Abstract
The most prevalent endocrine condition, diabetes mellitus, is characterized by aberrant protein, lipid, and carbohydrate metabolism as well as persistent hyperglycemia. It is a pathological condition that can result in long term conditions that harm various tissues and organs, including the brain. Metabolically, the brain is one of the most active of all organs of the body, and due to its high metabolic demands, its functions may be particularly sensitive to metabolic problems. Thus, under conditions with disturbances in systemic glucose homeostasis such as in diabetes, glucose supply to the brain may be affected and have deleterious impacts on brain metabolism. Melatonin, a small molecule neurohormone with special ability to freely cross biological membranes and the blood-brain-barrier, is known to possess anti-inflammatory potentials, acts as a free radical scavenger; and may be associated with diabetes due its functional inter-relationship with insulin. Researches have recently shown the capability of melatonin to reduce diabetic complications by ameliorating oxidative damage. Hence this review suggests a pharmacological dose of melatonin use to improving brain function owing to its capability in ameliorating complications in diabetic brain.
Introduction
Diabetes Mellitus (DM) is the most common endocrine disorder characterized by persistent hyperglycemia and abnormal carbohydrate, lipid, and protein metabolism. According to Mahmoud, 1 it is a pathological state which leads to long term complications causing damage of different tissues and organs, including the brain. The main complications of DM are nephropathy, retinopathy, and neuropathy. 2 Classification of the American Diabetes Association showed that DM is divided into different types according to the underlying cause, with type 1 and type 2 being the two main types and most frequent. 3
The brain is structurally and functionally the most diverse and intricate organ in the human body, and due to its plentiful lipid content and high energy requirement, it's metabolism places great demands on mitochondria and may be more susceptible to oxidative damage than the rest of other organs of the body.4,5 Metabolically, it is one of the most active of all organs of the body, and due to its high metabolic demands, brain functions may be particularly sensitive to metabolic problems. The brain is the central organ for adaptation to different stressors and is responsible for altering systemic functions through neuroendocrine, autonomic, immune and metabolic systems. 6 Normal brain function relies on efficient neuronal communication or neurotransmission. Therefore, maintaining neurotransmission is essential to ensure the fidelity of information transfer from one neuron to the next and the ability of the cell to respond to the local environment. 7 Under conditions with a disturbance in systemic glucose metabolism such as in diabetes, the supply of glucose to the brain may be affected and have impacts on brain metabolism and neurotransmission. 3 Defective neurotransmission and progressive neurodegeneration are reported to be triggered by diabetes. 8 It is increasingly evident that the brain is another site of diabetic end-organ damage. For instance, uncontrolled diabetes has a variety of adverse effects on brain metabolism, function and has been acknowledged as an important factor for brain disorders. 9 Other studies have demonstrated significant alterations in the cerebral metabolism, structure, and function during the development of DM and its complications9-11 In addition, Amer et al 12 reported that diabetic brain is a serious complication of diabetes, and it is associated with oxidative stress and neuronal injury. A plethora of studies in rodent models of diabetes suggests that both glucose neurotoxicity and deficient insulin signaling impair brain structure and function leading to behavioral and cognitive alterations.13,14 Interestingly, both types of diabetes may be associated with brain impairments such as cognitive decline and dementia. 3 Similarly, both hyper- and hypoglycemia affect brain function and may lead to cognitive dysfunction. 15
Melatonin, a tryptophan-derived endocrine hormone mainly synthesized by the pineal gland and locally by numerous other tissues,16,17 is known to be an anti-inflammatory agent and a powerful antioxidant. 18 Interestingly, melatonin may readily penetrate the blood-brain-barrier and enter the central nervous system and cerebrospinal fluid through the choroid plexus by virtue of its amphiphilicity. Studies have shown that melatonin may be associated with diabetes mellitus due to a functional inter-relationship between melatonin and insulin. Insulin is a key hormone that regulates glucose levels and cellular metabolism following meals in many tissues, including the brain. 19 For instance, melatonin has been found to influence insulin secretion both in vivo and in vitro, and night-time melatonin levels are related to night-time insulin concentrations in patients with diabetes. 18
A decreased level of melatonin secretion is independently associated with the increased risk of type 2 diabetes developing in a patient. Hence, it was recently reported that variants of melatonin membrane receptors and low secretion of melatonin aggravate the risk of developing type 2 diabetes, 19 suggesting that melatonin may be involved in the regulation of glucose homeostasis. An emerging body of evidence shows that melatonin reduces diabetic complications by ameliorating oxidative damage. 20 Mok et al, 21 also reported that melatonin administration is proven to bring other anti-diabetic effects such as reducing cellular apoptosis and promoting the production of antioxidants. Despite all available data and much recent advancement in the research on the role of melatonin in glucose homeostasis, the mechanisms that underlie its role in glucose homeostasis remain complex, and the available data and recent advancements in the field have produced inconsistent results. While some study suggests that melatonin plays a positive function in the prevention and treatment of T2DM, other studies have suggested it may work against insulin.22,23 In view of the foregoing, there is a conflicting report on the role of melatonin in diabetic conditions. This review therefore, provides an insight on the potential impact of melatonin on brain metabolism in diabetes.
Overview of Diabetes Mellitus
A chronic metabolic illness with a diverse pathophysiology, diabetes mellitus is variable and heterogeneous in nature. Elevated blood sugar levels, often known as hyperglycemia, are its defining feature. Hyperglycemia is caused by changes in insulin secretion, action, or both. Metabolic dysfunctions involving carbohydrates, fats, and proteins are caused by hyperglycemia, which can express itself in a variety of ways. Numerous microvascular and macrovascular problems of diabetes are frequently brought on by long-term hyperglycemia, and these complications are mostly to blame for the morbidity and death of people with diabetes. 24 These complications affect body organs, including the eyes, kidneys, heart, and nerves, causing organ damage, dysfunction, and finally organ failure. Neuropathy, which can be autonomic or peripheral, is caused by complications related to the nerves. Long term peripheral neuropathy is frequently accompanied by foot infections including ulcers and osteoarthropathy. Cardiovascular, gastrointestinal, and genitourinary (including sexual) dysfunctions are typical indications of autonomic neuropathy. 25
Diabetes has emerged as one of the most significant and difficult health problems facing the human population of the modern world due to its rising global prevalence. Currently, 387 million people are affected with diabetes; 26 The International Diabetes Mellitus Federation estimated that 592 million people will suffer from diabetes mellitus by the year 2035. In Nigeria, there has been an increase in the prevalence of DM. According to Uloko et al, 27 all regions of the country have been affected, with the highest prevalence seen in the south-south geopolitical zone and the lowest rate noted in the north-western zone. Accordingly, the pooled DM prevalence of 5.77% reported in the study by Uloko et al, 27 suggests that 11.2 million Nigerians are living with the disease. The increase in the prevalence of diabetes in Nigeria and globally has been paralleled by physical inactivity, advanced age, and rapid economic development; leading to urbanization and the adoption of modern lifestyle habits, including unhealthy dietary habits. 27 Thus, diabetes and diabetic complications are considered as leading causes of both morbidity and mortality in the world. 28
Classification of Diabetes and Pathophysiology
The classification currently in use, according to the American Diabetic Association, 29 is based on etiology and pathophysiology of the condition and is helpful in the clinical assessment of disease and in determining the necessary medications. Based on this classification, diabetes can be divided into four main types or categories: type 1 diabetes mellitus (T1DM), type 2 diabetes mellitus (T2DM), gestational diabetes mellitus (GDM), and diabetes caused or associated with certain specific conditions, pathologies, and/or disorders. The focus however, will be on type 1 and type 2.
Type 1 Diabetes Mellitus
T1DM often referred to as type 1A diabetes mellitus (DM), insulin-dependent diabetes mellitus (IDDM), or juvenile-onset diabetes, accounts for around 5-10% of all diabetes cases. It is an autoimmune condition marked by the T-cell-mediated apoptosis of pancreatic beta-cells, which causes an insulin shortage and ultimately leads to hyperglycemia. 30 Despite the fact that the pathogenesis of this autoimmunity is still not fully understood, it has been discovered that both hereditary and environmental variables play a role. Autoantibodies in particular are one of the immunological markers that distinguish T1DM from other autoimmune diseases. These autoantibodies are linked to the disease's distinctive immune-mediated cell death.
Type 2 Diabetes Mellitus
About 90%-95% of all instances of diabetes are T2DM, often referred to as non–insulin–dependent diabetes mellitus (NIDDM) or adult-onset diabetes in the former nomenclature. In this kind of diabetes, insulin resistance and β-cell dysfunction are the two main insulin-related abnormalities that are present. Disruptions in numerous cellular pathways cause insulin resistance, which reduces the sensitivity of cells in peripheral tissues, particularly those in the muscle, liver, and adipose tissue, to insulin. Increased insulin production to maintain normoglycemia is achieved in the early stages of the disease when impaired insulin sensitivity causes β-cells to hyper-function. Hyperinsulinemia, or increased amounts of circulating insulin, thereby prevents hyperglycemia. Nevertheless, over time, the increased insulin secretion by β-cells is unable to fully make up for the declining insulin sensitivity. Additionally, β-cell function starts to deteriorate, and its dysfunction finally results in insulin shortage.
This form of diabetes has a complicated pathophysiology and etiology that is caused by a number of known and unknown causes, which may be definitively attributed to a confluence of hereditary predispositions and significant environmental impacts. Ageing, obesity, family history of diabetes, sedentary lifestyles, prior gestational DM in women, and pathophysiological diseases including hypertension and dyslipidemia have all been linked to T2DM more frequently than other risk factors. In contrast to T1DM, no link between this illness and genes is implicated in the immune response, including autoimmunity, has been discovered, and as a result, there is no immune-mediated pancreatic β-cell death.
Brain Metabolism in Diabetes
Brain metabolism is highly active and accounts for 20% of the body's oxygen and 25% of glucose consumption in a subject at rest state. 31 Metabolically, the brain is an active organ in the body and does not store excess energy and derives almost all of its energy needs from aerobic oxidation of glucose. Thus, continuous supply of glucose and oxygen to meet its energy requirement is required. The major energy demand for the brain is to fuel neurotransmission. Concerning the substrates for the brain's mitochondria's oxidative phosphorylation, there is disagreement. 5 Glial cells use the remaining energy that is used by brain cells, with neurons using 70%-80% of it. Genes and metabolites involved in oxidative metabolism and mitochondrial functions are up-regulated in human brains, according to studies comparing the expression of genes and regulatory regions in the brains of different primates. Additionally, recent data suggests that the evolution of brain size and complexity depended on a rise in metabolic rate, together with a stronger propensity to store fat and changes in the allocation of energy resources. 32 In addition to advancing our understanding of how the brain works and adapts to environmental demands, understanding the metabolic signatures of various brain cells and their metabolic interactions may also shed light on the propensity of the human brain to age-related neurodegenerative disorders.
The majority of the energy metabolism in the brain is based on mitochondrial respiration, which is why oxygen and substrate delivery to this organ is crucial. Glucose has long been thought to be the main substrate for energy production in the central nervous system, and this belief has not changed over time. 33 The only point of contention among the researchers is whether glucose enters the brain directly or via lactate. However, it was also demonstrated long ago that fatty acids (majorly, octanoate) are oxidized in the brain (mainly in astrocytes), contributing to at least 20% of the overall energy produced in the brain. One must keep in mind that the structure and general functions of the cells these organelles are found in are closely related to the operations of mitochondria in order to understand the brain's energy metabolism.
Major Pathways of Glucose Intake in the Brain
The metabolic fate of glucose in the brain depends upon the cell type and the selective expression of metabolic enzymes. Neurons are predominantly oxidative, while astrocytes are mostly glycolytic. In addition to the production of adenosine triphosphate (ATP), glucose is also used to generate metabolic intermediates for the synthesis of fatty acids and other lipids required for membrane and myelin synthesis, amino acids for protein synthesis and neurotransmitter production; and 5-carbon sugars for synthesis of nucleotides; and to produce glycogen in astrocytes. In neurons, each molecule of glucose is oxidized via glycolysis, the pentose phosphate pathway, the tricarboxylic acid (TCA) cycle, and oxidative phosphorylation, with the production of carbon dioxide, water, and 30-36 molecules of ATP depending upon the rates of proton leakage in the mitochondria. 34
The glycolytic process metabolizes glucose to pyruvate, which can be actively transported into the mitochondria where it is converted to acetyl coenzyme A (Acetyl-CoA). Acetyl-CoA is complexed with citrate which undergoes a series of regenerative enzymatic reactions producing reduced nicotinamide adenine dinucleotide (NADH) and flavin adenine dinucleotide (FADH2) in the TCA cycle. The NADH and FADH2 produced during glycolysis and the TCA cycle are subsequently re-oxidized in the electron transport chain (ETC). Once inside the cell, glucose is irreversibly converted to glucose-6-phosphate by hexokinase. Although negligible compared to peripheral energy deposits, glycogen represents the largest energy reserve in the brain.
Through isoforms of equilibrative glucose transporters (GLUTs) that have different kinetic properties, glucose is reversibly transported from arterial blood across endothelial membranes into and out of brain cells. GLUT1 is localized in endothelium and astrocytes, GLUT3 and GLUT4 in neurons, and GLUT1 is also found in astrocytes and endothelium.35,36 The reduced concentration of glucose in the brain caused by the continual metabolism, is what propels net glucose absorption into the brain. The maximal transport capacity across the BBB is two to three times greater than the resting glucose utilization rate, with the brain-to-plasma distribution ratio for glucose in humans and rats being 0.2. More fuel is delivered than is used, which protects the brain from temporary energy failure that could have negative effects.
Although the precise pathways taken by glucose as it diffuses from arterial plasma into brain cells along its concentration gradient remain unknown, kinetic modeling suggests that the majority of glucose diffuses via interstitial fluid. Gandhi et al 37 who studied the diffusion of glucose through its concentration gradient from an astrocyte to a neuron in brain slices, showed that glucose can enter and exit cells depending on local concentration gradients and supply-demand ratios in specific cells. As a result, depending on local gradients, the total flow of glucose will be from source to sink through interstitial fluid and intracellular fluid via glucose transporters. After entering a cell, glucose can either be transported back to the blood and extracellular fluid, reduced to sorbitol, or phosphorylated by hexokinase to create glucose-6-phosphate (G-6-P), which is then retained inside the cell. Because a sizeable portion of the glucose that enters the brain is released back into the blood, the un-metabolized glucose in the brain forms an exchangeable pool. Thus, the three major pathways of glucose metabolism in the brain are (1) glycolysis, which produces three-carbon compounds, pyruvate, which can be used for biosynthetic processes or oxidized in the tricarboxylic acid cycle to produce energy as ATP through oxidative phosphorylation, or lactate for export; (2) glycogen synthesis, which stores glucose; and (3) the pentose phosphate pathway, which produces reducing equivalents. Each of these routes function in the brain in the same manner it does in the peripheral nervous system.
Diabetic Effects on the Brain Metabolism
Diabetes alters cerebral metabolism, structure, function and major alterations in the brain's signaling systems that impede metabolism as well as the functioning of the neurological and other systems.11,38 Diabetes results in downstream complications, including neuropathy. Since glucose is the primary source of metabolic fuel for the brain, any kind of dysglycemia (including hyperglycemia, hypoglycemia, and acute glycemic variations) may contribute to cerebral problems in diabetics and, acute glycemic fluctuation may also be responsible for the changes connected with diabetic brain. 39 Muscle cells may accept episodic glucose uptake while under the effect of insulin, neurons cannot because neurons have a continual high glucose requirement.
Glucose neurotoxicity, which can result from repeated bouts of hyperglycemia, is a condition in which the extracellular concentration of glucose interferes with the ability of neurons to absorb glucose. 40 Neuronal glucose levels can rise by up to fourfold in diabetes-related hyperglycemia. Intracellular glucose metabolism causes neuronal injury if this continues or if these episodes occur frequently. Diabetic neuropathy is the clinical condition that characterizes this dangerous long-term diabetes complication. The functional effects of these pathways are slowly becoming understood, but recent research has revealed the existence of harmful metabolic pathways downstream of elevated glucose to which neurons are particularly vulnerable. Thus, neurons are susceptible to glucose toxicity due to carrier-mediated glucose absorption with low hormonal control, which is likely a result of the fundamental necessity for a glycolytic substrate with minimal intracellular reserves. Although the blood-brain barrier provides some protection for the brain, there is evidence that it may not function as well for peripheral neurons because hyperglycemia is linked to significant increases in intracellular glucose levels. 40 It is unknown if more specialized brain cells, such as presumed appetite sensors, have more refined glucose affinities, making them more or less susceptible to glucose toxicity. The normal fate of intracellular glucose is phosphorylation of the carbon in the number-six position and entry into glycolysis, but when the insulin independent uptake is triggered by abnormally high glucose levels in the interstitial fluid, the glucose is diverted to metabolic pathways that can cause neurotoxicity. Other effects of diabetes on the brain metabolism are illustrated in Figure 1.

Effect of Diabetes on Brain Metabolism.
Diabetes and Cognition
Diabetes mellitus is a metabolic disease that can have worsening effects on many organs, including the brain. A growing body of evidence, both in animal models and epidemiological studies, has demonstrated that metabolic diseases such as diabetes are associated with alterations in the central nervous system, and is being linked with development of cognitive and memory impairments.
41
Also, according to Duarte,
10
diabetes is associated with decrements in cognitive function coupled with abnormalities in brain morphology
Type 1 diabetic patients have impairments in learning and memory, problem solving, and mental and motor speed. Patients with type 2 diabetes present moderate impairments in tasks involving verbal memory or complex information processing, increase in memory deficits, reduced frontal lobe/executive function, and reduction in psychomotor speed; while patients with Type 1 diabetes have consistently shown to do less well on assessments of information processing, attention, psychomotor efficiency, visual construction ability, and mental flexibility.
Although both diabetes types play a role in memory weakening, the mechanisms by which cognitive abilities are impaired in diabetes remains unclear. 44 Nevertheless, it is reported that factors such as neuronal apoptosis, altered neurogenesis, electrophysiological deficits, oxidative stress injury, neuroinflammation, reduced neuronal densities, and metabolic impairments are crucial in the pathogenesis of cognitive impairment, induction of structural changes and memory deficiency. 45 Diabetes-associated cognitive decline has been linked to learning and memory deficits, which in turn increase the risk for dementia. A study by Modi et al 46 recommended a significant role of oxidative stress in the pathogenesis of cognitive impairment. 46 The long-term hyperglycemia enhances glucose oxidation, which sequentially produces reactive oxygen species (ROS) causing increased oxidative stress; this leads to morphological and functional changes in the hippocampus due to the excessive production of malondialdehyde (MDA).
Overview of Melatonin
The word ‘melatonin’ (also referred to as N-acetyl-5-methoxytryptamine) is derived from a Greek word ‘melas meaning dark’ and ‘tonos meaning hormone of darkness’ is an indoleamine and ubiquitous molecule with wide distribution in nature and is produced by many living organisms- animals, humans, plants, fungi, and unicellular organisms. It was discovered and isolated from bovine pineal in 1958 by Aaron Lerner. Melatonin is primarily produced by the pineal gland in humans, with smaller amounts also produced by extrapineal sources such as the retina, platelets, Harderian gland, cerebellum, placenta, skin, lymphocytes, bone marrow, gastrointestinal system, and thymus, which extended the first-discovered roles of this indoleamine as a melanosome-concentrating agent in fish and amphibian and a mediator of the signal “darkness“ in the vast majority of vertebrates. 16
Notably, melatonin has been discovered in a wide range of foods, whether they come from edible plants or products derived from plants (such as tomatoes, strawberry, rice, corn, walnuts, black pepper, almond, cherry, and pomegranate). The plants and a select few food sources not only have melatonin but its precursors. In plants, melatonin presence is universal; however, the amounts can range from picograms to micrograms in plant tissue.
Relationship Between Melatonin and Diabetes
According to Bhat et al and Ali et al,47,48 diabetes mellitus, a condition of carbohydrate metabolism, is characterized by abnormalities in insulin secretion and action as well as high blood glucose levels. Because it helps regulate sleep and the circadian rhythm, melatonin has grown in significance. But recently, it has become more significant because of its impact on glucose tolerance and the risk or treatment of type 2 diabetes (T2D). This is brought on by the incomplete identification of T2D risk alleles in MTNR1B and the mixed effects of circadian rhythm disturbances on glucose metabolism. 49 According to Pulimeno et al 50 a circadian rhythm abnormality has been linked to the metabolic syndrome, which includes diabetes and obesity. Melatonin's role in insulin secretion and glucose homeostasis has been extensively studied. Melatonin levels have been found to be lower in T2D patients. For instance, in a study on the role of melatonin in glucose homeostasis in young Zucker diabetic fatty (ZDF) rats, a model for metabolic syndrome and T2D, it was discovered that oral melatonin administration reduces blood sugar levels in young ZDF rats by enhancing β-cell function. 51 According to research, the levels of the glucose transporters GLUT1 and GLUT2 and the pancreatic islet hormones have a direct impact on melatonin receptor insufficiency. 52 The relationship between melatonin and T2D is based on the discovery that insulin secretion is inversely correlated with plasma melatonin levels. 53 A key factor in the beginning of T2DM may be reducing melatonin secretion as a result of nighttime light exposure. Numerous studies have linked T2D, reduced glucose tolerance, and disrupted sleep patterns. In addition to boosting the positive effects of insulin, melatonin treatment modifies the synthesis of insulin in pancreatic beta-cells. 54
Antidiabetic Effects of Melatonin
Both animal and human research have demonstrated the usefulness of melatonin in regulating blood glucose. In pinealectomized mice, the expression level of the GLUT4 gene is decreased, which consequently causes insulin resistance and glucose intolerance. Melatonin therapy is used to treat these diseases. 55 For instance, an eight-week course of treatment with insulin and melatonin (0.2 mg/kg/day in drinking water) in diabetic rats with streptozotocin (STZ) improves glucose homeostasis and insulin sensitivity of white adipose tissue compared to utilizing one of these medications alone. 56 An available recent report by de-Sousa et al 57 suggested that 12-week melatonin administration had no effect on diabetes risk markers. Melatonin synthesis has been shown to decrease in diabetes animal models, and mice fed a high-fat diet who have insulin resistance respond better to melatonin treatment (100 mg/kg/day in drinking water for 8 weeks). 58
When compared to people without diabetes, people with type 2 diabetes mellitus produce less melatonin at night. 59 The glucose tolerance test in the morning and the evening gets worse after taking a single dosage of melatonin in healthy post-menopausal and pre-menopausal women (1 mg and 5 mg, respectively) according to several studies.60,61 On the other hand, long-term administration of prolonged-release melatonin (2 mg) over a period of 5 months lowers the HbA1c level and improves glycemic management. 62 Furthermore, patients with T2DM had better glycemic control after receiving melatonin (6 mg) for three months. 63 Adding zinc acetate (50 mg) and melatonin (10 mg) to metformin improved tissue responsiveness compared to metformin alone in patients with poorly managed T2DM. Melatonin (3 mg/day) treatment for 12 weeks reduced inflammatory state and insulin sensitivity in obese Acanthosis nigricans patients. 55 Melatonin may improve glycemic management by boosting insulin sensitivity and reducing fasting blood sugar, according to a recent comprehensive study. 64 Also, the findings of our latest research indicated an improvement in beta cell function, insulin sensitivity, and decreased insulin resistance following supplementation with melatonin. 65 We also suggested that in order to increase anti-hyperglycemic condition and strengthen the body's antioxidant defense system, treating type 2 diabetes with a pharmacological dose of melatonin is a crucial step in obstructing the mechanisms that cause hyperglycemia. Also, melatonin's anti-oxidative properties are very effective at reducing oxidative stress, which plays a significant part in the development of numerous problems of diseases like diabetes. Additionally, by scavenging reactive oxygen species, this hormone defends the pancreatic beta cells, which contain few antioxidants.66,67
Melatonin and Insulin Secretion
For glucose homeostasis and energy balance in the human body, the circadian regulation of carbohydrate metabolism is crucial. Obesity, metabolic syndrome, cardiovascular disease, and type 2 diabetes are just a few of the metabolic disorders that have been related to imbalances in glucose and insulin levels in tissue and blood. 68
First experimental injections of pineal extracts, according to the scientific literature, resulted in hypoglycemia, enhanced glucose tolerance, and hepatic and muscle glycogenesis. The metabolic disruption brought on by the lack of melatonin in the pinealectomized animals was classified as a diabetogenic syndrome represented by glucose intolerance and insulin resistance, both of which were centrally expressed at the level of the hypothalamus and peripherally in the hepatic, adipose, and skeletal muscle. Restricted eating or melatonin replacement therapy can reverse this abnormality. Along with this shocking discovery, some physiological or pathological conditions linked to lower blood melatonin levels, such as aging, diabetes, shift work, environmental illumination at night, and the so-called “phenomenon of light pollution,” can be seen to cause insulin resistance, glucose intolerance, and a number of other metabolic disorders. Most of these abnormalities are reduced by an adequate melatonin replacement medication. It is believed that the pinealectomy-induced insulin resistance and glucose intolerance are related to the mechanistic effects of the depletion of melatonin, which are perceived at the molecular level as a deficiency in the insulin-signaling pathway and a reduction in GLUT4 gene expression and protein content. This emphasizes the functional synergism between melatonin and insulin. According to Sharma et al, 69 the effects of melatonin on insulin secretion are mediated through MT1 and MT2 receptors which are expressed in pancreatic tissues. It decreases insulin secretion by inhibiting cAMP and cGMP pathways but activates the Phospholipase-C/IP3 pathway, which functions to mobilizes Ca2+ from organelles and concomitantly increase the secretion of insulin. Melatonin has also been demonstrated to successfully bypass several intracellular transduction steps of the insulin signaling pathway by inducing rapid tyrosine phosphorylation and activation of the tyrosine kinase beta-subunit of the insulin receptor through the action of MT1 membrane receptors. 70
Melatonin and Glucose Homeostasis
The role of melatonin in glucose homeostasis has become an active area of investigation. There is a growing body of evidence suggesting a link between disturbances in melatonin production and impaired insulin, glucose, lipid metabolism. 71 It is believed that maintaining the regularity of the circadian rhythm is the major purpose of activating the melatonin signaling pathway. However, a growing body of research is revealing complicated relationships between melatonin and the systems that control glucose management. 72 For instance, studies have linked melatonin to the activity of beta cells,72,73 and alterations in melatonin signaling may predispose people to the metabolic abnormalities connected with type 2 diabetes. 49 In investigations designed to establish a physiologic relationship between melatonin and glucose homeostasis, both decreased melatonin synthesis and secretion have been noted. 74
The pineal gland controls glucose homeostasis and insulin production from pancreatic β-cells. Patients with T2DM had higher levels of melatonin membrane receptor mRNA expression and lower levels of systemic melatonin circulating in the bloodstream. 75 The increased expression of melatonin membrane receptor mRNA is thought to make up for the low levels of melatonin found in T2DM patients. 76 Additionally, it has been demonstrated that polymorphisms in the MTNR1B gene harboring the risk allele G increase mRNA expression in β-cells, resulting in a reduction in insulin secretion because cAMP synthesis is reduced. As a result, this raises the plasma glucose level, causing T2DM. 77 Clinical studies have shown that melatonin improves glycemic control, and that melatonin deficiency may lead to T2DM. 51 Additionally, melatonin controls blood sugar levels by lowering fasting plasma glucose, not by raising insulin secretion. Melatonin reduces insulin production from β-cells and protects it from being functionally overworked in T2DM. 78 The elimination of MT1 has been linked to an increase in insulin resistance and glucose intolerance, according to a recent study. After a pinealectomy to reduce endogenous melatonin levels, the daily blood glucose rhythm was changed or disrupted, displaying rising glucose concentrations at night. 79 A study utilizing pinealectomized rats found that insulin levels declined and glucose and glucagon levels rose. Although glucose intolerance is present, it is lessened by melatonin therapy.
Melatonin and Diabetic Brain
Many structural and functional integrity issues pertaining to the brain are disrupted by diabetes. Neurological disorders such as schizophrenia, Alzheimer's disease, dementia, and impaired cognitive development stroke, T2DM, and T2DM-associated AD have been linked to decreased melatonin production and diabetes.80,81
Antioxidant therapy thus, may restore physiological capabilities since oxidative stress is a major factor in the development of diabetes problems. 82 Antioxidant therapy has been shown in numerous investigations to reduce diabetes-induced damage to hippocampus neuronal cells. In addition to its diverse physiological functions in endocrine and neurological problems, melatonin has been shown to have a wide range of safe actions in humans and animal models and also reported to have neuroprotective benefits in a variety of situations. 83 In neurodegenerative disorders where brain oxidative damage has been shown to be a common connection, melatonin serves as a neuroprotective agent. Melatonin has also been shown to influence insulin secretion, which is mediated by the MT1 and MT2 receptors, in both in vitro and in vivo studies. Additionally, melatonin provides protection to pancreatic β-cells, which are very susceptible to oxidative stress and the production of reactive oxygen species. For instance, in a streptozotocin-induced diabetic rat model, melatonin treatment decreases lipid peroxidation and inhibits glial fibrillary acidic protein (GFAP), a major indication of astrocyte dysfunction, according to the reports of Baydas et al 84 This is achieved by boosting the brain's defense mechanisms against oxidative stress, lowering brain nitric oxide synthase activity, and raising plasma cytokine levels that prevent cell death, melatonin works to preserve central nervous system homeostasis. 85
Several other researchers have documented the potential effects of melatonin on diabetes, and in combination with other therapies. For instance, Thomas et al 86 reported that the combination of melatonin and metformin had synergistic actions to modify progression of metabolic dysfunction in circadian disruption and diet-induced obesity (CDO) rats through improved adiposity, circadian activity, insulin sensitivity, and islet cell failure. Melatonin and metformin, the most often given medication for type 2 diabetes, have comparable antidiabetic effects because they both employ the same pathways to protect the pancreas and other organs from stress. Melatonin also enhances the effects of metformin. Melatonin and metformin are anticipated to have potentially synergistic effects, and it was highly recommended using both medications together therapeutically to manage diabetes. 87 Rahman et al, 88 reported that combining melatonin with exercise therapy efficiently scavenges dangerous free radicals, suggesting that melatonin administration counteracts oxidative pathways to provide its anti-diabetic benefits. Another study by de Oliveira, 56 described that streptozotocin-induced diabetic rats with the treatment of insulin and melatonin improved insulin sensitivity as well as glucose metabolism of white adipose tissues. Also, decreased melatonin production has been reported in diabetic animal models which were protected by exogenous melatonin administration in the high-fat diet-fed mouse with insulin resistance. 58 Amer et al 12 also reported that melatonin had a notable ameliorative impact against molecular and biochemical changes in the diabetic rats’ brains, an impact they attributed to its antioxidant properties. Accordingly, melatonin thus plays a vital role in preventing the development of diabetes through modulation of Aβ build-up, insulin resistance, glucose metabolism, and BBB permeability. 89 Studies of melatonin effects on diabetes and mechanisms are summarized in Table 1. Melatonin synthesis from the pineal gland is stimulated by the darkness and inhibited by light. The secreted melatonin can act on the brain function and metabolism leading to the up-regulation and down-regulation of some parameters as illustrated in Figure 2.
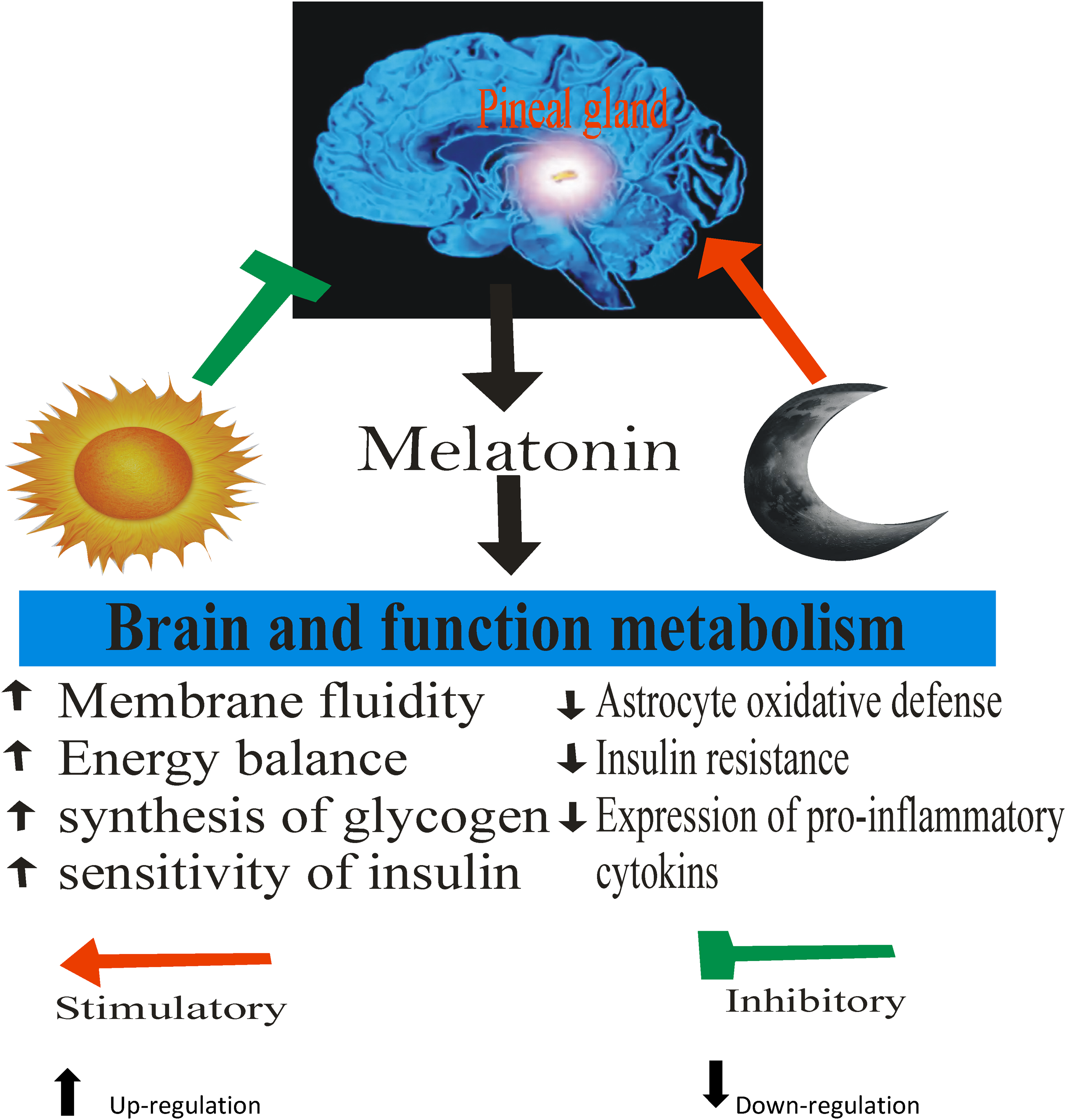
Effect of Melatonin on Brain Metabolism in Diabetes.
Studies of Melatonin Effects on Diabetes and Mechanisms.

I.P., intraperitoneal; PGC-1a, peroxisome proliferator-activated receptor gamma coactivator 1a; Nrf2, nuclear respiratory factor-2; GFAP, Glial Fibrillary acidic protein; NOS, nitric oxide synthase; NCAM, neural cell adhesion molecule.
The Melatonin Paradox
The underlying mechanisms connecting melatonin's role in glucose homeostasis remains complex and the evidence is sometimes contradictory. For example, although some studies have suggested that melatonin may have an adverse effect on insulin, other studies have found that melatonin may have a beneficial effect on the prevention and treatment of type 2 diabetes.22,23
The most widely accepted contemporary theory is that melatonin affects glucose homeostasis, which is mediated via its inhibitory effect on insulin secretion. The revelation that MTNR1B mRNA expression levels in human pancreatic islets of carriers of the common variant (a risk allele) are 4 times greater than in islets of those without this variant is a crucial result for this idea.
In light of this, it is anticipated that enhanced melatonin action may increase melatonin signaling in islets and decrease insulin release, resulting in hyperglycemia and a higher risk of T2DM.77,93 This approach first appears straightforward and consistent with the phenotype of risk allele bearers, which includes elevated FPG levels and T2DM risk. 94 Therefore, it would be recommended to restrict the melatonergic system's activity (by utilizing melatonin receptor antagonists) for T2DM patients who carry the risk gene. The situation is not as straightforward as that, however, as the decreased nighttime melatonin levels seen in T2DM patients 74 and the numerous rare MT2 mutants that cause a loss-of-function phenotype and are linked to an increased risk of developing T2DM,74,95 suggest rather the opposite, namely that dampening of the melatonin system results in the development of T2DM. 96
Numerous studies and commentaries,77,96-98 have addressed these contentious claims, but no explanation has been provided. Some have established a “equilibrium hypothesis” in an effort to explain the data, according to which deviation from the equilibrium (normal condition), caused by enhanced or muted melatonin activity in carriers of frequent and rare variants, respectively, becomes harmful for glucose homeostasis. 77 Others proposed a “age-related chronobiological hypothesis” that highlights the value of the circadian system and the decline of that system with age. 99 Others defended the “loss-of-function hypothesis” by disputing the significance of pancreatic MT2 receptors and by hypothesizing that melatonin has a significant indirect impact on glucose metabolism due to its well-established regulating role on the SCN, the body's main biological clock. 97 None of these possibilities are satisfying at the moment, or they require further experimental proof to be widely accepted.
In order to comprehend the impact of melatonin on insulin release in the early stages of T2DM, the issue of insulin sensitivity in the transition from pre-diabetes to T2DM should be taken into account. It may be possible to gain a better understanding of how this hormone works and the apparent contradictions to pave the way for a unifying model that can be accepted by all experts in the field by revisiting some specific aspects of the melatonin system, such as the various effects that melatonin has and the regulation (desensitization and sensitization) of melatonin responses. 23
Conclusion
From all the literatures reviewed, the capability of melatonin in ameliorating complications in diabetic brain has been proven. The impact mechanisms have been attributed to its antioxidant properties and by controlling the accumulation of Aβ, insulin resistance, glucose metabolism, and blood-brain-barrier permeability. However, there are still some disagreements in some aspects. Therefore, further studies are required to substantiate the impact of melatonin on brain metabolism in diabetes and molecular targets.
Footnotes
Acknowledgements
The authors are grateful to Dr Chinedum Emelike, Mr Sylvester Ibekailo, Dr Kenneth Oparaji and Miss Hauwa Ifogah of the Department of Physiology, for providing technical support.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The authors declare that the data supporting the findings of the study are available within the article
