Tang in Natural Product Communications
Footnotes
Ethical considerations
All experimental procedures were performed following the Animal Ethics Committee of North Sichuan Medical College (IRB number: 20230508001).
Author contributions
Lijuan Du and Shi-yu Tang designed the study, performed the experiment. Yu Zhan, Ge Yin, and Xue-gui Tang participated in the data analysis and the animal experiments. Xue-gui Tang participated in design, revised the manuscript. Lijuan Du and Xue-gui Tang wrote the manuscript. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Foundation of China, Upper Program (82074429).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data and Materials Availability
The datasets used or/and analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
