Abstract
Background
Hepatocellular carcinoma (HCC) has become one of the most common malignancies worldwide. Eriocalyxin B (EriB) is a natural, ent-kaurane diterpenoid isolated from Isodon eriocalyx that has been reported to exhibit remarkable antitumor and immunoregulatory activities.
Objectives
The aim of this study is to explore the role of autophagy in EriB-induced cell death in HCC cell lines.
Methods
The Cell Counting Kit-8 (CCK-8) was used to assess the EriB effects on HCC cell proliferation. Flow cytometry and confocal microscopy were performed to detect cell apoptosis and autophagic flux. Western blot and immunohistochemistry analyses were used to evaluate the expressions of related proteins. The antitumor effects were observed in a xenograft tumor model of HCCLM3.
Results
EriB significantly suppresses HCC cell proliferation while concurrently inducing apoptosis. Mechanistically, EriB triggers dose-dependent accumulation of autophagy markers LC3B-II and p62, accompanied by increased autophagosome formation. Paradoxically, EriB impedes late-stage autophagy by reducing acidified autophagic vesicles (RFP + GFP−) and attenuating autophagic flux, suggesting blockade of autophagosome-lysosome fusion. This autophagy-inhibitory effect is mediated through suppression of the mTOR/ULK1 signaling axis. In vivo, EriB markedly inhibits HCC tumor growth. Notably, EriB overcomes sorafenib resistance by antagonizing therapy-induced protective autophagy. Combination therapy with sorafenib synergistically enhances anti-HCC efficacy, as evidenced by significantly reduced tumor burden and weight compared to monotherapy regimens. The chemosensitizing effect of EriB is attributed to its ability to inhibit mTOR/ULK1-driven autophagy, thereby disrupting cancer cell survival mechanisms.
Conclusion
EriB offers a novel therapeutic strategy for natural product-based combination targeted therapy, demonstrating promising potential to overcome sorafenib resistance in hepatocellular carcinoma HCC.
Significance Statements
EriB suppressed cell viability and induced apoptosis in HCC cells.
EriB induced autophagy initiation by increasing autophagosome formation and inhibited autophagic flux by decreasing autolysosome maturation.
EriB inhibited tumor growth and autophagy via mTOR/ULK1 signaling pathway inactivation. This resulted in enhanced sensitivity of HCC cells to sorafenib in vitro and in vivo.
Introduction
Hepatocellular carcinoma (HCC) is the sixth most diagnosed type of cancer worldwide and the third leading cause of cancer-related mortality, according to Global Cancer Statistics 2020.1,2 Despite improvements in screening and therapy, the HCC incidence and mortality rate of HCC remain high. 3 Surgical resection is the primary treatment for patients with early-stage HCC.4,5 HCC is noted for its aggressive growth and late onset of symptoms, and a majority of HCC patients are diagnosed with advanced-stages and are not candidates for effective surgical or locoregional treatments. 6 Chemotherapy and radiation are ineffective in advanced HCC.2,7,8 Sorafenib is a dual-target inhibitor of serine-threonine kinases Raf and tyrosine kinases VEGFR/PDGFR, and it is the first FDA-approved targeted drug for patients with advanced unresectable HCC.9,10 However, sorafenib acquired resistance has become an obstacle to increase the overall survival benefits in HCC patients. The median survival for advanced HCC is approximately 9 months, and the 5-year overall survival rate is only 10%. 11 Therefore, the development of new treatment options is critical.
Eriocalyxin B (EriB) is a natural, ent-kaurane diterpenoid isolated from Isodon eriocalyx. EriB exhibits antitumor activity in several tumor types. EriB has been shown to induce apoptosis of lymphoma cells, colorectal cancer cells, and pancreatic cancer cells, in addition to the induction of angiogenesis and metastasis in breast cancer.12–15 It has been proposed that EriB induces autophagy in breast cancer cells. 13 As such, it has become clear that autophagy modulation is a promising cancer therapy target. Autophagy is a degradative and recycling process that is activated in HCC cells. Understanding the autophagy role in HCC is crucial for the development of autophagy inhibitors as pharmacological agents to target autophagy in HCC. These findings imply that autophagy modulation could be a rational strategy to enhance sorafenib sensitivity in HCC treatment.16,17 However, the specific underlying mechanisms of EriB in anti-HCC activity remain inconclusive. An understanding of the rational basis of the autophagy mechanism and the development of new strategies of autophagy regulation may provide a new approach for effective HCC treatment. In this study, we examine the effects of EriB on HCC cell death, and autophagy along with the mechanisms that underlie the synergistic anti-HCC effect of EriB combined with sorafenib both in vitro and in vivo.
Materials and Methods
Cell Culture, Reagents, and Antibodies
The HCC cell lines HCCLM3, HepG2 and Huh 7 were obtained from the Cell Bank of Type Culture Collection of the Chinese Academy of Sciences, and they were authenticated using short tandem repeat profiling. The HCCLM3 and Huh 7 cells were cultured in Dulbecco's Modified Eagle Medium (DMEM), whereas the HepG2 cells were cultured in minimum essential medium (MEM) supplemented with 10% fetal bovine serum (FBS) (CellorLab, Epizyme Co., Ltd.) in a 5% CO2 incubator at 37 °C.
EriB with a purity ≥98.5% was obtained from the BioBioPha Co., Ltd It was dissolved in dimethyl sulfoxide (Sigma-Aldrich, Merck KGaA).
The primary antibodies used in this study were caspase-3 (cat. no. ER30804), LC3B (cat. no. ET1701-65), ULK1 (cat. no. ET1704-63), p62 (cat. no. HA721171), mTOR (cat. no. HA500126), and Phospho-mTOR (S2448) (cat. no. HA600094). These were purchased from HuaBio, Inc. Phospho-AMPKα (Thr172) (cat. no. 50081), Phospho-ULK1 (Ser757) (cat. no. 14202), and cleaved caspase-3 (cat. no. 9661) were purchased from Cell Signaling Technology. PARP (cat. no. 381835) and β-tubulin (cat. no. R23622) were purchased from ZenBio Inc. (Chengdu, China). Ki-67 was obtained from the ZSGB Biotech Co., Ltd GAPDH (cat. no. KC-564) was obtained from the Kangchen Biotech Co., Ltd The β-Actin (cat. no. A2066) antibody was purchased from Sigma-Aldrich, Merck KGaA.
Cell Viability Assay
The human HCC cell lines HCCLM3, HepG2, and Huh 7 (3 × 103 cells/well) were seeded in 96-well plates and incubated overnight for cell attachment. The HCC cells were treated with the indicated EriB concentrations (0-4 µM) for 24 h to evaluate the cytotoxic effects of EriB. A total of 10 µL of the Cell Counting Kit-8(CCK-8) (Vazyme Biotech Co., Ltd) reagent was added to each well after incubation. This was followed by incubation at 37 °C for 1.5 h. The absorbance was then measured at 450 nm using a microplate reader (BioTek Instruments Inc.).
Cell Apoptosis Analysis
Cell apoptosis detection was performed using the Annexin V–fluorescein isothiocyanate (FITC)/ Propidium Iodide (PI) apoptosis detection kit (Vazyme Biotech Co., Ltd.). HCC cells (5 × 105 cells/well) were seeded in six-well plates and incubated with different concentrations of EriB (0, 1, 2, and 4 μM) for 24 h. Cells were collected and washed with ice-cold phosphate-buffered saline (PBS) and then stained with 5 µL of Annexin V-FITC and PI solution for 15 min while on ice. The stained cells were diluted with PBS and analyzed using a FACSCalibur flow cytometer (BD Biosciences). The procedure was performed according to a previous study by Xiang et al. 18
Stably Expressed GFP-LC3B
The lentiviral vectors expressing GFP-LC3B were purchased from Genechem (Shanghai, China). Huh7 cells stably expressing GFP-LC3B were selected using puromycin (2 μg/mL). After treatment with EriB for 24 h or Baf A1 (bafilomycin A1) for 4 h, the cells were fixed, and autophagic puncta formation was examined using Olympus FV1000 confocal microscope (Olympus).
Autophagic Flux Analysis
The autophagic flux assay was performed as previously described. 19 The HepG2 and Huh 7 cells (2 × 105 cells/well) were seeded in 6-well plates and transfected with lentivirus RFP-GFP-LC3B for 72 h and selected with puromycin to generate stable cell lines. The cells were treated with various concentrations of EriB (0, 1, 2, and 4 μM) for 24 h, and they were then collected and measured using a FACSCalibur flow cytometer (BD Biosciences). For autophagic puncta, Huh 7 cells expressing RFP-GFP-LC3B were seeded in glass dishes and treated according to the above conditions, fixed in 4% paraformaldehyde for 20 min and examined by Olympus FV1000 confocal microscope (Olympus).
Western Blot Analysis
Cells were lysed in a radioimmunoprecipitation assay (RIPA) (Beyotime Institute of Biotechnology Co., Ltd) buffer and supplemented with a cocktail of protease inhibitors. The total protein was then separated using sodium dodecyl sulfate–polyacrylamide gel electrophoresis and transferred to a nitrocellulose membrane. The membranes were incubated with primary antibodies after being blocked with 5% non-fat milk for 1 h (Sigma-Aldrich, Merck KGaA). They were incubated overnight at 4 °C and then with the secondary antibody (Sigma-Aldrich, Merck KGaA) for 1 h at room temperature. Signals were visualized by luminescence detection using a chemiluminescent substrate (Vazyme Biotech Co., Ltd).
Immunohistochemistry
Immunohistochemistry was performed to determine the expression of Ki67, p62, and cleaved caspase 3 expressions in the tumor sections.20,21 The primary antibodies Ki67, p62, and cleaved caspase 3, were used and then analyzed according to our previous study. 22
Animal Experiments
All animal procedures were conducted according to the National Institutes of Health (NIH) guidelines. The reporting of this study conforms to ARRIVE 2.0 guidelines. 23 Experiments were approved by and performed on July 04, 2024, in accordance with the Ethics Committee of the North Sichuan Medical College (Approval no. 2025019).
Male, BALB/C nude mice aged 6–8 weeks and weighing 18–22 g were purchased from the Huafukang Biotechnology Co., LTD (Beijing, China). The mice were maintained in a pathogen-free environment at 22 °C–26 °C on a 12-h light–dark cycle with sufficient food and water. The HCCLM3 cells (5 × 106 cells in 150 μL PBS) were implanted into the right flanks of the nude mice after 1 week of adaptation,. Once the tumor sizes reached approximately 60 mm3, the tumor-bearing mice with similar tumor volumes were randomly grouped (n = 5 per group). Groups were assigned to intraperitoneally receive the vehicle (2% dimethyl sulfoxide (DMSO)) or EriB (2.5 mg·kg−1·day−1) for 10 days prior to their sacrifice by cervical dislocation. Tumor volumes were measured using a caliper and calculated using the following equation: Volume (mm3) = width2 × length × 0.5. Tissue samples were collected and processed for further analysis.
For the tumor models with EriB with or without sorafenib treatment, 5 × 106 HCCLM3 cells in 200 uL PBS were subcutaneously injected into the flanks of the BALB/c mice (Huafukang). The tumor-bearing mice were randomly divided once the tumor sizes reached approximately 60 mm3 into the following groups (n = 6 per group): (1) control; (2) EriB; (3) sorafenib, or (4) sorafenib plus EriB. The sorafenib was administrated orally at a dosage of 30 mg/kg every day for 2 weeks. The EriB was administered via intraperitoneal injection at 3 mg/kg every 2 days for 2 weeks. The tumor volumes were measured using a caliper and calculated using the following equation: Volume (mm3) = width2 × length × 0.5. The tumor weights were recorded at the end of the experiment.
Statistical Analysis
All of the presented results are representative of at least three independent experiments. A one-way analysis of variance (ANOVA) was used to analyze multiple comparisons, and a student's t test was used to compare the two groups. Data are expressed as the means ± standard errors of the indicated number of experiments. P-values < .05 were considered statistically significant. Statistical analyses were conducted using GraphPad Prism 8.0 (GraphPad Software, Inc.).
Results
EriB Suppressed Cell Viability and Induced Apoptosis in HCC Cells
The EriB chemical structure is illustrated in Figure 1A. Three HCC cell lines (HCCLM3, Huh 7, and HepG2) were treated with various concentrations of EriB (0-16 µM) for 24 h to evaluate the viability of the HCC cells in response to EriB. The cell viability was then detected using a Cell Counting Kit-8 assay. The results demonstrated that the EriB treatment significantly inhibited the viability of the HCCLM3 (IC50 of 5.306 µM), Huh 7 (IC50 of 1.803 µM), and HepG2 (IC50 of 1.444 µM) cells after 24 h of treatment. These results are presented in Figure 1B. The IC50 values were calculated using GraphPad Prism 8.0. The HepG2 and Huh 7 cell lines were selected for the following experiments. In addition, the Annexin V-FITC/PI apoptosis assay was used to determine the EriB effects on cell death. The EriB treatment significantly increased the apoptosis of HepG2 and Huh 7 cells (Figure 1C and D) in comparison with the control group. In addition, the protein expression levels related to apoptosis were examined using western blot analyses. The cleaved PARP and cleaved caspase-3 expression levels exhibited enhanced expressions in a concentration-dependent manner when using the EriB treatment (Figure 1E). These results demonstrated that EriB inhibited HCC cell proliferation.
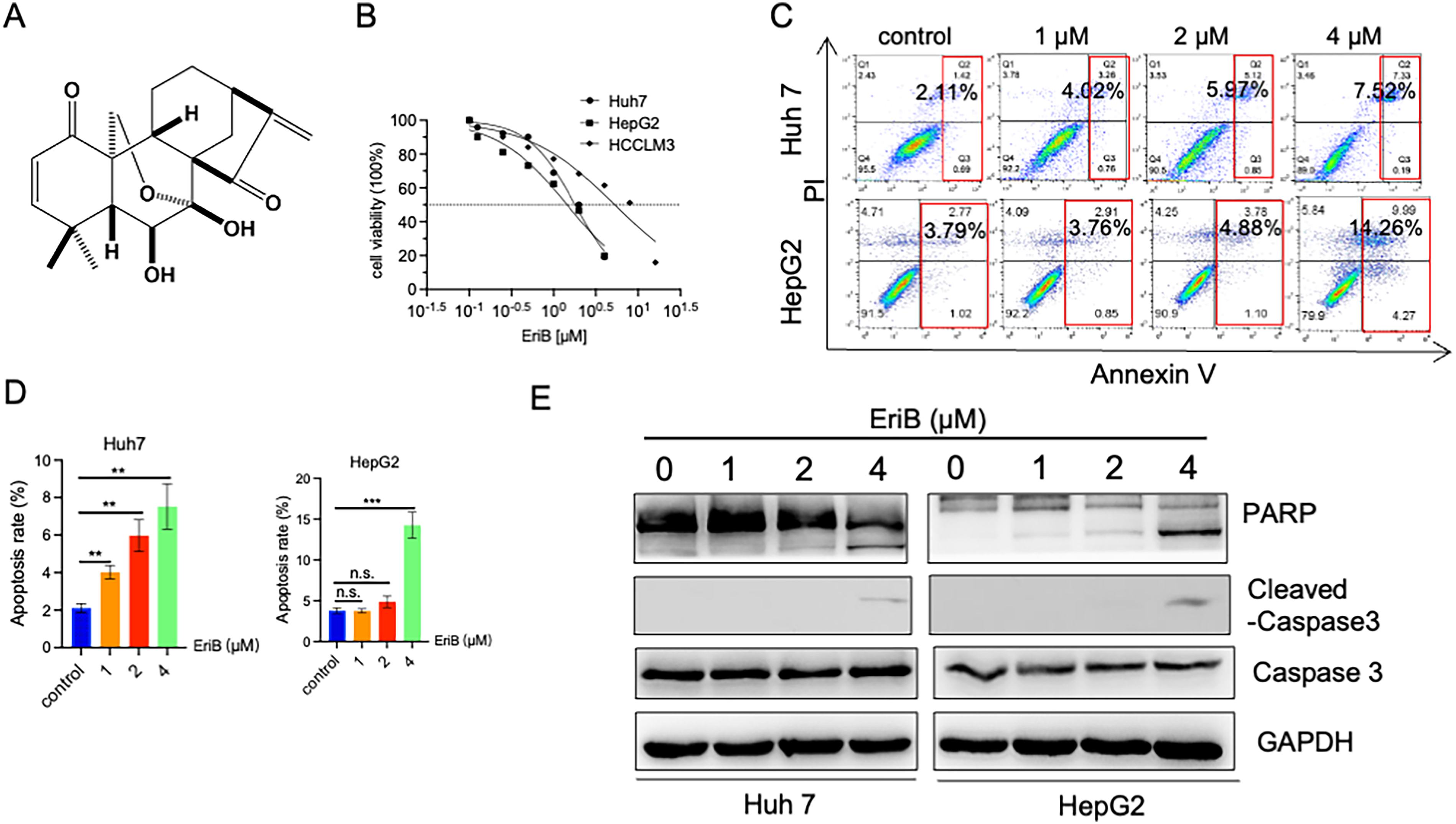
Erib Suppressed Cell Viability and Induced Apoptosis in HCC Cells. (A) Structure of EriB. (B) A CCK-8 Assay was used to Detect the Viability of the HCCLM3, HepG2, and Huh 7 Cells After Treatment with Various Concentrations of EriB (0-16 µM) for 24 h. (C, D) The HepG2 and Huh 7 Cells were Treated with the Indicated Concentrations of EriB for 24 h, and the Apoptotic Rates were Detected Using Annexin V-FITC/PI Staining and Flow Cytometry. ***P < .001 Compared with the Control Group. (E) Western Blot Analyses of PARP, Caspase-3, and Cleaved Caspase-3 After Treatment with 0–4 µM EriB for 24 h. GAPDH was used as an Internal Standard. Data are Means ± S.D. and are Representative of Three Independent Experiments. n.s., Not Significant. ** P < .01 and *** P < .001 Compared with the Control Group. HCC, HepatoCellular Carcinoma; EriB, Eriocalyxin B; CCK-8, Cell Counting Kit-8.
EriB Induced Autophagy Initiation and Inhibited Autophagic Flux in HCC Cells
Autophagy plays an important role in cancer by promoting cancer cell growth or death. The EriB effect on autophagy was determined by the transformation of LC3B-I to LC3B-II and the appearance of autophagic puncta in HCC cells with GFP-LC3B stable expression. Figure 2A shows that EriB enhanced the accumulation of LC3B-II and p62 in both the HepG2 and Huh 7 cells in a dose-dependent manner. Increased autophagy initiation or late autophagy inhibition could lead to LC3B accumulation. Autophagosome was accumulated by Baf A1, an autophagosome–lysosome fusion inhibitor that causes autophagosome accumulation and impairs the autophagic degradation of LC3B. The EriB and Baf A1 combined treatment synergistically induced an increased LC3B-II accumulation. This implied that EriB induced autophagy initiation (Figure 2B). A GFP-tagged LC3B construct was used to confirm this observation. 24 Figure 2C shows that the number of LC3B puncta was markedly increased in the EriB-treated cells, and co-treatment with Baf A1 increased the accumulation of autophagic puncta. These results suggested that autophagosome formation increased. According to these data, Huh 7 cells that stably expressed RFP-GFP-LC3B were used, in which the autophagosomes were labeled yellow and the autolysosomes were labeled red because of the quenching of GFP fluorescence in the acidic environment of lysosomes. Baf A1 was used as the negative control, and rapamycin was used as the positive control. The results indicated that the EriB treatment decreased autolysosome maturation (RFP + GFP−) (Figure 3A). the flow cytometric analyses revealed that the number of LC3B puncta and the percentages of the RFP + GFP− signal (autolysosomes) were markedly decreased in the EriB-treated cells (Figure 3B). These results indicated that EriB inhibited autophagic flux in HCC cells.
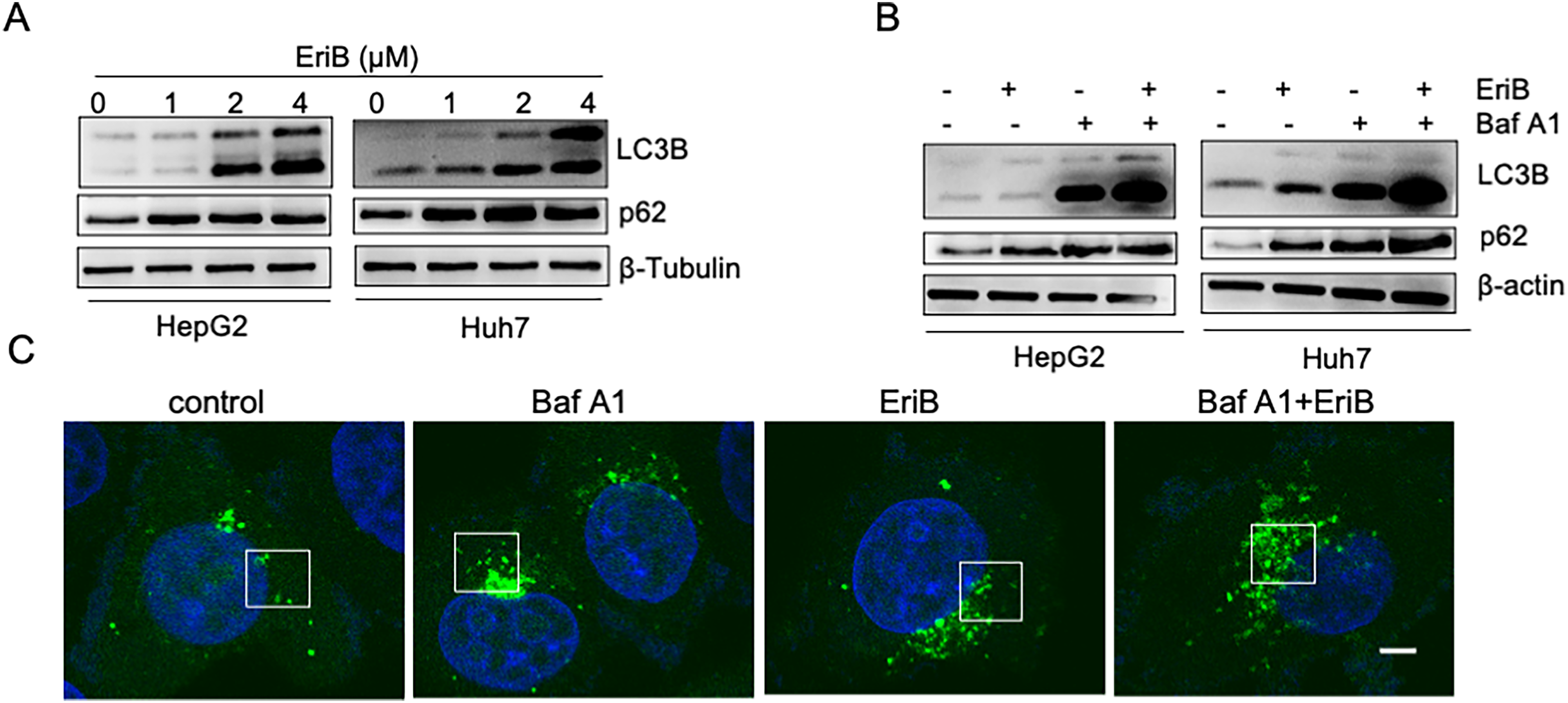
Erib Induced Autophagy Initiation in HCC Cells. (A) HepG2 and Huh 7 Cells were Treated with the Indicated Concentrations of EriB (0, 1, 2, and 4 µM) for 24 h. Protein Expression of LC3B and p62 Protein Expression Levels were Detected using Western Blot Analyses. β-Tubulin was used as the Loading Control. (B) The HepG2 and Huh 7 Cells were Treated with EriB (2 µM) for 20 h in Combination with or Without Baf A1 (100 nM) for 4 h. The LC3B and p62 Protein Expression Levels were Detected Using Western Blot Analyses. β-Actin was used as an Internal Control. (C) Representative Images of the Huh 7 Cells Stably Transfected with GFP-LC3B for 72 h and Treated as in (B). Scale Bar, 10 μm. Data are Representative of Three Independent Experiments. EriB, Eriocalyxin B; HCC, HepatoCellular Carcinoma.

Erib Inhibited Autophagic Flux in HCC Cells. (A) Representative Images of the Huh 7 Cells that Expressed Tandem RFP-GFP-tagged LC3B were Treated with EriB (2 µM) for 24 h, Baf A1 (100 nM) for 4 h, or Rapamycin (500 nM) for 3 h. Scale Bar, 10 μm. (B) The HepG2 and Huh 7 Cells that Stably Expressed RFP-sturbGFP-LC3B were Incubated with EriB for 24 h. The Autophagic Index was Analyzed Using Flow Cytometry. The Autophagic Index is the Ratio of the Areas of the RFP + GFP− Puncta (Autolysosomes) to the Areas of the RFP + GFP + Puncta (Autophagosomes) (n = 3). Data are Means ± S.D. and are Representative of Three Independent Experiments. **P < .01 and ***P < .001 Compared with the Control (One-Way ANOVA). EriB, Eriocalyxin B; HCC, HepatoCellular Carcinoma; ANOVA, Analysis of Variance.
EriB Inhibited the mTOR/ULK1 Signaling Pathway in HCC
The AMPK, mTOR, and the downstream target levels were detected using western blot analyses to investigate the underlying mechanisms of the inhibitory effects of EriB on the autophagy of HCC cells and because AMPK and mTOR are the major regulators of autophagy in HCC. The EriB treatment (0, 1, 2, and 4 μM) for 24 h decreased the p-AMPK, p-mTOR, and p-ULK1 expression in a concentration-dependent manner (Figure 4A). MHY1458 (10 μM), an mTOR activator, was used to treat the HepG2 cells with or without EriB to further determine the role of p-mTOR in the EriB-induced inhibition of autophagy. Figure 4B shows that the MHY1458 and EriB combination compromised the activated effect of MHY1458 on the phosphorylation of mTOR, and MHY1458 enhanced EriB-induced p62 accumulation. In addition, the mTOR inhibitor Torin 1 with EriB combination synergistically suppressed the phosphorylation of mTOR, and Torin 1 compromised the EriB-induced p62 accumulation (Figure 4C). These results indicated that EriB inhibited the autophagic flux of HCC cells via the mTOR/ULK1 signaling pathway.

Erib Regulated the mTOR/ULK1 Signaling Pathway in HCC Cells. (A) AMPK Expression and the Downstream Targets (mTOR and ULK1) were Detected Using Western Blot Analyses. The HepG2 and Huh 7 Cells were Treated with the Indicated Concentrations of EriB (0, 1, 2, and 4 µM) for 24 h. GAPDH was used as an Internal Control. (B) The Immunoblot Analysis of p62 in HepG2 Cells Incubated with EriB (2 μM) in Combination with or Without MHY1485 (10 μM) for 24 h. β-Actin was used as an Internal Control. (C) The Immunoblot Analysis of p62 in HepG2 Cells Incubated with EriB (2 μM) for 21 h in Combination with or without 100 nM Torin 1 for 3 h. β-Actin was used as an Internal Control. Data are Representative of Three Independent Experiments. EriB, Eriocalyxin B.
EriB Inhibited Tumor Growth in Vivo
The HCCLM3 subcutaneous xenograft tumor models were established in vivo to further investigate the anticancer effect of EriB on HCC. Figure 5A–C show that the EriB treatment significantly inhibited tumor growth and reduced the volume of HCCLM3 subcutaneous tumor xenografts compared with those in the control group. This inhibitory tumor growth effect was significantly enhanced by day 6 of treatment (<0.05). The western blot analysis results revealed that the EriB treatment decreased the p-AMPK, p-mTOR, and p-ULK1 expressions (Figure 5C). In addition, the immunohistochemical assays revealed that the EriB treatment markedly decreased the Ki-67 expression in the tumor tissues (Figure 5D). These results demonstrated that EriB inhibited tumor growth by inducing apoptosis and inhibited autophagy by preventing the activation of the mTOR/ULK1 signaling pathway in vivo.

Erib Effects on Tumor Growth and Suppression of Autophagy in HCCLM3 Tumor Xenografts. The HCC Mouse Model was Established by Creating Tumor Xenografts in Mice Followed by Treatment with the Vehicle (Control) or EriB (2 mg/kg) for 10 days. (A) Images of the Tumor Samples from Each Group. (B) Tumor Volumes were Measured and Calculated Every Day. (C) Western Blot Analyses of the Expressions of p-mTOR, mTOR, and p62 in the Tumor Xenografts. (D) Immunohistochemical Analysis of Ki-67 in the Tumor Tissues. Scale Bar, 100 µm. Data are Representative of Three Independent Experiments. EriB, Eriocalyxin B; HCC, HepatoCellular Carcinoma.
EriB Induced Sensitization of HCC Cells to Sorafenib by Inhibiting Autophagy
Sorafenib is the first-line chemotherapeutic drugs for HCC treatment. Autophagy regulation has been reported to restore drug sensitivity.25,26 We explored the EriB effect on autophagy and its contribution to sorafenib resistance by using sorafenib in combination with EriB to treat HCC cells. The combination group exhibited significant inhibition on HCC cell proliferation compared with sorafenib alone (Figure 6A). We also examined the p62 protein expression levels to clarify the role of EriB-mediated autophagy in sorafenib-induced apoptosis. The wertern blot results showed that EriB restored the sorafenib-induced decrease in p62. We then examined the apoptosis-related proteins expressions. The results showed that the expression of cleaved-caspase 3 and PARP expressions were significantly upregulated in Huh7 cells in the combination group compared with those of the single-drug treatments (Figure 6B).
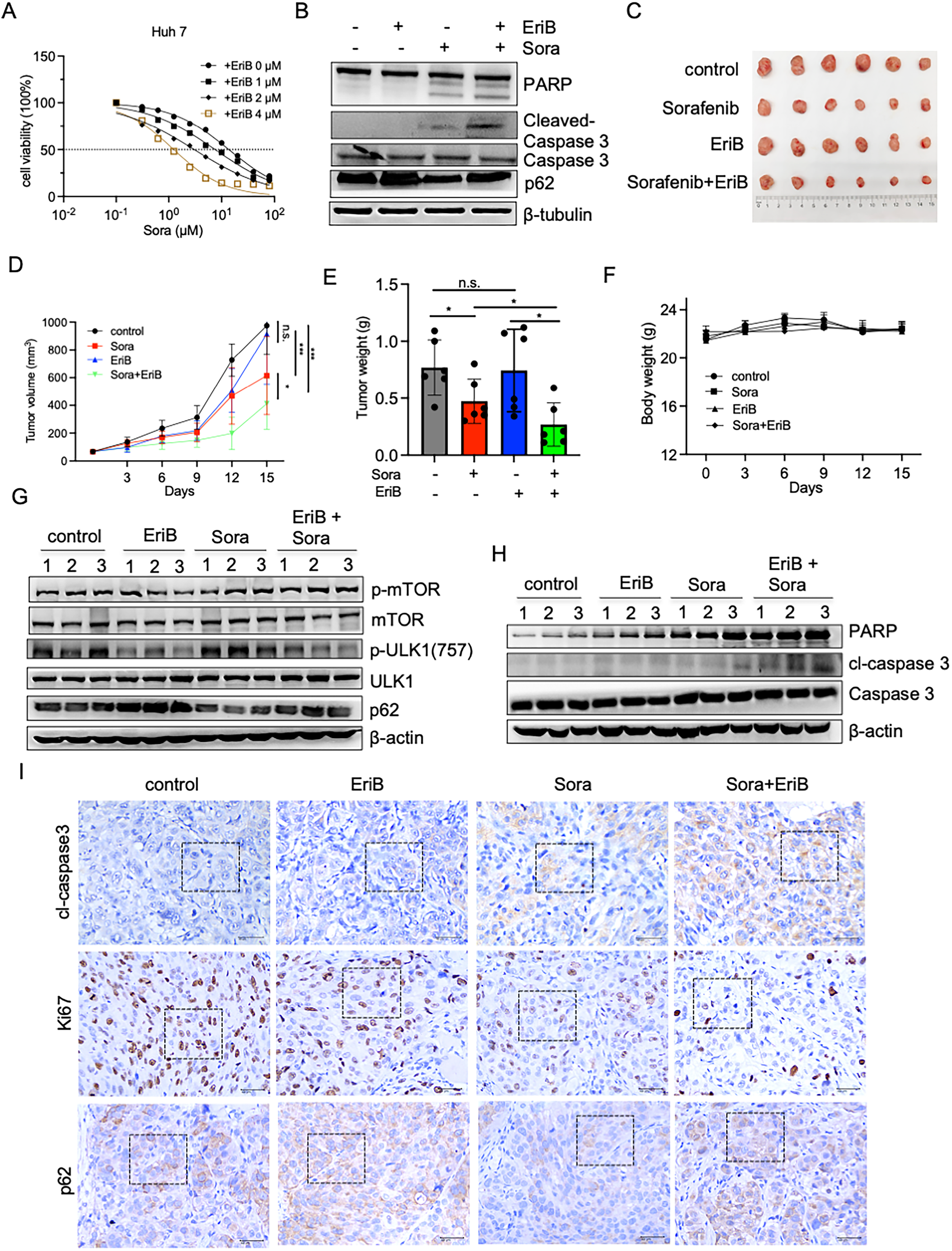
The EriB and Sorafenib Combination Inhibited the Growth of HCC Cells Both in vitro and in vivo. (A) Cell Viability of Huh 7 Cells Following Treatment with Sorafenib in the Absence or Presence of EriB for 24 h. (B) Western Blot Analyses of the PARP, Cleaved Caspase 3, Caspase 3, and p62 Expressions in Huh 7 Cells Following Treatment with EriB (2 µM) in the Presence or Absence of Sorafenib (2.5 µM) for 24 h. Nude Mice Bearing HCCLM3 Cells were Treated with the Control, EriB (3 mg/kg), Sorafenib (30 mg/kg), and EriB + Sorafenib Every 2 Days for 2 Weeks. (C–E) The HCCLM3 Tumor Images (C), Tumor Volumes (D), Tumor Weights (E), and Mouse Weights (F) were Measured for 2 Weeks. (G, H) Immunoblotting Detected the p-mTOR, mTOR, p-ULK1(757), ULK1, p62, PARP, Cleaved Caspase 3 and Caspase 3 Levels in the Control, EriB, Sorafenib, EriB + Sorafenib Groups. (I) Representative Immunohistochemistry (IHC) Staining Results for Ki-67, p62, and Cleaved Caspase-3 in the Control, EriB, Sorafenib, EriB + Sorafenib-Treated Nude Mice. Scale Bar, 40 µm. Data are Means ± S.D. and are Representative of Three Independent Experiments. n.s. No Significance, *P < .05, and ***P < .001 Compared with the Control Group (One-Way ANOVA).
EriB Enhanced Sorafenib Efficacy in Vivo
We established a subcutaneous xenograft model using HCCLM3 cells to determine the anti-tumor activity of the drug combination in vivo. Once the tumor size reached 60 mm3, the xenografted mice were then randomly assigned to four groups that received sorafenib (30 mg/kg) and/or EriB (3 mg/kg) for 14 days. The sorafenib and EriB combination therapy had a significant synergistic effect on tumor growth inhibition (Figure 6C–E). Notably, this indicated the treatment dose produced no detectable adverse effects on the mice or their body weights, indicating limited toxicity (Figure 6F). In addition, EriB reduced the expression of p-AMPK and p-mTOR expressions compared with those of the untreated group. EriB restored sorafenib-induced p62 down-regulation, and this result was consistent with the results of in vitro cellular experiments (Figure 6G). Significant upregulation of PARP and cl-caspase3 level upregulation showed that the combination therapy also strongly enhanced apoptosis (Figure 6H). The combination therapy inhibited tumor proliferation, autophagy, and induced apoptosis according to the Ki-67, p62, and cleaved caspase 3 staining. The in vivo study suggested that EriB may inhibit sorafenib-induced autophagy and increase sorafenib sensitivity in hepatocellular carcinoma cells by suppressing the mTOR/ULK1 signaling pathway.
Discussion
EriB is a natural diterpenoid isolated from I. eriocalyx, a Chinese herbal medicine that exhibits potent anti-inflammatory effects and antitumor activity. EriB has been reported to alleviate inflammation in experimental autoimmune prostatitis and experimental autoimmune encephalomyelitis.27,28 Previous studies have shown that EriB exerts various anticancer effects such as triggering apoptosis and cell cycle arrest in pancreatic cancer, 15 lymphoma cells, 14 and colorectal cancer cells. 29 It also suppresses of cell migration and metastasis in breast cancer cells 30 and activates of autophagy in breast cancer cells. 13 However, the association between EriB-induced cell death and autophagy has not been reported in HCC.
In this study, we investigated the anticancer effect of EriB on HCC cells and demonstrated the underlying mechanisms. Our results showed that EriB suppressed cell proliferation and induced apoptosis. Further evidence suggested that EriB blocked autophagic flux and enhanced autophagosome formation by downregulating the mTOR/ULK1 signaling pathway, and this was demonstrated in vitro and in vivo. These results provide a rationale for the development of EriB as a novel anticancer therapy for HCC.
Earlier reports showed shown that EriB induced apoptosis in several types of cancer.31,32 In this study, the HepG2 and Huh 7 cells were sensitive to EriB and were characterized by less cell survival and much cell apoptosis, which was consistent with the results of previous reports. Autophagy is a degradative and recycling process, during which damaged organelles, unfolded proteins and pathogens are engulfed in double-membrane vesicles and delivered to the lysosome to for digestion. This process contributes to the maintenance of homeostasis and promoting cell survival. 33
Autophagy degrades damaged organelles and subsequent genomic instability to suppress tumor growth during the earlier stage of cancer. 34 However, autophagy plays a critical role in tumor cell survival by recycling internal nutrients to the growing cancer cell during stress in advanced cancer. More recent preclinical and clinical evidence suggests that targeting autophagy can effectively inhibit cancer development and sensitize cancer to chemotherapeutic drugs.35,36 There is no specific autophagy inhibitor or inducer that has been approved to treat cancer. Therefore, it is necessary to develop new pharmacological agents to target autophagy. The results of this study indicated that EriB inhibited autophagy, as evidenced by the increased p62 and decreased the formation of matured autophagosomes in the HepG2 and Huh 7 cells. In addition, our data revealed that EriB promotes autophagosome formation, as evidenced by a massive accumulation of autophagosomes. Inconsistent with our study, studies have shown that EriB can induce autophagy in breast cancer cells, and this may be due to the dual role of autophagy in promoting survival or death in different cancer contexts. 13 Moreover, we demonstrate that EriB blocks autophagic flux to suppress HCC cell growth. These results suggested a cytotoxic role of autophagy arrest in the anti-HCC effect of EriB.
Mechanistic analyses were performed to investigate the mechanisms of EriB regulation in HCC cell autophagy. The AMPK and mTOR signaling pathways are classical regulators of autophagy, and numerous studies have reported that AMPK promotes autophagy, while mTOR inhibits autophagy.37,38 mTORC1 inhibition resulted in the activating ULK1 complex in which phosphorylate activates vacuolar protein sorting 34 complex, leading to the conjugating of LC3 to lipid constituents that form autophagosome. 34 However, some studies found that mTOR inhibition results in enhanced autophagosome accumulation rather than autophagy induction. 38 Our previous study showed that fangchinoline induced autophagy via regulation of AMPK/mTOR/ULK1 pathway in colorectal cancer cells, and costunolide inactivates the AMPK/mTOR signaling pathway to suppress autophagy in ovarian cancer cells.18,24 Notably, EriB inactivated AMPK and mTOR signaling concurrently. This result indicated that EriB-induced autophagosome formation was AMPK-independent but was regulated by the inactivation of the mTOR/ULK1 pathway. Consistent with this finding, EriB significantly decreased mTOR phosphorylation, and the decrease in mTOR phosphorylation was blocked by MHY1458. We hypothesized that EriB induces HCC cell death through autophagy modulation and inhibition of the mTOR/ULK1 signaling pathway. EriB is a natural diterpenoid that can simultaneously inhibit AMPK/mTOR and block autophagy flow, and its multi-target properties may be superior to single-target autophagy inhibitors (for example, mTOR inhibitors are prone to trigger AKT feedback activation). While traditional autophagy regulatory drugs usually target a single node (such as the mTOR inhibitor rapamycin), EriB's simultaneous inhibition of AMPK and mTOR may more completely block compensatory pathway activation through a “double-lock mechanism” (such as AMPK inhibition can avoid AMPK feedback activation after mTOR inhibition). In contrast to other natural products (eg, berberine targeting AMPK, triptolide targeting mTOR), the dual action of EriB may reduce the risk of drug resistance.
Sorafenib has been reported to induce protective autophagy and contribute to chemotherapy resistance in HCC cells.39,40 The target of autophagy is a promising strategy for surviving chemoresistance.41–43 EriB may contribute to overcoming sorafenib resistance due to the function of EriB in the inhibition of autophagy flux. Furthermore, EriB and gemcitabine synergistically promote apoptosis in pancreatic cancer cells. 15 Consistent with these findings, we found that EriB promoted sorafenib-induced apoptosis in vitro and in vivo by inhibiting sorafenib-induced protective autophagy. In addition, further investigation of EriB and sorafenib for the synergistic treatment of HCC is warranted. We demonstrated that EriB had a significantly superior effect on HCC compared with sorafenib, the most used first-line drug. If these findings are successfully translated into clinical practice, the rational application of EriB may be an alternative therapy for HCC treatment. The inhibitory effect of EriB on autophagy in HCC may be more suitable for advanced HCC patients with high mTOR activation, and the combination of EriB with sorafenib can accurately overcome drug resistance. However, this new therapeutic approach concept requires validation in further preclinical models (such as patient-derived xenograft models) and well-designed clinical trials. This study requires further clinical sample validation and in vivo pharmacokinetic studies.
Conclusion
In summary, the results of this study revealed a novel antitumor activity of EriB in HCC. In addition, it was demonstrated that EriB inhibited autophagy through mTOR/ULK1 signaling that affected the sensitivity of HCC cells to sorafenib. These findings provide strong evidence that EriB may be a potential and effective candidate against human HCC due to its prominent anticancer efficacy.
Supplemental Material
sj-tiff-1-npx-10.1177_1934578X251339276 - Supplemental material for Eriocalyxin B Inhibits Autophagy in Hepatocellular Carcinoma Cells via mTOR/ULK1 Signaling Pathway Inactivation
Supplemental material, sj-tiff-1-npx-10.1177_1934578X251339276 for Eriocalyxin B Inhibits Autophagy in Hepatocellular Carcinoma Cells via mTOR/ULK1 Signaling Pathway Inactivation by Mingxia Tong, Xiaojin Gao, Maowei Lian, Xiaocong Xiang and Xiaobing Gong in Natural Product Communications
Supplemental Material
sj-tiff-2-npx-10.1177_1934578X251339276 - Supplemental material for Eriocalyxin B Inhibits Autophagy in Hepatocellular Carcinoma Cells via mTOR/ULK1 Signaling Pathway Inactivation
Supplemental material, sj-tiff-2-npx-10.1177_1934578X251339276 for Eriocalyxin B Inhibits Autophagy in Hepatocellular Carcinoma Cells via mTOR/ULK1 Signaling Pathway Inactivation by Mingxia Tong, Xiaojin Gao, Maowei Lian, Xiaocong Xiang and Xiaobing Gong in Natural Product Communications
Supplemental Material
sj-tiff-3-npx-10.1177_1934578X251339276 - Supplemental material for Eriocalyxin B Inhibits Autophagy in Hepatocellular Carcinoma Cells via mTOR/ULK1 Signaling Pathway Inactivation
Supplemental material, sj-tiff-3-npx-10.1177_1934578X251339276 for Eriocalyxin B Inhibits Autophagy in Hepatocellular Carcinoma Cells via mTOR/ULK1 Signaling Pathway Inactivation by Mingxia Tong, Xiaojin Gao, Maowei Lian, Xiaocong Xiang and Xiaobing Gong in Natural Product Communications
Supplemental Material
sj-tiff-4-npx-10.1177_1934578X251339276 - Supplemental material for Eriocalyxin B Inhibits Autophagy in Hepatocellular Carcinoma Cells via mTOR/ULK1 Signaling Pathway Inactivation
Supplemental material, sj-tiff-4-npx-10.1177_1934578X251339276 for Eriocalyxin B Inhibits Autophagy in Hepatocellular Carcinoma Cells via mTOR/ULK1 Signaling Pathway Inactivation by Mingxia Tong, Xiaojin Gao, Maowei Lian, Xiaocong Xiang and Xiaobing Gong in Natural Product Communications
Supplemental Material
sj-tiff-5-npx-10.1177_1934578X251339276 - Supplemental material for Eriocalyxin B Inhibits Autophagy in Hepatocellular Carcinoma Cells via mTOR/ULK1 Signaling Pathway Inactivation
Supplemental material, sj-tiff-5-npx-10.1177_1934578X251339276 for Eriocalyxin B Inhibits Autophagy in Hepatocellular Carcinoma Cells via mTOR/ULK1 Signaling Pathway Inactivation by Mingxia Tong, Xiaojin Gao, Maowei Lian, Xiaocong Xiang and Xiaobing Gong in Natural Product Communications
Supplemental Material
sj-tiff-6-npx-10.1177_1934578X251339276 - Supplemental material for Eriocalyxin B Inhibits Autophagy in Hepatocellular Carcinoma Cells via mTOR/ULK1 Signaling Pathway Inactivation
Supplemental material, sj-tiff-6-npx-10.1177_1934578X251339276 for Eriocalyxin B Inhibits Autophagy in Hepatocellular Carcinoma Cells via mTOR/ULK1 Signaling Pathway Inactivation by Mingxia Tong, Xiaojin Gao, Maowei Lian, Xiaocong Xiang and Xiaobing Gong in Natural Product Communications
Footnotes
Acknowledgements
We thank Prof. Chunlei Yu at the North Sichuan Medical College for guiding the animal experiments.
Ethical Approval
The animal experiments protocols were approved by the Animal Experimental Ethics Committee of the North Sichuan Medical College (Approval no. 2024070) on July 04, 2024, in compliance with the National Institutes of Health guidelines for the care and use of laboratory animals.
Author Contributions
Conception and design: Xiaobing Gong, Mingxia Tong. Acquisition of data: Mingxia Tong. Xiaobing Gong, Xiaojin Gao. Analysis and interpretation of data: Mingxia Tong, Maowei lian, Xiaobing Gong. Writing, review, and/or revision of the manuscript: Mingxia Tong, Xiaobing Gong, Administrative, technical, or material support: Xiaojin Gao, Xiaocong Xiang.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Applied Basic Research Program of Sichuan Province, Health Commission of Sichuan Province, (grant number 20YYJC2624, No.21PJ192).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All of the experimental procedures that involved animals were conducted in accordance with the Institutional Animal Care guidelines of the North Sichuan Medical College, China and approved by the Animal Experimentation Ethics Committee of the North Sichuan Medical College (No. 2024070), Sichuan Province, China. This article does not contain any experiments that used human subjects.
Statement of Informed Consent
There were no human subjects that were used in this study, and informed consent was not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
