Abstract
Introduction
The cocoa tree (
In general, the traditional extraction processes often use organic solvents involving acids and alkalis, which have been restricted in the food and pharma industry due to their inherent toxicity and environmental hazards. Although these approaches had a high extraction efficiency and ease of implementation, high solvent consumption, time-consuming, and the degradation of bioactive compounds was reported. 4 To address these concerns, the alternative treatment approach emerging with the green solvent called Deep Eutectic Solvent (DES) combined with low-cost techniques has been investigated to mitigate the issues related to conventional processes.5,6 Being introduced in the early twenty-first century, DES is a low toxicity, reusability, cost-effective, and good biodegradability extraction solvent system inheriting and optimizing the characteristics of ionic liquids. Briefly, DES is often formed by combining at least two solid components through the formation of hydrogen bonds between a hydrogen bond acceptor (HBA) and a hydrogen bond donor (HBD). 7 Among the potential mixtures, DES composed of choline chloride (ChCl) and glycerol (Gly) has garnered significant attention due to its unique properties. They demonstrate high solubility for a wide range of compounds, from polar to non-polar substances, along with excellent thermal stability and low volatility, making them suitable for various applications. Furthermore, these components are non-toxic, renewable, and cost-effective.8,9 Previous works found that phenolic compounds can dissolve spontaneously into a solvent consisting of ChCl/Gly.10,11 However, DES solvents are often characterized by high viscosity, which limits the mass transfer diffusivity between the material and the extraction phase. To address this issue, combining DES with advanced techniques such as ultrasound-assisted extraction can enhance mass transfer and expedite the diffusion of compounds through cavitation, mechanical action, and thermal effects.5,6 Patil et al reported achieving a maximum curcuminoid yield of 77.13 mg/g from Curcuma longa using ultrasound-assisted DES-based extraction, outperforming traditional methods. 12 Recently, some studies have explored the effect of sonication on the extraction of various cocoa by-products, such as cocoa shells and CPH, using ethanol/water as solvents and identified their potential polyphenolic contents.13,14
To the best of our knowledge, there are very few published studies investigating the production of phenolic compounds from CPH residues through ultrasound-assisted DES extraction. We hypothesized that UAE-DES method would enhance the phenolic contents and allow us an optimal process of extraction. Therefore, this study aimed to model and optimize the extraction conditions to enhance polyphenols, flavonoids, and catechins from CPH using green solvent as DES. The experimental parameters comprised the use of different solid-to-liquid ratio, time, and temperature. Response surface methodology (RSM) was used to optimize processes when non-linear and interactive effects between independent variables exist. The phenolic profile of CPH extract and antioxidant activities were investigated by LC-UV-MS. The optimal extraction process not only considered the content of bioactive compounds but also the antioxidant capacity of the extract.
Experiment
Chemicals and Materials
Cocoa fruits (
Sample Preparation and Experimental Design
The DES was prepared with choline chloride as the HBA and Glycerol as the HBD at a ratio of 1:4 (w
Identification of Phenolic Compounds
A qualitative analysis of phenolic constituents from cocoa pod husk (CPH) extract was performed using the Ultra-High Performance Liquid Chromatography with Diode Array Detection and Quadrupole Mass Spectrometry UHPLC-DAD-MSQ system (Thermo, USA) equipped with a Phenomenex Ultracarb ODS (30) column (150 × 4.6 mm, 3 µm). The UV detector was set to 280 nm. The mobile phase employed a gradient elution system consisting of two solvents: solvent A (HPLC-grade water with 0.1% formic acid) and solvent B (acetonitrile with 2% (v/v) formic acid). The gradient elution ran for 20 min, followed by a switch to pure acetonitrile. The flow rate was maintained at 1 mL/min, and the injection volume was set to 20 µL. The eluted compounds were subsequently desolvated through electrospray ionization (ESI) and analyzed in the mass spectrometer. The mass spectrometer operated in negative ionization mode, scanning a mass range of m/z 150–2000, with an injection duration of 150 ms and two micro-scans per measurement. To confirm structural identities, the deprotonated ions [M − H]− were subjected to a second mass analysis step (MS/MS), providing detailed fragmentation patterns for accurate compound identification. 16
Chemical Analysis
The total polyphenols content (TPC) was spectrophotometrically determined using Foin-Ciocalteu's phenols reagent, according to the method described by Benítez-Correa et al
8
with some modifications. Firstly, 1 mL of the extract was added into the test tubes followed by adding 2.5 mL of Folin-Ciocalteu reagent 10% (
The total flavonoid content (TFC) followed the method described by Mohaddese et al, 17 in which quercetin was used as the standard substance for flavonoid compounds. Results were expressed in g of quercetin equivalent (QE)/100 g dry weight of substrate.
The catechin concentration (CC) was determined by the vanillin assay method (Rebollo-Hernanz et al 2021). 18 Catechin was used as the standard substance. Results were expressed in g of catechin equivalent (CC)/100 g dry weight of substrate
Antioxidant Activity Analysis
The ABTS free radical (ABTS●+) scavenging method was conducted following the procedure described by Le et al.
19
Firstly, the stock solution ABTS●+ was prepared by mixing 7 mM ABTS●+ solution with 2.45 mM potassium persulfate solution (1:1,
The DPPH a radical scavenging capacity assay was adopted from Brand-Williams et al.
20
and Nguyen et al.
21
with some modifications. DPPH reagent solution was prepared by dissolving DPPH into 100 ml of methanol. 150 μL of the extract was mixed with 3 ml of DPPH reagent and reacted in the dark for 30 min at room temperature. Then the absorbance was measured at 517 nm using a UV-Vis spectrophotometer. The DPPH radical scavenging activity of the extracted sample was determined using the following formula.
In which, Abscontrol represents the absorbance of the DPPH reagent solution and Abssample represents the absorbance of a mixture of sample extract and DPPH reagent solution.
The total antioxidant activity of the extract was determined using a modified version of the ferric-reducing (FRAP) assay. 22 The FRAP reagent solution was freshly prepared by mixing 30 ml of acetate buffer (300 mM), 3 ml of TPTZ (10 mM in HCl 40 mM), and 3 ml of FeCl3 (20 mM) and incubated at 37 °C before testing. The mixture of 0.15 mL extract sample and 3 mL FRAP reagent was kept in the dark for reaction in 30 min before being measured at 593 nm. A calibration curve with FeSO4 standards was measured in the range of 0.5–2.5 mM and the result was expressed in g of FeSO4 /100 g dry weight of substrate.
Experiment Design and Statistical Analysis
A Box–Behnken design, a spherical and rotatable experimental design, was employed to investigate the relationship between multiple independent variables and extraction efficiency, and to optimize the process to achieve the maximum extraction efficacy. The design comprised 13 experimental runs, with each independent variable evaluated at three coded levels (−1, 0, + 1). The independent variables included reaction time (X1, 20-60 min), reaction temperature (X2, 40-60 °C), and substrate-to-solvent ratio (X3, 20%–5%, w/w). The response variables measured were total phenolic content (TPC, g GAE/100 g dry weight of substrate), (TFC, g GE/100 g dry weight of substrate), and catechin content (CC, g CC/100 g dry weight of substrate). The optimization process was conducted using JMP 17.0, a statistical software platform, in conjunction with the Box-Behnken design. RSM was applied to identify the optimal conditions for the extraction process.
The statistical equation for RSM typically involves a second-order polynomial model to describe the relationship between the response variable (Y) and the independent variables (Xi). For a Box-Behnken Design, the general form of the equation was reported by Araujo,
23
as follows: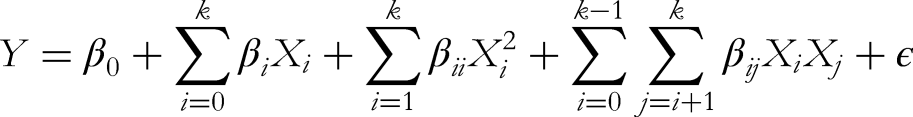
In which,
Y is the response variable. β0 is the intercept term. βi are the linear coefficients for the independent variables Xi. βii are the quadratic coefficients for the squared terms Xi2. βij are the interaction coefficients for the interaction terms XiXj. ɛ is the random error term. k is the number of independent variables.
Experimental data were analyzed using Excel 2016 and SPSS 20.0, with results expressed as means ± standard deviation of triplicate measurements. Analysis of variance (ANOVA) was performed to compare sample means, and the effects of factors and their interactions were considered statistically significant at a threshold of p < 0.05.
Results
Optimization of the Extraction Conditions to Recover Bioactive Compounds from CPH
RSM combined with Box-Behnken experimental design was applied to optimize the UAE process of bioactive compounds from CPH. According to the preliminary experiments, investigated factors included reaction time (20-60 min), reaction temperature (40-60 °C), and the substrate: extraction solvent ratio (20% – 5%, w/w). The yields extract of bioactive compounds (TPC, TFC, and CC) were depicted in Table 1 while their multiple regression analysis was summarized in Table 2. Depending on extracting conditions, TPC of the extract was in a range of 2–12 gGAE/100 g dry weight of substrate. This result was in line with the finding in a recent study using DES to extract bioactive compounds from cocoa husk. 24 For flavonoids and flavanols content, the yield extract was in the ranges of 0.5–2 gQE/100 g dry weight of substrate and 17–63 gCE/100 g dry weight of substrate, respectively. Similar to the findings in previous studies,18,25 it could be seen that CPH was the potential source for catechin while its flavonoid content was quite small.
Values of Bioactive Compounds From CPH Extracts Obtained From Box-Behnken Design to Optimize The Ultrasonic-assisted Extraction Process With DES Solvent.

TPC = Total phenolic content; TFC = Total flavonoid content; CC = Catechin concentration.
Regression Coefficients for Extraction Factors and Statistics for the Fit from Multiple Regression for Response Variable Analysis.
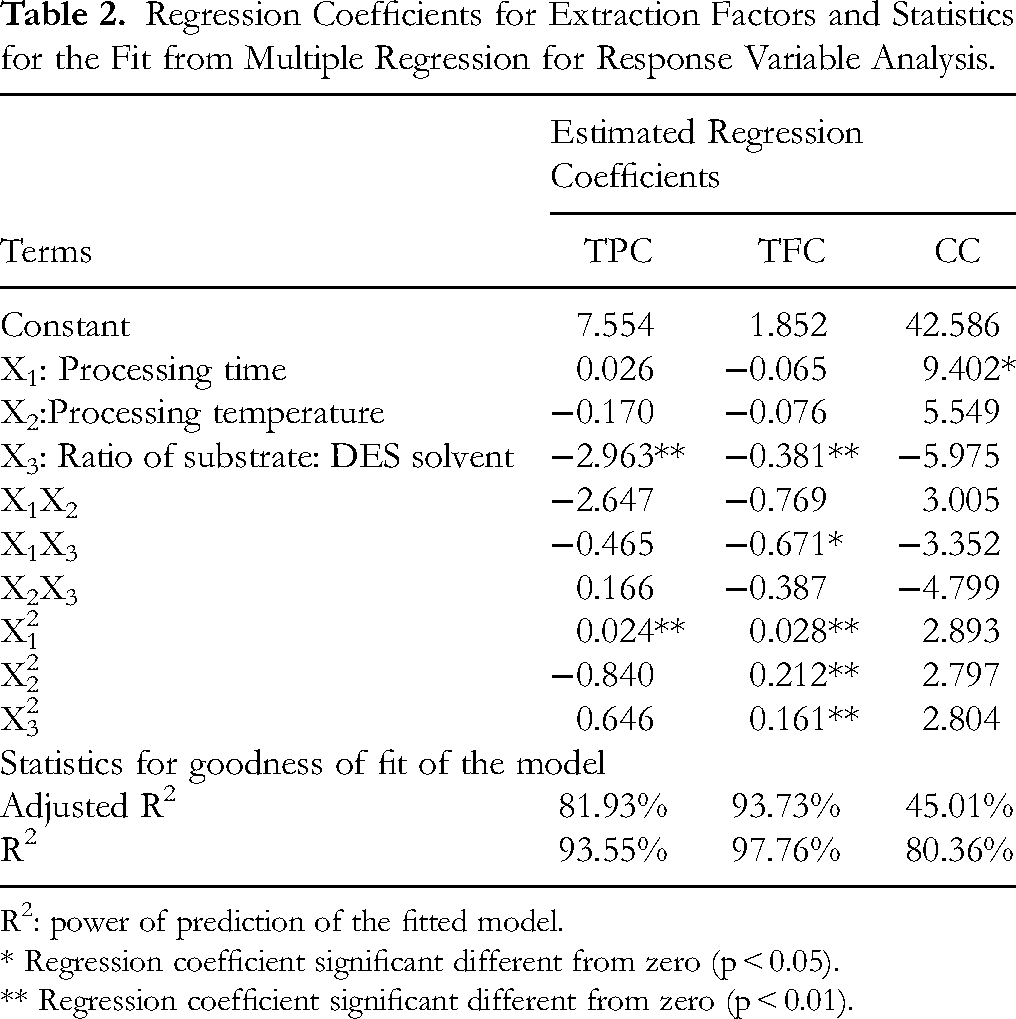
R2: power of prediction of the fitted model.
* Regression coefficient significant different from zero (p < 0.05).
** Regression coefficient significant different from zero (p < 0.01).
According to data analysis, TPC, TFC and catechin content of the extracts showed a well-fit with second-order models (R2 = 93.55%; 97.76%; and 80.36%, respectively), and established models were statistically reliable and reasonable (p < 0.05). Therefore, these regression models could be applied to predict the amount of extracted bioactive compounds from CPH based on extraction time (X1), extraction temperature (X2), and the ratio of substrate to solvent (X3).


The Antioxidant Capacity of the Extract
Pearson's correlations were studied to analyze associations between in vitro parameters and the antioxidant capacity of CPH extracted with DES. The analysis showed a tight correlation between antioxidant activity and TPC, TFC, and CC, with corresponding coefficients of 0.78, 0.63, and 0.68, respectively (Figure 1). Besides, Figure 2 shows the
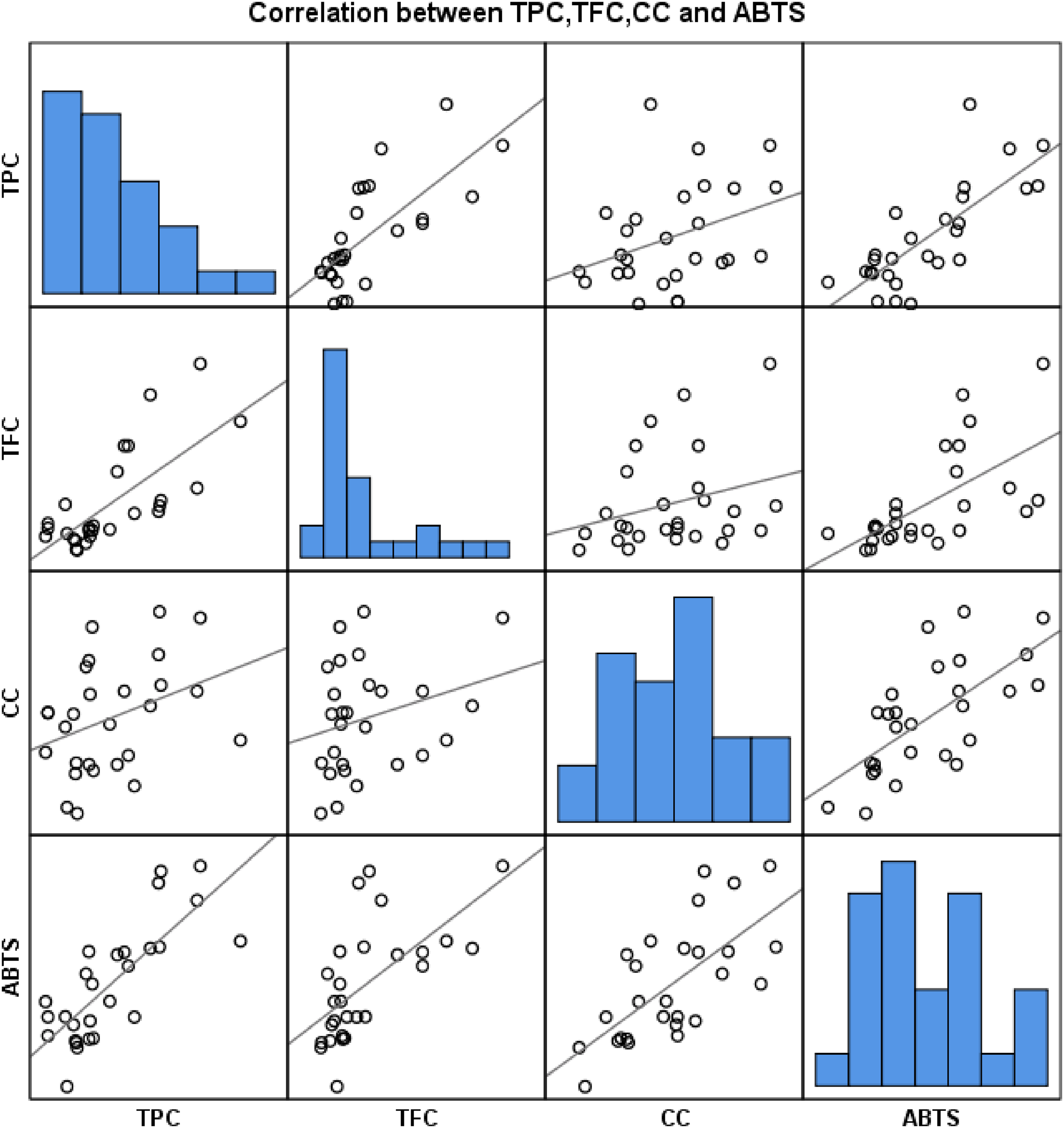
The Correlation Between Bioactive Contents and Antioxidant Activity.

Antioxidant Activity by ABTS Assay (A); DPPH Assay (B) and FRAP Assay (C) of Cocoa-pod Husk Extracts at Substrate: Solvent Ratio of 5%. Values are the Mean Of Triplicate Data ± Standard Deviation.
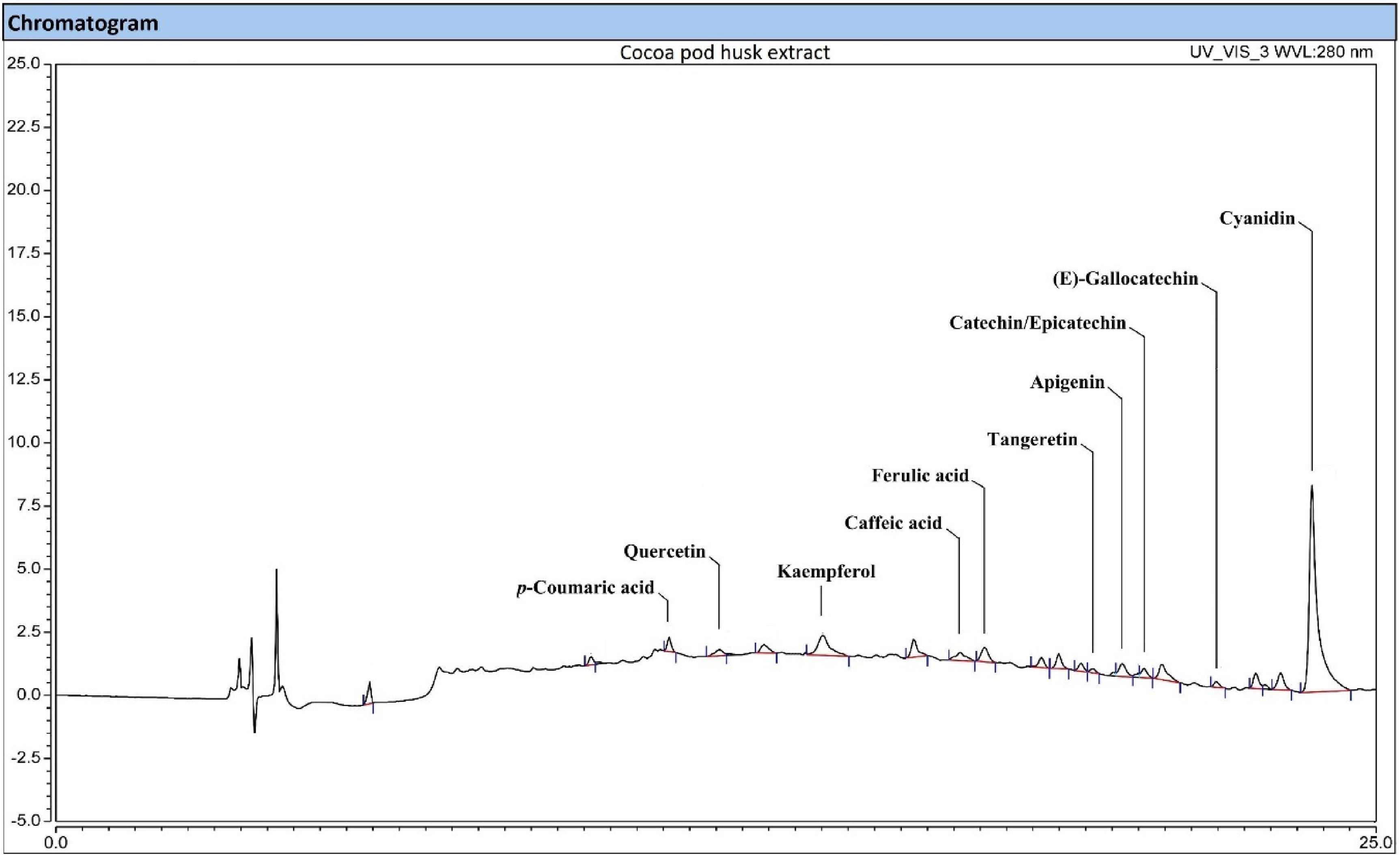
UHPLC-DAD-MSQ Chromatogram of DES Extract of CPH.
The Phenolic Compounds Profile of the CPH Extract Using UAE-DES Method.
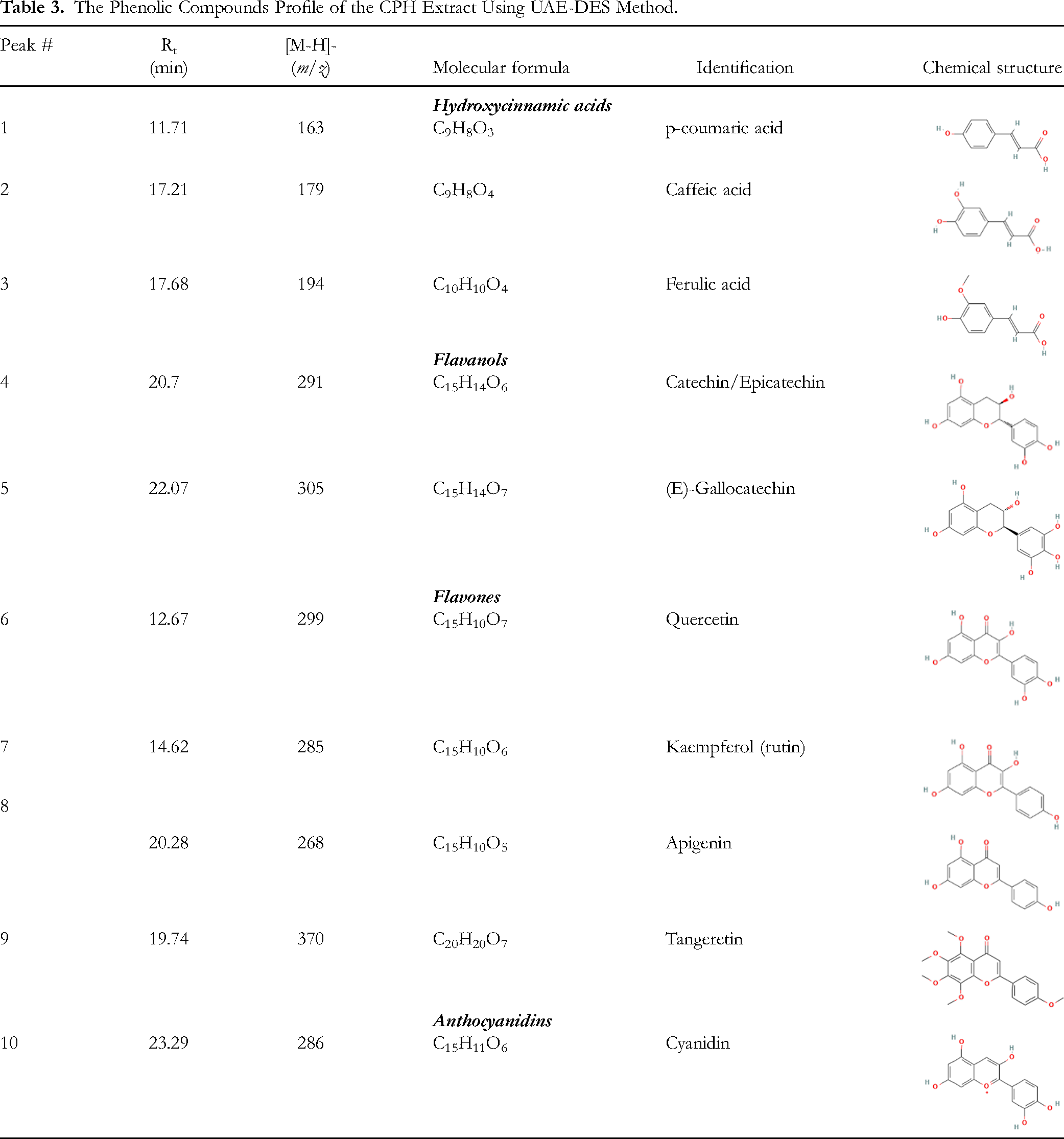
Discussion
In general, increasing extracting temperature and time significantly improved the extraction of phenolic and flavonoid compounds in fruit biomass.27,28 For instance, a recent study by Pal and Jadeja 29 found higher phenolic compounds extracted from onion peels by microwave-assisted DES when prolonging the irradiation time from 5 to 15 min. However, these bioactive compounds are also sensitive to heat treatment, therefore, their contents could be reduced due to oxidation under intensive heat. Our study found that three factors of the extracting process had varying effects on three bioactive compounds. The ANOVA of the experimental design indicated the linear effect of substrate: solvent ratio (X3) and quadratic effects of extracting time (X12) on the extract yield of phenolic and flavonoid compounds. According to the mathematical model obtained for TPC, it was seen that the highest phenolic content of 12.22 gGAE/100 g dry weight of substrate was achieved by operating the extraction process at 50 °C for 40 min. Similarly, samples also remained with the highest antioxidant capacity at the same extraction condition. Catechin content, on the other hand, acquired the intensive extracting temperature and longer extraction time in comparison with phenolic compounds because this flavanol had a higher thermal stability. Our findings were in agreement with the study of Bajkacz et al., 30 which found a higher mass transfer of catechin when increasing the extraction time and temperature, leading to a decrease in adsorption and interaction between analytes and matrices. Besides, the assessment of DES solvent could be different when DES extraction seems more suitable for catechin, an active compound owning many hydroxyl groups and exhibiting higher polarity than phenolic compounds. 31 For TFC, it was mainly affected by ultrasonic temperature and the ratio of substrate to solvent with a quadratic effect, as well as by the interaction of X1-X3 (p < 0.05).
Besides extracting temperature and time, TPC, TFC, and CC of the cocoa extract were highly influenced by the ratio between the substrate and DES solvent. The extract with the ratio of substrate: DES solvent of 20% exhibited the lowest concentration of bioactive compounds. As illustrated in Figure 4, the phenolic contents showed an upward tendency with an increase of added solvent. The greater amount of solvent could cause an increase in concentration gradient and hence, more bioactive compounds could be released. 32 Besides, the addition of extraction solvent could enable proper hydration and solvation of active compounds more efficiently. Therefore, after considering the actual process and all relevant technical aspects, the optimum conditions of the extraction process should be 50 °C for 40 min with a substrate-solvent ratio of 5% (w/w). At these optimal extraction conditions, the concentrations of TPC, TFC, and CC were found to be 10.14 gGAE/100 dry weight of substrate, 2.18 gGE/100 g dry weight of substrate, and 64.80 gCE/100 g dry weight of substrate, respectively while the antioxidant capacity was 13.725 g TE/100 g dry weight of substrate, 67.1% OR and 0.887 gFeSO4/100 g dry weight of substrate assayed with ABTS, DPPH and FRAP, respectively.
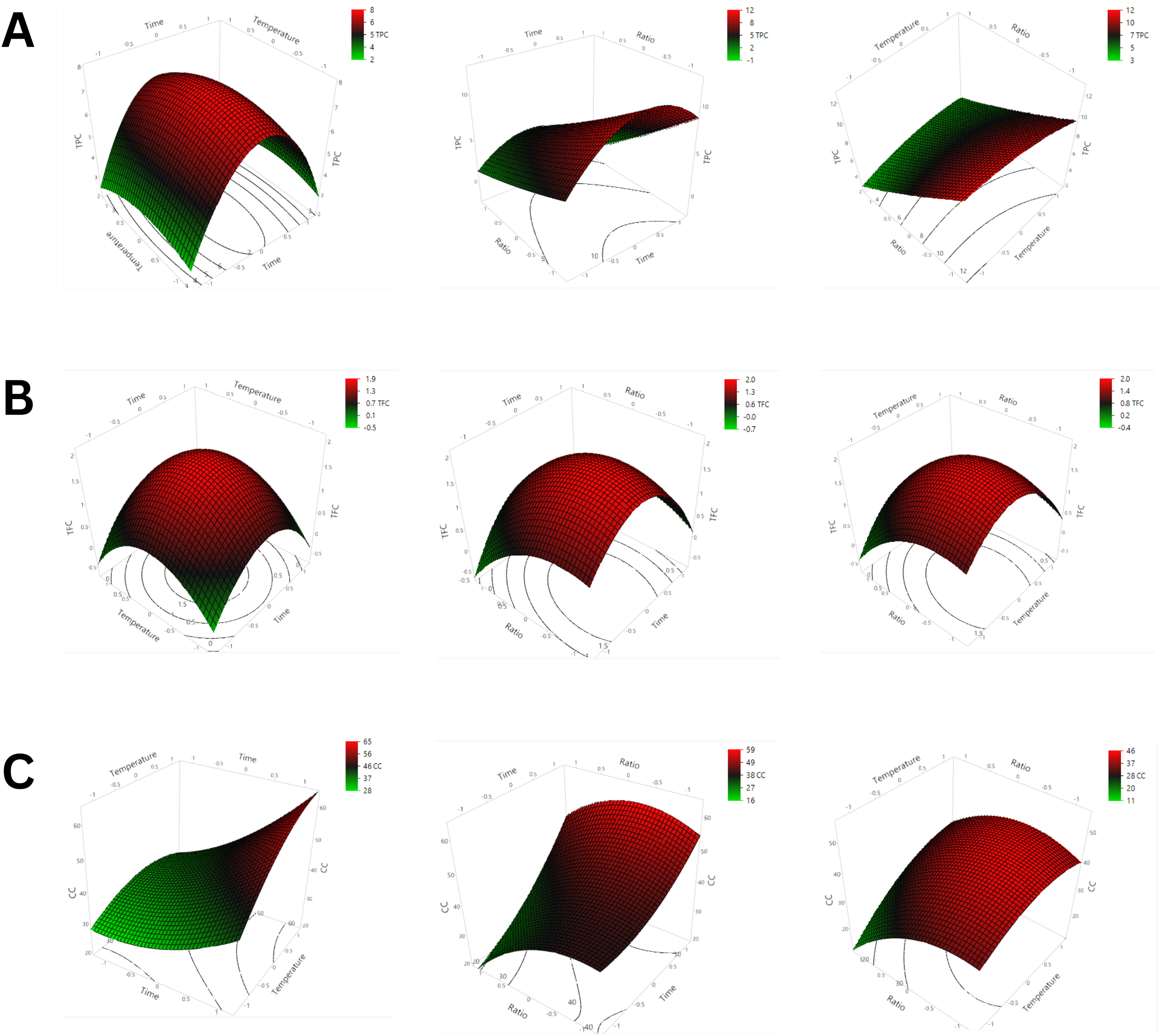
Illustrates the Effect of Pair Factors (Time-temperature, Ratio-time and Ratio-temperature) for Each Response Variable (TPC, TFC, CC) by Response Surface 3D Plot.
When considering the antioxidant aspect, numerous studies investigated the extraction of cocoa by-products with conventional solvent (such as methanol-HCl extraction coupled with sonication), unveiling a rich profile of bioactive compounds including protocatechuic acid, epicatechin, procyanidin B2, and catechin.18,33,34 However, traditional solvents are not eco-friendly and often are negatively effects on the consumer. Green solvents like DESs are often favored by the low toxicity but their efficiency on the antioxidant capacity of the extract is not well understood. When replacing ethanol by DES (choline chloride – lactic acid mixture) in extracting cocoa bean shell, Mansinhos et al recognized a significant increase of antioxidant activity
35
but the sonication technology was not applied and therefore, the extraction time is quite long. Return to our work, the combination of DES and UAE in extracting CPH showed potential benefits in recovering bioactive compounds. For instance, the antioxidant capacity of the CPH extract using DES-UAE is equivalent with the traditional solvent
26
while the extraction temperature is significantly lower (50 °C
Conclusion
This study focused on modeling and optimizing extraction conditions to maximize the yield of polyphenols, flavonoids, and catechins from CPH using DES-assisted ultrasound techniques. The optimization process prioritized not only the content of bioactive compounds but also the antioxidant capacity of the extracts. It demonstrates that ultrasound-assisted deep eutectic solvent (UAE-DES) extraction is an effective and promising method for extracting phenolic compounds from cocoa by-products. The results revealed that CPH extracts obtained using green solvents—DES—are rich in phenolics and exhibit high antioxidant capacity, making them suitable for potential applications in the food and pharmaceutical industries. In the food sector, phenolic-rich extracts can enhance the nutritional value, shelf life, and sensory properties of products. In pharmaceuticals, these extracts hold promise for their therapeutic properties, contributing to drug development and disease prevention. Furthermore, future research could explore the performance of different types of DES or investigate emerging extraction and purification technologies, taking into account not only extraction efficiency but also environmental and economic considerations.
Footnotes
Acknowledgment
This study was supported by Nong Lam University, Ho Chi Minh City, Vietnam.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Ethical Considerations
Ethical Approval is not applicable to this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Nong Lam University under Grant number CS-SV23-HHTP-03.
Conflict Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
