Abstract
Steroidal alkaloids are derived from the steroid skeleton with one or two nitrogen atoms. They are widely distributed in tropical and subtropical regions and possess a range of biological activities. The structures of steroidal alkaloids are comparable to those of anabolic steroids, steroidal hormones, and corticosteroids, making them a valuable source for drug discovery. Taxonomically, steroidal alkaloids are limited in distribution to certain plant families, predominantly the Apocynaceae, Buxaceae, Solanaceae, and Liliaceae. This review highlights the steroidal alkaloids from the Apocynaceae family and their biological activities. The articles published from 1919 to 2021 were included in this review. A total of 163 steroidal alkaloids and 12 biological activities were reported from plant species belonging to the Apocynaceae family in this period. Of the 410 genera in the Apocynaceae, only 10 contain steroidal alkaloids. Although some alkaloids from the Apocynaceae family were also reported in the Buxaceae family, especially tetracyclic triterpenes with a pregnane side chain, most steroidal alkaloids can only be found in several genera of the Apocynaceae family.
Keywords
Introduction
A steroidal alkaloid has a basic steroid skeleton and contains at least one nitrogen atom where the nitrogen atom may be attached to either the side chain or the polycyclic system. 1 In addition, this nitrogen atom may be part of an additional ring such as a lactam ring, lactone ring, pyrrolidine ring, or pyrroline ring. Steroidal alkaloids are found in several plant families, notably Apocynaceae, Solanaceae, Buxaceae, and Liliaceae. Many steroidal alkaloids have become targets of pharmacological investigations due to their structural similarities with anabolic steroids, steroidal hormones, and corticosteroids. 2 In this review, the steroidal alkaloids from the Apocynaceae family are summarized. This family comprises woody climbers, tropical trees, shrubs, and herbs. 3 Plants of the Apocynaceae are predominantly distributed in tropical and subtropical regions although are also well established in temperate zones. 4 Steroidal alkaloids of the Apocynaceae are classified into four main structural groups: conanine, pregnane, paravallarine, and amino-glycosteroids (Figure 1). 5 By high performance liquid chromatography (HPLC), some of the alkaloids can be detected at around 200 to 245 nm, but most can only be detected at around 200 nm.6–8 The use of acetonitrile and water as mobile phases allows the detection at that wavelength. Of 410 genera in the Apocynaceae family, 9 only 10 namely Beaumontia, Chonemorpha, Dictyophleba, Funtumia, Holarrhena, Kibatalia, Malouetia, Tabernaemontana, Vahadenia, and Wrightia, have been reported to contain steroidal alkaloids. Some steroidal alkaloids in the Apocynaceae family can also be found in the Buxaceae family. Steroidal alkaloids from these genera showed acetylcholinesterase (AChE),10,11 antibacterial,11–15 anticercarial, 16 antidiarrheal, 17 antifeedant, 18 antimycobacterial, 19 antiplasmodial,20–22 antitrypanosomal, 23 cytotoxic,20,21,23–26 hypotensive, 27 leishmanicidal activities, 28 and antibiotic efficacies. 29

Four structural groups of Apocynaceae.
Biosynthesis of steroidal alkaloids
The steroidal alkaloids isolated from the genus Holarrhena are biosynthesized from cholesterol.
30
According to Kumar et al., all pregnane-type steroidal alkaloids are derived from cholesterol (

Biosynthesis of pregnenolone.
A steroidal alkaloid such as 3β-holaphyllamine (

Biosynthesis of some steroidal alkaloids.
Distribution of Steroidal Alkaloids
Beaumontia Species
There is only 1 report on steroidal alkaloids from the Beaumontia genus. A pregnane steroidal alkaloid, namely beaumontamine (
Chonemorpha Species
There are 5 reports on steroidal alkaloids from the genus Chonemorpha. The roots of Chonemorpha fragrans (Moon) Alston yielded 3 pregnane-type steroidal alkaloids, namely N-formylchonemorphine (
Steroidal Alkaloids From the Apocynaceae Family.

Note. aPregnane.
Conanine.
Paravallarine.
Amino-glycosteroids.
Dictyophleba Species
There are 4 reports on steroidal alkaloids from the Dictyophleba genus. Investigation of the roots of Dictyophleba lucida yielded dictyodiamine (
Funtumia Species
Two species of Funtumia were reported to contain steroidal alkaloids. From the stem bark of Funtumia africana (Benth.) Stapf (synonym: F. latifolia), conanine-type steroidal alkaloids were characterized, namely 12α-hydroxy-norcona-N(18),1,4-trienin-3-one (
Alkaloid
Holarrhena Species
All plants in the genus Holarrhena produce steroidal alkaloids. The leaves of Holarrhena congolensis Stapf contained the new alkaloid kisantamine (
Phytochemical investigation of the leaf extract of Holarrhena curtisii King & Gamble yielded N-formylconkurchine (
Methylholaphylline (
Investigation of the leaves of H mitis R.Br. revealed N-demethylmitiphylline (
Phytochemical studies on the seed extract of H pubescens (Buch. Ham) (synonym: H. antidysenterica and H. febrifuga), yielded holarricine (
Kibatalia Species
Steroidal alkaloids were reported from five species of the Kibatalia genus. Kibatalia gitingensis (Elmen) Woodson yielded kibataline (
N-methylgitingensine (
Malouetia Species
Steroidal alkaloids were reported from four species of the genus Malouetia. Malouetia arborea Miers. yielded malarborine (
Tabernaemontana Species
It is surprising that of 118 Tabernaemontana species, only one has been reported to contain steroidal alkaloids. Two pregnane-type steroidal alkaloids were isolated from the roots of Conopharyngia pachysiphon (synonym: T. pachysiphon Stapf), and deduced as 20α-amino-3,5-pregnadiene (
Vahadenia Species
A study on the roots of V. laurentii (De Wild) Stapf resulted in the characterization of irehdiamine F (
Wrightia Species
Only two species of Wrightia contain steroidal alkaloids. Investigation of Wrightia tomentosa (synonym: W. arborea (Dennst.) Mabb.) resulted in the isolation of
In conclusion, 163 steroidal alkaloids have been successfully isolated from the 10 genera of the Apocynaceae family so far. Table 1 summarizes all the steroidal alkaloids obtained from the Apocynaceae family.
Chemotaxonomic Significance of Steroidal Akaloids in the Apocynaceae Family
Steroidal alkaloids are limited in distribution to particular plant families, for example, the cholestane type of steroidal alkaloids can be found in the families Solanaceae, Liliaceae, Berberidaceae, Asteraceae, Buxaceae, Alliaceae, and Verbenaceae. Pregnane, cyclopregnane, and conanine types of steroidal alkaloids are found in the Buxaceae and Apocynaceae. The paravallarine type of steroidal alkaloids is exclusively found in the genus Kibatalia of Apocynaceae.25,121–127 The genera within the subfamily Apocynoideae mainly contain the pregnane and conanine types of steroidal alkaloids, except for the steroidal alkaloids in the tribe Apocyneae. Beaumontia sp. and Chonemorpha sp. in this tribe contain mainly pregnane-type steroidal alkaloids. Steroidal alkaloids from the tribes Malouetieae and Wrightieae are mainly pregnane and conanine types, except for the Kibatalia sp. Only K. arborea was reported to contain a pregnane alkaloid, namely holafebrine (
Conessine (
Several steroidal alkaloids in the Apocynaceae family were also reported in the Buxaceae family, a small plant family with only five genera. Steroidal alkaloids found in both plant families are mainly pregnane tetracyclic triterpenes
138
with C-4 methyls, a 9β, 10β-cycloartenol system, and a degraded C-20 side chain.
139
Holadienine (
Biological Activities
AChE Activity
Conessine (
Holarrhena-type steroidal alkaloids were found to bind to the allosteric site of the AChE enzyme. 10 The presence of an N-methyl moiety and an unsubstituted N-H on the pyrrolidine ring are essential for AChE inhibitory activity, while cleavage of 1 or 2 N-methyl groups at C-3 reduces this inhibitory activity. In contrast, Cheenpracha et al. suggested that the cleavage at N,N-dimethyl group at C-3 significantly increases AChE activity of Hollarhena-type steroidal alkaloids. 11
Antibacterial Activity
Conimine (
Conessine (
Anticercarial Activity
Conessine (
Antidiarrheal Activity
Conessine (
Antifeedant Activity
Conessine (
Antimycobacterial Activity
Holadysamine (
Antiplasmodial Activity
Conessine (
Antitrypanosomal Activity
Seventeen alkaloids isolated from the leaves and stem bark of H floribunda, namely 3β-holaphyllamine (
Cytotoxic Activity
Irehline (
Hypotensive Activity
Holamide (
Leishmanicidal Activity
Holacurtine (
Restoration of Antibiotic Efficacy
The combination of conessine (
Bioactive Steroidal Alkaloids
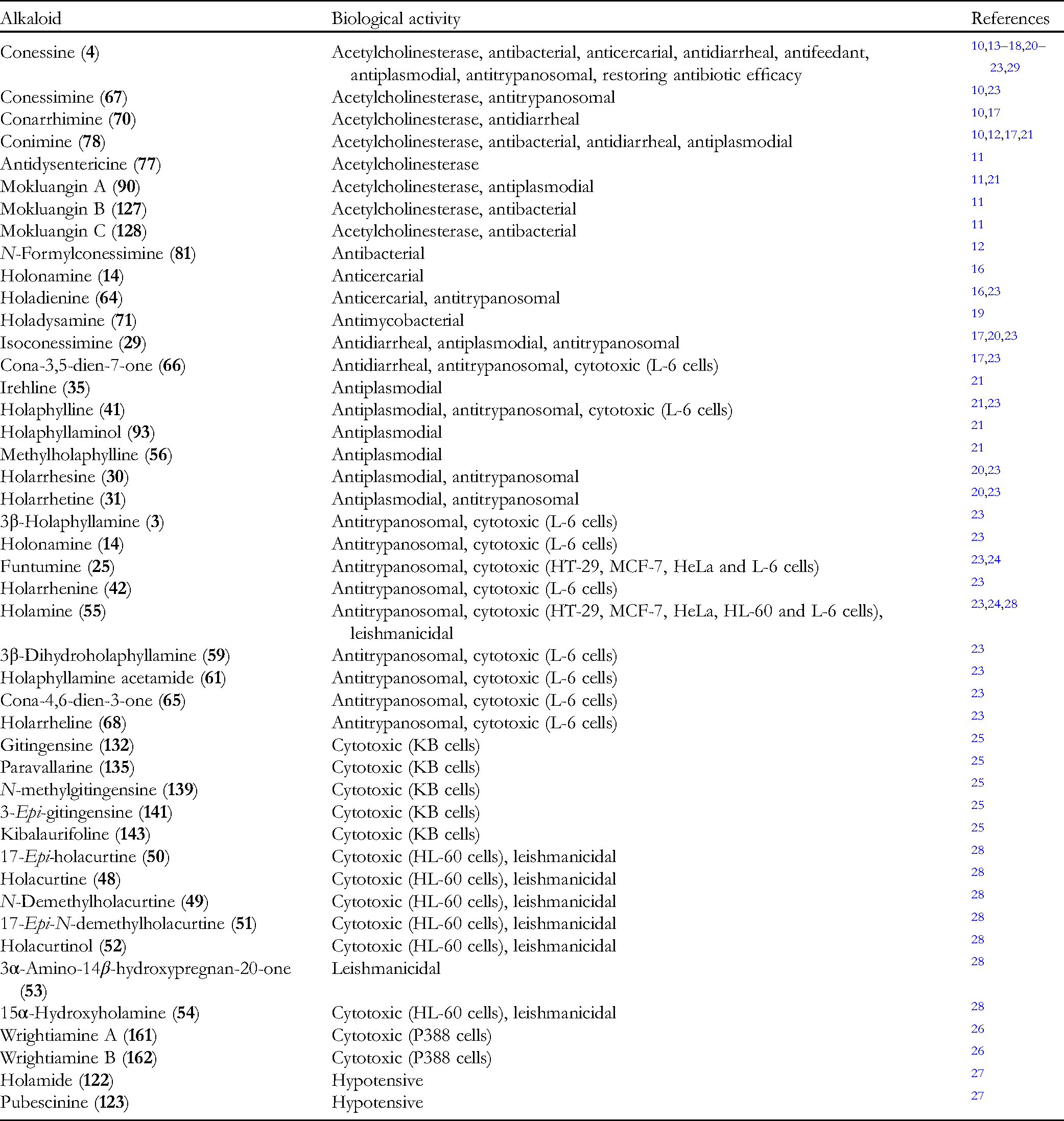
Table 2 summarizes the biological activities of steroidal alkaloids from the Apocynaceae family. Steroidal alkaloids with low basicity due to N-demethylation at C-3 showed better bioactivity compared to other alkaloids. 21 This trend was observed in several bioassays.11,23 The effect of methylation on the nitrogen of the pyrroline and pyrrolidine moieties varies, depending on the individual bioactivity being examined. Most bioactivities were reported as alkaloids derived from Holarrhena spp., and a few from Funtumia and Wrightia spp. As the most studied, Holarrhena steroidal alkaloids exhibited a wide spectrum of biological activities.
Biological Activities of Steroidal Alkaloids From Apocynaceae Family.
Conclusion
The isolation of steroidal alkaloids was first reported in 1919. These compounds are found in a limited number of plant families, including the Apocynaceae. A total of 163 steroidal alkaloids have been reported from the Apocynaceae family from 1919 to 2021. Of the 410 genera in the Apocynaceae, only 10 contain steroidal alkaloids. Holarrhena is the most well studied compared to the other genera, while conessine (
Footnotes
Acknowledgements
The authors would like to thank Universiti Teknologi MARA for financial support via MyRA Road to HiCOE Research Grant (600-RMC/GPM COE 5/3 (052/2021)) and for the facilities provided. The authors would also like to thank Professor Emeritus Geoffrey A. Cordell for proofreading this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The research was supported by Universiti Teknologi MARA via MyRA Road to HiCOE Research Grant (600-RMC/GPM COE 5/3 (052/2021)).
