Abstract
Objective
The detrimental effect of gamma radiation on testicular tissue is an intricate problem in the field of nuclear medicine. Therefore, this study aimed to investigate the potential mechanism of carvacrol against testicular toxicity induced by γ-rays based on bioinformatics and in vivo experiments.
Methods
The carvacrol targets were retrieved from the SwissTargetPrediction database. In addition, the targets for radiation-induced testicular damage were identified from the GeneCards database. However, the common potential targets were identified. Subsequently, GO and KEGG enrichment was conducted by using the KOBAS server. However, animals in the in vitro experiment were used to validate the in-silico analysis. In particular, the oxidative stress markers, including malondialdehyde (MDA), reduced glutathione (GSH), total antioxidant status (TAS), superoxide dismutase (SOD), and catalase (CAT), as well as testis and sperm characteristics and serum testosterone level were examined to unveil the crucial of carvacrol in γ-rays induced testicular damage.
Results
The findings explored that γ-rays induced testicular damage by markedly increasing oxidant markers, MDA, and substantially decreasing antioxidant markers, including GSH, TAS, SOD, and CAT. Additionally, radiation adversely affected testis and sperm characteristics and serum testosterone levels. However, bioinformatic analyses explored that the carvacrol target regulates the oxidation-reduction and testosterone biosynthesis processes. Additionally, experimental analysis explored that carvacrol improved the testis and sperm characteristics and oxidative stress.
Conclusion
The study revealed that carvacrol could reduce testicular toxicity in γ-irradiated rats via its antioxidant activity. Additionally, the study could benefit patients receiving radiotherapy.
Introduction
Radiation, particularly ionizing radiation, is present in the contaminant environment and causes damage to cells, tissues, and organs. Furthermore, radiation is employed extensively in clinics as a medical technical tool. However, Radiotherapy is an efficient remedy for different tumors, including cervical, rectal, lung, prostate, and breast cancers. 1 However, the reproductive system, particularly male reproduction, is susceptible to radiation-induced damage. Notably, testicular tissue exhibits heightened radiosensitivity, rendering spermatogenesis particularly vulnerable to radiation-induced impairment, potentially leading to infertility. 2 Furthermore, oxidative stress induced by radiation is recognized as a critical factor influencing testicular function. 3 Specifically, excessive generation of reactive oxygen species (ROS) can overwhelm endogenous antioxidant defenses, damaging testicular spermatozoa cell membranes. 4 Furthermore, the productive disorders, including a reduction in sperm count and motility, testosterone level, and the diversity of the testicle antioxidant enzymes, are consequences of γ-rays exposure in rats and mice.5,6
The sorbitol dehydrogenase (SDH) enzyme is a particular marker correlated with testicle dysfunction. 7 Sperm found in the seminiferous tubules has SDH that helps in its development; besides, it catalyzes the transformation of sorbitol to fructose and delivers energy to the rat sperm cells to convey regular activity. 8 Moreover, the generation of estrogens and androgens is regulated by 17β-hydroxysteroid dehydrogenases (17β-HSDs), which catalyze the reduction of 17-ketosteroids and the oxidation of 17β-hydroxysteroids. In particular, HSD17B3 is a gene that encodes17β-HSD3. However, HSD17B3 is expressed almost exclusively in testicular Leydig cells and contributes to the growth of sex traits in men by converting androstenedione (A4) to testosterone (T). In addition, 17β-HSD3 deficiency is a condition that affects male sexual development. Consequently, the potential damage to sexual development could result from the inhibition of 17β-HSD3, which results in impaired testosterone synthesis. The 17β-HSD enzyme plays an essential role in the biological activity of steroid hormones such as testosterone. 9 A decline in its activity causes a reduction in the serum testosterone level, total sperm count, sperm motility, and significant sexual dysfunction. 10 Despite the enzyme's critical role in early developmental and prepubertal processes, the interaction between 17β-HSD3 and environmental toxicants remains largely unexplored. 11
Natural herbal plants or their fractions are an optimistic hope in the prevention/treatment of testicular injury arising from exposure to γ-radiation.12,13 Clinically, carvacrol is safe when used against inflammation and oxidation and acts as an antimicrobial. 14 It protects against renal, hepatic, and malignant toxicity without toxic effects.15,16 Carvacrol exists in several food and aromatherapy plants of the Lamiaceae family, such as thyme, rosemary, and marjoram.17-19
However, the precise molecular pathways underlying the therapeutic effects of carvacrol in mitigating radiation-induced testicular damage remain incompletely understood. Consequently, a thorough exploration of the molecular mechanisms through which carvacrol modulates radiation-induced testicular toxicity is warranted. Network pharmacology, an evolving interdisciplinary field, offers a powerful approach for comprehensively dissecting the functional mechanisms of carvacrol. 20 In this study, we aimed to systematically elucidate the molecular mechanisms by which carvacrol counteracts oxidative and reproductive dysfunctions induced by 5 Gy whole-body γ-irradiation in the testes, utilizing a combination of bioinformatics analysis and experimental validation.
Materials and Methods
Predicting Related Targets
The carvacrol 2D structure was obtained from PubChem. The SwissTargetPrediction platform was employed to predict possible targets for carvacrol based on the structures of compounds uploaded in SDF format. 21 The probability was set to greater than 0. Moreover, the targets of radiation-induced testicular damage were identified via the GeneCard database with the keyword radiation-induced testicular damage. 22
Determination of Common Targets Between Carvacrol and Radiation-Induced Testicular Damage
The carvacrol targets and radiation-induced testicular damage genes were imported into Venny 2.1 (https://bioinfogp.cnb.csic.es/tools/venny/) to acquire common targets as potential targets for further analyses. 23
The GO Function and KEGG Pathway Enrichment Analyses
Gene ontology (GO) is extensively employed to investigate gene functions, including the biological process (BP), molecular function (MF), and cell component (CC). The Koto Encyclopedia of Genes and Genomes (KEGG) is a comprehensive database that includes genomic, chemical, and system functional information. It is frequently employed to identify considerably enhanced biological pathways. However, GO and KEGG for carvacrol targets were performed using the KOBAS platform. 24 However, the results were visualized by R-package software. 25
Drug and Chemicals
The chemicals were purchased from Sigma-Aldrich Co., USA. However, 3 mL of distilled water was used as an oral vehicle for carvacrol. The carvacrol dosage was 75 mg/kg/day for the succeeding 8 weeks. 26
Animal Groups
Thirty male Albino rats (age 10-11 weeks), weighing 260-270 g, were obtained from the Egyptian Organization for Biological Products and Vaccines, Helwan, Cairo, Egypt. The animal was kept at a controlled room temperature (23 ± 2 °C) and humidity (55%–65%) on a 13:11 h light/dark photo-cycle at NCRRT, Nasr City, Cairo, Egypt. They had free access to clean water and normal standard food. The reporting of this study conforms to ARRIVE 2.0 guidelines. 27
After adaptation for a week, rats were randomly divided into five groups (n = 6) as follows: Group I (control): The rats received the vehicle (3 mL distilled water/day orally), Group II (Carvacrol): The rats received carvacrol (75 mg/kg body weight/day orally), Group III (5 Gy γ-rays): The rats received the vehicle and then exposed to 5 Gy γ-rays after 30 min of the last dose, Group IV (Carvacrol + γ-rays): The rats received carvacrol doses and then exposed to 5 Gy γ-rays after 30 min of the last dose, and Group V (γ-rays + Carvacrol): The rats exposed to 5 Gy γ-rays and then received carvacrol doses for 8 weeks.
Gamma-Irradiation
The γ-irradiation was performed at the NCRRT, Nasr City, Cairo, Egypt, by a gamma cell-40, Canada. Animals were subjected to a single dose of whole-body 5 Gy at a dose rate of 0.38 Gy/minute during experimentation to induce testicular toxicity for the tested animals.28,29
Specimen Collection
All the animals fasted overnight after 3 days of the end of the experiment on day 59. Subsequently, they were sacrificed by cervical dislocation under anesthesia (87 mg ketamine/kg and 13 mg xylazine/kg of body weight). However, the effect of testicular toxicity at the biochemical and histopathological levels exhibited after 3 days of radiation.30,31 The blood samples were collected by heart puncture and kept in glass test tubes for clotting. Then, they were centrifuged at 6000 rpm for 3 min, and the obtained sera were harvested for biochemical analysis. Coda epididymis and testes organs were removed, washed with ice-cold saline solution, and then the right testes were weighed. Immediately, the testicular tissue sample fragments were collected, and the right testes were kept to investigate biochemical indices.
Testis Tissue Preparing
The testicular tissues (1:10 w/v) were homogenized in tris-HCI buffer (50 mM; pH 7.4) by Teflon homogenizer (MNW−302, Poland) for 3 min. The homogenates were then centrifuged at 3000 rpm for 10 min. The testes homogenates samples were frozen at −70 °C for oxidative stress investigations. 32
Evaluating Oxidative Stress and Enzymatic/Nonenzymatic Antioxidant Markers
In the testicular tissue samples, TAS, SDH, and 17β-HSD were estimated according to Erel, 33 Bergmeyer, 34 and Jarabak et al, 35 respectively. MDA 36 was examined by using an MDA assay kit (BioDiagnostics Co., Giza, Egypt; Cat. No. MD25-29). Additionally, GSH 37 was examined using commercial kits provided by BioDiagnostic, Giza, Egypt (Cat. No. GR25-11), following the manufacturer's method. Moreover, SOD 38 activity was assessed by SOD assay kit Biodiagnostic (Cat. No. SD 25-21). CAT 39 activity was examined using commercial kits provided by BioDiagnostic, Giza, Egypt (Cat. No. CA25-17), following the manufacturer's method.
Serum Testosterone Level in Blood
The testosterone level in the male rat serum was measured using rat testosterone ELISA kits (Catalog number CSB-E05100r) obtained from Cusabio Biotech Company (Houston, TX, USA) according to manufacturers’ instructions. 40
Analysis of Sperms
Right, cauda epididymis was put in a Petri dish with 10 mL of 0.1 M phosphate-buffered saline (PBS; pH 7.4) for sperm counting and motility assessment. Analysis of sperms was achieved using the Sahin et al 41 method. Sperm counting and sperm motility were performed using a hemocytometer under a conventional microscope. A cover slide was put on the hemocytometer slide. Subsequently, a drop of 10 μL of caudal epididymis sperm suspension was loaded under the cover slide for sperm counting or motility.
Statistical Analysis
Data are represented as mean ± standard deviation of the mean (SD). The normality of data distribution was assessed using the Shapiro-Wilk test. Statistical significance of differences between groups was determined by one-way analysis of variance (ANOVA), followed by Fisher's Protected Least Significant Difference (PLSD) post-hoc test. The level of statistical significance was set at P < .05. 42
Results
Novel Targets for Carvacrol
The 2D structure in SDF format was retrieved for the Pubchem platform. A total of 100 candidate targets of carvacrol were explored from SwissTargetPrediction that target different classes, including cytochrome P450, oxidoreductase, and kinase (Figure 1).

Overlapping Common Targets of Carvacrol
A total of 3664 radiation-related targets were identified from GeneCards databases, and 100 candidate targets for carvacrol were identified from SwissTargetPrediction. However, 56 intersection targets were obtained among these gene sets and were used for further analysis (Figure 2).

Functional Enrichment Analysis
GO terms explored that carvacrol target genes enriched in the oxidation-reduction and testosterone biosynthesis processes (Figure 3).
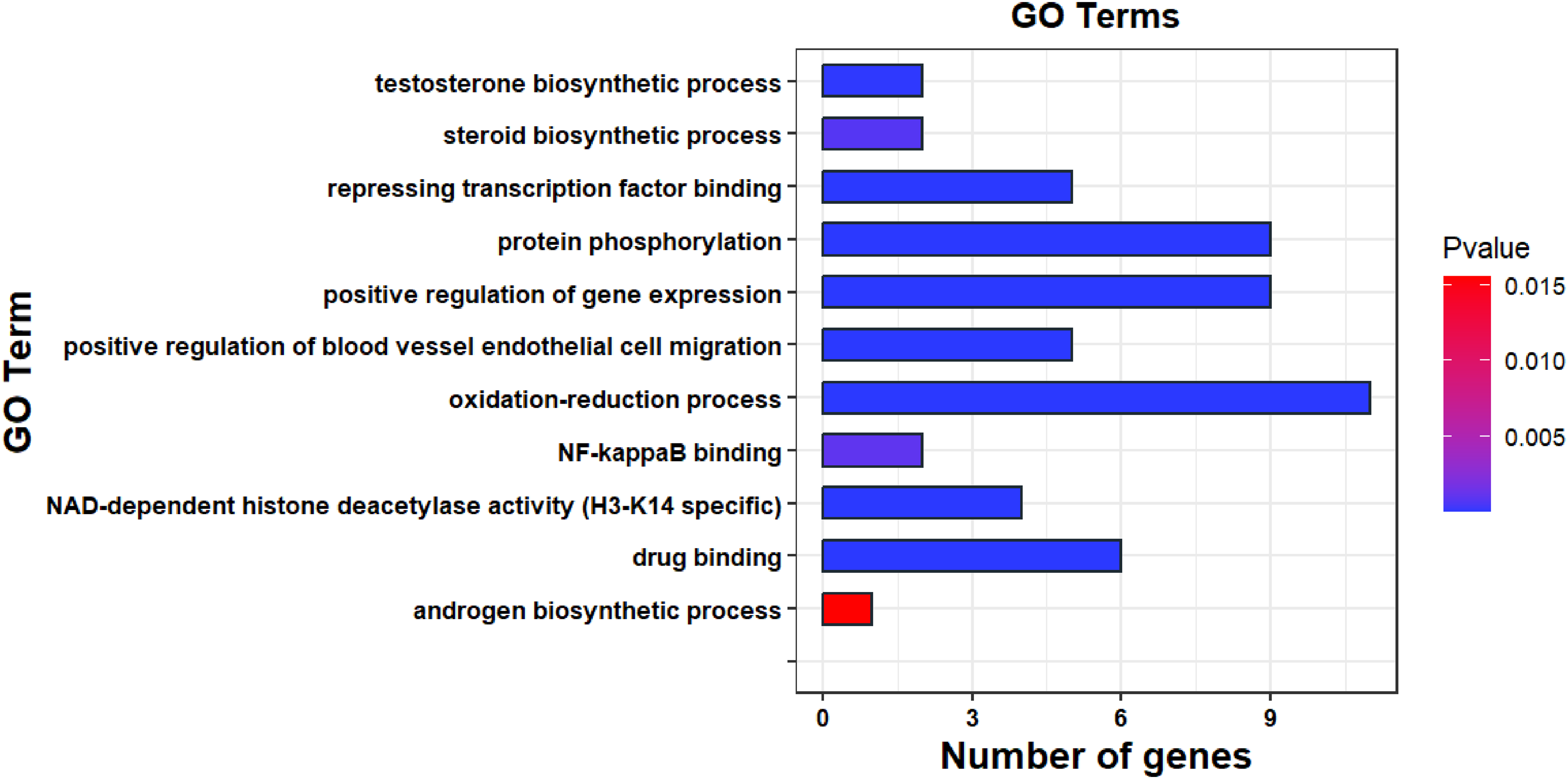
Moreover, the KEGG results showed that carvacrol target genes are enriched in different pathways, including metabolic pathways, endocrine resistance, inflammation, and hypoxia-inducible factor (HIF) signalling pathways (Figure 4). Thus, the effect of carvacrol in regulating oxidative and testosterone biosynthesis was studied to validate the dry data analysis.

The Impact of Carvacrol in Radiation-Induced Testicular Oxidative Stress
The current results explored that radiation evoked oxidative stress by significantly increasing the MDA level (P < .05) and significantly decreasing the antioxidant marker levels, including GSH (P < .05), TAS (P < .05), SOD (P < .05), and CAT (P < .05), in Group III compared to Group I (Figure 5). In addition, carvacrol (pre- and post-treatment) significantly decreased the oxidative damage in the brain tissues by decreasing MDA level and significantly increasing the antioxidant marker levels, including GSH (P < .05), TAS (P < .05), SOD (P < .05), and CAT (P < .05) in Groups IV and V compared to Group III (Figure 5).

Carvacrol Improved Radiation-Induced Testicular Oxidative Stress. Values Were Expressed as Means ± SD. Data are Significantly Different at P < .05. Statistical Analysis was Performed Using one-way ANOVA.
The Impact of Carvacrol in Radiation-Induced Testicular Damage
The current results explored that radiation significantly increased testicular damage by decreasing the activities of SDH (P < .001) and 17β-HSD (P < .001) and the testosterone levels (P < .001) in Group III compared to Group I (Figure 6). In addition, pre-treatment of carvacrol significantly decreased the testicular damage by increasing the activities of SDH (P < .001) and 17β-HSD (P < .001) and testosterone levels (P < .001) in Group IV compared to Group III (Figure 6). Similarly, carvacrol (post-treatment) significantly decreased the testicular damage by increasing the activities of SDH (P < .001) and 17β-HSD (P < .001) and testosterone levels (P = .0002) in Groups V compared to Group III (Figure 6).

Carvacrol improved radiation-induced testicular damage. Values were expressed as Means ± SD. Data are significantly different at P < .05. Statistical analysis was performed using one-way ANOVA.
The Impact of Carvacrol in Radiation-Induced Infertility
The current results explored that radiation significantly increased testicular
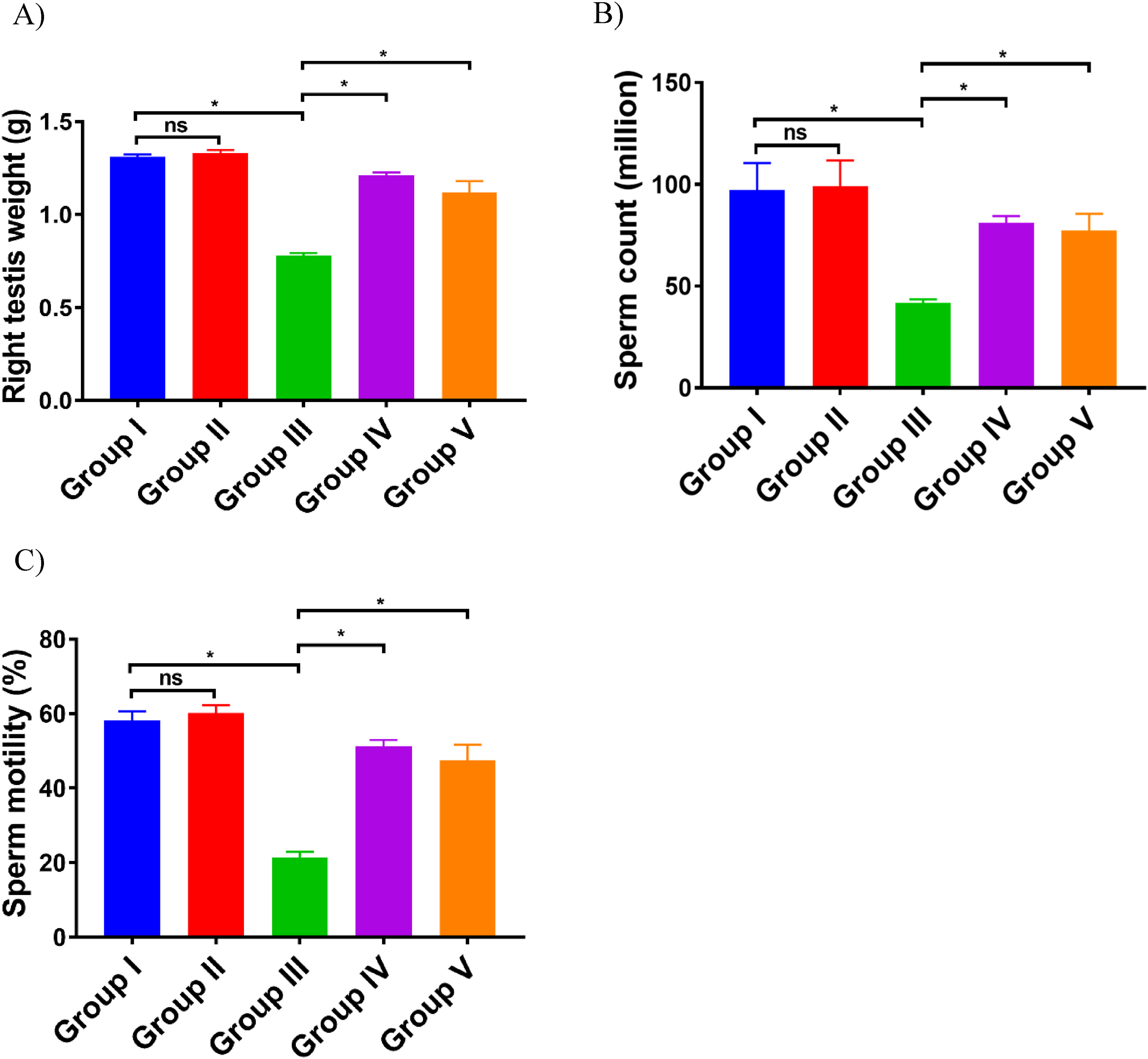
Carvacrol Improved Radiation-Induced Infertility. Values Were Expressed as Means ± SD. Data are Significantly Different at P < .05. Statistical Analysis was Performed Using one-way ANOVA.
Discussion
The mechanism of carvacrol in the treatment of radiation-induced testicular damage is not fully understood. Thus, network pharmacology and bioinformatics have superiority in the systematic elucidation of the mechanism of carvacrol at the molecular level and the representation of interactions between carvacrol, potential targets, and various pathways. However, the current bioinformatic analysis explored that carvacrol is crucial in regulating oxidation-reduction and testosterone biosynthesis processes. Additionally, experimental studies explored that carvacrol improved the testis and sperm characteristics and oxidative stress induced by gamma-irradiation. In particular, carvacrol treatment markedly decreased radiation-induced oxidative damage in the brain tissues via markedly decreasing MDA levels and considerably increasing the antioxidant marker levels (GSH, TAS, SOD, and CAT). In addition, carvacrol significantly decreased radiation-induced testicular damage by substantially increasing the activities of SDH and 17β-HSD and testosterone levels. Moreover, carvacrol improved radiation-induced testicular infertility by increasing the right testis's weight, sperm count, and sperm motility percentage.
Ionizing radiation, directly and indirectly, causes biological effects. It has been shown that γ-irradiation could damage biological tissues following the generation of ROS or free radicals. In the current study, rats exposed to a single radiation dose showed a significant increase in the MDA levels of their brain tissues. Furthermore, our findings showed that irradiation of the rats reduced CAT and SOD activities and GSH and TAS levels in their brain issues, probably due to the oxidative inactivation of the antioxidant enzymes through increased ROS production. It was recognized that mammal testicles are very sensitive to oxidative stress. 43 In particular, our findings agree with previous findings that showed radiation evoked reproductive damage in male rats via increasing oxidative stress.4,43-46
However, carvacrol improved oxidative stress due to its antioxidant activities, 47 which is related to the hydroxyl group “OH−” covalently linked to the aromatic ring. 48 The oxidant-antioxidant stability is delivered by different types of oxidants that arise in the animals, and appropriate antioxidant medications compensate for these oxidants. 49 Furthermore, carvacrol has protective and therapeutic effectiveness due to its anti-inflammatory and anti-apoptotic properties.50,51 Determining TAS is a beneficial, fast, and undoubtedly routine technique to estimate the complex oxidative exploration of a disease. 52 In the present study, the TAS data revealed that carvacrol protects/treats testis from γ-rays-induced oxidative stress. Carvacrol was found to alleviate testicular tissue damage in diabetic rats, probably via its antioxidant effects, 51 and improves TAS in sodium arsenite-induced oxidative stress in rats. 53 Moreover, carvacrol is a novel natural antioxidant extract. 54
In the present study, we found that acute exposure to γ-rays caused lessening in both SDH and 17β-HSD, possibly related to the decline in testosterone level.45,55 However, the SDH and 17β-HSD activities were significantly improved by carvacrol in the current study. Carvacrol has a wide assortment of pharmacological activity comprising antioxidant and anti-inflammatory properties, and it protects germ cells against oxidative stress.26,50 In addition, the bioinformatic results in the current study explored that carvacrol regulates the 17β-HSD3 activity, which governs testosterone biosynthesis.
Furthermore, ionizing radiations decreased sperm production, motility, and weight of testes in mice and rats. 56 However, the diagnostic indicators of radiation-induced testicular toxicity were reductions in testis’ weight and size.57,58 In addition, increased free radicals following γ-rays-induced testicular toxicity led to oxidative stress and deterioration of the antioxidant defense system. 29 Oxidative stress is defined as one of several mediators of male infertility, which causes sperm dysfunction. 59 Carvacrol can effectively prevent damage to the testis and epididymis sperm characteristics.50,51 Carvacrol has been approved as a potential stimulator of testosterone construction. 60 Moreover, it was revealed that carvacrol administration significantly enhanced testes weights, sperm count and motility, and serum testosterone, enhanced the testicular antioxidant enzymes activity, and attenuated the degenerative alterations and apoptosis in testes of intoxicated rats. 61 Additionally, previous studies showed that carvacrol exerted a protective influence against testicular toxicity in adult diabetic rats where carvacrol is intermediated by its antioxidant activity and stimulating testosterone production. 62
Limitation
While the present study offers insights into the potential protective effects of carvacrol against γ-ray-induced testicular lesions in rats, several limitations warrant consideration. Firstly, the precise mechanisms underpinning these protective effects remain to be fully elucidated. Further research is necessary to explore these mechanisms in other animal models, particularly those exhibiting more remarkable physiological similarity to humans and, ultimately, human subjects. Secondly, the study focused on adult rats, and thus, the protective efficacy of carvacrol across different developmental stages of male reproductive organs—from juvenile to mature—remains unknown. Consequently, future studies should investigate the impact of carvacrol on testicular toxicity throughout the lifespan.
Conclusion
The combined bioinformatic and experimental findings suggest that carvacrol demonstrates promising radioprotective and radiotherapeutic potential against γ-ray-induced testicular damage in rats. However, additional studies are imperative to translate these findings into clinical applications for radiotherapy patients, particularly in the context of fertility preservation. These future investigations should employ animal models that more closely resemble human physiology to validate and expand upon the current observations.
Footnotes
Acknowledgments
We thank our colleagues, the Radiation Health Research Department for providing laboratory conveniences, and members of the National Center for Radiation Research and Technology (NCRRT), Egyptian Atomic Energy Authority for providing the necessary irradiation facilities. This work was performed in the NCRRT.
Ethical Considerations
The research experiment protocol was officially approved no. (F/ 22A/24) by the Research Ethics Committee (REC- NCRRT).
Informed Consent/ Patient Consent
There are no human subjects in this article, and informed consent is not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
The research experiment protocol was officially approved no. (F/ 22A/24) by the Research Ethics Committee (REC- NCRRT).
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Significance Statement
Radiation induced testicular damage. Network pharmacology and experimental validation exhibited that carvacrol reduces testicular toxicity in γ-irradiated rats via its antioxidant activity. However, it could benefit patients receiving radiotherapy.
