Abstract
Radix paeoniae alba is the dried root of Paeonia lactiflora Pall (PLP) of ranunculaceae, with the effects of anti-depressants, anti-inflammatory, regulate menstruation and protect central nervous system et al Total glycoside of paeony (TGP), extracted from the dried root of PLP, is the major active components concluding paeoniflorin and albiflorin. A systematic review of relevant research on the absorption, distribution, metabolism, excretion (ADME process) and toxicity of major components such as paeoniflorin and albiflorin was conducted. The results show that paeoniflorin and albiflorin were both absorbed swiftly with varying Cmax and Tmax values. Both paeoniflorin and albiflorin exhibited swift and extensive distribution across various tissues throughout the body, with a particular concentration in the stomach, small intestine and kidney. The study revealed that the primary metabolic pathways included hydrolysis, oxidation and conjugation et al Toxicological studies have shown that TGP has certain adverse reactions, and the side effects of albiflorin are less. At present, there are some differences in the research results of ADME related to TGP in different literatures. This study can provide reference for further elucidating the mechanism, efficacy evaluation and clinical application of TGP in vivo.
Introduction
Paeonia lactiflora Pall (PLP), commonly known as Baishao in China, is extensively spread throughout various regions globally.1,2 The medicinal root of PLP is Paeoniae Radix Alba, which holds significant worth as a conventional herbal remedy in Chinese medicinal practices. 3 Modern pharmacology studies demonstrated that PLP displays a wide spectrum of medicinal actions, including, anti-depressants, 4 anti-inflammatory,5,6 regulate menstruation, 3 protect central nervous system, 7 antiviral 8 and anti-tumor 9 et al Total glucosides of Paeony (TGP) is the effective part of PLP. It mainly contains moniterpenoid glycosides such as paeoniflorin and albiflorin (Figure 1), the two most abundant components (make up nearly half of TGP's composition) which is the pharmacodynamic material basis of PLP.10‐12 Like PLP, TGP has effective treatment of inflammatory arthritis, 13 acute kidney injury, 14 constipation, 15 and parkinsonism 16 et al Furthermore, TGP has some superior features with efficacy and safety.

Structures of Paeoniflorin and Albiflorin.
Besides pharmacology studies, information on the absorption, distribution, metabolism, excretion and toxicity of TGP have a major impact on further clinical utilization of PLP. However, there is no literature to summarize pharmacokinetics of TGP. Including both Chinese and English terms for “total glucosides of paeony” “paeoniflorin,” “albiflorin,” “pharmacokinetics” “absorption,” and “distribution,” “metabolism,” “excretion,” and “toxicity” as keywords, we consulted current research outputs documented in PubMed, SciFinder, Google Scholar, Springer, Wiley, Science Direct, Elsevier, and CNKI from 2004 to 2024. Drawing on extensive academic resources, the purpose of the current review is to give a comprehensive summary and analysis of ADME (Absorption, Distribution, Metabolism and Excretion) of TGP, providing reference for further elucidating the mechanism, efficacy evaluation, new drug development and clinical application of the main chemical components of PLP in vivo.
Pharmacokinetics
Pharmacokinetics of TCM is the study of the dynamic changes in the ADME of active ingredients or marker components of TCM in the body. By integrating with toxicology, it elucidates the safety of TCM and provides a basis for scientifically explaining the material basis and mechanism of action through which TCM exerts its pharmacological effects. It is a pivotal discipline in interpreting the theories and mechanisms of TCM. The analysis of components and metabolites of TCM is characterized by complex compositions, numerous interferences, low concentrations, wide dynamic ranges of concentration changes and substantial data processing workloads. This necessitates analytical techniques with high separation efficiency, sensitivity, and selectivity, such as time-of-flight mass spectrometry (TOF), Fourier transform ion cyclotron resonance (FT-ICR), and Orbitrap mass analyzers.
Absorbtion
So far, many scholars have investigated the pharmacokinetic characteristics of paeoniflorin and albiflorin in animals following TGP treatment. Research demonstrates that paeoniflorin and albiflorin conforms to the first-order kinetic process in rats, and is absorbed, distributed and eliminated quickly. The peak concentration can be reached within a short time after oral administration of TGP.17‐20 Detailed pharmacokinetic parameters are shown in the Table 1.
Mean Pharmacokinetic Parameters of TGP.

Concurrent analysis of paeoniflorin pharmacokinetics from TGP in Sprague-Dawley (SD) rats and spontaneously hypertensive rats (SHR) using high-performance liquid chromatography-tandem mass spectrometry (HPLC-MS) revealed marked differences in drug exposure profiles. Specifically, SHR demonstrated notably reduced clearance (CLz/F) coupled with significantly elevated area under the concentration-time curves (AUC(0-t) and AUC(0-∞)) and prolonged time to maximum concentration (Tmax) relative to SD rats (P < 0.05). Furthermore, the values of MRT and T1/2z for paeoniflorin in SHR exceeded those observed in SD rats, ranging from 1 h to 2 h.17 Jiang et al establish an LC-MS/MS technique utilized for quantifying paeoniflorin and albiflorin in hepatic injury rat serum after oral administration of TGP capsule, 18 finding that the MRT and T1/2z of the two analytes were higher than other pharmacokinetics study in normal rats. It is also demonstrated that the presence of liver injury pathologies can extend the duration of residence for both analytes.
Can Gong et al investigated the pharmacokinetics differences of albiflorin and paeoniflorin, following oral administration of PLP and Zengmian Yiliu prescription (administered alongside cisplatin during ovarian cancer chemotherapy) in rats using a sensitive LC-MS/MS. 19 The results show that albiflorin and paeoniflorin were both taken up swiftly by varying Cmax and Tmax values.
Minmin Zhao and colleagues have established and verified a LC-MS/MS technique coupled with solid-phase extraction, for the concurrent measurement of paeoniflorin and albiflorin levels in rat plasma and tissue following the administration of TGP. They concluded that the pharmacokinetic profiles of paeoniflorin and albiflorin are comparable due to their analogous chemical structures. 20 To a certain extent, the pharmacokinetic behavior of TGP can be largely inferred from the characteristics of paeoniflorin and albiflorin.
The rate and degree of absorption of compounds directly affect the concentration it reaches in the body, which in turn affect the efficacy and duration of action of compounds. To sum up, absorption is of great significance to drug research. It not only affects the efficacy and duration of drug action, but also provides an important basis for drug design and optimization, new drug development and evaluation, and personalized drug use and drug interaction.
Distribution
Research shows that both paeoniflorin and albiflorin exhibited swift and extensive distribution across various tissues throughout the body, with a particular concentration in the stomach. 20 Paeoniflorin reached its peak levels in the stomach (33.66 mg/mL), trailed by the intestine (19.57 mg/mL) and heart (11.70 mg/mL). Meanwhile, albiflorin displayed the highest concentrations in the stomach (6.070 mg/mL), followed by the heart (1.830 mg/mL) and liver (1.460 mg/mL). 20 Additionally, they demonstrated the capacity to penetrate the blood-brain barrier.
In an experiment, normal rats were given TGP by intragastric administration in a dosage of 2.82 g/kg, and tissue samples of heart, liver, spleen, lung, kidney, stomach, small intestine and large intestine were collected at 1 h, 3 h and 6 h time points, respectively. 21 The concentrations of paeoniflorin and albiflorin in these tissues were measured using HPLC, and the specific concentrations of these two constituents in each tissue were calculated to further analyze and explore the tissue distribution characteristics of TGP in normal rats. The outcomes revealed that paeoniflorin and albiflorin could be detected in all tissues at 1 h. At 3 h, higher concentrations were found in the small intestine, stomach, large intestine, as well as in the kidney, spleen, and liver. At 6 h, the concentrations remained high in the small intestine, large intestine, and stomach, while the concentrations in other tissues were lower. The concentration of paeoniflorin and albiflorin in the small intestine, stomach and large intestine was the highest, possibly because the small intestine was the main absorption organ, followed by the kidney, liver, lung and other tissues, possibly because the blood flow in these tissues was larger.
After prolonged administration of TGP, Ma Zhe et al examine the distribution of paeoniflorin and albiflorin in the tissues of rats with collagen-induced arthritis (CIA). 22 Following long-term administration, the primary distribution of paeoniflorin and albiflorin was observed in the small intestine and kidney with significant presence, secondly in the spleen, thymus, synovium, liver, lung and heart. The contents of paeoniflorin and albiflorin in small intestine exceeded those in additional tissues, indicating that small intestine may be the main site of action of TGP. Paeoniflorin and albiflorin are more distributed in the kidney, which supports the conclusion that total paeoniflorin is eliminated quickly and has a short retention time in rats.
The pharmacokinetics of paeoniflorin in brain tissues of normal rats and rats with cerebral ischemia-reperfusion injury were analyzed and compared by HPLC-MS/MS. 23 The findings revealed that paeoniflorin was present in both blood and brain tissue samples of rats within 5 min after intragastric administration of Huangqi Guizhi Wuwu Decoction, suggesting its ability to cross the blood-brain barrier. This excellent permeability across the blood-brain barrier could be attributed to paeoniflorin's small molecular size and its rate of protein binding.
In a study, Feng Liu et al elucidated the variations in tissue distribution of albiflorin between normal rats and those with chronic gastritis following oral administration of Weikangling capsules (WKLCs). 24 Albiflorin was found extensively in the heart, liver, spleen, lung, kidney, stomach and small intestine in normal rats. This study also reveals that higher levels of albiflorin were present in the small intestine and stomach. The highest concentration of albiflorin in the small intestine occurred at the 4 h mark, indicating a gradual accumulation of albiflorin in this region. One hour after the administration of WKLCs, rats with chronic gastritis exhibited significantly elevated concentrations of albiflorin in their small intestines compared to normal rats (P < 0.05), indicating a marked improvement in the distribution of albiflorin.
By studying the distribution of compounds in animals, it is possible to predict the possible distribution of compounds in the human body(For details, please refer to Figure 2), thereby guiding the formulation design of compounds, so that compounds can reach the target organs more effectively, improve efficacy and reduce toxicity. Meanwhile, by studying the distribution changes of compounds in specific disease states, we can understand the effects of diseases on the distribution of compounds, and thus provide new ideas for the treatment of diseases
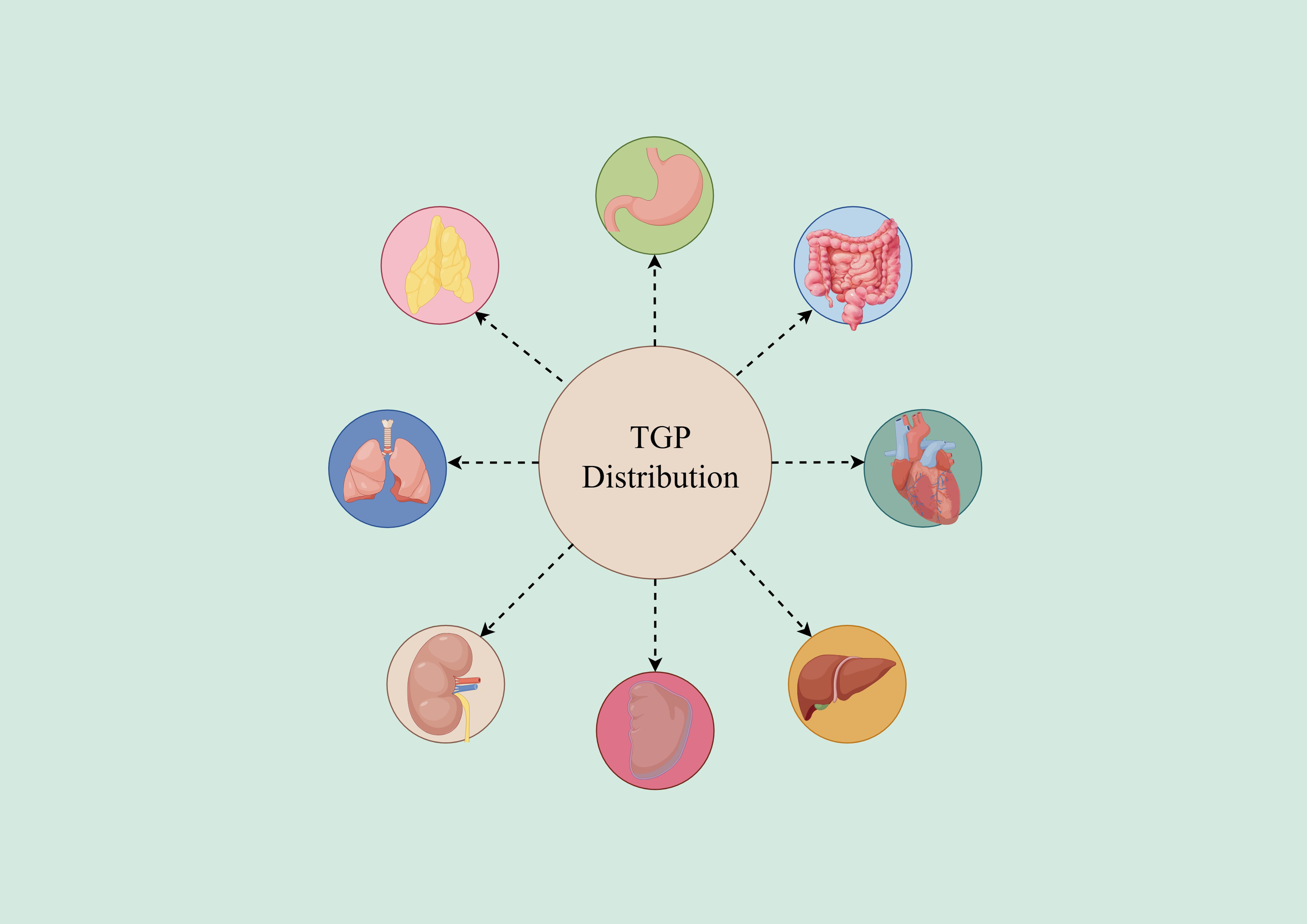
The Tissue Distribution of TGP.
Metabolism
Intestinal bacteria are considered a “hidden organ” that significantly influences drug metabolism, transformation, and the exertion of pharmacological effects. Ke Zhong et al added paeoniflorin to the intestinal flora of rats cultured in vitro for anaerobic incubation, analyzed the change process of paeoniflorin within 48 h, and explored the metabolic transformation of paeoniflorin by intestinal flora of rats in vitro. 25 The metabolic products were analyzed and identified by Ultra high performance liquid chromatography-four-pole time-of-flight tandem mass spectrometry (UPLC-Q-TOF-MS /MS). The isolated gut microbiota of rats first converts paeoniflorin into albiflorin, and then further metabolizes it through multiple pathways such as deglucosylation, debenzoylation, and rearrangement involving quaternary ring cleavage, gradually transforming paeoniflorin into various metabolites including albiflorinaglycone, deacylate albiflorin and paeonilactone-B, which are more readily absorbed by the intestine. Zhao utilized models of in vitro liver microsomal incubation and intestinal bacterial metabolism to investigate the metabolic conversion of albiflorin. 26 After 12 h of incubation in the intestinal bacterial metabolism model in vitro, the prototype drug of albiflorin was completely metabolized and transformed, accompanied by the substantial production of benzoic acid. In a study by Shanshan Sun, the metabolic products of paeoniflorin in the human gut microbiota (HGM) was studied based chemical profiling method through UPLC-Q-TOF-MS/MS. The study also revealed that the primary metabolic pathways included hydrolysis, oxidation, and conjugation, while the metabolic sites involved the glycosidic linkage, ester bond, benzene ring, and pinane within the structure. 27
The liver holds a pivotal role in drug biotransformation because it possesses a large number of highly abundant drug-metabolizing enzymes, notably cytochrome P450 (CYP450), which can promote processes such as breakdown, oxidation and coupling. 28 L.J. Zhu and colleagues investigated the metabolism of paeoniflorin using rat liver microsomes in vitro, resulting in the identification of 20 paeoniflorin metabolites. 29 Research indicated that the primary metabolic routes for paeoniflorin in rat liver microsomes involve hydrolysis and oxidation, with the configurations of pinane and the ester linkage serving as the primary sites of metabolism.
Conducting metabolic studies is crucial for uncovering the bioactive components of herbal remedies in vivo, as well as the metabolites involved in their metabolic pathways. Lijun Zhu discovered metabolites of paeoniflorin, determined the metabolites of TGP through the identification of paeoniflorin's metabolites, and contrast the metabolic distinctions between paeoniflorin and TPG following oral gavage administration by the use of UPLC-FT-ICR MS and UPLC-Q-TOF MS2. 28 They discovered a total of 42 metabolites of paeoniflorin, among which they successfully identified 18. The findings suggest that the primary metabolic routes for paeoniflorin involve the hydrolysis of its ester and glucosidic bonds, as well as conjugation with glucuronide. Furthermore, the number of detected and identified metabolites of TGP was lower than that of paeoniflorin due to the influence of other monoterpene glucosides present in TGP, such as albiflorin and oxypaeoniflorin, which affected the metabolism of paeoniflorin. 30 Liu et al employed in vivo biopharmaceutical analysis to screen for quality markers in Xiaoer Chaige Tuire Oral Liquid with albiflorin as a major component. 31 Through in vivo analysis of rat plasma, benzoic acid was detected, leading to the speculation that albiflorin underwent a dual hydrolysis process, encompassing both ester and glycosidic hydrolysis, ultimately yielding benzoic acid.
There are few detailed research data on the pharmacological effects of metabolites of TGP. However, combined with the pharmacological effects of TGP itself and the characteristics of metabolism, it can be inferred that their metabolites may affect pharmacological effects in the following ways. The metabolites of TGP may affect the pharmacological action by inheriting the pharmacological activity of the original drug, changing the intensity and duration of pharmacological action, and affecting the metabolism and excretion of the drug. However, further scientific research is needed to confirm these assumptions.
Excretion
Excretion refers to the process by which organisms eliminate waste products and surplus substances from their bodies. This typically involves the removal of metabolic waste, toxins and other unwanted materials through various bodily systems, such as the urinary system (via urination), the digestive system (via feces), the respiratory system (via exhalation) and the skin (via sweating). It is a crucial biological function that helps maintain homeostasis and overall health. During the experiments focused on the excretion of paeoniflorin in rats, the compound underwent primary excretion via benzoic acid configuration through both urine and feces, while the elimination pathway of paeoniflorin via its original, unmetabolized form was minimal. 32 Furthermore, the urinary excretion of benzoic acid was notably greater than that in feces, suggesting efficient absorption of benzoic acid. In other words, after paeoniflorin was metabolized, benzoic acid was absorbed and subsequently excreted through urine in rats.
The pharmacokinetic profiles of paeoniflorin following both single and repeated intravenous administrations of Huoxuetongluo lyophilized powder in healthy Chinese subjects was investigated. 33 After seven consecutive days of once-daily dosing, no buildup of paeoniflorin was observed. Roughly 48.4% of the administered paeoniflorin was excreted by the kidneys as it was originally. The mean renal clearance of paeoniflorin, at 5.3 L/h, was lower than its mean total clearance of 11.3 L/h, suggesting the involvement of renal tubular reabsorption in its excretion process.
The research focused on studying the pharmacokinetics of paeoniflorin and albiflorin in rats following oral administration of TGP was studied by Fei Fei et al 34 The entire volume of urine and feces excreted was collected at different points in time following the oral administration of 180 mg/kg of TGP. Meanwhile, bile samples were gathered from rats at different time intervals following the insertion of a cannula into the bile duct. About 46% of paeoniflorin was excreted in total, with 43.06% in feces, 1.39% in urine and 1.53% in bile. Similarly, about 44% of albiflorin was excreted, with 40.87% in feces, 0.99% in urine and 1.59% in bile. These findings suggest that both compounds are primarily excreted in their original form through feces.
The varying descriptions of paeoniflorin's excretion form in vivo found in different literature may stem from several reasons. Firstly, differences in research methodologies, sample selection and experimental conditions employed in various literature may lead to varying observations of the excretion form of paeoniflorin. Secondly, when paeoniflorin is co-administered with other drugs, drug interactions may occur, affecting the metabolism and excretion of paeoniflorin. Certain drugs may inhibit or induce the metabolic enzymes of paeoniflorin, thereby altering its excretion form. Additionally, the metabolic pathways of paeoniflorin in the body may be relatively complex, involving multiple enzymes and metabolic routes. Different studies may focus on various metabolic pathways and metabolites, leading to discrepancies in the descriptions of its excretion form.
Toxicity
Every medication will produce effects other than its intended therapeutic purpose, known as adverse drug reactions. Luo Jing et al summarized the findings from systematic reviews (SRs) regarding the advantages and safety of TGP, a commonly prescribed medication for rheumatoid arthritis (RA) in China, for individuals suffering from RA. 35 The most frequently observed unfavorable results of TGP ranged from slight to moderate diarrhea. The animal toxicity studies of TGP have shown that TGP can cause an increase in platelet count, but it has no significant toxic effects on blood and urine routine tests, as well as liver and kidney function. 36 It has been reported that TGP can cause hemorrhagic rash, and the mechanism may be related to allergy. 37 Experimental studies on the mutagenicity and teratogenicity of TGP have shown that when large doses of TGP are given to rats, they have an embryonic-toxic effect on fetal and placental development (mainly manifested as a marked reduction in fetal and fetal disk weight). 38 However, no significant teratogenic effects of TGP have been observed on fetal appearance, visceral and skeletal morphology.
The possible harmfulness of albiflorin was assessed using traditional methods, involving in vivo acute oral harmfulness tests on rats and dogs. 39 For these conventional toxicity evaluations, a dose of 5000 mg/kg of albiflorin was administered, which is much higher than the clinical dose for humans. The findings indicated that albiflorin administration did not significantly alter the food intake of beagle dogs. None poisonous outcomes were seen in dogs, and also, no significant difference was noted between SD rats in the albiflorin-treated and control groups. The utilization of metabolomics for evaluating medication poisonousness within the medicine-making sector and medicinal plants commenced in the early 2000s. 40 LC-MS/MS technology was applied to conduct a metabolomics examination of the urine samples of rats from different groups, including the albiflorin-treated and control groups. Through the analysis of metabolites in the two groups, no significant difference was found, indicating that albiflorin had no effect on the metabolism of rats. 40 In summary, the toxicity of albiflorin is relatively low, and its safety in clinical application has been ensured.
Clinical Application
At present, there are relatively few clinical studies on TGP. Some studies have shown that anti-tumor drug methotrexate combined with TGP is significantly better than methotrexate and TGP alone in the treatment of juvenile idiopathic arthritis. 41 Song et al 42 explored the effects of morphine combined with paeoniflorin on the dosage of antitumor drugs and related side effects in patients with middle and advanced cancer pain, and found that paeoniflorin could significantly reduce morphine consumption in patients with cancer pain and reduce the occurrence of morphine related side effects. TGP combined with conventional treatment can improve pain and swelling in patients with RA and coronary heart disease by inhibiting the release of inflammatory factors, regulating Th17/Treg balance, reducing immune indexes and improving coagulation function. 43 TGP can improve the clinical efficacy and quality of life of patients with allergic rhinitis by correcting Th1/Th2 immune imbalance. 44 Therefore, the combination of TGP and other drugs can often produce a synergistic effect, by enhancing the therapeutic effect. At the same time, this combination can reduce the toxicity of drugs and the occurrence of chemical drug adverse reactions.
Conclusions and Future Directions
Studying the ADME process of TCM ingredients in the body is conducive to elucidating the interaction between drugs and the organism as well as its underlying mechanisms. Pharmacokinetic characteristics of TGP emphasize its effective absorption and excretion, indicating good bioavailability. In terms of distribution, TGP exhibit a certain degree of tissue affinity, enabling them to accumulate in specific tissues and thereby exert local pharmacological effects. Significant advancements have been made in the metabolic research on TGP and its main component paeoniflorin. These studies have not only unveiled the crucial role of intestinal bacteria in drug metabolism but also delved deeply into the liver's capacity for drug biotransformation. In summary, TGP has certain toxicity, while the toxicity of albiflorin is relatively low. This research enhances bioavailability and provides a more scientific theoretical foundation for the clinical application of TGP.
Although the ADME of action of TGP has been partially elucidated, there are still many unknown areas awaiting exploration. Metabolomics is a high-throughput analytical technique used to study changes in metabolites in living organisms.Metabolomics also enables the discovery of biomarkers associated with TGP efficacy, which can help evaluate the therapeutic effect of TGP and predict the patient's response to TGP. Proteomics is a technique that studies the composition, structure, function and interactions of all proteins in living organisms. In TGP studies, proteomic techniques can be used to explore the effect of TGP on protein expression profiles and to reveal the relationship between its pharmacological action and protein expression.
Future research can further investigate the mechanisms of TGP action at cellular and molecular levels. This will support for a more comprehensive knowledge of understanding of the pharmacological effects of TGP and offer a theoretical foundation for its application in more disease areas. Currently, TGP is primarily administered in oral formulations, but its bioavailability and stability still need improvement. Future research can focus on developing novel formulations, such as microcapsules and subnano emulsion liposomes, to enhance the efficacy of TGP and reduce adverse reactions. In summary, as a drug component with broad prospects, TGP requires continued in-depth research into its mechanism of action, optimization of formulations and drug delivery methods, expansion of clinical application areas and strengthening of quality control in its future development. This will contribute to promoting the widespread clinical use of TGP and the advancement of TCM.
Footnotes
Author Contributions
Design of the work: Jie Ning; literature review for Pharmacokinetics: Jie Ning; literature review for Toxicity: Jie Ning; drafting the manuscript: Jie Ning; critical revision of the manuscript: Jie Ning, Yizhou Xin.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Traditional Chinese Medicine Science and Technology Project of Shandong Province (grant number Z-2023052) and Qilu Health and health leading talent project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
