Abstract
Background
Panax ginseng Meyer (P. ginseng) is a well-known herb of traditional Chinese medicine that has been used to revitalize the body and mind. A recent study revealed that P. ginseng exhibits neuroprotective activity against brain damage. P. ginseng also enhances cognitive function. However, the protective effects of P. ginseng berry against hepatic encephalopathy (HE) have not yet been studied. We investigated the protective effect of ultrasonication-processed GBE (UGBE) compared to GBE on mild bile duct ligation (MBDL) to verify the effect of P. ginseng berry on HE.
Methods
After ultrasonication, the component ratios of ginsenosides Rh1, Rh4, Rg2, Rg3, Rk1, Rk3, and F4 in the extract increased. In this study, the protective effect of the newly developed UGBE against neuronal damage was evaluated in an MBDL model. Silymarin was used as a positive control. UGBE, GBE, and silymarin were orally administered for 6 weeks after MBDL surgery.
Results
MBDL rats demonstrated delayed pain response and impaired motor coordination, mobility, spatial learning, and related memory errors. However, UGBE administration ameliorated HE symptoms, whereas treatment with GBE or silymarin did not show a recovery effect. The recovery effects of UGBE are due to a reduction in neuronal damage caused by MBDL surgery. Microglial activation and the production of inflammatory cytokines were decreased in the UGBE treatment group.
Conclusion
UGBE has a protective effect against MBDL-induced HE in rats through the inhibition of microglial activation in the brain.
Introduction
Hepatic encephalopathy (HE) is a neuropsychiatric condition associated with chronic or acute liver disease. 1 It is associated with a wide range of neuropsychiatric disorders. These psychomotor symptoms and other cognitive and behavioral dysfunctions are the main factors that lower the overall quality of life in patients with HE.2,3
As HE is a common complication of liver failure that leads to complex neuropsychiatric syndromes, several treatment strategies for HE have been developed according to the causes of HE symptoms.4,5 The inhibition of ammonia production is currently considered a representative treatment strategy for HE. However, the benefits of these treatments remain limited and still controversial.6,7 Although liver transplantation is considered the only fundamental treatment for HE, there are many considerations for transplantation, and it is a costly procedure.8,9 Thus, developing new treatments and proper strategies for HE that focus on i) reducing ammonia levels and ii) neuropsychiatric symptoms may be interesting.
Panax Ginseng Meyer (P. ginseng) is known as one of the most commonly used medicinal herbs in the long history of medicine in East Asia. 10 P. ginseng may act favorably against obesity and brain-, cardiac-, and liver-associated diseases.11–13 Previous studies have shown that extracts of P. ginseng have potent neuroprotective effects against brain damage. Furthermore, P. ginseng extract has been shown to ameliorate learning and memory deficits. 14 These pharmacological functions are attributed to ginsenosides, which are major compounds in P. ginseng.15–17
The roots and leaves of P. ginseng are often used to prepare crude drugs or functional foods because they have various physiological effects of P. ginseng.18,19 It has recently been shown that P. ginseng berries may have antidiabetic and anti-obesity effects, which are assumed to be derived from ginsenoside Re, the major compound of the berry. 15
Ginsenoside Re is known to inhibit neuroinflammation by acting on the CAMK/MAPK/NF-kB signaling mechanism in microglia. 20 Since ginsenoside Re acts on the nervous system in this way, we are confident that it will be a very interesting study to elucidate whether HE is closely related to the nervous system by determining its mechanism using an HE model.
Ultrasonication is primarily used to increase ginsenoside Re in ginseng berry. 21 To modify the composition and content of ginsenosides in the extract, ultrasonication is currently used instead of the traditional way. 22 Ultrasonication enhances the hydrolysis reaction and is performed under variable conditions (temperature changes and catalysts) to produce the targeted materials. 23 Ultrasonication can transform ginsenosides and increase the active prosapogenin content, which induces diverse physiological effects. 24
In the present study, the ultrasonication-processed P. ginseng berry extract (UGBE) was prepared by the newly developed ultrasonication procedure from P. ginseng berry extract (GBE). With this UGBE, we explored the protective effects of UGBE under rat HE conditions.
Materials and Methods
Materials
The anti-major histocompatibility complex (MHC) class II antibody was purchased from Novus Biologicals (Littleton, USA). The MHC class II ELISA kit was obtained from MyBioSource (San Diego, CA, USA). The UGBE and GBE was provided using Professor Sung Kwon Ko's patent technic (Korean Patent No 10-1416669, Ginseng berry preparation containing high concentration of ginseng prosapogenin using ultrasonic treatment and its manufacturing method), working at the Department of Oriental Medical Food & Nutrition, Semyung University, Chungbuk, Korea). Ginsenoside standards were obtained from Chromadex (Irvine, CA). Dulbecco's phosphate-buffered saline was acquired from Welgene Inc. (Seoul, Korea).
Preparation of UGBE
Ethyl alcohol 2000mL was added to 200 g of dried P. ginseng berry (4-year grown). GBE was prepared from P. ginseng berry-ethyl alcohol mixture by reflux extraction and filtration twice, followed by concentration using vacuum evaporation. The prepared GBE was oscillated and vibrated using an ultrasonicator (KODO, Hwaseong, Korea) with 600 W at 100˚C for 10 h. The remaining solutions were concentrated by vacuum evaporation and freeze-dried to produce UGBE as a brownish extract.
Then, 2 g of the UGBE and GBE extracts was extracted with diethyl ether (50 mL) three times using an ultrasonicator (KODO, Hwaseong, Korea), and the supernatant was removed for further analysis. The residue was treated three times with 50 mL of n-butanol. The filtration and concentration of the n-butanol fraction during ultrasonication were measured using a vacuum evaporator (Fig. S1).
Analysis of Ginsenosides in UGBE
To analyze the total content and composition of ginsenosides in UGBE and GBE, a Waters 1525 binary high-performance liquid chromatography (HPLC) system (Milford, USA) was used (Fig. S2). Separation of UGBE was conducted using gradient elution with Eurospher analytical column (100-5 C18, 250 mm×3.0 mm, 5 μm, Knauer, Berlin, DE) under room temperature (The eluent A; acetonitrile, The eluent B; distilled water). The elution process was conducted under the condition as follows: 0 min, 17% of A; 25 min, 25% of A; 50 min, 40% of A; 105 min, 60% of A; and 110 min, 100% of A. With a flow rate of 0.8 mL/min and an injection volume of 20 μL, the chromatograms were conducted with a UV/Vis Waters 2478 Dual λ Absorbance Detector (Milford, USA) at 203 nm.
Animals and Experimental Design
In this study, 72 male Sprague–Dawley rats (200-250 g) were obtained from Samtako Bio Korea (Osan, Korea). The air condition was set as 24 ± 2°C with 70 ± 5% humidity. A 12:12 h light/dark cycle was used. Pathogen-free food and water were provided and sterilized bedding was used for housing. All animals were fasted for 24 h prior to surgery and then sacrificed. In this study, all animal experiments were approved by the Institutional Animal Care and Use Committee (IACUC) of Chung-Ang University (IACUC-2016-00095). This study conformed to the ARRIVE 2.0 guidelines. 25
The rats were randomly allocated to eight groups as follows (n = 9): control, sham operation, MBDL, GBE, silymarin, UGBE1, UGBE2, and UGBE3. MBDL surgery was conducted in all rats except those in the control and sham operation groups. Normal saline (2 mL/kg) was orally administered to rats in the control, sham operation, and MBDL groups for 6 weeks, while 250 mg/kg GBE was administered to the GBE group, 250 mg/kg silymarin to the silymarin group, and 100 mg/kg, 250 mg/kg, and 500 mg/kg of UGBE to the UGBE1, UGBE2, and UGBE3 groups, respectively. During the administration period, motor coordination, mobility, spatial learning, and pain response were measured every 2 weeks. All rats included in this study were sacrificed the day after the last administration using CO2 inhalation, immediately followed by the collection of brain samples for further assays.
MBDL Model
In this study, the MBDL rat model was used to evaluate the long-term effects of UGBE on HE. 26 As it is difficult for rats to survive if all bile ducts are ligated, MBDL was performed. The detailed procedure for inducing the MBDL rat model has been described in a previous study. 27 Briefly, only three of the five bile ducts were ligated after a midline abdominal incision in the MBDL surgical procedure. The middle of the bile duct, the site between the three upper bile ducts (proximal) and two lower bile ducts (distal), was ligated. The sham operation group underwent only a midline incision without MBDL surgery.
Spatial Learning and Motor Function
Assessment of spatial learning and related errors, such as reference memory error (RME) and working memory error (WME), was conducted with an eight-arm radial maze and an Ethovision Video tracking system (Noldus, NED), as described previous research.28,29 The rats were trained for six days, with five trials per day, prior to the experiments. Four of the eight arms were randomly selected, and a small pellet was placed on each selected arm at the end of the arms (distal). These arrangements were set specifically for each rat and maintained during training. The rat was placed in the wall, which blocked its sight in the center of the 8-arm radial maze. The trial ended when the rat bit the pellets in the baited arm or 5 min had elapsed. The number of RME (number of visits to unbaited arms) and WME (number of visits to arms already visited in the same trial) were calculated. Measurements of rats that finished the test within 6 min were conducted during the 6-day trial.
Motor function of the rats was evaluated using an EthoVision video tracking system (Noldus, NED). The rats were placed in a 40 × 40 cm×40 cm open black box and allowed to walk freely in the box for 30 min. The velocity and duration of movement were calculated.
Motor Coordination
Motor coordination in rats was evaluated using the RotaRod treadmill (Ugo Basile, ITA) test. 30 The diameter of the rod is 75 mm and the velocity of revolution is 19.4 cm/1 round/2 s. The rat was placed on the rod, and riding time was recorded automatically when the rat fell.
Preparation and Biochemical Assay of Brain Samples
Brain perfusion was conducted using phosphate-buffered saline (PBS) (pH 7.4). Whole brains were removed and dissected into four parts (cerebral cortex, cerebellum, striatum, and hippocampus). After dissection, brain samples were washed in saline and flash frozen immediately at —80 °C for further assays (ammonia level, TNF-α, and IL-1β). Further assays were performed according to manufacturer's instructions.
Immunohistochemistry
Transcardial perfusion of rat brains was performed for immunohistochemistry using PBS (pH 7.4), followed by fixation with a 4% (w/v) paraformaldehyde solution. After being removed, brain samples were dissected in the same way as described above and immersed for 24 h under 4 °C with the same fixative solution. The samples were then immersed in a dextrose solution in PBS for 24 h in a concentration-dependent manner (30-20%, w/v). Immersed brain samples were flash-frozen in liquid nitrogen at —80 °C for cry cutting. Coronal sections of frozen brain samples with a thickness of 5 μm were obtained using cry cut machine. For staining microglial activation, an MHC class II antibody (1:500) was used, and the secondary antibody was a Dako (Santa Clara, USA) RealTM EnVisionTM Detection System Rabbit/Mouse (1:500). The sections were developed using diaminobenzidine, mounted on poly-lysine gelatinized glass slides, and dehydrated using graded ethanol solutions before coverslipping. The stained tissues were observed using a Leica DMR 6000 microscope, and images were captured using a Leica DM 480 camera at 20X magnifications (Wetzlar, Germany).
Cresyl Violet Staining
Cresyl violet staining was performed to detect neuronal damage in the brain. Sectioned 5-μm thick tissues were stained with 0.1% (w/v) cresyl violet solution to detect neuronal damage. The number of Nissl-stained cells was then analyzed. The stained tissues were observed under a Leica DMR 6000 microscope and images were captured using a Leica DM 480 camera at 20X and 40X magnifications (Wetzlar, Germany).
Data Analysis
In this study, the collected data were expressed as mean ± SEM One-way analysis of variance test was performed for statistical analysis of differences between each group. Statistical significance was set at a p-value < 0.05.
Results
HPLC Analysis of UGBE
Our previous study demonstrated the composition ratio of GBE and UGBE using HPLC systems 27 (Fig. S2). Ultrasonication for 10 h at 100˚C significantly increased the levels of the ginsenosides Rg2, Rg3, Rh1, Rh4, Rk1, Rk3, and F4. Additionally, the ginsenosides Rg3 and Rk1, which were not identified in GBE, were detected in UGBE. Ginsenosides that were increased by ultrasonication were found to have fewer polar residues than ginsenoside Re decreased by ultrasonification. 27
Effect of UGBE on Impaired Spatial Learning
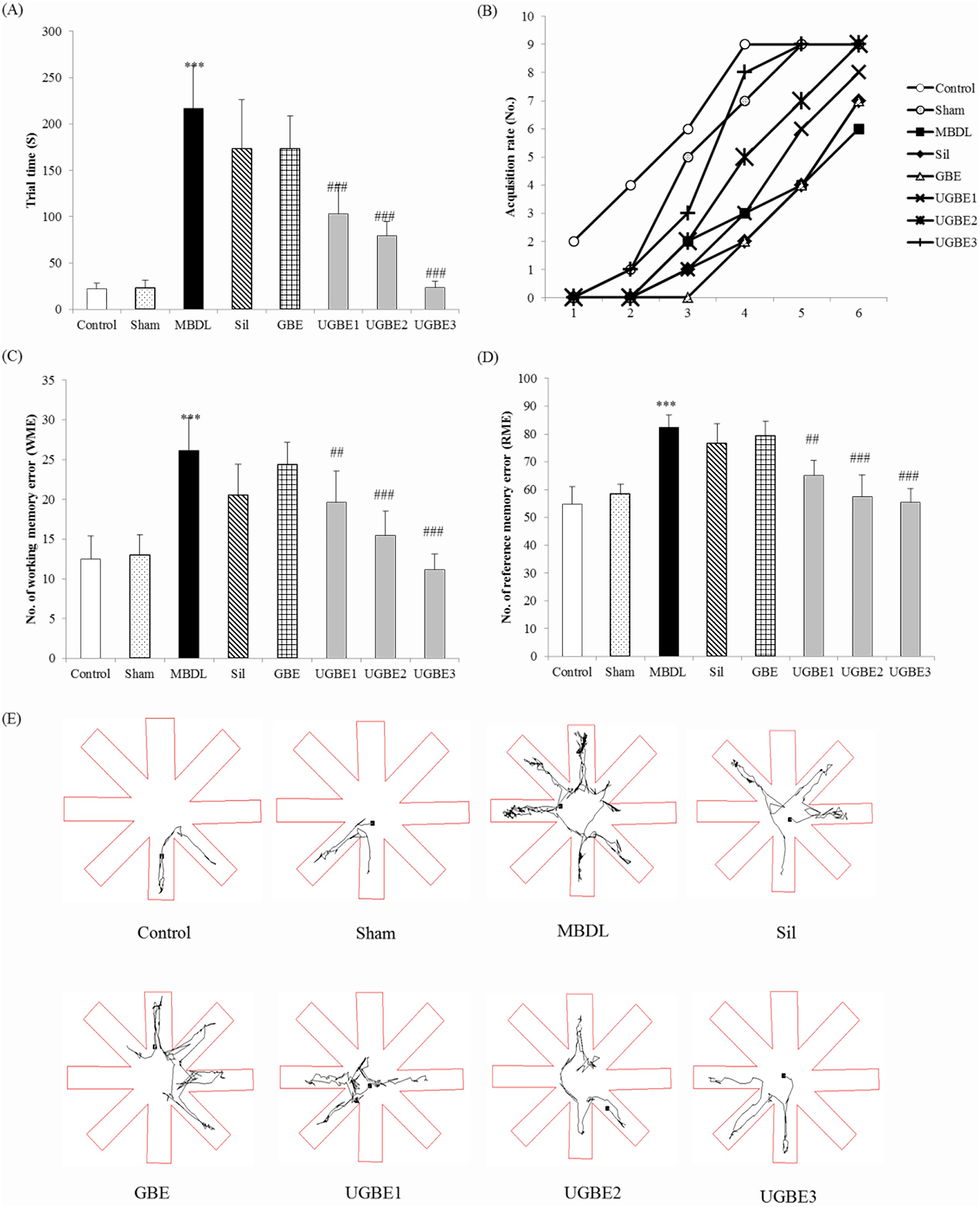
To evaluate the effect of the UGBE on impaired spatial learning induced by MBDL surgery, we conducted a radial maze test (Fig. 1). Four parameters were evaluated: trial time, RME, WME, and the number of rats that finished the test within 6 min. The trial time, RME, and WME were significantly higher in the MBDL group than in the control group. UGBE treatment decreased these parameters in a dose-dependent manner (Fig. 1A, 1C, and 1D). A rapid increase in the number of rats that completed the test within 6 min was observed in the UGBE group during the 6 days of the trial (Fig. 1B). Interestingly, silymarin treatment did not improve spatial learning in MBDL rats. GBE treatment had no effect on the impaired spatial learning.
The motor functions of the rats were recorded using a video-tracking system. The distance moved and the total duration of movement were measured as indicators of mobility (Fig. 2A and 2 B). UGBE treatment improved indicators of mobility in rats. Treatment with UGBE also normalized the decreased rearing and ambulatory counts in MBDL rats (Fig. 2C and 2D). Treatment with silymarin had no effect on impaired motor function in rats. Treatment with UGBE and silymarin normalized the impaired motor coordination in MBDL rats (Fig. 3). Treatment with GBE did not show the effect on impairment in motor function or coordination induced by MBDL surgery.


To evaluate the effect of UGBE on microglial activation, the protein expression of MHC class II molecules in each brain section was measured (Fig. 4). Treatment with UGBE also decreased the protein expression of MHC class II molecules in four brain sections (cerebral cortex, cerebellum, hippocampus, and striatum). Conversely, treatment with silymarin did not reduce MHC class II overexpression in the cerebral cortex or striatum (Fig. 4B and 4H). Immunohistochemical analysis was conducted to confirm the protein expression of MHC class II (Fig. 4A, 4C, 4E, and 4G). The number of MHC class II cells that manifested as positive cell counts in UGBE treatment group was significantly lower than that in the MBDL group. GBE treatment did not affect MHC class II overexpression caused by MBDL surgery.

Table 1 demonstrates the TNF-α level in each brain sections in each treatment group. Treating UGBE was found to reduce the protein expression of TNF-α in four brain sections (cerebral cortex, cerebellum, hippocampus, and striatum). On the other hand, the treatment of silymarin did not reduce TNF-α production in the cerebral cortex and striatum. Moreover, the ammonia levels of each brain section in each treatment group were measured. Ammonia levels in the brain and protein expression of TNF-α in each brain section showed the similar tendency of protein expression of MHC class II. Interestingly, silymarin treatment only affected the ammonia levels in the experimental groups. The treatment of silymarin was found to have no effect on protein expression of TNF-α in the cerebral cortex and striatum. GBE treatment did not affect the neuroinflammation caused by MBDL surgery.
Effect of UGBE on Neuroinflammation.

Effect of UGBE on Neuroinflammation.

Control, control; sham, sham operation control rats; MBDL, MBDL rats; Sil, MBDL rats treated with silymarin (150 mg/kg); GBE, MBDL rats treated with GBE (150 mg/kg); UGBE1, MBDL rats treated with UGBE (100 mg/kg); UGBE2, MBDL rats treated with UGBE (250 mg/kg); UGBE3, MBDL rats treated with UGBE (500 mg/kg). Data are expressed as means ± SEM (n = 9). ***P < 0.005 compared to control, #P < 0.05, ##P < 0.01, ###P < 0.001 compared to MBDL.
Cresyl violet staining of brain sections was performed to determine neuronal degeneration. Neurons appeared more pyknotic and condensed (Fig. 5). A significant decrease in Nissl-stained cells (normal neurons) was observed in the four brain sections induced by MBDL surgery. However, treatment with UGBE increased the number of Nissl-stained cells in four brain sections (the cerebral cortex, cerebellum, hippocampus, and striatum). Furthermore, silymarin only affected the number of Nissl-stained cells in the cerebellar and hippocampal sections. GBE treatment did not affect the neuronal damage caused by MBDL surgery.

P. ginseng exerted various physiological effects. 31 Ginsenoside Re, the major component of P. ginseng berries, was found to have antidiabetic and anti-obesity effect.15,32 However, the protective effects of ginsenosides in HE remain unclear. This study aimed to analyze the components of UGBE and investigate their protective effects in a rat model.
P. ginseng has been used as a medicinal herb in Asia for a long time and has been verified for its safety and effectiveness. 33 Generally, the LD 50 value in rats is 750 mg/kg; however, in other studies, no hematological or histological abnormalities were found, even when administered at a dose of 4000 mg/kg for 20 days. 34 However, in the case of pregnant women, experimental results reveal that it should be administered with caution during the first 3 months of pregnancy. 35
In our previous study, we confirmed that the composition ratios of Rh1, Rh4, Rg2, Rg3, Rk1, Rk3, and F4 were significantly higher in UGBE than in GBE. Ginsenoside Re levels were lower in UGEB than in GBE. In the case of UGBE used in this study, although the ginsenoside RE content was lower than that of GBE, the neuronal protective effect was superior to that of GBE. Hence, follow-up studies on the neuroprotective effects of ginsenosides are required. These components, such as Rh1, Rg2, Rg3, Rk1, and F4, are known to have anti-inflammatory, antioxidative, anti-carcinogenic effects.36–41 Hence, UGBE could exert protective effects against HE through these components increased by ultrasonication. There are limitations in conducting experiments on the direct effects of the components involved in the protective effects in the HE models. As the current experiments are industrialized utilization technologies that use patents, research at the component level will be challenging in terms of time and cost in developing industrial materials. Additional cell studies will determine whether biomarkers or factors that mediate inflammatory signaling pathways are altered.
Microglia are the glial cells that mainly participate in the active immune response of the central nervous system (CNS), including the brain and spinal. 42 Microglia transform into their active form (reactive microglia or microglial activation) in response to injuries or pathogens and phagocyte-damaged cells and debris, which causes neuroinflammation.43,44 Excessive ammonia production leads to hyperammonemia. Under our experimental HE conditions, the microglia were activated by hyperammonemia. Ammonia, a representative cytotoxic agent in the CNS, plays a key role in the neurological pathophysiology of HE. 45 Several studies have reported that ammonia induces microglia activation, astrocyte swelling, migration, and production of inflammatory cytokines through the blood–brain barrier.46–48 Neuronal damage is known to have detrimental effects on cognitive function, spatial learning, and locomotor ability. 49 Ammonia overproduction by MBDL was found to be significantly decreased in the brain after treatment with silymarin or UGBE. Based on the fact that UGBE treatment reduced ammonia more than silymarin treatment, it is assumed that UGBE is more potent than silymarin in the treatment of hyperammonemia.
Impaired spatial learning and locomotor activity are the representative symptoms of HE. 50 The 8-arm radial maze is commonly utilized as a behavioral paradigm for spatial learning and cognitive function.28,29 In our experiments, the 8-arm radial maze test revealed a significant increase in the trial time in the MBDL-induced HE model, indicating impaired memory acquisition and spatial learning. However, UGBE treatment restored the RME and WME. Moreover, treatment with UGBE improved the acquisition rate of spatial learning, whereas other treatment groups showed no effect on spatial learning and cognitive function.
In the HE condition, rats usually exhibited a decline in spontaneous motor activities, including distance traveled, time traveled, number of rearing, and ambulatory count. 51 Treatment with UGBE normalized the impaired motor function paradigms. Although there are still some controversies regarding the effects of MBDL surgery on motor coordination, 52 the impairment in motor coordination, which was measured using the rotarod test in our MBDL models, was recovered through treatment with UGBE.
All these behavioral impairments in MBDL surgery are related to locomotor and cognitive functions in the brain, including the cerebral cortex, cerebellum, hippocampus, and striatum. In the present study, neuroinflammation in each part of the brain was analyzed based on the results of behavioral tests. To investigate microglial activation in the brain, protein expression of MHC class II, a representative surface molecule of reactive microglia for antigen presentation, was measured. 53 Several studies reported that the microglia activation facilitated TNF-α-mediated motor neuron death.54,55 In this study, a significant increase in the expression of MHC class II and TNF-α by MBDL surgery was found in all brain sections. In the cresyl violet assay, neuronal death was observed by visualization of Nissl-stained cells, indicating the normal viability of the cells in each section.
Silymarin was used as the positive control in the present study. Silymarin is widely known to have substantial hepatoprotective effects that have led to its commercialization by pharmaceutical companies. 56 Silymarin also reduced hepatotoxicity caused by MBDL surgery in rats. However, the protective effect of silymarin on neuronal damage was somewhat questionable compared to that of silymarin in the liver and was less potent than that of UGBE. In all behavioral tests, the silymarin-treated group demonstrated improved motor coordination compared to the MBDL group. Silymarin did not affect spatial learning, memory, cognitive function, or motor function. Moreover, the treatment of silymarin did not affect the microglia activation and production of TNF-α in the cerebral cortex and striatum. The neuronal damage pattern in silymarin-treated group reveals a similar pattern to that of MHC class II and TNF-α expression. Motor coordination is regulated by activity in the hippocampus and cerebellum.57,58 The cerebral cortex is closely related to spatial learning and cognitive function, 59 and the striatum controls spontaneous motility under physiological and pathophysiological condition. 60 Thus, silymarin exerts protective effects on the hippocampus and cerebellum. Further studies are required to understand the exact protective mechanisms of silymarin against neuronal damage.
Based on these results of the present study, we suggest that oral administration of UGBE reduces MBDL-induced neuronal damage in the brain. Additionally, brain ammonia levels were reduced following UGBE administration. This protective effect is mediated by the suppression of microglial activation in the brain. Consequently, UGBE showed a significant protective effect in the MBDL-induced HE rat model and has the potential to be developed as a new remedy for HE.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251318889 - Supplemental material for Ultrasonicated Ginseng Berry Extract mediated protective effect against Mild Bile Duct Ligation induced Hepatic Encephalopathy in rats
Supplemental material, sj-docx-1-npx-10.1177_1934578X251318889 for Ultrasonicated Ginseng Berry Extract mediated protective effect against Mild Bile Duct Ligation induced Hepatic Encephalopathy in rats by Won Seok Choi, Yoonjin Nam, Ji-Yun Lee, Jong Hyuk Lee and Uy Dong Sohn in Natural Product Communications
Supplemental Material
sj-jpeg-2-npx-10.1177_1934578X251318889 - Supplemental material for Ultrasonicated Ginseng Berry Extract mediated protective effect against Mild Bile Duct Ligation induced Hepatic Encephalopathy in rats
Supplemental material, sj-jpeg-2-npx-10.1177_1934578X251318889 for Ultrasonicated Ginseng Berry Extract mediated protective effect against Mild Bile Duct Ligation induced Hepatic Encephalopathy in rats by Won Seok Choi, Yoonjin Nam, Ji-Yun Lee, Jong Hyuk Lee and Uy Dong Sohn in Natural Product Communications
Supplemental Material
sj-jpeg-3-npx-10.1177_1934578X251318889 - Supplemental material for Ultrasonicated Ginseng Berry Extract mediated protective effect against Mild Bile Duct Ligation induced Hepatic Encephalopathy in rats
Supplemental material, sj-jpeg-3-npx-10.1177_1934578X251318889 for Ultrasonicated Ginseng Berry Extract mediated protective effect against Mild Bile Duct Ligation induced Hepatic Encephalopathy in rats by Won Seok Choi, Yoonjin Nam, Ji-Yun Lee, Jong Hyuk Lee and Uy Dong Sohn in Natural Product Communications
Footnotes
Acknowledgments
This research was supported by the High Value-Added Food Technology Development Program, Ministry of Agriculture, Food and Rural Aggairs (113021-03) and the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Science, and Technology [Grant NRF-2019R1F1A1062070].
Author Contributions
Experiments were done by Won Seok Choi and Yoonjin Nam in pharmacology laboratory in Chung-Ang University. Won Seok Choi and Yoonjin Nam analysed raw data following Ji-Yun Lee's instructions. Manuscript is written by Won Seok Choi, Yoonjin Nam and Jong Hyuk Lee. Jong Hyuk Lee and Uy Dong Sohn organized all procedure related to this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
Animal experiments in this study were approved by the Institutional Animal Care and Use Committee (IACUC) of Chung-Ang University (IACUC-2016-00095).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
